Abstract
Our study describes the clinical, epidemiologic, pathologic, immunohistochemical, and molecular aspects PCR of a case of cutaneous pythiosis in a donkey (Equus asinus) from Brazil. During a dry period, the animal grazed for 4 months around a pond where the vegetation remained green. Skin lesions were nodular, multifocal, and disseminated, mainly involving the legs, ventral chest, and mammary gland. On cut surface, there were multifocal to coalescent discrete yellow foci, and occasional small cavitations with a few kunkers. Ulcerative nodular pyogranulomatous and eosinophilic dermatitis with folliculitis and furunculosis were observed histologically. Hyphae were observed in sections stained with Gomori methenamine silver. Immunohistochemistry with Pythium insidiosum antibodies yielded strong immunostaining of hyphae. P. insidiosum DNA was extracted from tissues in paraffin blocks by amplification of a fragment of 105 bp, which targets the 5.8S ribosomal gene. After the diagnosis of pythiosis, the larger skin lesions were excised and treated as second intention healing wounds, which were completely healed 30 days after resection. Small skin lesions regressed spontaneously in ~60 days. The granulomatous inflammation and outcome of the disease in this donkey were similar to cases of pythiosis in cattle.
Pythiosis is a granulomatous disease caused by the oomycete Pythium insidiosum, which affects different species of wild and domestic animals, and even humans, in various regions of the world.3,7 The clinical presentation can vary according to the affected species. In animals, the infection may develop in subcutaneous or cutaneous, gastrointestinal, nasal, pulmonary, or bone tissue. 3 The cutaneous form is considered most common and has been reported frequently in horses. The main feature of the lesion in this species is the presence of branched cavitations filled with necrotic, yellow-white, firm, granular material, known as kunkers, seen on the cut surface of the affected tissue.3,7 Sporadic cases with cutaneous involvement and absence of kunkers have been described in cattle,4,6,12 sheep, 14 goats, 2 dogs, 5 cats, 5 birds, 8 and dromedary. 16 Pythiosis in donkeys, causing skin lesions similar to those seen in horses, was reported in Colombia in 2013. 1
In Brazil, to date, pythiosis has been reported to affect horses,9,11 cattle,4,12 sheep,10,14 goat, 2 dogs, and cats. 5 The objective of this study is to report clinical, epidemiologic, pathologic, immunohistochemical, and molecular features of a case of cutaneous pythiosis in a donkey in the semiarid region of Brazil.
The disease occurred in a mixed-breed, female, adult, pregnant donkey referred to the Veterinary Hospital of the Federal University of Campina Grande (Patos, Paraíba, Brazil) in July 2014. The donkey had multiple skin nodules that progressed for ~30 days to ulcerated, exudative areas. The donkey, along with 15 other donkeys and 3 horses, was from a farm of native pastures (named caatinga) in the municipality of Jucurutu, Rio Grande do Norte. However, only this donkey was grazing in a paddock around a pond where the vegetation remained green. The animal was frequently observed grazing within the pond, which had abundant aquatic vegetation on its surface. The disease was not observed in the other equids.
Clinical examination revealed multiple nodular, rounded, firm skin lesions ranging from 1 to 11 cm in diameter, some ulcerated and others hairless, distributed in the ventral region of the chest and the abdomen, forelimbs, hind limbs, and mammary gland. The largest lesions were typically ulcerated, with serosanguineous exudate (Fig. 1A). Skin biopsy of the ulcerated and non-ulcerated lesions in the chest and limbs was performed for histologic examination. Macroscopically, the cut surface of these fragments showed discrete multifocal to coalescing yellow foci, 0.2–0.4 cm in diameter, surrounded by white, smooth, shiny tissue. Infrequently, adjacent to the foci, small cavities were seen containing kunkers (Fig. 1B).
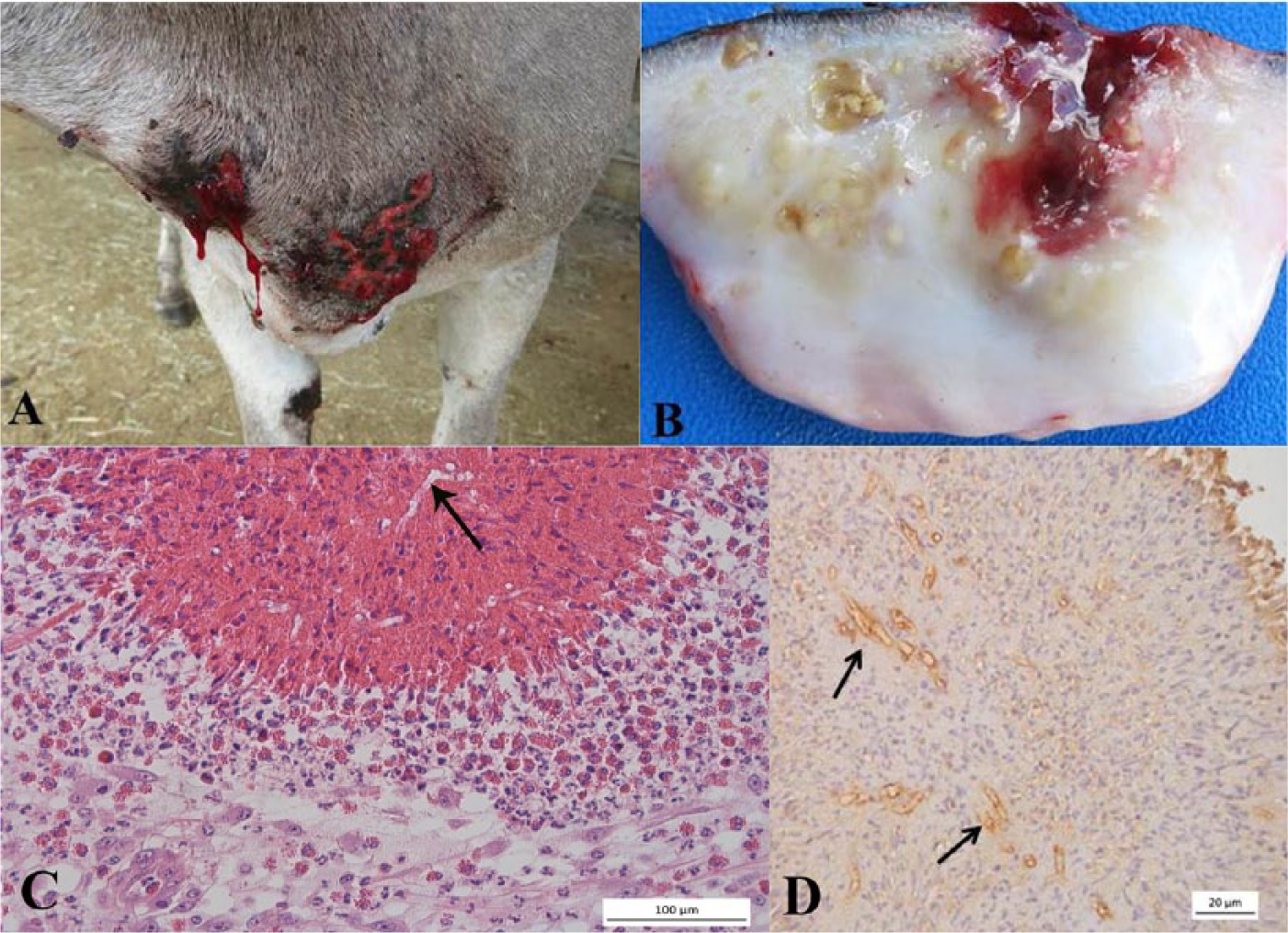
Microscopic lesions were characterized by pyogranulomatous and eosinophilic ulcerative nodular dermatitis with folliculitis and furunculosis. The superficial and deep dermis showed multifocal to coalescing pyogranulomas, with a central area of necrotic eosinophils surrounded by eosinophils, lymphocytes, plasma cells, macrophages, and multinucleated giant cells (Fig. 1C). The granulomas were surrounded by fibrous tissue, frequently infiltrated by eosinophils and, in lesser numbers, lymphocytes and plasma cells. Transverse and longitudinal sections of negatively stained hyphae, occasionally surrounded by small amounts of Splendore-Hoeppli–like reaction, were observed in the necrotic centers. The central area of necrosis was absent in some granulomas. Some hair follicles were ruptured and partially replaced by large areas of necrosis that were infiltrated by large numbers of eosinophils and small numbers of negative-staining hyphae. Macrophages, lymphocytes, and eosinophils infiltrated the walls of some follicles, causing mural folliculitis as well as discrete furunculosis. In sections impregnated with Gomori methenamine silver stain, rarely septated hyphae with parallel walls and irregular branches were observed. Hyphal diameters, obtained through a microscope a with a digital camera, a controlled by the manufacturer’s software, ranged from 2 to 8 µm.
To confirm the etiologic agent and make the diagnosis, immunohistochemistry was performed using polyclonal anti–P. insidiosum serum diluted 1:800, streptavidin–peroxidase complex, and chromogen 3.3′-diaminobenzidine (DAB). As a positive control, histologic sections from a confirmed case of equine pythiosis were used. Sections from the same donkey were used as a negative control, with the primary antibody replaced by phosphate buffered saline containing 0.5% polysorbate 20. This technique yielded strong immunostaining of brown hyphae (Fig. 1D), confirming the diagnosis of pythiosis. DNA extraction was performed using paraffin-embedded tissue samples, after heating in alkaline pH buffer, followed by phenol–chloroform extraction, ethanol precipitation, and resuspension in ultrapure water, as previously described. 13 Polymerase chain reaction (PCR) was performed using primers targeting PI1 (5′-TTCGTCGAAGCGGACTGCT-3′) and PI2 (5′-GCCGTACAACCCGAGAGTCATA-3′), which amplified a 105-bp fragment for the first ribosomal internal transcribed spacer region (ITS1) DNA of P. insidious. DNA extracted from paraffin-embedded tissue samples from a confirmed case of equine pythiosis was used as positive control. Ultrapure water was used as negative control. The samples from this donkey and the positive control samples were PCR positive.
The largest skin lesions were surgically excised, and treated by secondary intention healing, with topical application of chlorhexidine and insect repellent. The smaller nodules were not surgically removed. Healing of the surgical wounds was observed by 30 days after the procedure, and the smaller, untreated, nodules disappeared after 60 days.
Gross cutaneous lesions observed in this donkey were similar to those described in ruminants. Small cavities filled with kunkers were seen on the cut surface of some nodules. Kunkers are considered pathognomonic of pythiosis in horses, and have also been reported in donkeys with skin lesions 1 and camels with vulvar involvement. 15 In the donkey observed in our study, the presence of small kunkers associated with negative staining of intralesional hyphae allowed the presumptive diagnosis of pythiosis. However, kunkers were not a predominant feature in the lesions in this donkey, in contrast to other cases in horses 7 and donkeys. 1
Microscopically, granulomatous inflammatory reaction and/or pyogranulomatous lesions with discrete central areas with necrotic eosinophils predominated in the lesions of this donkey. Similar granulomatous lesions have been reported in cattle and are thought to be responsible for the self-limiting condition of pythiosis in this species.4,12 In our study, the remission of smaller nodules and the quick healing of the posttreatment wounds may be attributed to the type of granulomatous inflammatory response observed, characterized by mononuclear cells, macrophages, and many multinucleated histiocytic giant cells. Development of granulomatous inflammation requires predominance of a Th1-type cellular immune response involving lymphocytes and cytokines, such as interleukin-2 and interferon-γ, which damage and destroy the hyphae. 12 Although in this case the donkey had a benign form of the disease, in a previous report from Colombia, several donkeys had a more severe form of pythiosis with gross and histologic lesions similar to those observed in horses. 1 The differences compared with the clinical picture observed in the previous report may be the result of different resistance to the infection between donkeys or to genetic diversity of P. insidiosum.
It has been reported that pythiosis mainly occurs through direct contact of zoospores or infective forms of P. insidiosum with a skin wound or mucous membrane. The zoospores are attracted by chemotaxis and penetrate the skin of animals through preexisting lesions, where the skin infection begins. 7 However, although considered rare, the possibility of penetration of the agent through hair follicles, without a preexisting skin lesion, was suggested based on observation of hyphae in the hair follicles of affected cattle. 12 Furthermore, under laboratory conditions, mobile zoospores of P. insidiosum were strongly attracted to the hair of the animal species tested (horses, cattle, rabbits, mice), particularly the part of the hair found within the hair follicle. 12 Thus, it is possible that, in this donkey, the infective form of P. insidiosum penetrated the dermis through hair follicles because hyphae were seen in hair follicles associated with folliculitis and furunculosis, and preexisting traumatic lesions were not reported.
In the semiarid region of northeastern Brazil, several factors are considered to be predisposing factors for pythiosis in horses, sheep, and goats: periods of water scarcity, leading to use of water reservoirs during the dry season; high water temperatures, favoring proliferation of the infective form of the agent; and shortage of feed in the dry season, which leads the animals to intensify grazing around and within reservoirs in search of food. 9 In the case of the donkey reported herein the disease occurred during a long dry period (Grande do Norte Agricultural Research Company [EMPARN], http://189.124.135.176/monitoramento/monitoramento), with severe shortage of forage, forcing the animal to stay for long periods in the water grazing the only available vegetation located around and within the pond. In addition to environmental factors, different patterns of behavior may also influence the development of pythiosis in different animal species.9,11 In horses, the habit of grazing in flooded areas is the major risk factor. 11 In contrast, donkeys, goats, and cattle prefer dry environments, avoiding lengthy stays in flooded areas. 9 In a previous report of pythiosis in donkeys in Colombia, 1 the environmental conditions in which the disease occurred were not mentioned.
Footnotes
Authors’ contributions
LA Maia contributed to conception and design of the study; contributed to acquisition and interpretation of data; and critically revised the manuscript. RG Olinda, F Riet-Correa, and AFM Dantas contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and critically revised the manuscript. L Nakazato and EG Miranda Neto contributed to conception and design of the study; contributed to analysis and interpretation of data; and critically revised the manuscript. TF Araújo and PR Firmino contributed to conception of the study and contributed to acquisition of data. All authors drafted the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Axio Scope.A1, AxioCam MRc 5, ZEN 2012 software; Carl Zeiss MicroImaging GmbH, Göttingen, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received financial support through CNPq, no. 474602/2012-5.
