Abstract
In transfusion medicine, blood typing is an integral part of pretransfusion testing. The objective of the current study was the clinical evaluation of an automated canine cartridge dog erythrocyte antigen (DEA) 1.1 blood-typing method (QuickVet®/RapidVet®) and comparison of the results with a gel column–based method (ID-Gel Test Canine DEA 1.1). Ethylenediamine tetra-acetic acid–anticoagulated blood samples from 11 healthy and 85 sick dogs were available for typing. Before blood typing, all samples were tested for agglutination and hemolysis. All samples were tested once or multiple times with both methods according to the manufacturer’s guidelines. With the gel method, 53 dogs tested DEA 1.1 positive and 42 dogs DEA 1.1 negative; blood typing was not possible due to erythrocyte autoagglutination in 1 dog. With the cartridge test, 53 samples tested DEA 1.1 positive, 34 samples tested DEA 1.1 negative, and 6 results were inconclusive (3 samples were not included due to autoagglutination or severe hemolysis). Without taking the inconclusive samples into account, the agreement between both methods was 96.5%. The sensitivity and specificity for samples that were definitively typed by both methods were 100% and 91.9%, respectively. The cartridge test was suitable for in-clinic canine DEA 1.1 blood typing, although some discrepancies compared to the gel method existed. The cartridge test is software-directed, is easy to use, and does not require user interpretation, but preanalytical guidelines (sample evaluation for agglutination and hemolysis) have to be followed. For inconclusive results, an alternate blood-typing method should be performed.
Introduction
Red blood cell (RBC) transfusions have become an integral part of intensive care treatment in critically ill dogs. To ensure safe and efficient transfusions, blood group determination is recommended not only in human beings but also in dogs. 17 Blood types are genetic markers on the surface of RBCs; a set of 2 to several alleles at 1 gene locus makes up a blood group system. The canine blood type classification uses the dog erythrocyte antigen (DEA) system; a number is used to represent the blood group system or the blood type (Vriesendorp HM, Westerbroek DL, D’Amaro J, et al.: 1973, Joint report of 1st International Workshop on Canine Immunogenetics. Tissue Antigens 3:145–163; Vriesendorp HM, Albert ED, Templeton JW, et al.: 1976, Joint report of the 2nd International Workshop of Canine Immunogenetics. Transplant Proc 8:289–314). More than 12 blood group systems have been recognized in dogs.3,6,9 Most of these blood group systems have 2 alleles, which means a dog can be positive or negative for that blood type. The DEA 1.0 system contains 3 antigens, DEA 1.1, DEA 1.2, and DEA 1.3, and a null type (equivalent to Aa1, Aa2, Aa3, and A–).15,16 A null phenotype means that the dog does not carry genes for expression of the antigen. Dog erythrocyte antigen 1.1 is the most common antigen in this system and is expressed by the RBCs of approximately 45%–64% of dogs in different countries.12,14
In dogs, no clinically important, naturally occurring alloantibodies against RBC antigens have been detected. An acute hemolytic transfusion reaction in a dog receiving its first transfusion has not been reported. However, if blood type–incompatible RBC transfusions are administered, alloantibody production can be induced. 3 Dog erythrocyte antigen 1.1 seems to be the most antigenic canine blood type. Transfusion of DEA 1.1–positive erythrocytes to a DEA 1.1–negative dog will lead to the formation of alloantibodies within approximately 4 days. 1 An acute life-threatening hemolytic transfusion reaction has been documented in a sensitized DEA 1.1–negative dog transfused with DEA 1.1–positive blood. 7 If a female dog has received a mismatched RBC transfusion prior to whelping, alloantibodies in the colostrum might possibly cause alloimmune hemolytic anemia (neonatal isoerythrolysis) in neonatal puppies.4,10,18
Blood types other than DEA 1.1 seem to be less antigenic. However, severe hemolytic transfusion reactions have been reported in response to DEA 4 and to an unidentified common RBC membrane antigen.5,13 A fatal hemolytic transfusion reaction occurred in a dog receiving 3 DEA 1.1–compatible transfusions; a cross-match prior to the third transfusion was not performed (unpublished observation). Therefore, a cross-match test prior to transfusion is strongly recommended in dogs that have received their first transfusion more than 4 days previously.
The availability of canine blood-typing reagents is limited, and typing against blood types other than DEA 1.1 is only offered by specialized laboratories (Animal Blood Resources International, Stockbridge, Michigan). Since the mid-1990s, blood-typing cards for DEA 1.1 a have been commercially available, enabling typing for DEA 1.1 in clinical practice (Seth M, Winzelberg S, Jackson KV, Giger U: 2008, Comparison of gel column, card, and cartridge techniques for DEA 1.1 blood typing of dogs. J Vet Intern Med 22:775. Abstract).8,11 The principle of this card-based agglutination test and all other DEA 1.1 blood-typing methods is a visible hemagglutination reaction resulting from the binding of the DEA 1.1 RBC surface antigen to antibodies. The antibody used for the card test is a monoclonal antibody developed by the Kansas State University. 2 In the last decade, additional canine blood-typing tests were made commercially available. A gel column microtube agglutination test b is based on agglutination of RBCs in microcolumns containing DEA 1.1 monoclonal antibodies (Seth M, et al.: 2008, Comparison of gel column, card, and cartridge techniques). 8 An immunochromatographic membrane dipstick technique c is based on the migration of RBCs on a monoclonal antibody–containing membrane; agglutinating erythrocytes form a visible line on the membrane (Seth M, et al.: 2008, Comparison of gel column, card, and cartridge techniques).
In 2011, a new canine blood-typing method was developed that consists of a one-time-use cartridge inserted into an analyzer and in need of only minimal user interpretation. d The monoclonal antibody used for the cartridge test is the same as the antibody used for the card test. a The objective of the current study was the clinical evaluation of this canine cartridge blood-typing system and comparison of the results with the gel column test.
Material and methods
Blood samples
Ninety-six blood samples were collected from 96 dogs of 40 different breeds and of 30 mixed-breed dogs presented at the Small Animal Clinic, Faculty of Veterinary Medicine, Freie Universität Berlin (Berlin, Germany) from May through July 2011. Eleven of the dogs were healthy blood donors, and 85 dogs suffered from various diseases, including anemia (immune-mediated, blood loss, and nonregenerative anemias); orthopedic, neurological, gastrointestinal, and respiratory diseases; trauma; neoplasia; and others. Blood was drawn from the cephalic, saphenous, or jugular vein and was placed in a tube containing ethylenediamine tetra-acetic acid (EDTA) as anticoagulant. The blood samples were analyzed immediately or were stored up to 6 days at 4°C until blood type determination. The hematocrit value (Hct) or packed cell volume (PCV) of these samples ranged from 0.13 to 0.64 l/l (mean: 0.45 l/l, SD ±11.6). Twenty-five dogs were anemic (PCV 0.13–0.36 l/l, mean: 0.28 l/l, SD ±5.5).
Before blood typing, all samples were tested for agglutination and hemolysis. To test for agglutination, a drop of EDTA blood was placed on a slide and mixed with a drop of phosphate buffered saline (PBS) solution. If there was severe agglutination present, the RBCs were washed 3 times with PBS; if the agglutination persisted, it was considered as persistent autoagglutination (n = 1). For all samples, a PCV was established; based on the color of the plasma, the sample was graded as no (n = 59), mild (n = 35), or severe (n = 2) hemolysis.
Cartridge test
The cartridge typing system consists of a one-time-use cartridge d with 3 capillary channels (Fig. 1) that is used with an analyzer. e The cartridge is inserted into the analyzer, where it is heated to 37°C. After measuring the Hct, the blood sample was diluted with PBS according to the on-screen instructions. The proper dilution (number of diluent drops) was calculated by the analyzer based on the measured Hct. Afterward, 100 µl of the diluted sample was added to the preheated cartridge (Fig. 2). The sample advances through the channels by capillary action. At a single site for each of the reaction channel and reference channel, the analyzer continuously measures the intensity of light transmitted through the blood sample as the blood flows through each channel. By comparing the curve of transmitted light for the reaction channel and reference channel, the software determines whether agglutination has occurred and reports the results within 5 min as positive, negative, or inconclusive based on the presence or absence of agglutination. Inconclusive results are reported when the analyzer cannot make a certain classification of the sample as either DEA 1.1–positive or DEA 1.1–negative based on features in the measured signal (Fig. 3). All blood-typing results are automatically archived in the analyzer and can be printed or exported via a network connection to a Central Laboratory Information System.
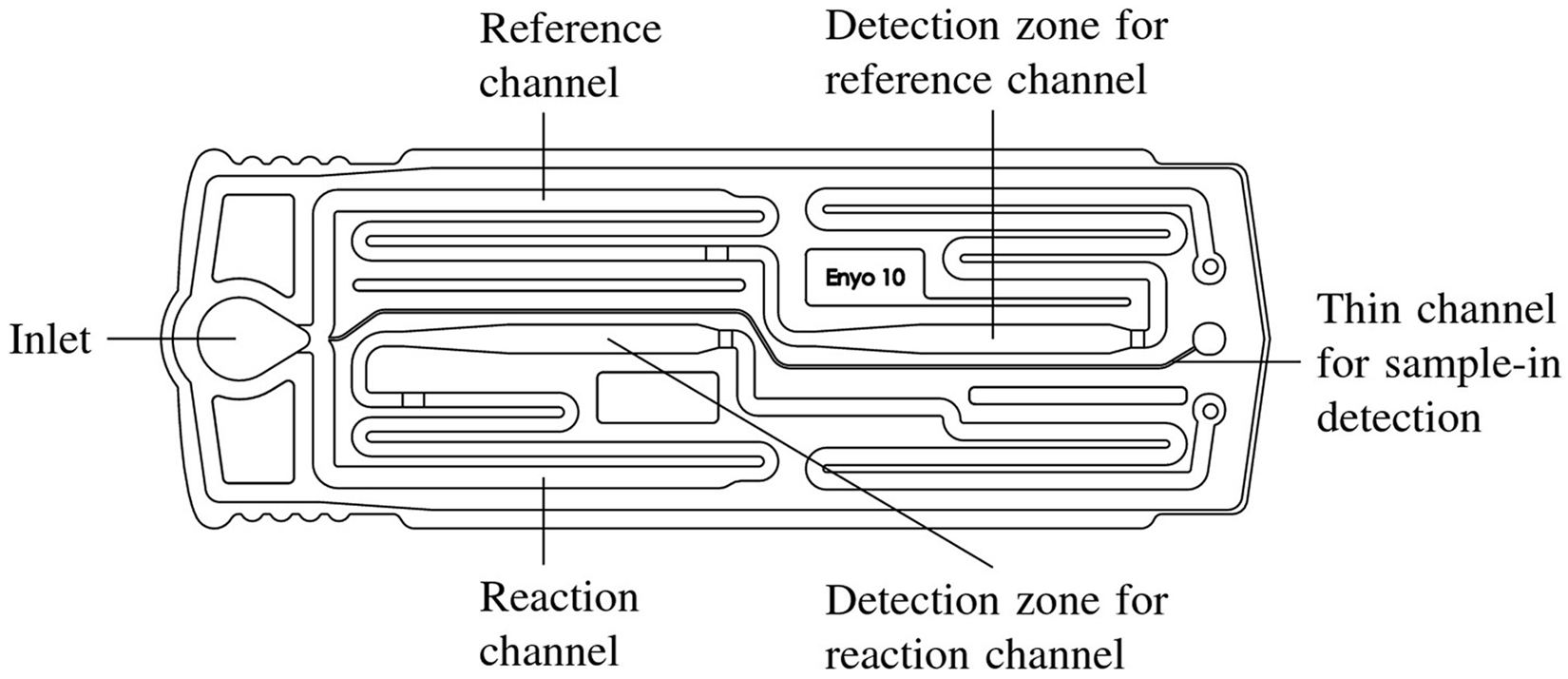
QuickVet®/RapidVet® dog erythrocyte antigen (DEA) 1.1 blood-typing cartridge with 3 channels: a thin channel to detect when adequate sample has been added to the cartridge; a reaction channel with DEA 1.1–specific monoclonal antibodies and immunoassay stabilizer; and a reference channel with immunoassay stabilizer (contains no antibodies).

The diluted blood sample is added to the cartridge preheated in the analyzer to 37°C.
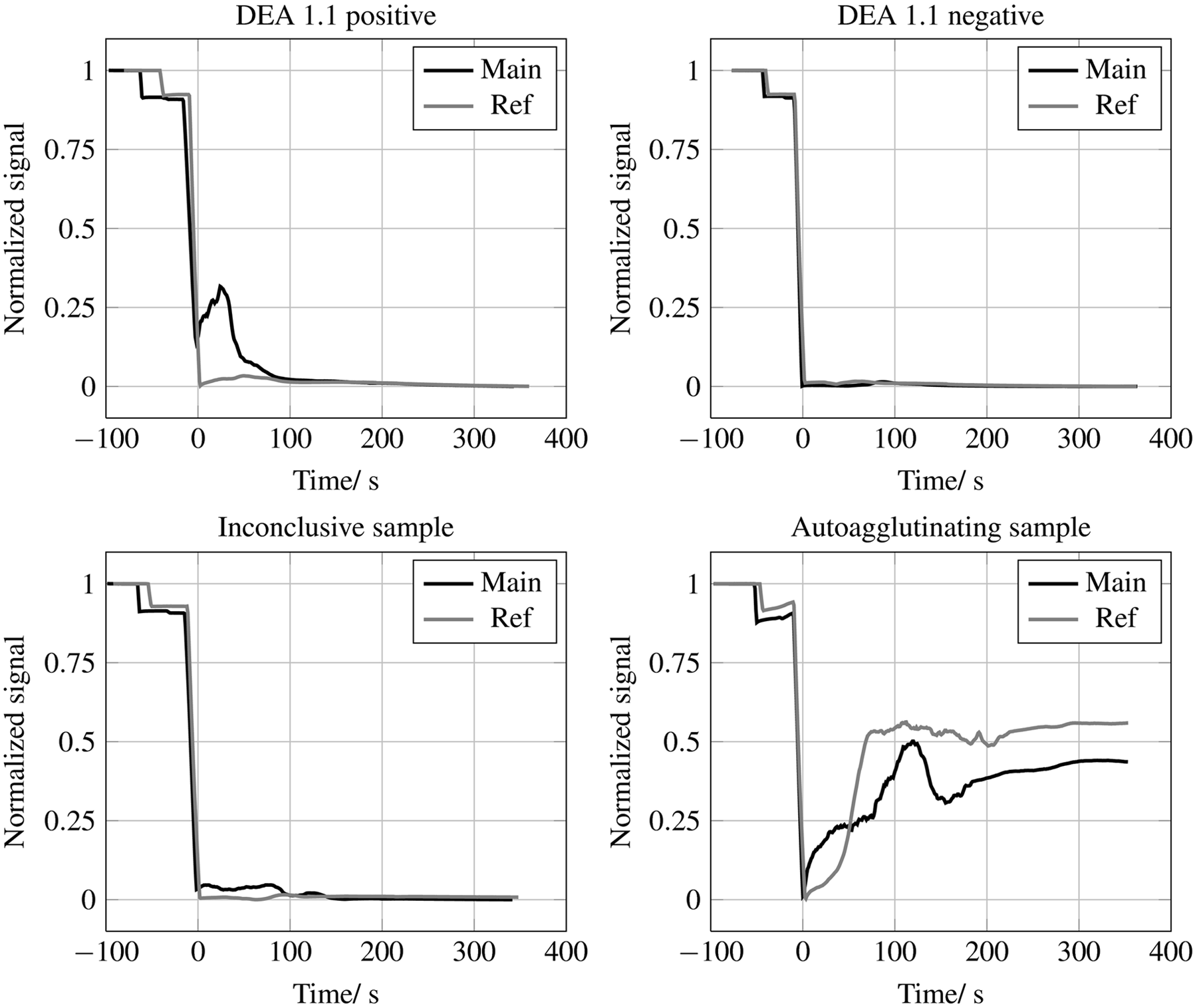
Cartridge test signals from dog erythrocyte antigen (DEA) 1.1–positive and DEA 1.1–negative blood samples as well as inconclusive and autoagglutinating blood samples. DEA 1.1–positive samples have an increased signal in the reaction (Main) channel and no signal in the reference channel (Ref). DEA 1.1–negative samples have almost identical signals in the reaction (Main) channel and the reference channel. Inconclusive samples have insufficient difference between the signals in the 2 channels for the analyzer software to make a definite decision about the blood type of the tested sample. In case of autoagglutination, there is an increased signal in the main channel as well as in the reference channel.
Gel column test
One gel column card b contains 6 polypropylene gel columns, allowing typing of 3 blood samples. The gel columns labeled as DEA 1.1 contain monoclonal anti–DEA 1.1 antibodies (cell line 5B6) in the gel matrix. The gel matrix of the control column does not contain any reagents and serves as the negative control (autoagglutination control). For preparation of a 5% RBC suspension, 500 µl of diluent f was mixed with 25 µl of packed RBCs. The RBC suspension was incubated for 10 min at room temperature. Thereafter, 12.5 µl of the RBC suspension was added to the DEA 1.1 column and to the control column. The card was centrifuged at 80 × g for 10 min in a centrifuge provided by the company. g The cards were visually interpreted as follows: (0) negative, when all RBCs are at the bottom of the tube; 1+, when very few RBC agglutinates are dispersed in the lower part of the gel, with most RBCs at the bottom of the tube; 2+, when all RBCs are agglutinated and dispersed in the gel; 3+, when some RBC agglutinates are dispersed in the upper part of the gel and most of the RBCs formed a red line on the surface of the gel; and 4+, when all RBCs formed a red line on top of the gel. Retention of ≥2+ was considered a positive test result (Seth M, et al.: 2008, Comparison of gel column, card, and cartridge techniques). 8
Testing design
All tests were performed by the same laboratory technician. At the time of performing 50 of the 96 cartridge tests, the technician was blinded to the gel test result, the samples being labeled only by a bar code. In performing the remaining 46 tests, the technician was not blinded to the gel test results first, but since the result of the cartridge test is determined by software, she could not affect those results.
All samples were tested first with the gel test (85 tests once, 10 tests twice, 1 test 3 times). Then the cartridge test was performed once or several times using the same samples (61 samples were tested once, 23 samples twice, 5 samples 3 times, 3 samples 4 times, 2 samples 5 times, 1 sample 6 times, and 1 sample 14 times). The positive and negative samples that were retested were chosen randomly. All the inconclusive samples were retested once or several times. Two analyzers were available, which were used alternately. For direct comparison of the results between the gel test and the cartridge test, the result from the first gel and first cartridge test typing was always used.
Agreement, sensitivity, and specificity
The sensitivity and specificity of the cartridge test were determined in comparison with the gel test as follows: sensitivity by index of the performance of the cartridge test, calculated as the percentage of dogs with a DEA 1.1–positive test result that are correctly classified as being positive; specificity by index of the performance of the cartridge test, calculated as the percentage of dogs with a DEA 1.1–negative test result that are correctly classified as being negative. Agreement was defined as the percentage of concordant results between the cartridge and the gel test. The 95% confidence interval (CI) was calculated using BIAS. h
Results
Gel test
With the gel method, 53 dogs typed as DEA 1.1–positive (4+: n = 46; 3+: n = 5; 2+: n = 2) and 42 dogs as DEA 1.1 negative (0: n = 40; weak 1+: n = 2). For 1 dog with immune-mediated hemolytic anemia, blood typing was not possible due to persistent autoagglutination (control column positive). In 10 cases, the gel test was repeated; the results were always in agreement.
Cartridge test
With the cartridge test, 53 samples tested DEA 1.1 positive, 34 samples tested DEA 1.1 negative, and 6 results were inconclusive (first measurement). According to the manufacturer’s instructions, severely hemolyzed or agglutinating samples cannot be used for typing with the cartridge test. Two blood samples were severely hemolyzed and 1 sample was autoagglutinating; therefore, these samples were typed but the results were not used for further evaluation. The dog with persistent agglutination of RBCs and inconclusive typing result with the gel test was positive with the cartridge test (Fig. 3). The 2 samples that were severely hemolyzed tested negative with the cartridge test.
Seventeen DEA 1.1–positive samples were repeated between 1 and 5 times. Sixteen samples always tested positive on repeat, 1 sample was positive once and inconclusive once, and no positive sample tested negative on repeat. Eleven negative samples were repeated 1 or 2 times; all samples tested negative on repeat. Six blood samples were initially inconclusive. When retested between 1 and 13 times, 3 were consistently inconclusive (tested 1 or 2 times), 1 was negative once and inconclusive 2 times, and 1 was positive twice and inconclusive (once) on repeat. The sixth sample was 7 times positive, once negative, and 5 times inconclusive on repeat. If samples were tested on the 2 different analyzers available in the laboratory, there was no difference between the results.
Comparison of cartridge and gel test results
Table 1 shows the results of the comparison between gel test and cartridge test results (first measurement). Only the results of 93 measurements were used for the statistical evaluation because 2 samples were severely hemolyzed and 1 sample was autoagglutinating. The dog with persistent agglutination of RBCs and inconclusive typing result with the gel test was positive with the cartridge test. The 2 samples that were severely hemolyzed were negative and positive with the gel test, respectively. Both samples tested negative with the cartridge test.
Dog erythrocyte antigen 1.1 blood typing results of the gel test and the cartridge test (results from the first gel and first cartridge test typing were used).*

Numbers in parentheses are percentages. The gel test consisted of the ID-Gel Test Canine DEA 1.1 (DiaMed AG, Cressier sur Morat, Switzerland). The cartridge test consisted of the QuickVet®/RapidVet® DEA 1.1 Blood Typing Cartridge (Scandinavian Micro Biodevices ApS, Farum, Denmark).
The cartridge test was in agreement with the gel test in 90.3% (84/93) of all measurements (95% CI: 0.82–0.95). Considering only the positive or negative cartridge test results, the agreement between both test methods amounted to 96.5% (84/87; 95% CI: 0.90–0.99). The sensitivity was 96.2% (50/52) with a 95% CI of 0.87–1.00. The specificity was 82.9% (34/41; 95% CI: 0.68–0.93) if all measurements were taken into account. The sensitivity and specificity for samples that were definitely typed by both systems were 100.0% (50/50; 95% CI: 0.94–1.00) and 91.9% (34/37; 95% CI: 0.78–0.98), respectively.
Practicability of the cartridge test
To perform the cartridge test, the PCV has to be determined because the blood sample has to be diluted accordingly. Moreover, the samples have to be tested for hemolysis and agglutination. The procedure itself takes 10 min (preheating, reading of the results). User test performance and interpretation of the cartridge test are software-directed and it is therefore easy to use. The test result is given by the analyzer and does not require user interpretation.
Discussion
The cartridge test evaluated in the current study is the first automated blood-typing test in veterinary medicine. The cartridge test principle is based on the difference in the absorption properties of plasma and RBCs (hemoglobin). In the reference channel, there are RBCs suspended in plasma in a homogeneous way. In the test channel, for samples negative for DEA 1.1, there are also RBCs suspended in plasma in a homogenous way. In a test channel for samples positive for DEA 1.1, the fact that agglutinates exist means there are plasma segments in the channel that contain fewer or no RBCs.
In contrast with the other test systems available, no user interpretation is necessary. However, the user has to follow strict preanalytical guidelines. Determination of Hct is needed to perform correct dilution of the sample, and the user is required to visually inspect the plasma for hemolysis. If a blood sample has not been sufficiently diluted prior to being added to the cartridge, the analyzer might not be able to distinguish between a high concentration of RBCs in the reference channel and the agglutinates in the test channel. This can lead to a false-negative test result. According to the manufacturer, the test is robust against deviations of 1 drop from the number of droplets calculated to be required (not evaluated in the present study).
Severe agglutination or hemolysis will impede correct interpretation of the results because autoagglutination can lead to a false-positive result and severe hemolysis to a false-negative result. According to the manufacturer, the software identifies severe agglutination and aborts the test with an advisory note. This software was not available at the time of testing. The RBCs can be washed with physiological saline; if the agglutination breaks up, the blood-typing test can be performed. Severely hemolytic samples should not be used, as the analyzer is unable to distinguish between the absorption of the hemolyzed plasma in the reference channel and any agglutinates in hemolyzed plasma in the test channel. In the present study, mildly hemolytic samples led to accurate results. A severely hemolytic sample, however, was typed negative when the gel test was positive.
The agreement between the cartridge and the gel test, which is considered the standard typing method for comparison, amounted to 90.3% (84/93); when the inconclusive samples were not taken into account, the agreement was 96.5% (84/87). In a 2008 published abstract (Seth M, et al.: 2008, Comparison of gel column, card, and cartridge techniques), 3 canine DEA 1.1 blood-typing assays (the gel test used in the current study, a card agglutination assay, and an immunochromatographic method) were compared. Identical typing results were only obtained in 84% of cases by all methods.
When the inconclusive samples were not considered, the specificity of the cartridge test compared with the gel test was 91.9%. The correct identification of DEA 1.1–negative recipients is very important to prevent sensitization of a negative dog with DEA 1.1–positive blood. When screening blood donors, the sensitivity, which was 100% with the cartridge test, is more important, as DEA 1.1–positive donors should not be misidentified as being DEA 1.1 negative.
Overall, there was a good agreement between the 2 methods in the present study, but test result discrepancies were seen. Differences in the monoclonal antibodies used by the 2 systems may be the cause for contradictory results. In 3 cases, the gel test was negative when the cartridge test was positive. Two of these samples were also tested with the card test, which uses the same antibody as the cartridge test. 2 The results were in both cases positive (2+, 3+; data not shown). When 23 canine samples were tested with the card test and the gel tube test in an earlier study, 8 13 were positive with the card but only 9 were positive with the gel tube test. The MSU (Michigan State University) test classified these 4 dogs as DEA 1.2 positive. The conclusion of this study was that the card test may produce weak reactions with blood from DEA 1.2–positive dogs. 8 In the present study, however, the card test results were strong; therefore, a DEA 1.2 reaction causing the positive result with the card and cartridge test and the negative result with the gel tube seemed less likely.
Six samples were tested as inconclusive. If the user is convinced that the sample quality is sufficient and the testing (e.g., the dilution step) was performed correctly, it is recommended to test the dog with another method (e.g., in a reference laboratory). A repetition of a sample for which the cartridge test was correctly performed is not recommended based on the present data. For inconclusive samples, the software of the cartridge test analyzer gives a clinical guideline regarding transfusion decisions. If a donor is tested inconclusively, it should be considered DEA 1.1 positive in order to prevent sensitization of a DEA 1.1–negative patient. However, if a recipient is tested inconclusively, the dog should only receive DEA 1.1–negative RBCs. 6 Three of 6 inconclusive samples were consistently inconclusive. One sample with a negative gel test result was inconclusive twice and negative twice with the cartridge test. Another sample (gel test negative) was inconclusive twice and positive twice. The sixth sample was negative with the gel test and either inconclusive, positive, or negative with the cartridge test. Inconclusive tests might be analogous to a 0.5+ or 1+ result in other typing systems such as gel tube or card systems (Seth M, et al.: 2008, Comparison of gel column, card, and cartridge techniques).8,11 The 2 dogs that had a weak 1+ reaction with the gel test had inconclusive results with the cartridge method.
In conclusion, the cartridge test was suitable for in-house DEA 1.1 blood typing, but there were some discrepancies compared with the results of the gel method. The cartridge test is software directed, is easy to use, and does not require user interpretation, but strict preanalytical guidelines have to be followed. For inconclusive results, an alternate blood-typing method should be performed.
Footnotes
Acknowledgements
The authors thank Dr. Gisela Arndt, Institute for Biometrics and Data Processing, Freie Universität Berlin, for her help with the statistical analysis.
a.
DMS RapidVet-H®, DMS Laboratories Inc., Flemington, NJ.
b.
ID-Gel Test Canine DEA 1.1, DiaMed AG, Cressier sur Morat, Switzerland.
c.
Quick Test DEA 1.1, Alvedia, Lyon, France.
d.
QuickVet®/RapidVet® DEA 1.1 Blood Typing Cartridge, Scandinavian Micro Biodevices ApS, Farum, Denmark.
e.
QuickVet® Analyzer, Scandinavian Micro Biodevices ApS, Farum, Denmark.
f.
ID-Diluent VET 1: modified bromelin solution, DiaMed AG, Cressier sur Morat, Switzerland.
g.
ID-Centrifuge 12 S II, DiaMed Microtyping System, Cressier sur Morat, Switzerland.
h.
BIAS for Windows, Version 9.04, Epsilon-Verlag, Düsseldorf, Germany
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Kohn received research support in the form of analyzers, cartridges, reagents, and reimbursement for the gel column cards used from Scandinavian Micro Biodevices ApS and DMS Laboratories Inc.
