Abstract
We investigated CD47 expression in cryopreserved sections of equine cutaneous masses and normal skin. CD47 is a cell surface protein expressed on many cell types and overexpressed in some tumors. Interaction of CD47 and signal regulatory protein–alpha (SIRPα) inhibits phagocytosis by macrophages. Formalin-fixed tissues from horses prospectively enrolled in the study were used to establish a histologic diagnosis. Immunohistochemical assays were performed on cryopreserved tissues using anti-CD47 antibodies or IgG control antibodies. CD47 was not expressed on equine normal skin but positivity to CD47 was present in 13 of 24 (54%) masses. Immunotherapy with anti-CD47 antibodies for equine cutaneous tumors that express CD47 warrants further investigation.
Cluster of differentiation (CD)47, or integrin-associated protein, is a transmembrane protein expressed in many cell types.1,10,11 CD47 has been linked to normal and pathophysiologic processes, including apoptosis, proliferation, cell adhesion, inflammation, and immunity. 12 CD47 is a marker of self and interacts with signal regulatory protein–alpha (SIRPα), a molecule expressed on monocytes, macrophages, and dendritic cells.1,10 Binding of CD47 with SIRPα enforces tolerance and prevents phagocytosis by macrophages.3,22 Loss of CD47 leads to phagocytosis of aged or damaged cells. 12 The role of CD47 in regulating phagocytosis and its mechanism of protection are illustrated by its function on erythroid cells. Expression of CD47 is necessary to protect transfused red blood cells, platelets, and lymphocytes from phagocytosis by splenic macrophages. 11 CD47 has also been found to be expressed on multiple tumor types.2,7,24 It is well established that many tumors in humans express increased levels of CD47 compared with their normal cell counterparts, and protection of tumor cells is afforded by the high levels of expression.3,7,24 There are many reported actions of CD47 and its binding partners, including inhibition of phagocytosis, proinflammatory effects, and cell spreading and migration effects.19,20 When activated, CD47 can induce caspase-independent cell death (type-III programmed cell death). 20 Interruption of the interaction of CD47 and SIRPα could therefore be useful in malignant diseases. In fact, anti-CD47 antibodies have been used in vitro and in vivo to inhibit growth and prevent the spread of tumors.2–5,24 Binding of CD47 using monoclonal antibodies has been shown to be effective in increasing phagocytosis of cancer cells by macrophages in experimental models of leukemia, lymphoma, and carcinomas.2,7,24
Approximately 30% of horses presented for dermatologic disease have been reported to have cutaneous neoplasia. 23 A survey of 2 veterinary diagnostic laboratories, over a 10-year period, found cutaneous neoplasia in 65% of submitted samples from 3,272 equids. 15 The 3 most common tumors in the survey were equine sarcoid, squamous cell carcinoma, and melanoma. 15 Even before masses get large enough to affect function, they are cosmetically undesirable to the owner. Tumor type, tumor stage, prior treatment, and tumor location influence the choice of treatment modality and the response to treatment. Wide surgical excision is curative for many tumors,13,17 but when a mass is located near vital structures, then nonsurgical options (e.g., intralesional cisplatin, cryotherapy, laser ablation, intratumoral hyperthermia, radiotherapy) that have the potential to damage surrounding tissues may be the only option. A therapy that reduces the tumor mass without damaging the surrounding tissues, like immunotherapy that targets CD47, is an appealing alternative. To our knowledge, there are no reports concerning CD47 expression in normal tissues or tumors in the veterinary literature. In this study, expression of CD47 in equine normal skin and cutaneous masses was investigated.
Thirty, client-owned equids that presented to the Lloyd Veterinary Medical Center (Iowa State University, Ames, Iowa) for incisional biopsy and/or surgical excision of cutaneous masses were prospectively enrolled in the study. Samples were collected over a period of 1.5 years. Owners were informed of the study aims, procedures, risks, and aftercare and indicated their consent for inclusion in this study by signing a consent form approved by the Institutional Animal Care and Use Committee. Signalment and tumor location were recorded. A physical examination was used to assess general health and ensure accordance with standards outlined in the Institutional Animal Care and Use Protocol.
Patients were sedated with 0.01 mg/kg detomidine a intravenously, and local anesthesia was achieved with mepivacaine. b Two 6-mm punch biopsies of normal skin were removed from an inconspicuous area under the mane on the neck. Two tissue samples of the cutaneous masses were collected, as well. If biopsy rather than mass removal was requested by the client, a sample considered the minimal amount necessary for study purposes was removed from the mass by punch biopsy (6 mm). If mass removal was requested or preferred, the tumor was removed in its entirety if possible, and tissue samples of ~2–3 cm in diameter were collected from the mass. If complete removal was not feasible, as much of the tumor was removed as possible without compromising function of surrounding tissues. One section from the cutaneous mass and 1 normal skin biopsy were each placed in 10% formalin and snap-frozen in a cryomold that contained optimal cutting temperature compound (OCT). d Samples were labeled and placed into sterile sampling bags e and transferred immediately to a −80°C freezer until CD47 expression was evaluated. Snap-frozen samples were stored on average for 1 week (maximum storage time: 45 days) prior to evaluation for CD47 expression.
Hematoxylin and eosin–stained sections of the paraffin-embedded, formalin-fixed biopsies from normal skin, and tissue from the cutaneous masses, were evaluated by a veterinary pathologist (JM Hostetter), and a histologic diagnosis made. Histologically, 7 of the cutaneous lesions were non-neoplastic despite clinically appearing to be tumors, and were excluded from the statistical analysis. Expression of CD47 in tissue from masses and normal skin was evaluated by immunohistochemistry (IHC) on cryosections.3,21 There was minimal to absent staining for CD47 using formalin-fixed tissues, therefore IHC for CD47 expression was evaluated on cryopreserved sections of the normal skin and cutaneous masses. OCT-embedded tissues were sectioned to 5 μm and fixed with methanol. Sections were blocked with 10% goat serum and probed with anti-CD47 antibody f (anti-bovine monoclonal antibody, mouse host) or immunoglobulin (Ig)G control antibody at 1:25 and incubated for 2 h at room temperature. Detection was performed using a biotinylated anti-Ig secondary antibody g (polyclonal, goat host) at 1:80 and incubated for 15 min at room temperature. This was followed by streptavidin–horseradish peroxidase (HRP), and then colorimetric detection using 3,3′-diaminobenzidine (DAB). Sections were counterstained with hematoxylin. Concentrations of primary and secondary antibodies were optimized for staining for human and equine neoplastic tissues. Human ovarian adenocarcinoma was used as a positive control as it has previously been reported to highly express CD47 (Fig. 1). 24 Sebaceous glands demonstrated strong CD47 positivity (Fig. 2); nonspecific background staining of sebaceous glands has been previously reported. 8 To validate this, we performed negative control staining of both skin and tissue from masses with only the secondary antibody (no anti-CD47 antibody). This staining revealed that the sebaceous glands were strongly positive, whereas tissues from the masses were not.
Cryopreserved human ovarian adenocarcinoma. CD47 monoclonal antibody–based immunohistochemistry with hematoxylin counterstain. Bar = 50 μm.
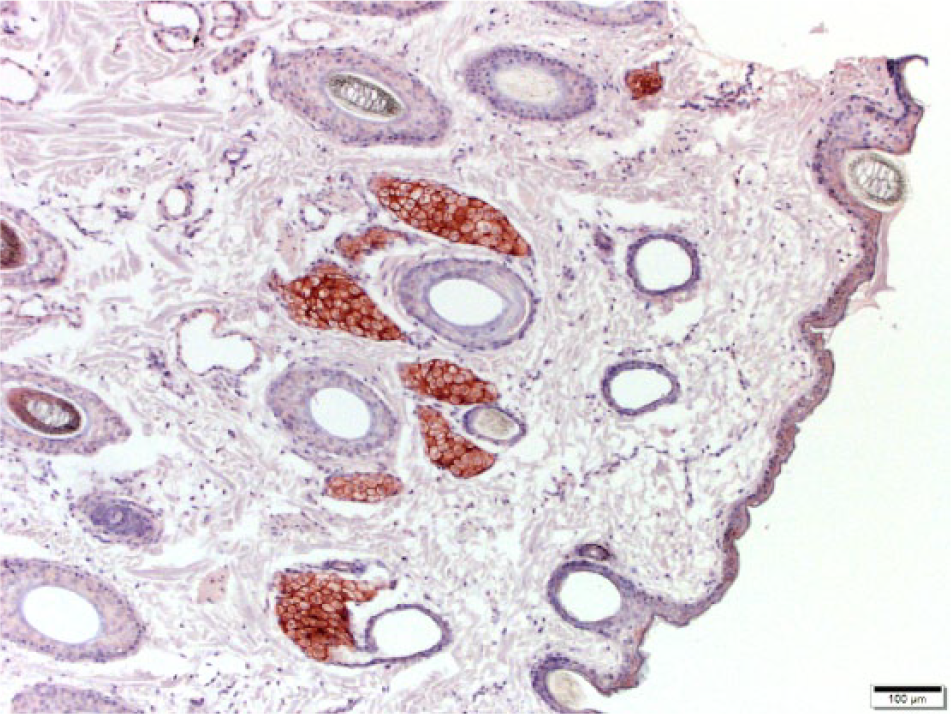
Cryopreserved equine normal skin. Dark brown to red coloration of the sebaceous glands can be seen; this staining is nonspecific. There is a lack of immunostaining in other epithelial cells. CD47 monoclonal antibody–based immunohistochemistry with hematoxylin counterstain. Bar = 100 μm.
CD47-stained sections of normal skin and cutaneous masses were scored by a veterinary pathologist (JM Hostetter) using a semiquantitative score 6 (Supplemental Table 1, available at http://vdi.sagepub.com/content/by/supplemental-data). In our study, there was no need for correlation to prognosis or to response to treatment; however, the semiquantitative scoring system was used to document the variation of CD47 expression in various equine tissues. The section was scanned at 400× magnification. Scores for IHC were determined by frequency and intensity of staining. Frequency was scored from 0 to 5 with 0 = no immunoreactivity, and 5 = diffuse immunoreactivity. Intensity of stain was scored from 0 to 5 with 0 = no immunoreactivity and 5 = intense immunoreactivity. A total combined score was obtained by adding the 2 scores (maximum value of 10). A positive result was a total combined score of 2 or greater. Wilcoxon signed-rank test was used for comparison of nonsebaceous skin versus tumor staining. Nonparametric analysis of variance (Kruskal–Wallis test) was used for comparison of tumor type to combined IHC score; tumor type to positive IHC score (any positive score or not); breed to positive IHC score; and tumor location to positive IHC score. Although the scoring system was used to assign a level of intensity and distribution of staining to mass sections, all skin samples were negative. As such, analysis of skin versus mass was performed using only a positive or negative result (no score number) for tumors. Analysis of tumor type versus score was completed. Logistic regression was performed to examine age versus positive IHC score.
Thirty equids ranging in age from 2 to 33 years (median 13 years) were enrolled in the study. Breeds or species enrolled in the study included 13 American Quarter Horses, 8 American Paint Horses, 2 Grant zebras, 2 mixed-breed ponies, and 1 each of Thoroughbred, Arabian, Shetland pony, Fjord, and donkey. A summary of results is provided in Table 1. A total of 30 samples of normal skin and 32 cutaneous mass samples were obtained (note: 2 equids had 2 masses in different locations). One mass was excluded from further evaluation because the frozen samples were misplaced. Of the remaining masses, 24 were tumors and were included in analysis. Tumor types included equine sarcoid (
Summary of immunohistochemistry (IHC) results.*
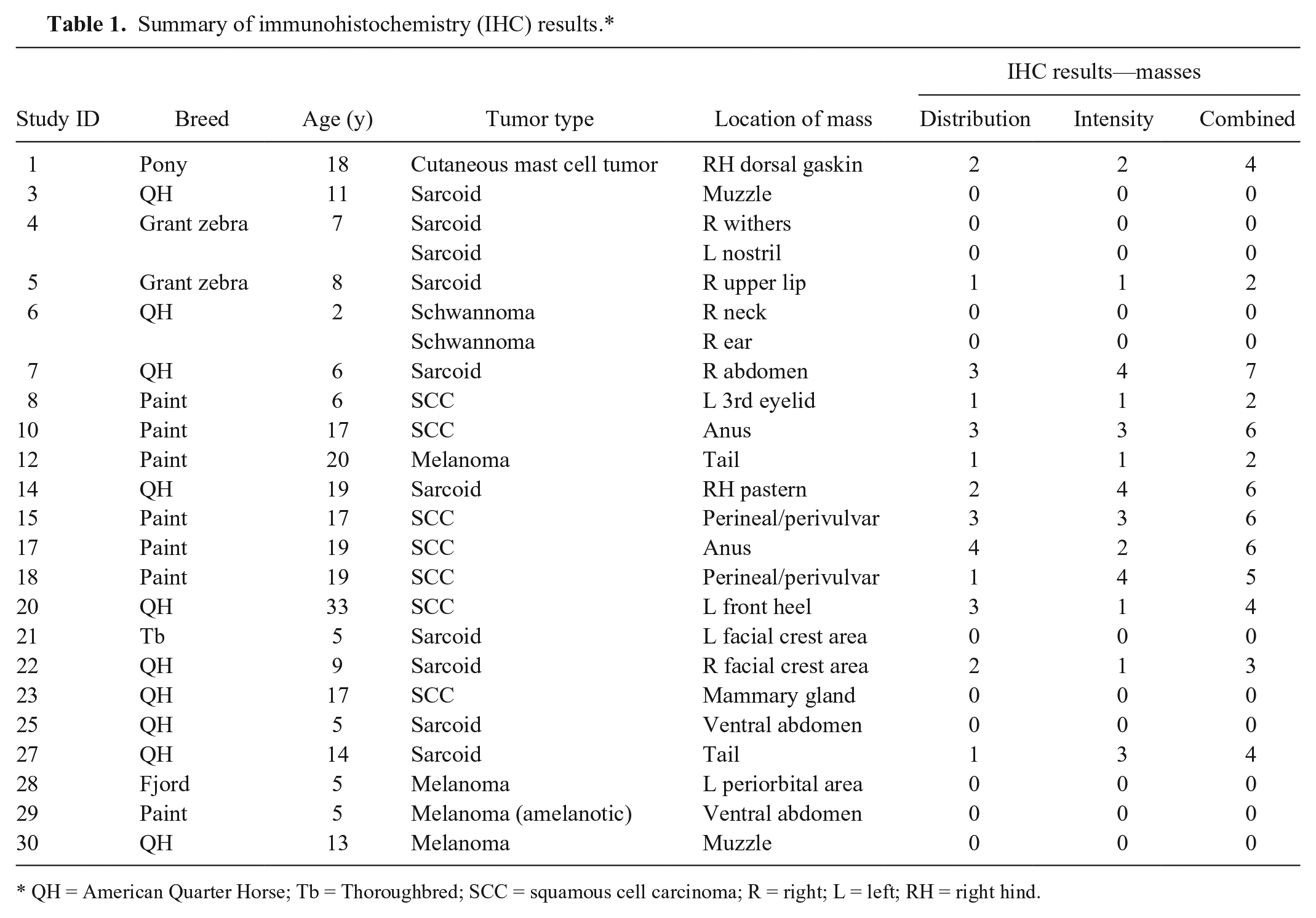
QH = American Quarter Horse; Tb = Thoroughbred; SCC = squamous cell carcinoma; R = right; L = left; RH = right hind.
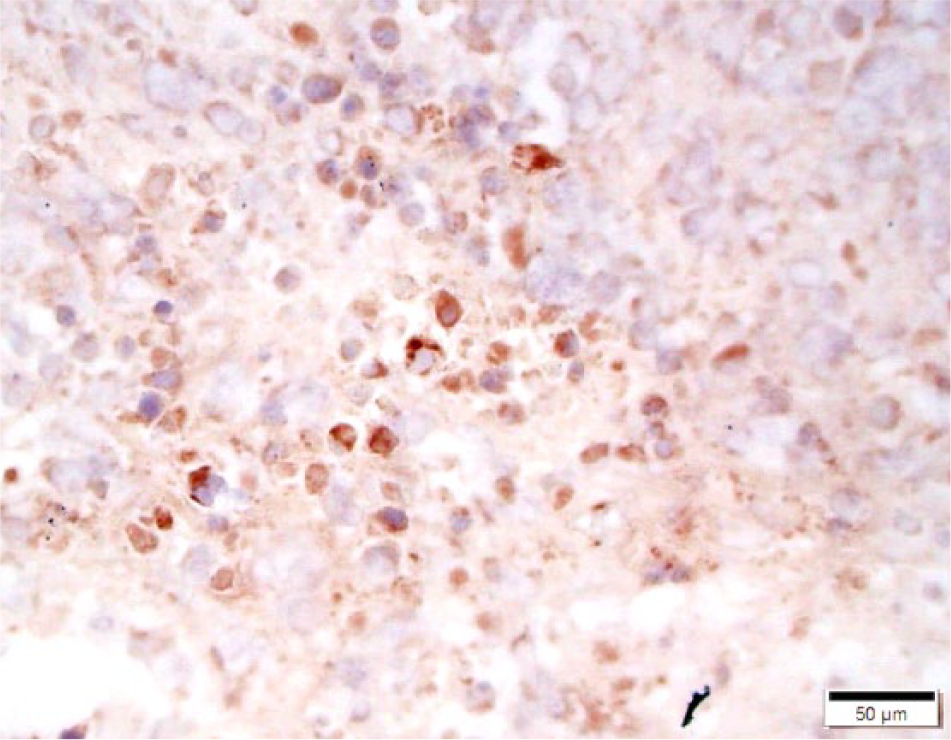
All skin lacking sebaceous glands had distribution and intensity scores (and thus combined scores) of 0 (Figs. 2, 3). In the samples of tissue from masses, 13 of 24 (54%) samples were positive for CD47 (Figs. 4, 5). Frequency scores ranged from 0 to 4 and median score was 1; intensity scores ranged from 0 to 4 and median score was 1. The combined scores ranged from 2 to 7 and the median combined score was 2. A significant difference (
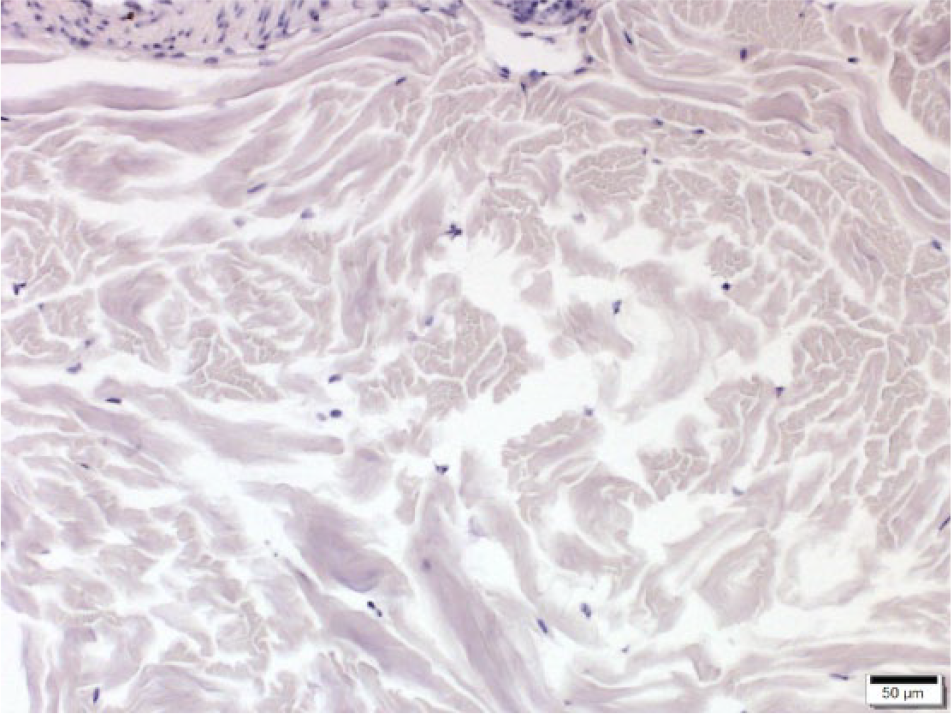
Cryopreserved equine normal skin. No sebaceous glands are present in this field. There is a lack of immunostaining. CD47 monoclonal antibody–based immunohistochemistry with hematoxylin counterstain. Bar = 50 μm.
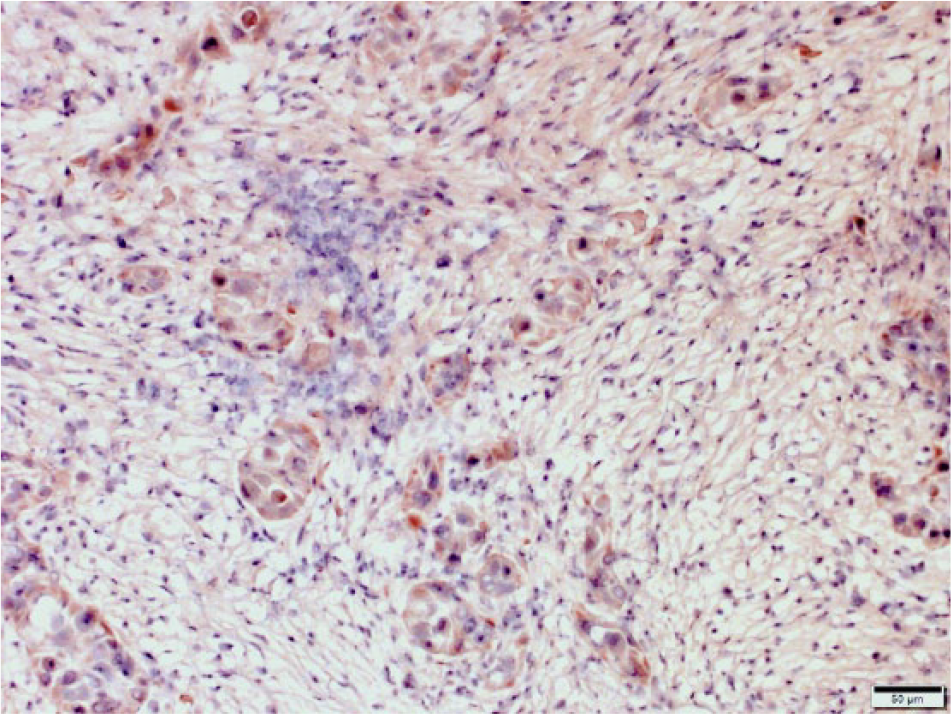
Cryopreserved equine squamous cell carcinoma. Neoplastic cells stain dark brown to red indicating expression of CD47 by neoplastic cells. CD47 monoclonal antibody–based immunohistochemistry with hematoxylin counterstain. Bar = 50 μm.
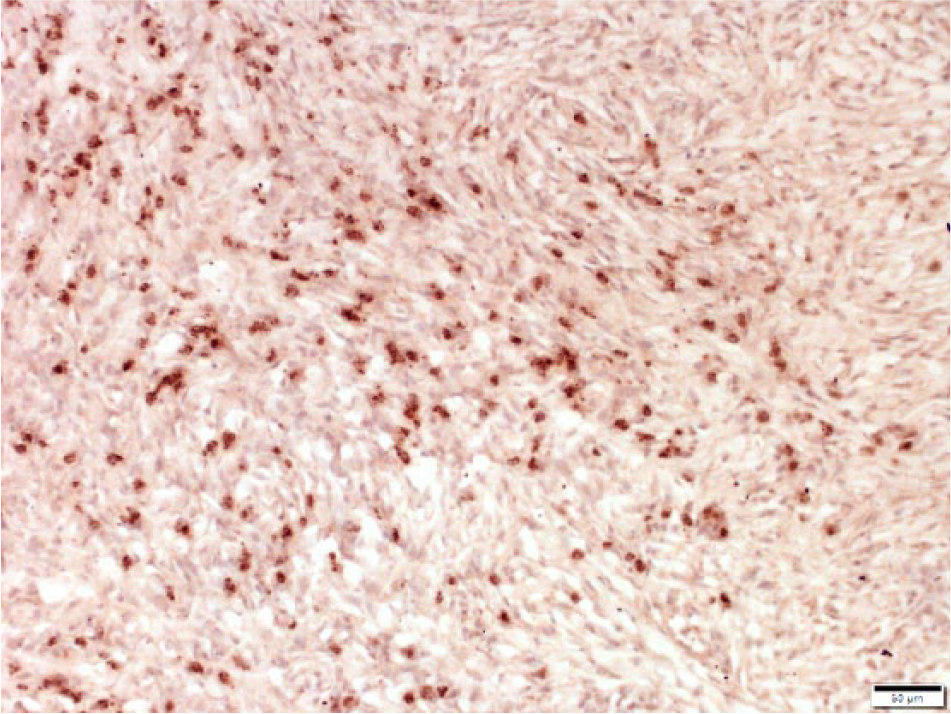
Cryopreserved equine sarcoid. Neoplastic cells stain dark brown to red indicating expression of CD47 by neoplastic cells. CD47 monoclonal antibody–based immunohistochemistry with hematoxylin counterstain. Bar = 50 μm.
The investigators expected some expression of CD47 in normal equine skin, but that it would be low compared to that of the cutaneous masses. 21 In our investigation, CD47 was expressed in cryopreserved tissues from several different equine cutaneous masses but was undetectable in normal skin. The reason for this is unclear; We expected that expression in normal skin would be present but less than in abnormal tissues and tumors. Although there is binding of the monoclonal antibody used for staining (P-HUH71A f ) to equine tissues in this study, it was not specifically raised to the equine CD47 protein and therefore may not provide the sensitivity needed for detection of CD47 expression in normal equine skin. Of interest was the finding that some of the nonneoplastic lesions that were excluded from the statistical analysis did express CD47 (4/7 cases). This finding warrants further investigation of CD47 in nonneoplastic lesions, as there may be implications for diagnosis, prognosis, and therapeutic interventions.
Although our IHC assay for equine CD47 is a new protocol, development of the assay was routine. Antibody-based assays can be nonspecific and empirical; however, our use of positive and negative controls, including isotype antibodies, improved the specificity of our assay. There is only a small amount of data as to the species specificity of the anti-CD47 antibodies with respect to equine tissues. Evaluation of human, mouse, rat, pig, and cow CD47 proteins reveals that the percentage of identity of the Ig domain of CD47 between humans and mice is 62%, whereas it is 70% for bovine. 21 There is evidence that Ig domains are relatively resilient against multisite mutations. 21 The extent to which the bovine and equine amino acid sequences for CD47 are the same in a BLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi) alignment is 75%; human and equine are 69%. This supports observations by others that CD47 protein sequence is highly conserved, and equine cell cross-reactivity to human CD47 antibodies has been documented as well. 14
Our evaluation of expression of CD47 in equids is novel in veterinary medicine and offers avenues to innovative approaches for treatment of skin tumors by using an immunotherapy aimed at reducing tumor mass. The CD47-SIRPα interaction is thought to block phagocytosis by several mechanisms, and binding CD47 on tumor cells with anti-CD47 antibody would be expected to interfere with the CD47-SIRPα interaction, thereby enhancing macrophage phagocytic activity. 4 Those tumors with increased or more intense staining for CD47 might be expected to respond better to treatment with anti-CD47 antibodies, as increased expression has been shown to strengthen phagocytic inhibitory signals. 9
Development of a specific antibody treatment aimed at CD47 to be used alone or in combination with other treatments may support resolution of some types of tumors while limiting damage to normal structures.3,24 Blockade of SIRPα-CD47 interaction may also promote immune system action against the tumor, particularly in cases where CD47 is highly expressed, as it is the only identified negative regulator of phagocytosis. 20 CD47 also has effects on dendritic cells, including inhibition of maturation of dendritic cells. As a whole, these effects and other reported effects that promote tumor growth and survival likely decrease the efficiency of immune responses against tumor cells that highly express CD47.20,22
Investigation of the presence and behavior of CD47 in equine patients would be expected to help provide valuable information for other veterinary species, and may be a useful animal model for the study of CD47 immunotherapy to treat human tumors. If so, it may benefit researchers as well as veterinarians attempting to better treat these tumors. Additionally, some cutaneous tumors in horses are similar to human tumors, which may provide opportunities for translational studies. If therapeutic applications are developed, there are many tumor types and species that may benefit from their use alone or in combination with other treatments already employed such as radiation therapy and chemotherapy. This study demonstrates that CD47 is expressed in several equine tumors and offers a possible therapeutic target.
Footnotes
Authors’ contributions
SS Caston contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; critically revised the manuscript; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. EE Cooper contributed to acquisition, analysis, and interpretation of data, and drafted the manuscript. P Chandramani-Shivalingappa contributed to acquisition, analysis, and interpretation of data, and critically revised the manuscript. BA Sponseller and JM Hostetter conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and critically revised the manuscript. Y Sun contributed to acquisition, analysis, and interpretation of data. All authors gave final approval.
a.
Dormosedan, Zoetis Inc., Florham Park, NJ.
b.
Carbocaine, Pharmacia & Upjohn Co., Peapack, NJ.
c.
Monocryl, Ethicon US LLC, Blue Ash, OH.
d.
Tissue-Tek OCT compound, Sakura Finetek USA Inc., Torrance, CA.
e.
Whirl-Pak bags, Nasco International Inc., Fort Atkinson, WI.
f.
Anti-CD47 antibody P-HUH71A IgG1, Monoclonal Antibody Center, Washington State University, Pullman, WA.
g.
MultiLink, BioGenex Laboratories Inc., Fremont, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for a portion of the work reported here was received as a grant from The American Quarter Horse Foundation.
