Abstract
Encephalitozoon cuniculi is an important microsporidian pathogen that is considered an emergent, zoonotic, and opportunistic. It infects both domestic and laboratory rabbits, generating severe chronic interstitial and granulomatous nephritis with fibrosis and granulomatous encephalitis. Encephalitozoonosis is diagnosed in paraffin-embedded sections by examining the spores in the host tissues. The spores are difficult to observe when the samples are stained with hematoxylin and eosin (H&E), particularly when there is an inflammatory reaction and tissue damage. The spores are easily mistaken for other microorganisms, such as fungi (yeasts), protozoa, and bacteria. In our study, we used kidney samples from E. cuniculi–positive rabbits and employed 14 recommended histologic stains for detecting microsporidia spores: alcian blue, calcofluor white, Giemsa, Gram, Grocott, H&E, Luna, Luxol fast blue, Masson trichrome, modified trichrome stain (MTS), periodic acid–Schiff reaction (PAS), Van Gieson, Warthin–Starry (WS), and Ziehl–Neelsen (ZN).We concluded that MTS and Gram stain, detected by light microscopy, and calcofluor white stain, detected by ultraviolet light microscopy, are the best stains for detecting spores of E. cuniculi in paraffin-embedded tissues from infected rabbits. These stains were superior to WS, ZN, Giemsa, and PAS for identifying spores without background “noise” or monochromatic interference. Also, they allow individual spores to be discerned in paraffin-embedded tissues. MTS allows observation of the polar tube, polaroplast, and posterior vacuole, the most distinctive parts of the spore.
Introduction
Encephalitozoon cuniculi is an obligate, spore-forming, intracellular eukaryotic microorganism that belongs to the phylum Microsporidia.3,5 Molecular analysis of this parasite has suggested that its taxonomic position may be among fungi rather than protozoa. 51 Encephalitozoon cuniculi is a common pathogen of rabbits (Oryctolagus cuniculus) causing renal and central nervous system chronic disease characterized by granuloma formation and fibrosis. 29 However, it also affects other mammals, including rodents, carnivores, herbivores, nonhuman primates, and humans. 16 Infected rabbits eliminate the spores in urine and feces; thus, infection of the host generally occurs after ingestion of water or food contaminated with infective spores. 36 Intrauterine infection and the inhalation of microsporidian spores also have been suggested as possible routes of infection.2,17,39 In many rabbits, the infection can persist subclinically for a long time. 20
Currently, E. cuniculi is considered to be an emergent, zoonotic, and opportunistic pathogen in immunocompetent and immunocompromised individuals.1,48 This microsporidian was first reported in 1923 as causing encephalitis and renal disease in domestic rabbits.49,63 Since then, many studies have been undertaken to detect and identify the infective spores of E. cuniculi in tissue of infected individuals. Encephalitozoon cuniculi infection, as an emerging disease, has become a new chapter in infectious diseases and an important theme in animal and human health. 49 There are 3 genotypes based on polymorphism studies: I (rabbit strain), II (mouse strain), and III (dog strain), of which genotypes I and III have been found in humans.9,62 There is an increasing number of reports regarding the presentation of encephalitozoonosis in pet stores and laboratory animal centers.10,13,44 In most cases, definitive detection of E. cuniculi is made by postmortem and histologic study of the infected rabbits. Several organs are sampled, such as the brain and liver, but the kidneys are the only organs that macroscopically show the typical pitted gray or red areas of necrosis accompanied by fibrosis on their surface. Trained pathologists are required for proper detection. Usually, the spores appear in fixed samples stained with hematoxylin and eosin (H&E) as undefined small oval structures. 56 Most of the studies describing the morphologic appearance of other microsporidian spores are performed using histologic samples from corneal biopsies or wet mounts from feces and urine. 21 To date, there are no studies describing the microscopic features of E. cuniculi spores from naturally infected individuals using different stains.
Two of the organs most affected by E. cuniculi in rabbits are the brain and the kidneys, which are microscopically characterized by chronic granulomatous encephalitis and severe chronic granulomatous and interstitial nephritis with fibrosis, respectively. In most cases, detection of this microorganism is made by electron microscopy, coprodiagnosis, cytology, indirect fluorescent antibody test (IFAT), molecular biology analysis, and histology. 15 The spores are ovoid, ~1.5–2.5 μm in length, and have a thick wall composed of an outer proteinaceous exospore, an endospore made of chitin, and an inner plasma membrane. 19 The spore contains a unique nucleus and a polar tube coiled 5–6 times. Examination of E. cuniculi spores in infected samples generally requires the use of a light microscope with 100× magnification for definitive detection.
Significant improvement has been made in the development of complementary staining methods for the detection of microsporidians. In the literature, the majority of the staining techniques are performed on cytologic and wet preparations, 47 and results are presented as general tables, without specifying genera of microsporidia. The stains that are used in these preparations may not give the same results in histologic samples because of the tissue features and the different procedures used for tissue processing. The microscopy techniques commonly used to detect different microsporidian spores are divided into 2 categories: 1) stains or techniques detected by ultraviolet light and 2) stains detected by ordinary light. Microsporidian spores can be mistaken for bacteria, yeasts, protozoa, necrotic tissue, and even artifacts. In the case of E. cuniculi–infected tissues, there is limited information regarding the use of different stains for detecting and confirming the spores. The purpose of our study was to describe the main morphologic features of E. cuniculi spores, as well as to explain the advantages and disadvantages of 14 different staining techniques in the kidneys of naturally infected rabbits. The stains or techniques used for detecting the spores are those routinely employed in veterinary diagnostic laboratories. These results can be used as a guide when examining suspected E. cuniculi–infected tissues.
Materials and methods
Animals for study
Ten New Zealand white rabbits were examined because they had clinical signs suggestive of encephalitozoonosis, including torticollis, lethargy, rolling, dehydration, and urine scalding of hindlimbs. The rabbits came from a semi-intensive farm located in the municipality of Galeana, Nuevo León, México (24°50′N 100°04′W). The problem at the farm occurred from June to August 2014. The animals were ~3 mo of age, weighing ~1.5 kg (normal = 2.5–3 kg). Sera were collected and sent to the Immunology Department of the Faculty of Veterinary Medicine, Autonomous University of Nuevo Leon (FMVZ-UANL; Mexico), and subsequently, the affected animals were submitted for postmortem examination to the Pathology Department.
Encephalitozoon cuniculi detection
Enephalitozoon cuniculi infection was confirmed by the recognition of typical granulomatous lesions in the brain and kidney, as well as by carbon immunoassay testing (CIA test), IFAT (rat antiserum against E. cuniculi), and calcofluor white (CFW) stain. In the CIA test, sera from rabbits containing agglutinated spores that stained gray-brown against a carbon particle background were considered positive for E. cuniculi infection. In IFAT, the kidney samples with green ovoid structures measuring 1–2 μm, thus compatible with E. cuniculi, were considered positive.
Samples and staining
The rabbits were euthanized, and a routine postmortem examination was performed. All animal procedures were approved by the Centro de Investigación y Desarrollo en Ciencias de la Salud-CIDICS-UANL. Samples from several organs, including the kidneys, were fixed in 10% buffered formalin for 48 h. The samples were embedded in paraffin, sectioned at 5 μm, and initially stained with H&E. The main lesions observed in the kidneys were severe chronic granulomatous and interstitial nephritis, accompanied with glomerular synechiae, inter- and intratubular hemorrhage, and severe interstitial fibrosis and necrosis. Other staining methods used included alcian blue (AB), CFW, Giemsa,16,23 Gram, Grocott, H&E, 21 Luna, Luxol fast blue (LFB), 23 Masson trichrome (MaT), modified trichrome stain (MTS), periodic acid–Schiff (PAS), Van Gieson (VG), Warthin–Starry (WS), and Ziehl–Neelsen (ZN) 32 (Table 1). Samples were considered positive for infection with E. cuniculi when microsporidian spores were observed in the granulomatous lesions stained with H&E.6,18,30
Summary of commonly used stains for histologic examination of microsporidian spores.
Results
Encephalitozoon cuniculi detection
All of the sampled rabbits were assayed for anti–E. cuniculi spore antibodies and were seropositive at titer 1:64 (10 of 10). In IFAT, kidney samples containing green ovoid structures measuring 1.5–2 μm were considered positive.
Histopathology
Grossly, the most important lesions observed in the kidneys had a pitted appearance with irregular white to gray depressions, which occasionally coalesced. Histologic examination revealed severe granulomatous interstitial nephritis with macrophage and lymphocyte infiltration and severe fibrosis. Cysts containing abundant E. cuniculi spores were mostly seen in the renal medulla, although they were occasionally seen in the cortex. Degenerative changes in epithelial renal tubules and extensive areas of necrosis and hemorrhage in the renal parenchyma were observed as well.
Staining results
Hematoxylin and eosin
Although this is the routine stain used when examining paraffin-embedded samples, E. cuniculi infection could only be suggested after observing the associated inflammatory response, granulomas, and fibrosis. The spores looked slightly basophilic, transparent, and poorly defined (Fig. 1A).
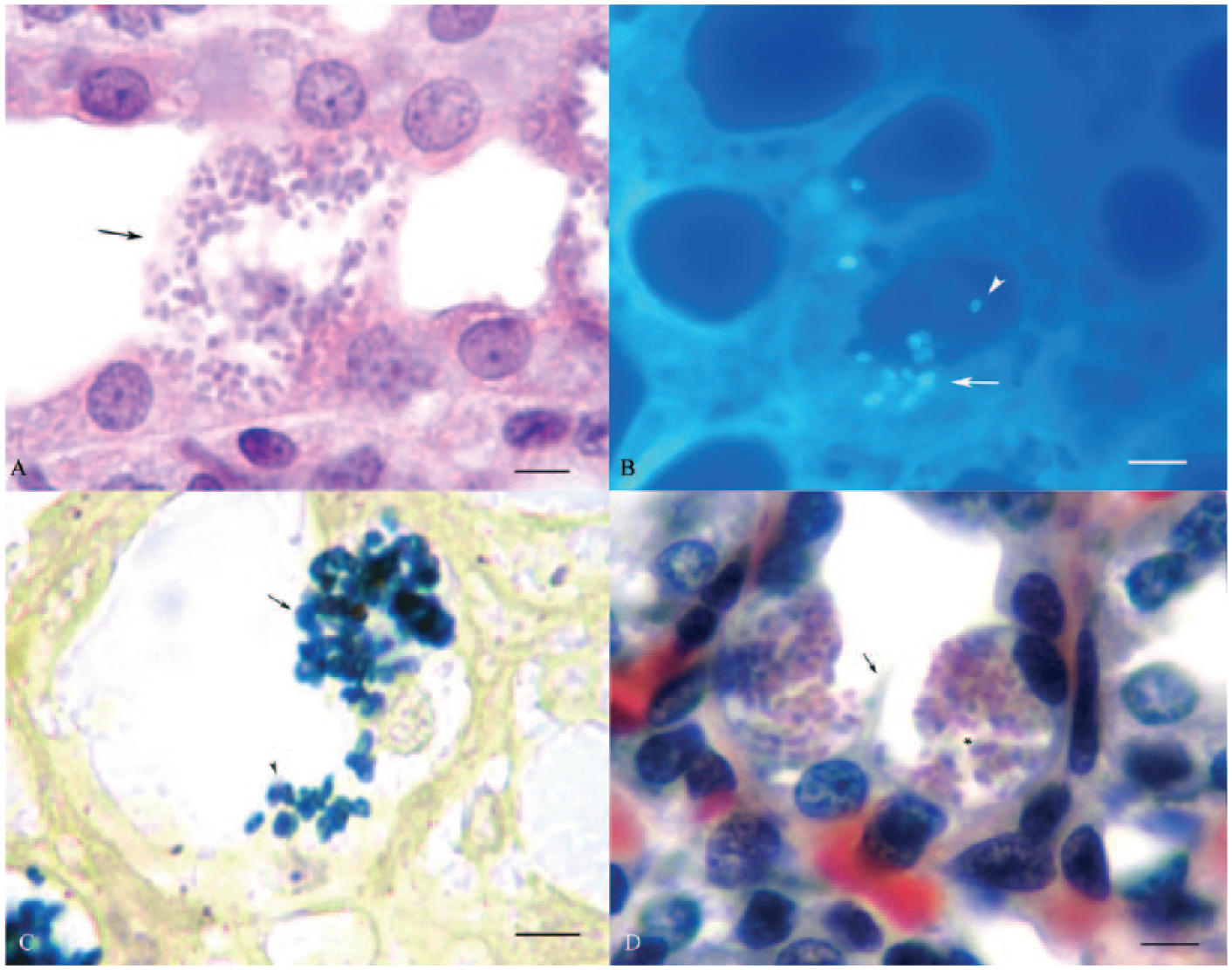
Renal tissue sections from New Zealand white rabbits naturally infected with Encephalitozoon cuniculi.
Alcian blue
One remarkable feature of this stain was the well-defined cell membrane of the infected host cell. The contour of the cyst wall was dark and contrasted with the surrounding purple tissue and the neighboring healthy renal tubule epithelial cells. The spores of E. cuniculi appeared as indistinct and blurry basophilic structures.
Calcofluor white
The spores appeared bright blue with well-defined oval structures against a dark background (Fig. 1B). The mature spores appeared as complete ovoid structures, whereas immature or empty spores appeared as ring-like structures.
Gram
The spores appeared as dark blue ovoid structures and were visible against an amber background (Fig. 1C). The internal structures of the spores were rarely seen.
Giemsa
The E. cuniculi spores were difficult to distinguish against a bluish background. There was a lack of definition of the infected cell membrane and the adjacent tubule epithelial cells.
Grocott
The spores appeared as dark-brown rounded structures and were masked in the brown-to-green tissue background. There was not a clear distinction between the infected cell membrane and the adjacent healthy tubule epithelial cells.
Luna
The background tissue was well contrasted, which allowed the identification of microsporidian cyst walls. The stages of the spore development were observed in different colors, from red to black (Fig. 1D).
Luxol fast blue
This stain easily allowed observation of the stages of the microsporidian differentiation in different colors (violet to dark-blue) and the microsporidian cyst wall was well outlined in blue (Fig. 2A). The blue background of the tissue allowed the observer to distinguish the microsporidian cysts and the adjacent unaffected tubule epithelial cells.
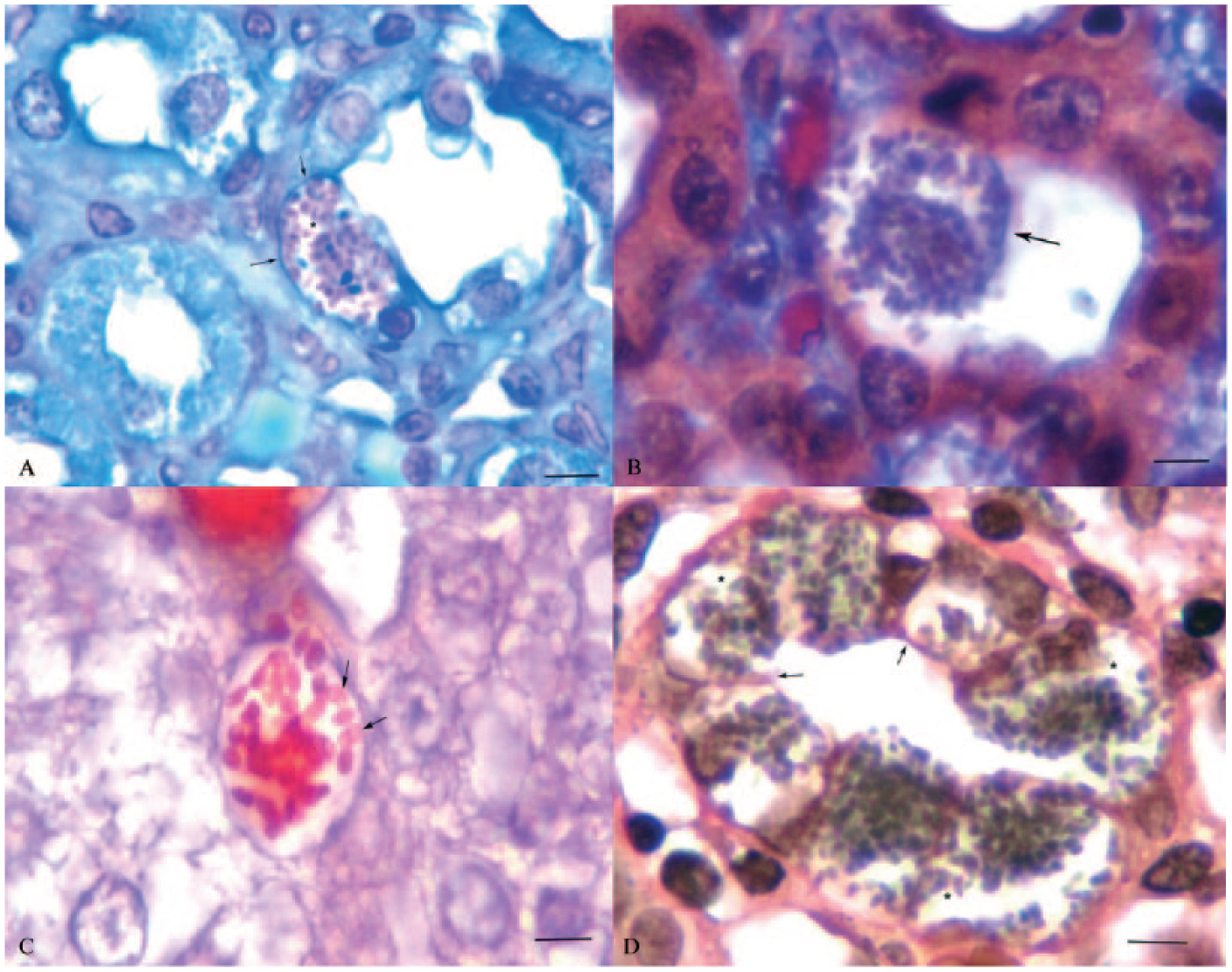
Renal tissue sections from New Zealand white rabbits naturally infected with Encephalitozoon cuniculi.
Masson trichrome
This stain provided excellent tissue contrast background, with renal epithelial tubules in red. The spores were observed as purple, rounded, and oval structures without definition of internal morphology. The cyst walls were not well defined (Fig. 2B).
Modified trichrome stain
The spores were observed as ovoid or round red structures. The polaroplast, posterior vacuole, and polar tube were perfectly visible (Fig. 2C). The polar tube could be recognized by the typical belt-like stripe in the equatorial plane of the spore, and the cell membrane of the infected host cell was discernible.
Periodic acid–Schiff reaction
The spores appeared purple and transparent, and the internal morphology was absent. However, the tissue background and the basal lamina of renal tubules were easily recognized as pink or purple structures.
Van Gieson
The spores were observed as dark ovoid structures against a pink background (Fig. 2D). The internal morphology was not clearly discernible. The cyst cell walls were seen as pink outlined margins, making a clear differentiation between infected and healthy cells.
Warthin–Starry
The spores were observed as gold oval structures, which were stained at exactly the same level as the background tissue.
Ziehl–Neelsen
It was difficult to identify E. cuniculi spores because they were indistinguishable from the blue background. Spores appeared poorly differentiated, blurred, and sometimes transparent.
Discussion
Encephalitozoon cuniculi is a common pathogen of rabbits and other domestic and wild mammals, including primates. 7 In rabbits, E. cuniculi causes a chronic infection mainly characterized by severe damage of the central nervous system and renal tissue, producing severe granulomatous meningoencephalitis and a granulomatous interstitial nephritis, respectively. 27 It has been proposed that the inflammatory granulomatous response is caused by a structural component of the endospore, chitin. 42
Recognition of the spores by light microscopy is the method most often employed for the detection of E. cuniculi in most Latin American veterinary diagnostic laboratories. Although this technique is robust, histologic identification of spores using routine stains is difficult to interpret. This is partially because there is insufficient information about E. cuniculi histochemical examination in paraffin-embedded tissues. The histologic detection of this pathogen has been difficult because of the small size of the spores and their similar morphology to fungi. Thus, they are confused with bacteria, protozoa, artifacts, or are ignored because of lack of expertise. 6 In addition, some microsporidian spores have variable color properties according to their developmental stages.51,59 Most of the histochemical information in the literature comes from ophthalmologic biopsies or conjunctival smears, without confirmatory detection, using different stains and then extrapolating that information to other microsporidia. 47
We are not aware of any previous literature pertaining to histochemical studies of rabbits infected with E. cuniculi. In our study, renal tissue was selected because E. cuniculi spore-laden cysts are defined more frequently in this organ than in any other tissue. Despite the severity of the inflammatory reaction at the renal cortical level, the presence of spores is easily detected because of the minimal inflammatory response in the renal medulla. In other organs, the severity of the granulomatous reaction and fibrosis mask the presence of the microsporidian spores, blocking observation of the cysts. Normally, the histologic detection of E. cuniculi is suggested by the identification of suggestive lesions in the most common sites of infection, such as renal parenchyma, in sections stained with H&E. The most recognizable lesion is a severe granulomatous reaction accompanied with fibrosis. Nevertheless, H&E stain is perhaps more suitable for the recognition of the chronic inflammatory response rather than for spore identification. Identification of the internal spore morphology is not possible; therefore, experienced diagnosticians are required to recognize the indistinct shape of the spores.
We have classified the histochemical examination of E. cuniculi spores into 3 categories: internal spore morphology, spore shape, and stages of parasite maturity:
1) Internal spore morphology is visible with MTS because it is specific for chitin.26,43,60 This stain provided excellent definition of the polaroplast, the polar tube, and the posterior vacuole. MTS efficiently identified the microsporidian spores, allowing the differentiation of these parasites from other microorganisms and helping to confirm the diagnosis of E. cuniculi infection in rabbits. One of the advantages of using MTS stain is the enlargement of the spores, facilitating the observation of their internal morphology.
2) Spore shape can be observed with CFW, Gram, MaT, and VG stains. One important benefit of using CFW in paraffin-embedded tissues was its rapid time to detection 8 and its sensitivity to recognize both complete and empty spores, the latter of which are observed as bright-blue rounded structures. CFW belongs to a group of optical brighteners with affinity for chitin, which microsporidia spores contain in the endospore layer.33,54 The mature spores appeared completely stained, whereas immature spores appeared as ring-like structures. CFW can be used in fecal samples, corneal scrapings, conjunctival smears, or paraffin-embedded tissues. One disadvantage of using CFW is that it can stain other fungi, such as yeasts, because they contain chitin. However, microsporidian spores can be differentiated from these microorganisms given that they are uniform in size and nonbudding.14,15 This stain, when used in combination with others, makes the confirmation of microsporidiosis much easier. Gram stain reacts with peptidoglycans, which could suggest their presence within E. cuniculi spores.14,53 With this stain, the E. cuniculi spores appear as gram-positive microorganisms against a very clear background, which facilitates visualization. The frequency of spore detection was high, conferring reliable results for the detection of microsporidians. 22 In fact, Gram stain has not only been used in mammalian tissues, but also to detect microsporidian spores in zebrafish (Danio rerio).45,46 In our study, color did not vary between immature (red) and mature (purple) spores, as reported by others,28,61 and the internal details of E. cuniculi spores, such as the belt-like stripe, were not observed. MaT stain is routinely used for demonstrating connective tissue. It has been employed to detect the capsule of collagen surrounding microsporidian cysts in hepatic tissue from teleosts. 58 In our study, MaT stain visualized the healthy renal tubular epithelia, leaving the cysts containing the spores as protruding structures without the cyst wall detected, contrasting with the formation of collagen observed around the cysts in other studies. 35 On the other hand, this stain defines the morphology of the spores, shown as dark-blue rounded structures; however, confirmation of microsporidian spores would require additional stains. One of the remarkable features of using VG stain is its preference for the E. cuniculi cyst walls, which are observed as red boundaries contrasting with the gray ovoid spores. The affinity of this stain is for collagen fibers, 32 thus, in our study, it may have recognized the reticular fibers of the cell membrane around the E. cuniculi cyst or a subtle formation of collagen fibers, which are produced for the support of parasite proliferation, as previously reported in fish with microsporidian xenomas. 31
3) Stages of parasite maturity are demonstrated with Luna 40 and LFB 52 stains, which can distinguish the different stages of parasite development. Luna is a stain used to demonstrate eosinophil cytoplasmic granules and erythrocytes. It has also been suggested to have affinity for the chitin of Encephalitozoon spp. and Enterocytozoon sp. in humans and fish. 40 The red positive reaction of this stain could indicate a high concentration of chitin in the mature spores, whereas the black spores indicate lower amounts of chitin in immature spores. LFB stain is commonly used for demonstrating phospholipids and myelin in tissue sections 53 and to study the differentiation of the central nervous system for morphologic purposes. 38 This stain is among the least cited for demonstrating microsporidian spores in tissue sections, but was used to detect the phospholipids of the inner plasma membrane of spores. 39 This stain not only reflected the amount of phospholipids within the spores (blue for mature spores and purple for immature stages of spore development) but also differentiated the cyst wall, observed as blue margins, from the adjacent healthy tubule epithelial cells, suggesting an active production and/or replacement of phospholipids within the infected host cell.
The stains unsuitable for clear detection of E. cuniculi included AB, Giemsa, Grocott, H&E, PAS, WS, and ZN because the internal morphology and external features of the spores were not clearly discernible. The spores are easily mistaken with microorganisms, tissue debris, or artifacts. AB is a basic stain that detects mucosubstances. It has been used to detect the sporogony stages of Nosema and Thelohania in simuliid larva.12,37 However, it was difficult to distinguish the morphology of the E. cuniculi spores, suggesting that they may contain less mucinous content. Giemsa stain is used for the demonstration of protozoa in bodily fluids and for detecting microsporidians in intestines of humans infected with HIV/AIDS.34,41 Nevertheless, in our study, this stain did not achieve good results because of the monochromatic appearance of both the tissue background and the spores. 50 Our results contrast with another study using immunosuppressed BALB/c mice, where Giemsa stain was suitable for detecting E. cuniculi spores.28,55 Grocott stain detects glycogen, mucin, and argyrophilic components in fungi. 46 This stain has been found to be useful for the detection of microsporidian spores in human ocular tissue.24,57 These results contradict those from our study. The observed E. cuniculi spores were not distinct from the background tissue, thus providing an unreliable confirmation of microsporidians spores. H&E is considered the routine stain for examination of paraffin-embedded tissues infected with E. cuniculi. However, the E. cuniculi spores stain weakly with H&E, 42 and definitive recognition of the spores requires a highly experienced pathologist. 25 PAS reaction is used for detecting glycogen and mucinous substances. 53 In some genera of microsporidia, it detects a structure rich in polysaccharides, such as the anchoring disk, which is located at the anterior end of microsporidian spores. This structure is part of the extrusion apparatus and appears under light microscopy as a small red dot. 4 The anterior granule is key for the identification of microsporidian spores in ocular biopsies.23,55 However, PAS reaction was not valuable for the detection of E. cuniculi because the anterior granule was not identifiable. WS silver stain has been used to detect microsporidian spores in the digestive system of humans11,14 and teleosts. 25 Nevertheless, detection of E. cuniculi spores was not feasible in our study because the spores appeared as very compact clusters of brown ovoid structures, making it hard to differentiate them from the background. In some cases, staining with WS enhances the birefringence of microsporidian spores in tissue sections,14,25 but this staining feature was not observed with E. cuniculi spores. ZN stain has been used for demonstrating acid-fast bacteria and lipogenic components in tissue sections,32,53 and for demonstrating microsporidian spores in ocular specimens24,57 and fish samples. 41 The acid-fast microsporidian spores appear as round clumped structures with a dark band at the tip. 47 Unfortunately, this stain was impractical for detecting E. cuniculi spores in our study, possibly because the spores have limited lipid content. 23
We have proposed 3 stain categories based on their affinity and sensitivity in detection of E. cuniculi spores. We believe this work will provide direction and help confirm the diagnosis of suspected encephalitozoonosis cases. Whenever feasible, further studies such as polymerase chain reaction or IFAT should be considered for validating a precise identification; however, such methods are expensive, time consuming, require expertise, and sometimes provide contradictory results. As cases of E. cuniculi infection increase in Mexico, we recommend employing MTS and Gram stains concurrently with H&E stain. This practice would increase the possibility of identification and allow confirmation of detection of this pathogen.
Footnotes
Acknowledgements
We thank César Dávila-Martínez and Ivonne Salinas-Navarro for technical assistance with the CFW stain and IFAT.
a.
Carbon immunoassay testing, Medicago, Quebec, Canada.
b.
Rat antiserum against E. cuniculi, Aniara Diagnostica, West Chester, OH.
c.
Calcofluor white stain M2R, Sigma-Aldrich, St. Louis, MO.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
