Abstract
A diagnosis of neoplasia was noted in 125 of 357 donkeys (35%) in our review of medical records from 5 veterinary schools in the United States and Canada. Equine sarcoid was the most common tumor in our study, accounting for 72% of all tumors and 82% of cutaneous tumors. Soft-tissue sarcomas were the second most common skin tumors. All other types of neoplasia were rare. Important differences in the occurrence of neoplasia in donkeys compared to horses included the rarity or absence of squamous cell carcinoma in any organ system and gray horse melanoma. Lymphosarcoma, the most common malignant tumor in horses, appears to be very rare in donkeys. We report several tumors in donkeys including melanocytoma, peripheral nerve sheath tumor, and gastrointestinal stromal tumor. Our data demonstrate commonalities as well as differences in neoplastic diseases of donkeys and horses. Understanding differences in carcinogenesis among these 2 closely related species can inform researchers pursuing pathogenic mechanisms of equine disease and inform veterinary diagnosticians regarding tumor prevalence.
Introduction
The veterinary literature lacks a review of neoplastic diseases in donkeys (Equus asinus). Aside from a 2011 review of autopsy cases from 1,444 geriatric donkeys in the United Kingdom, 35 reports of neoplastic disease in donkeys are largely limited to individual case reports. Donkeys generally are treated as horses in clinical settings and, although this can serve the clinician well, consideration of salient differences between the species may further benefit donkey health and help elucidate details of the pathogenesis of some common equine diseases. Donkeys account for a small minority of the equine population in the United States and Canada and generally are perceived to have limited economic value. However, members of the species Equus asinus have unique characteristics that make them of physiological and medical interest to scientists, veterinarians, and equine enthusiasts. The purpose of our survey is to report neoplastic diseases diagnosed in donkeys at 5 veterinary schools in the United States and Canada and to draw comparisons and contrasts to neoplastic diseases of horses.
Materials and methods
We reviewed computerized and/or paper autopsy and biopsy reports for all donkey and burro accessions from 5 veterinary schools in the United States and Canada. Time periods included in the survey varied among institutions given the availability and accessibility of medical records (Table 1). One of the authors (CR Davis) reviewed each medical record in detail. All diagnoses of neoplasia were tabulated, with anatomic location or source of the neoplasia recorded along with patient signalment when available. When an individual animal was rechecked or rebiopsied and the same diagnosis was made, the diagnosis was only recorded once. Sex was recorded as jenny (female donkey), jack (intact male donkey), or gelding (castrated male donkey). For summary purposes, anatomic locations of sarcoids were grouped as follows: head included face, ears, lips, periocular, maxilla, and mandible; inguinal included prepuce, scrotum, and teat area; and trunk included neck, shoulder, pectoral area, ventrum, ventral flank, and caudal abdomen. Although there is some discussion among pathologists as to whether equine pars intermedia lesions represent hyperplasia or neoplasia, for simplicity in this survey they are grouped together as adenomas. Because donkey breeds are not well defined in donkeys in the United States and Canada, with the possible exception of miniatures, and because many medical records lacked breed information, the only breed designations recorded in this survey were miniature and “other.” Ages were compared between sexes via Student t-test. The medical records reviewed for our study were not always complete, thus signalment information, patient history, gross description, and ancillary tests were not always available for each case. Many samples were submitted from sources outside of the veterinary schools, and these submissions frequently lacked complete patient signalment, history, and gross anatomic findings. We include additional detailed information on 2 lesions, to our knowledge, not previously reported in the donkey: peripheral nerve sheath tumor and gastrointestinal stromal tumor.
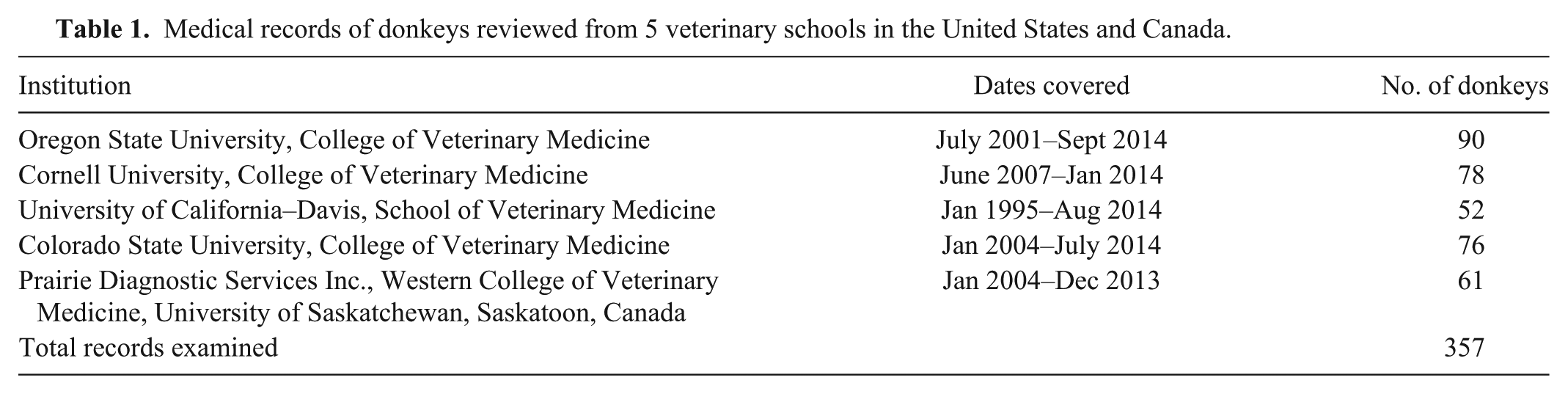
Medical records of donkeys reviewed from 5 veterinary schools in the United States and Canada.
Results
We reviewed records for 357 donkeys. The mean age of donkeys with tumors was 13 years, with a range of <1 to 30 years. A total of 126 tumors was diagnosed in 125 donkeys for a neoplasia prevalence of 35% in this survey population (Table 2).
Diagnoses of neoplasia in donkeys by breed and age.*
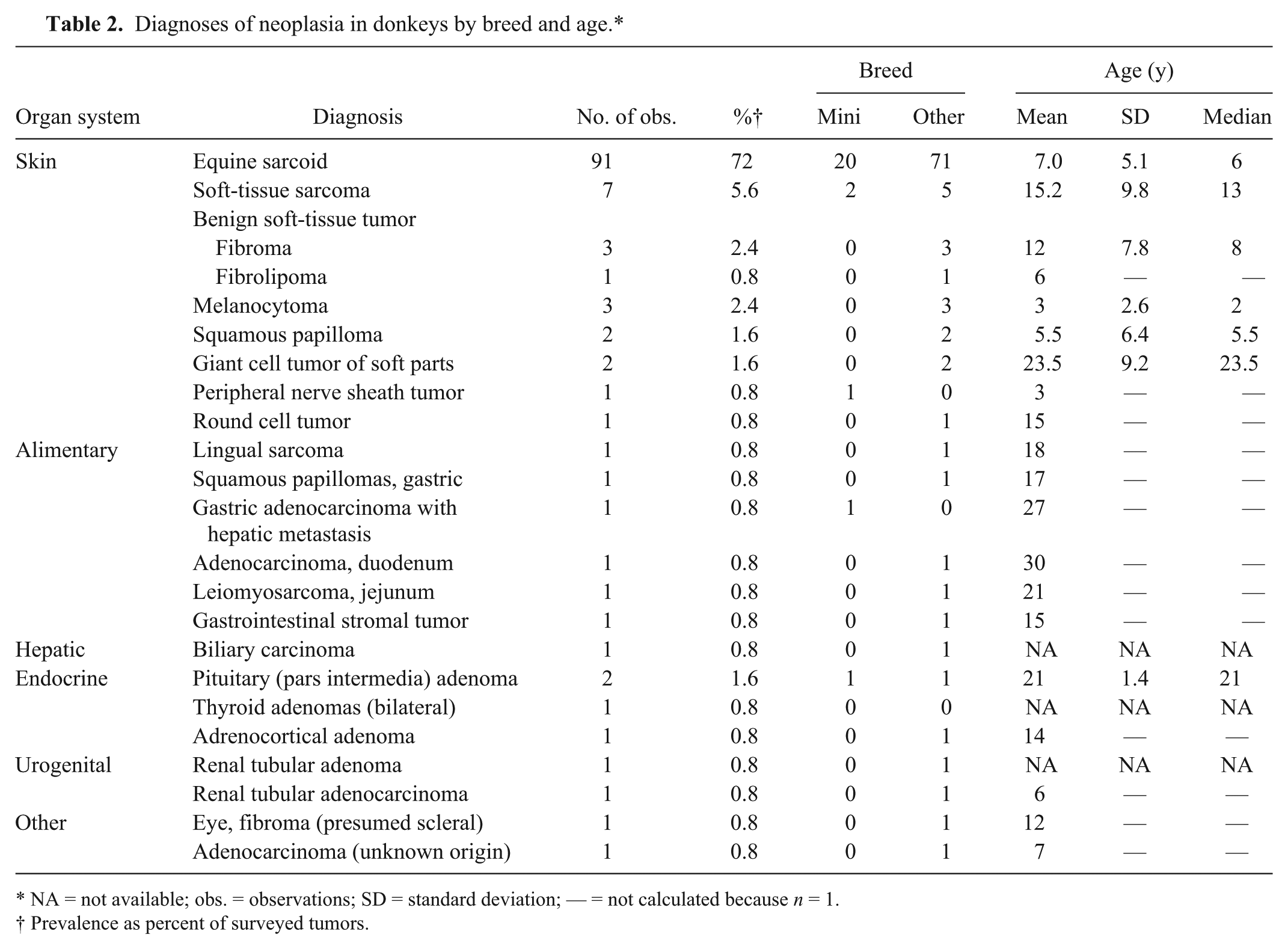
NA = not available; obs. = observations; SD = standard deviation; — = not calculated because n = 1.
Prevalence as percent of surveyed tumors.
Skin
Equine sarcoid
Equine sarcoid was the most common neoplasm observed in this survey (91 of 126), representing 72% of all neoplasms from any organ system, 82% of all cutaneous neoplasms, and 46% of all skin submissions. Age data were available for 93% of donkeys with equine sarcoid. The mean age of donkeys (sexes combined) at the time of diagnosis was 7 years, with a range of 1–30 years (Table 2). No statistical difference in age was observed between geldings and jacks. However, jennies were significantly younger at time of diagnosis (p < 0.05) compared with geldings and jacks combined. Our data show a sex bias, with higher numbers of sarcoids diagnosed in males by a factor of 1.8 to 1. The anatomic location of the sarcoid was recorded for 60 cases. The most common locations for sarcoids in this study were the head (50%) and inguinal region (23.3%), followed by trunk (15%), limbs (8.3%), and tail (3.3%). Of those sarcoids on the head, 14 (47%) were periocular. A sex difference in sarcoid location was observed only in the inguinal area, with 13 of 14 inguinal sarcoids observed in males: 12 of 14 inguinal sarcoids were located on the prepuce of 6 jacks and 6 geldings, and 1 sarcoid was located on the scrotum of a jack. Only 1 jenny had a sarcoid in the inguinal region, lateral to a teat. Diagnoses were divided almost evenly between jacks and geldings, which does not support the notion that surgical trauma associated with castration plays a role in the induction of equine sarcoids.
Dermal sarcomas
Soft-tissue sarcomas of the skin were observed in 7 donkeys, with a range of 2–28 years and a mean of 15 years. Two sarcomas were periocular, 1 mandibular, 1 preputial, 1 pectoral, and 2 on limbs (antebrachium and thigh). These locations, especially periocular and preputial, are reminiscent of sarcoids, and equine sarcoid was listed as a differential diagnosis for one of the periocular sarcomas.
Benign soft-tissue tumors
Three fibromas and 1 fibrolipoma were observed. The fibromas were located on the lateral thorax of a 21-year-old jenny, the shoulder of an 8-year-old gelding, and the neck of a 7-year-old jack. The fibrolipoma was located in the paralumbar fossa of a 6-year-old gelding.
Melanocytic tumors
Three melanocytomas were observed in 3 separate adult donkeys: 1 within the same mass as a sarcoid on the lower hind leg, 1 on the hock, and 1 on the dorsal mid-line of the rump.
Squamous papilloma
Two squamous papillomas were observed, 1 on the lip of a juvenile (specific age not recorded) jack, and 1 on the nostril of a 10-year-old jenny; no histologic evidence of viral infection was observed in these papillomas. No molecular studies were performed.
Giant cell tumor of soft parts (GCTSP)
One GCTSP observed in a 17-year-old jenny was described as a circumscribed, fast-growing mass on the lateral rib cage. A second GCTSP was located on the elbow of a 30-year-old gelding.
Peripheral nerve sheath tumor (PNST)
One PNST reported in a 3-year-old miniature jenny was located in the dermis of the dorsal hock. The tumor was 4 cm in largest dimension, poorly circumscribed, nodular, and expansile. Features that indicated neural derivation included small, delicate spindle cells in a loosely arranged, myxoid ground substance. These cells had delicate, wavy bipolar cell processes and were arranged in closely spaced small fascicles and frequent storiform configurations. Several half-Verocay bodies were observed. Approximately 60% of cells were immunoreactive for S100 antigen.
Round cell tumor
A single round cell tumor of undetermined origin associated with extensive inflammation and necrosis was observed on a hind leg extending from the stifle to the ventral abdomen of a 17-year-old jenny. Tumor cells were negative for T- and B-cell markers and for toluidine blue staining for mast cell granules. Histologic characteristics of plasma cell or histiocytic differentiation were not evident on hematoxylin and eosin–stained slides. Further characterization was not available.
Alimentary and hepatic
The mean age of donkeys with alimentary or hepatic neoplasia was 21 years, with a range of 15–30 years. No sex or breed predilections were observed, although the number of tumors reported is small.
Lingual sarcoma
One lingual sarcoma, 5 mm diameter, was recorded in an 8-year-old gelding. Diagnosis was based on histologic features made from a small punch sample; the submucosal mass was composed of sheets of spindle cells extending to the margins of the sample; no mitoses were observed.
Gastric squamous papillomas and adenocarcinoma
Multiple squamous papillomas were observed in the non-glandular gastric mucosa of a 17-year-old jenny. One gastric adenocarcinoma was described as well-circumscribed, multinodular, occluding the pyloric lumen, with hepatic metastasis in a 27-year-old miniature jenny.
Intestinal adenocarcinoma and leiomyosarcoma
One multinodular, verrucous, ulcerated duodenal adenocarcinoma was described as well-differentiated but invasive in a 30-year-old gelding. One leiomyosarcoma almost completely obstructed the distal jejunum of a 21-year-old jenny that had been presented because of colic of 24-h duration. Tumor cells were strongly immunoreactive for smooth muscle actin (SMA) and desmin and nonreactive for CD117 and S100, confirming smooth muscle origin.
Gastrointestinal stromal tumor (GIST)
One GIST in the rectal wall was observed in a 15-year-old jenny presented for rectal prolapse and a history of depression of 24-h duration. An 8 × 8 × 10 cm firm, cavitated, bilobular fibrotic mass with areas of necrosis and hemorrhage was found in the rectal wall. Microscopically, the mass was unencapsulated and poorly delineated, composed of lobules of pleomorphic polygonal to spindyloid cells in a background mucinous matrix. Mitoses were not observed. Edema, fibrin, and necrosis were observed throughout the mass. Approximately 25% of the tumor cells were strongly immunoreactive for S100, but no tumor cells were immunoreactive for SMA, desmin, or CD117 (c-kit). Coexpression of neurogenic (S100, neuron-specific enolase) and myogenic (SMA) differentiation antigens (but not desmin) with CD117 expression is the gold standard for the immunohistochemical diagnosis of a GIST. However, the histology and immunophenotypes of equine GISTs are variable as a result of divergent lines of differentiation along neural and/or smooth muscle lineages. S100 positivity and the lack of SMA suggest neural differentiation of this spindle cell tumor that arose in the muscular layers of the intestine. Thus, despite being negative for CD117, the immunohistochemistry and histopathology results were most compatible with a GIST.
Biliary carcinoma
One biliary carcinoma was reported, but no signalment or other supporting data were available.
Endocrine
Pituitary hyperplasia or adenoma
Mild diffuse hyperplasia or adenoma of the pituitary pars intermedia was observed in 2 jennies, aged 14 and 20 years, respectively. Both lesions were considered incidental postmortem findings and were not associated with clinical signs of endocrine dysfunction. No further phenotypic characterization of pituicytes was available.
Thyroid and adrenal adenomas
Bilateral thyroid adenomas were diagnosed in an adult jenny of unknown age presented for autopsy because of severe laminitis. An adrenocortical adenoma was diagnosed in a 14-year-old gelding presented because of heart and liver failure. In both cases, these were considered incidental postmortem findings.
Urogenital
Renal tubular adenoma and adenocarcinoma
Two renal tumors were observed—1 tubular adenoma with no patient age or sex information, and 1 tubular adenocarcinoma in a 6-year-old jenny. No primary genital tract tumors were identified.
Other neoplasms
A single fibroma was reported in the sclera of a 12-year-old gelding. No other ocular adnexal or orbital tumors were observed. One adenocarcinoma was observed in the abdominal cavity (carcinomatosis) of a 7-year-old jenny. Pancreatic duct origin was suspected but an adenocarcinoma from another tissue (ovary, uterus) or mesothelioma could not be ruled out.
Discussion
Equine sarcoid is recognized as the most common skin tumor of horses20,42,53,56 and donkeys1,39,54 worldwide. In the present survey, sarcoids accounted for 72% of all neoplasms, 82% of all cutaneous neoplasms, and 46% of all dermatologic submissions. In previous retrospective equine studies (reporting predominantly horses) in the United States and Canada, sarcoids accounted for 46% 42 and 51% 53 of all cutaneous tumors, respectively, and 42% of all dermatology submissions. 56 Mean age at diagnosis (7 years) and age range (1–30 years) in our survey are similar to previous reports in horses.42,56 Consistent with our data, the head and neck have been reported as the most common sites for sarcoid in horses. Our data show higher relative numbers in the inguinal region than reported in horses.42,56 We observed a sex bias, with higher numbers of sarcoids diagnosed in males by a factor of 1.8 to 1, with the vast majority of lesions in males occurring in the inguinal region. This is consistent with the previous impression that young male donkeys are at higher risk of sarcoid development. 9 Our survey also showed jennies were significantly younger at time of diagnosis (p < 0.05) compared to geldings and jacks combined. Reasons for this difference are not apparent from available information, but follow-up in larger groups of donkeys will be of interest. A much lower prevalence of sarcoids (1%) was reported in a survey of postmortem findings for geriatric donkeys in the United Kingdom. 35 Many factors may contribute to this difference. The U.K. study comprised a geriatric population of donkeys that may have experienced attrition of animals with severe sarcoid or resolution of sarcoid with appropriate therapy. In our study, equine sarcoid as a percentage of all neoplasms varied among institutions from 22% to 95%. Only 2 sarcoids were diagnosed at the University of California–Davis, likely because a donkey with a sarcoid would be routinely diagnosed by a private practitioner through a private diagnostic laboratory, thus bypassing a referral to the teaching hospital. In contrast, laboratories diagnosing the most equine sarcoids were Oregon State University (Corvallis, Oregon) and Western College of Veterinary Medicine (Saskatoon, Saskatchewan, Canada), both institutions that provide diagnostic laboratory services to referring veterinarians. Exposure to cattle is another important factor that may vary by region.
A large body of evidence supports an etiologic role for bovine papillomaviruses (BPV-1 and -2) in the development of equine sarcoid.7,9,19,39 Papillomavirus DNA similar to BPV-1 and -2 was demonstrated in equine sarcoids removed from donkeys, suggesting that the disease in the donkey is similar to that in the horse and that the E5 open reading frame may be involved in equine sarcoid oncogenesis. 39 High prevalence of one particular BPV sequence variant in equine sarcoids suggests that this variant shows a fitness advantage in equids. 52 None of the sarcoids in our survey were tested for the presence of BPV. Routine testing for the presence of BPV in veterinary diagnostic settings is not practical, largely because immunohistochemistry is not applicable given the lack of viral replication in equine sarcoids. PCR and in situ hybridization are expensive and time-consuming tests that are typically not employed in clinical settings, especially in animals of perceived low economic value.
Breed predispositions to development of equine sarcoid have been reported in horses. American Quarter Horses, Appaloosas, and Arabians were affected more often than Thoroughbreds, and Standardbreds had an even lower prevalence.34,53 Breed predisposition may be related to certain major histocompatibility complex class II (MHC II) haplotypes that have been shown to be associated with development of equine sarcoid.5,9, 20 The high prevalence (53%) of sarcoids in South African Cape Mountain zebras (Equus zebra zebra), an inbred population derived from no more than 30 individuals, may be associated with a bottleneck in MHC II haplotype diversity. 33 A higher relative risk for development of sarcoids in donkeys compared to horses was first reported in a survey from western Canada. 56 However, any breed predisposition within the donkey population in North America is difficult to determine because breeds are not well defined and are largely classified according to size alone (miniatures, standards, and mammoths). In our study, breed information was most often not available, but when it was, miniature was most often recorded. Our data do not elucidate any breed predisposition among donkeys, but they confirm that donkeys are susceptible to development of equine sarcoid. In determining whether or not donkeys are truly predisposed to development of equine sarcoid one must keep in mind that donkeys do not commonly develop other cutaneous tumors that are relatively common in horses, (i.e., squamous cell carcinoma [SCC], melanoma, and lymphosarcoma). Thus, the relative prevalence of equine sarcoid is high in donkeys. Further research into predisposing factors such as husbandry practices (co-housing with cattle) and the role of MHC II characteristics in donkeys is warranted.
Soft-tissue sarcomas were the second most common skin tumor in our study. In a 2013 survey, donkeys (including burros) and mules were considered 2.5 times more likely to develop soft-tissue sarcomas than any breed of horse represented in the study. 42 Because differentiation of sarcoids, sarcomas, and fibromas can be difficult when the epidermal component is absent, it is tempting to hypothesize that some soft-tissue sarcomas and fibromas are actually sarcoids lacking the distinctive epidermal component. In situ hybridization with BPV probes could provide a definitive diagnosis in these cases; however, this is not a viable routine test option.
Benign soft-tissue skin tumors (3 fibromas and 1 fibrolipoma) were uncommon in our survey. We found no reports of fibroma or fibrolipoma in donkeys in the literature, although low prevalences of 2.4% 42 and 2.1% 48 were reported for soft-tissue fibromas in horses. Screening this group of tumors for BPV would also be of interest.
Melanocytomas were uncommon in our data set and were not observed in the typical locations for gray horse melanomas (underside of tail, perianal, lips, and periocular). We found no reports of melanocytic tumors in donkeys in the literature. Melanomas are relatively common equine skin tumors ranking after sarcoid and SCC.42,53 Age-related graying in horses from several breeds, including Arabians, American Quarter Horses, Thoroughbreds, and Lipizzaners, is strongly associated with development of melanoma and is attributed to an autosomal dominant trait.10,41 Although some donkeys progressively gray with age (a roan color pattern referred to as “frosted”), 47 development of melanomas has not been associated with this color trait in donkeys. Melanoma of gray horses is speculated to be an unintended consequence of human selection pressure for color, selective pressure that has not been applied to the donkey. 41
Two squamous papillomas in our study were located on the head, reported as one of the most common sites for squamous papillomas in horses 42 ; multiple squamous papillomas were observed in the stomach of 1 jenny. Papilloma has been reported rarely in the horse esophagus, 48 but not in the stomach. Because of the presence of a stratified squamous epithelium in a portion of the equine stomach, development of papillomas in this tissue is not surprising. Occurrence of esophageal or gastric papillomas in any species may be vastly underappreciated given the mild disease nature and potential for spontaneous regression. Papillomas of the upper alimentary tract in cattle are linked to specific strains of BPV, 8 and human esophageal squamous papilloma has been linked to specific strains of human papillomavirus infection. 24 Equus caballus papillomavirus (EcPV) is associated with papillomatosis in young horses, most commonly characterized by papillomas on the face, lips, and ears, which regress spontaneously, and with aural plaques, which do not regress. Recently, EcPV-2 has been implicated in the development of genital SCC in horses.28,30,37,57 The relationship between EcPV and gastrointestinal squamous papillomas in the horse is unknown. Although donkeys most likely get papillomatosis similar to horses, we could find no references in the published literature, and the specific papillomaviruses involved are not described.
GCTSP has not been described in the donkey (although the gelding in our report was previously included in a survey of equine tumors in the United States). 53 GCTSP, first described in a mass removed from the fascia over the jugular groove of an Arabian mare, 17 is well documented as a rare tumor of aged horses and mules, occurring most commonly on a hind limb, but also reported from the flank, abdomen, muzzle, thorax, and neck.6,40,42
PNST has not been described previously in the donkey, to our knowledge. The tumor in the present report was not well encapsulated, and ~60% of the tumor cells were S100 positive, indicating that this mass may have been a neurofibroma rather than a schwannoma, both subtypes of PNST. Schwannomas, considered uncommon tumors in horses, are typically well-encapsulated benign tumors in which almost all cells express S100, whereas neurofibromas may be poorly demarcated and contain a mixture of Schwann cells, perineurial cells, and fibroblasts. 43 The specific cell of origin was undetermined for a single round cell tumor identified in our survey. Because round cell tumors are generally rare in donkeys, application of additional diagnostic modalities will be important to elucidate the origin of these tumors in future cases.
Sixteen nondermatologic tumors observed in the present study were all single diagnoses, except for 2 cases of pituitary (pars intermedia) adenoma or hyperplasia. Among nondermatologic tumors, alimentary neoplasia comprised the largest group. The only oral tumor observed was a lingual sarcoma. Lingual tumors are uncommon in the horse and include SCC, rhabdomyosarcoma, salivary adenocarcinoma, chondrosarcoma, mast cell tumor, and lymphosarcoma. 31 Lingual and, more generally, oral tumors are previously unreported in donkeys, to our knowledge. In our study, gastric squamous papillomas were observed in 1 jenny and are discussed above with skin papillomas. We report 1 gastric adenocarcinoma with hepatic metastasis, 1 duodenal adenocarcinoma, and 1 intestinal leiomyosarcoma. Gastric adenocarcinoma has been reported in geriatric donkeys, 35 but this diagnosis was combined with all gastrointestinal neoplasia, so the precise number of cases was unclear. Gastric adenocarcinomas are rare in horses, whereas gastric SCC accounts for the vast majority of stomach tumors in this species.38,50 To our knowledge, gastric SCC has not been reported in the donkey. Duodenal adenocarcinomas in horses are the second most common intestinal neoplasm after lymphosarcoma, 49 but no reports could be found for this neoplasm in donkeys. A retrospective analysis of geriatric U.K. donkeys reported gastrointestinal neoplasia to be rare (3.7%) and included leiomyoma, lipoma, undifferentiated malignancy, and gastric or cecal adenocarcinoma. 35
We report 1 donkey with GIST associated with significant clinical signs. GIST is a rare gastrointestinal tumor of the horse, having been reported in the stomach, small intestine, cecum, and colon.11,21 The majority of GISTs reported in horses were incidental findings, although a large tumor in the stomach and greater omentum with cardiac metastasis was associated with clinically significant hypoglycemia. 22
We observed only a single hepatic neoplasm and a suspected metastatic pancreatic neoplasm with carcinomatosis. Primary hepatic and pancreatic tumors are very rare in both horses 3 and donkeys.26,35,46 A retrospective analysis of geriatric U.K. donkeys reported benign (biliary adenoma, cystadenoma, and cholangioma) and malignant (cholangiosarcoma and bile duct carcinoma) liver neoplasia at 0.7% and 4.2% of diagnoses, respectively. 35
Endocrine neoplasms found in our survey were few, benign, and not associated with clinical endocrine dysfunction. In aged horses and ponies, hyperplasia of the pituitary pars intermedia is associated with equine Cushing’s syndrome (pituitary pars intermedia dysfunction) characterized by hirsutism, hyperhidrosis, muscle atrophy, laminitis, polydipsia, polyuria, and opportunistic infections. Although pars intermedia hyperplasia or adenoma is relatively common in aged horses, we found only a single published report in donkeys. 35 Equine Cushing’s syndrome may be an under-reported syndrome in donkeys. Further characterization of the lesions in donkeys may elucidate the types and functionality of the pituicytes affected. We also report a single donkey with bilateral thyroid adenomas and another with an adrenocortical adenoma. In the survey of geriatric U.K. donkeys, pituitary hyperplasia, adenoma and adenocarcinoma, thyroid adenoma and follicular carcinoma, and adrenal adenoma, carcinoma, sarcoma, and pheochromocytoma were reported. Although specific prevalence of each condition was not reported, malignant thyroid disease prevalence was 0.1%. 35 A single case report documents a large adrenocortical carcinoma with metastases to liver, heart, and lung in an 8-year-old jack. 45
Our survey revealed only 2 primary and no metastatic renal tumors. Malignant renal neoplasia (no specified type) was observed in only 0.4% of diagnoses in the U.K. survey of geriatric donkeys. 35 Primary kidney tumors are also rare in horses, with renal carcinoma the most common type, although various tumor types have been reported. 51
Ocular tumors are uncommon in horses, although SCC is well documented to occur in the nictitating membrane of horses,44,53 and a variety of primary and metastatic orbital tumors have been reported in horses.2,14 In our survey, only a single benign scleral fibroma was reported. Similarly, no ocular tumors were observed in the large group of geriatric U.K. donkeys, 35 and no reference to ocular tumors in donkeys was found in the literature.
Several rare tumors of donkeys not observed in our survey but documented in the literature include: a mastocytoma in the jugular groove of a 10-year-old male donkey 25 ; bilateral Leydig cell tumors incidentally found in a donkey at slaughter 18 ; uterine fibrosarcoma associated with colic in a jenny successfully treated with ovariohysterectomy 4 ; fibrous dysplasia or ossifying fibroma in the maxillary sinus of a 9-year-old gelding 12 ; a collision tumor with glandular and spindle cell components in the paranasal sinus of a 28-year-old male miniature donkey 55 ; and 2 osteosarcomas, 1 of the distal phalanx in an 18-year-old gelding 36 and 1 of radius and ulna with metastases to lymph nodes and liver in a 10-year-old gelding. 27 Both osteosarcomas occurred in bones of the limb, whereas osteosarcomas in the horse, although very rare, are usually found in the skull or mandible, and the involvement of 2 adjacent bones (radius and ulna) with metastasis to lymph node and liver is not typical of osteosarcoma in any species. 27 In the retrospective analysis of geriatric U.K. donkeys, several tumors of the female reproductive tract were reported: thecoma, granulosa cell tumor, and uterine leiomyosarcoma and adenocarcinoma (specific site unspecified). Malignant neoplasia of the female reproductive tract was rare, with a prevalence of 0.5% in this analysis; no neoplasms of the male reproductive tract were reported. 35
Lymphosarcoma is the most common malignant neoplasm in the horse 16 but has been reported only rarely in donkeys.13,32,35 One case report in a 36-year-old donkey described a pulmonary angiocentric lymphosarcoma with characteristics of lymphoid granulomatosis similar to that observed in humans. 13 A second case report described a gastric lymphosarcoma with metastasis to liver and spleen in a 15-year-old gelding donkey. 32 In the retrospective analysis of geriatric U.K. donkeys, lymphosarcoma was reported affecting lymph nodes, but the number of cases observed was not presented. 35 In a 2013 review of 203 cases of equine lymphosarcoma from 8 veterinary institutions, no donkeys were represented. 16
SCC was not observed in donkeys in the present report and was similarly lacking in the survey of 1,444 geriatric U.K. donkeys. 35 Only 1 report of SCC in a donkey was found in the published literature (Kerr KM, Alden CL. Equine neoplasia—a ten year survey. Proc Ann Meeting Am Assoc Vet Lab Diagnosticians; 1974;17:183–189) but no anatomic location was provided. In 2 retrospective surveys of equine cutaneous neoplasia and 2 surveys of all equine neoplasia, SCC was the second most common skin tumor (one study included nictitating membrane and conjunctiva) after equine sarcoid, accounting for ~18% of skin tumors in each study in the United States and/or Canada.42,53 SCC was the second most common tumor of any organ system after sarcoid, accounting for 19% of all tumors in a 2016 U.K. study 29 and 25% in an older U.S. study. 48 The vast majority of animals in these reports were horses, but SCC was not observed in donkeys that were included in these surveys. SCC is the most common tumor arising in the periocular (eyelids, nictitans, conjunctiva, limbus) 15 and penile areas of horses. 37 With the recent elucidation of a role for EcPV-2 in equine genital SCC28,30,37,57 and head and neck SCC, 23 one may speculate that, given species differences known to be important with papillomavirus pathogenesis, donkeys are less (or not at all) susceptible to this infection, accounting at least in part for the scarcity of SCC in donkeys. SCC are more common in Appaloosa and American Paint Horses,42,53 possibly because of increased ultraviolet light exposure in animals with poor or absent dermal and mucous membrane pigmentation. Although most donkeys are fully pigmented, there are many white-patterned donkeys, 47 and male donkeys with no white patterning may have pink penile skin, thus pigmentation differences alone do not account for differences in SCC occurrence between horses and donkeys.
Our survey demonstrates commonalities as well as differences in neoplastic diseases of donkeys and horses. Equine sarcoid is the most common tumor in both donkeys and horses, although sarcoids in the donkeys of our report accounted for an even higher percentage (72%) of overall tumors compared with the highest reported percentage of sarcoids in horses (44%). 48 This high relative prevalence of sarcoids in donkeys could be attributed to the absence of SCC and melanoma, both common skin tumors in horses. Lymphosarcoma, the most common malignant tumor in horses, is very rarely reported in donkeys. Understanding the differences in carcinogenesis between these closely related species may prove useful to veterinary diagnosticians as well as to researchers pursuing pathogenic mechanisms of equine disease.
Footnotes
Acknowledgements
We thank Derrick Padar at the College of Veterinary Medicine, Oregon State University library for his assistance with literature searches.
Authors’ contributions
CR Davis contributed to conception and design of the study; contributed to analysis and interpretation of data; and drafted the manuscript. BA Valentine, E Gordon, SP McDonough, PA Schaffer, AL Allen, and P Pesavento contributed to acquisition and interpretation of data. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
