Abstract
Reproductive pathology of domestic guinea pigs is underreported to date. To provide a comprehensive review of uterine disease in guinea pigs, we performed a retrospective study of the pathology archives of the University of Tennessee, College of Veterinary Medicine. By histology, 13 of 37 uterine lesions in 23 animals were neoplastic; the other 24 nonneoplastic lesions included cystic endometrial hyperplasia (16 of 24), endometrial hemorrhage (3 of 24), pyometra (2 of 24), polyp (2 of 24), and mucometra (1 of 24). The most common guinea pig uterine neoplasms were uterine leiomyomas (6 of 13), followed by adenomas (3 of 13) and leiomyosarcomas (1 of 13). Other neoplasms included anaplastic tumors of unknown origin (2 of 13) and choriocarcinoma (1 of 13). Both anaplastic tumors and the choriocarcinoma were positive for vimentin. The choriocarcinoma was positive for HSD83B1, indicating a trophoblastic origin and its final diagnosis. All were negative for cytokeratin and smooth muscle. In multiple animals, more than 1 tumor or lesion was reported. Estrogen receptor and progesterone receptor expression was nearly 100% in uterine neoplasms. Nearly all animals for which data were available had cystic rete ovarii (18 of 19); the animal with no cystic rete ovarii had paraovarian cysts. In our study, female pet guinea pigs had a tendency to develop cystic endometrial hyperplasia and uterine neoplasia. Factors for the development of these lesions could be cystic rete ovarii, hormone dysregulation, and/or age. Other factors could contribute to the development of uterine lesions. As in other species, early ovariohysterectomy could decrease the prevalence of uterine lesions.
Keywords
Introduction
Guinea pigs (Cavia porcellus) are increasing in popularity as companion animals. More than one million pet guinea pigs were determined to reside in U.S. households in 2007. 21 However, most publications on neoplasia in this species focus on spontaneous or induced tumors in laboratory guinea pigs. Historically, neoplasia was thought to be rare in guinea pigs.20,23 From more recent publications, we know that spontaneous neoplasms are rare in guinea pigs <1 year of age, but prevalence can reach 30% in some groups of guinea pigs after 3 years of age.20,23 The misconception of low incidence of neoplasia in this species may have arisen because guinea pigs are usually housed under controlled laboratory conditions and often euthanized early in their lives, whereas a pet guinea pig can reach geriatric status of 12 years of age. 13 To our knowledge, there is only one other published report of uterine neoplasia in guinea pigs, 5 and that publication focuses mainly on spontaneous leiomyomas.
The goal of our retrospective study was to describe the prevalence of uterine lesions in pet guinea pigs submitted to either the autopsy or surgical biopsy service at the University of Tennessee from 2000 to 2015. Benign and malignant tumors, as well as nonneoplastic uterine lesions, were included in the study.
Materials and methods
Case selection
We reviewed all records that included specimens from pet guinea pigs submitted to the autopsy and/or biopsy services of the University of Tennessee, College of Veterinary Medicine between years 2000 and 2015. Only records that documented female reproductive tract lesions were included and hence prevalence in the general population cannot be determined from this study. None of the included cases were from laboratory guinea pigs. Available patient data included age, sex, and clinical history.
Gross pathology
Descriptions of uterine tissues included color, dimension, and the presence of masses or fluids. Additional gross pathologic findings that were described by the submitting clinician or pathologist were also listed for each case.
Histopathology
Tissues were preserved in 10% neutral-buffered formalin and processed routinely. Paraffin-embedded tissues were sectioned at 5 μm, mounted on frosted glass slides, stained with hematoxylin and eosin (H&E), and examined by light microscopy. For each case, previously reported cases were reviewed by a pathology resident (T Veiga-Parga), and confirmed by a board-certified pathologist (SJ Newman).
For our study, sections of ovaries and uteri with lesions were evaluated histologically. Smooth muscle tumors were classified as leiomyoma or leiomyosarcoma based on mitotic index, degree of pleomorphism, presence of necrosis and hemorrhage, local invasion, and metastasis. Diagnosis of malignancy included a mitotic index of ≥1 mitotic figure per 10 high-power fields (hpf, 400×) and a mitotic index of 6–9 per 10 hpf with increased pleomorphism, local invasion, or metastasis. 9 Leiomyomas had a mitotic index of 0–1 mitoses per 10 hpf. Adenomas were discrete nodules with low anisocytosis and anisokaryosis, few mitotic figures, and without invasion of the surrounding tissue. Features consistent with adenocarcinoma include greater pleomorphism and mitoses, peripheral invasion, and/or scirrhous response. The criteria for diagnosis of anaplastic tumors were more complicated. Anaplastic tumors were those in which histologic features did not include any of the previously described tumors.
Immunohistochemistry
In cases in which a definitive histologic diagnosis could not be determined based on the H&E slide, immunohistochemistry (IHC) was performed. IHC for smooth muscle actin a (SMA, clone 1A4), cytokeratins AE1/AE3 a (CK, clone AE1/AE3), vimentin a (clone V9), and hydroxyl-C-5-steroid dehydrogenase b (HSD3B1) was performed on a subset of the tumors. Positive controls included smooth muscle, epidermis, and connective tissue. Negative controls were produced by not applying the primary antibody. SMA was applied for 30 min at a 1:1,000 dilution, CK was applied for 30 min at 1:800, and vimentin was applied for 30 min at 1:2,500. Positive controls for HSD3B1 monoclonal antibody were cotton rat (Sigmodon hispidus) placenta and guinea pig endometrium, and the procedure performed was as reported previously. 12
Histologic sections of all tumors and polyps had IHC performed for estrogen receptor c (ER-α, clone CC4-5) and progesterone receptor (PR, clone PR10A9). d Concentrations for ER were at a 1:50 dilution and for PR were at a 1:100 dilution. Positive controls included normal guinea pig uterus; negative controls did not receive the primary antibody. Incubation times were 60 min for ER and 30 min for PR.
Results
Tissues from 23 guinea pigs were examined and yielded 37 lesions, including 24 nonneoplastic and 13 neoplastic. Cystic endometrial hyperplasia (CEH; Fig. 1A) was the most prevalent uterine change (16 of 23) and the most frequent nonneoplastic lesion (16 of 24; Tables 1 and 2). Other nonneoplastic lesions in decreasing incidence were endometrial hemorrhage secondary to uterine prolapse (3 of 24; Fig. 1B), pyometra (2 of 24), polyp (2 of 24), and mucometra (1 of 24). One guinea pig had concurrent CEH and multiple uterine tumors (Tables 1 and 2). The median age of guinea pigs with uterine lesions was 3.8 years and for uterine neoplasia was 4.2 years. The most common tumor of guinea pigs was uterine leiomyoma (6 of 13; Fig. 1C), followed by adenomas (3 of 13; Fig. 1D). Only 1 tumor was classified as a leiomyosarcoma. There were no adenocarcinomas. Leiomyomas and adenomas were concurrent in 2 cases, and 1 animal had multiple leiomyomas. Smooth muscle tumors were within the serosa and myometrium of the uterine horns. A surprising finding was that 3 of the neoplasms were diagnosed as anaplastic tumors by routine microscopy and postulated to be choriocarcinomas based on histologic criteria (Fig. 1E). Subsequent IHC revealed all anaplastic tumors to be negative for SMA and CK, and positive for vimentin (Table 3). Only 1 anaplastic tumor was positive for HSD3B1 (Fig. 1F) and consequently diagnosed as a choriocarcinoma.
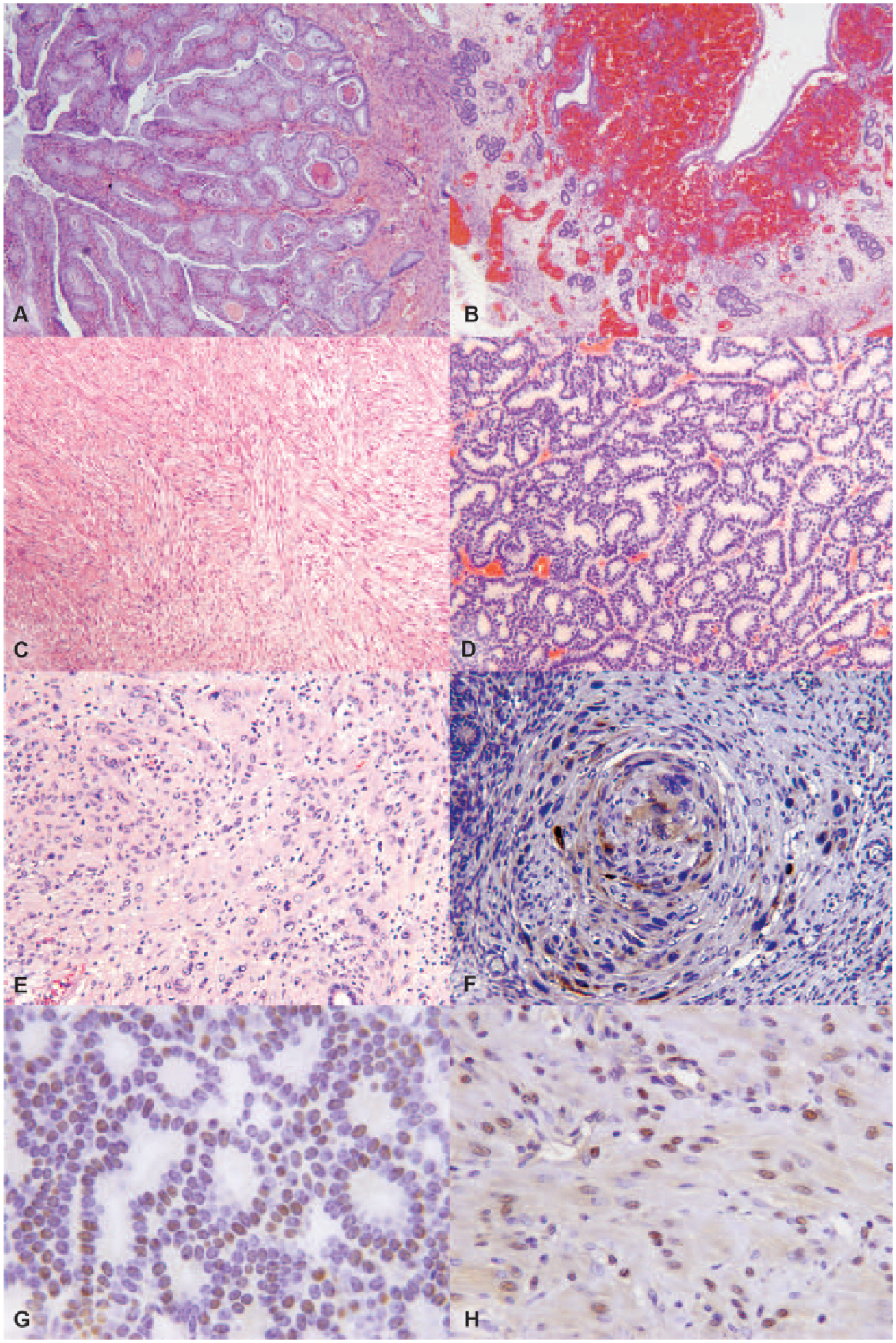
Guinea pig uterine lesions.
Uterine lesions of guinea pigs.
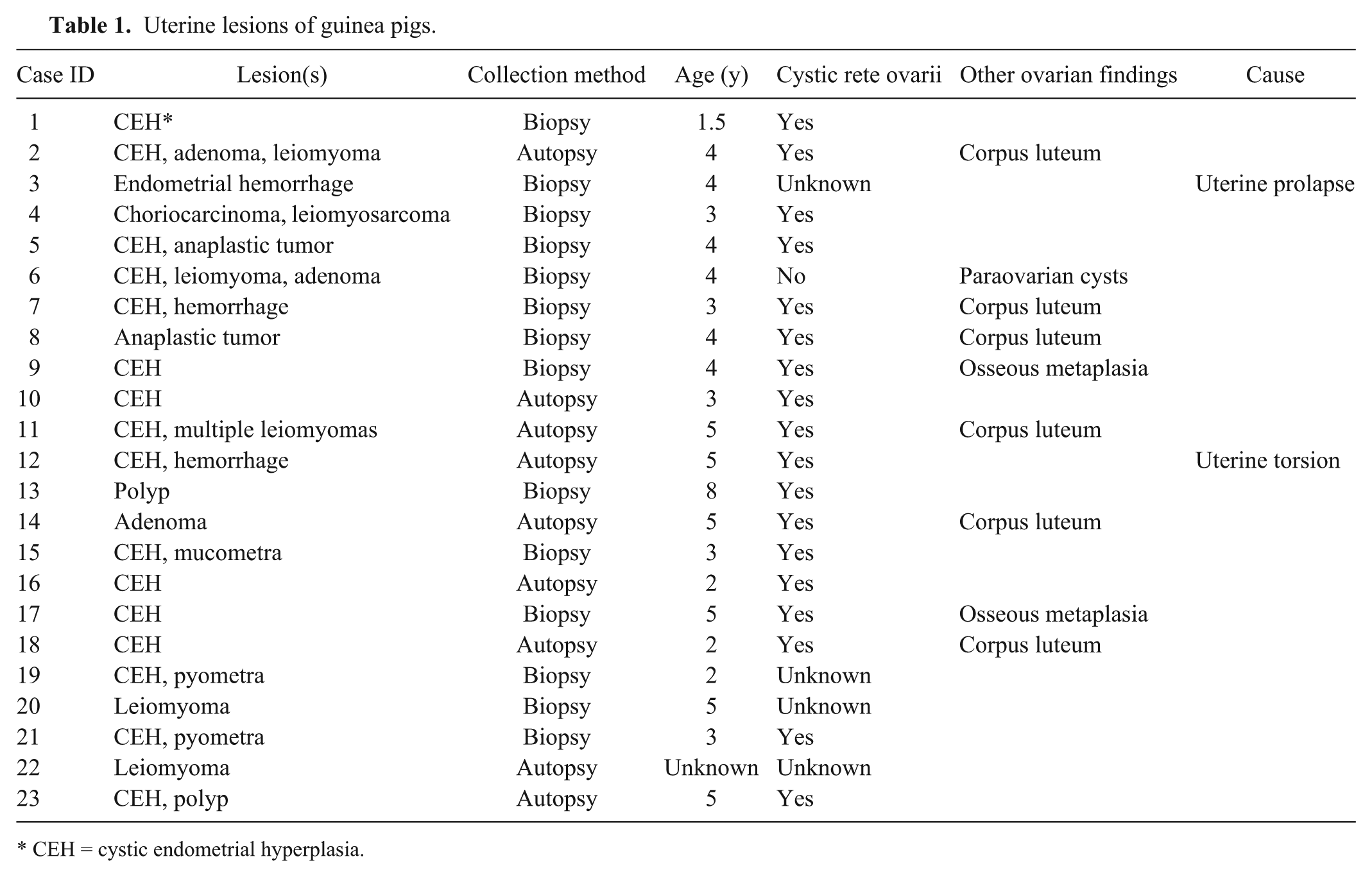
CEH = cystic endometrial hyperplasia.
Comparison of uterine lesions in guinea pigs (n = 23).
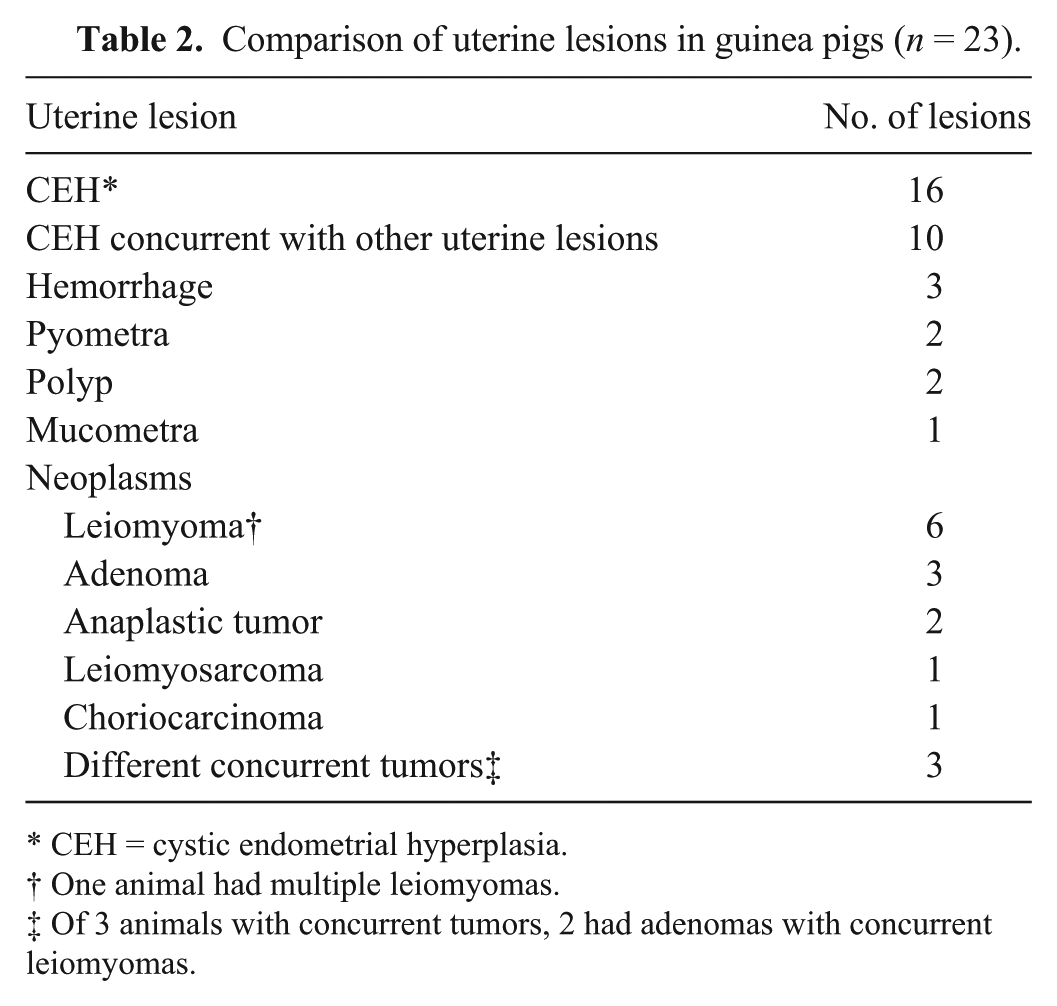
CEH = cystic endometrial hyperplasia.
One animal had multiple leiomyomas.
Of 3 animals with concurrent tumors, 2 had adenomas with concurrent leiomyomas.
Immunohistochemistry of anaplastic uterine tumors in guinea pigs.*
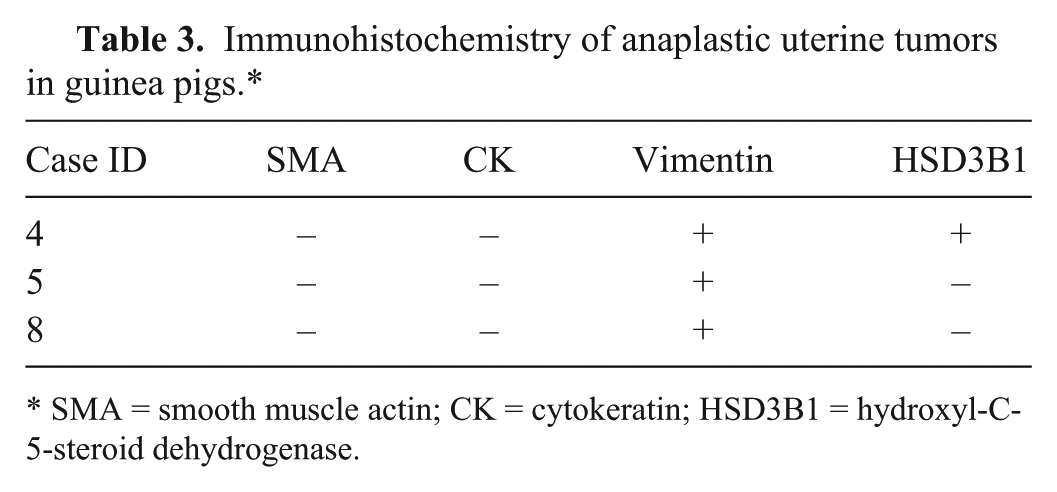
SMA = smooth muscle actin; CK = cytokeratin; HSD3B1 = hydroxyl-C-5-steroid dehydrogenase.
IHC for ER and PR was done for all tumors and polyps (Table 4). Most tumors, with the exception of 2 cases (Table 4), were positive for PR (Fig. 1G) and ER (Fig. 1H). Both polyps were positive for both markers. There was no difference in hormone receptor expression in benign versus malignant smooth muscle tumors. Only 1 adenoma was negative for both markers. The intensity of staining differed among the tumors, and was graded (–) no expression, 1 (mild), 2 (moderate), and 3 (marked; Table 4).
Immunohistochemistry results for all uterine tumors and uterine polyps in guinea pigs (n = 11). Nuclear expression of estrogen receptor (ER) and progesterone receptor (PR) were measured on a scale from 1 (low expression) to 3 (strong expression).*
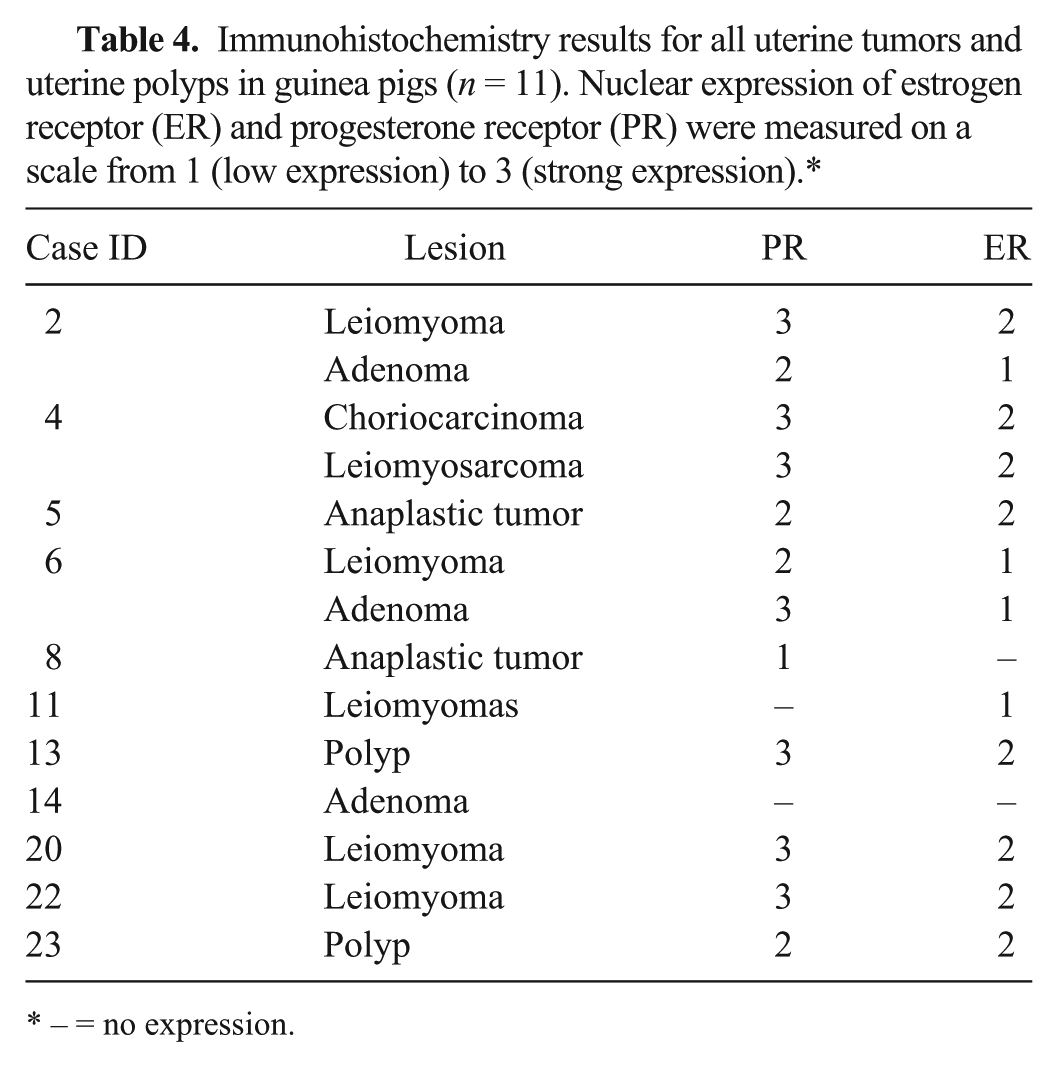
– = no expression.
We evaluated a histologic section of 1 ovary for 19 of 23 guinea pigs. Microscopic findings included follicles at different stages, regressing follicles, and corpora lutea. Nearly all guinea pigs with uterine neoplasia in which ovaries were available had cystic rete ovarii (18 of 19; Table 1), as did all guinea pigs with non-neoplastic uterine lesions. Concurrent follicular development and presence of corpora lutea were occasionally observed. One animal without cystic rete ovarii had paraovarian cysts.
Discussion
The results of our study indicate a high prevalence of CEH (16 of 24) and uterine neoplasia (13 of 37) in pet guinea pigs. In an earlier study, 5 only 8% of guinea pigs submitted to the autopsy service were reported to have uterine leiomyomas, and the only other reported uterine neoplasms were a cavernous hemangioma and a teratoma. In our sample population, uterine leiomyoma was also the most frequent uterine tumor, representing 6 (of 13) of all uterine tumors, followed by adenomas (3 of 13). Leiomyomas and adenomas were concurrent in 2 cases, and 1 animal had multiple leiomyomas, as has been reported in other species. 9 Additionally, 3 of 13 uterine neoplasms were initially diagnosed as anaplastic tumors, and characterization with IHC could not reach a further diagnosis for 2 of them. The other anaplastic tumor was positive for HSD3B1, based on control tissues, including cotton rat placenta for which it was optimized, and guinea endometrium under the influence of progesterone, which acted as an internal control, indicating a presumptive trophoblastic origin, and hence a choriocarcinoma. 12
Choriocarcinoma is a rare, highly malignant, hormonally functional, biphasic, trophoblastic tumor composed of cytotrophoblastic- and syncytiotrophoblastic-like cells. Choriocarcinomas involve 3 distinct neoplastic conditions: 1) gestational choriocarcinoma of trophoblastic origin primarily located in the uterus and arising after a gestational event, 2) gonadal or extragonadal (usually midline location) nongestational choriocarcinoma of germ cell origin, and 3) nongestational choriocarcinoma arising within a poorly differentiated endometrial carcinoma.2,3 Choriocarcinomas have been reported to occur either in the uterus or ovaries. Spontaneous occurrence of this tumor is very rare in animals, as well as in humans. 8 Other species diagnosed with ovarian or uterine choriocarcinomas include macaques,4,14,22 rabbits, 11 mice,1,2 and a rat. 18 Primary gastric choriocarcinoma has been reported in a dog. 19 To our knowledge, there has been no prior publication of spontaneous choriocarcinoma in pet guinea pigs. In the current case, the tumor was considered nongestational because, to our knowledge, this guinea pig had never mated. In most cases, nongestational choriocarcinomas arise from germ cells in the ovaries. 3 All anaplastic tumors in the current study had concurrent cystic rete ovarii. None of them had evidence of neoplastic germ cells in the ovaries. Moreover, nongestational choriocarcinomas can rarely arise from trophoblastic differentiation within poorly differentiated endometrial carcinomas. 3 In our case, there was no evidence of endometrial carcinoma.
Age is likely a risk factor for the development of uterine neoplasia in guinea pigs, given that all animals with uterine tumors in our study were 3 years of age or older, similar to those from previous reports in pet guinea pigs with spontaneous neoplasms. 7
In our study, the most frequent uterine lesion was CEH. Previous reports showed that in multiple domestic animal species, estrogen stimulation played a role in the development of cystic uterine changes. 9 Moreover, endogenous hormones can play a role in the development of tumors in female and male reproductive organs. 9 In our study, all but 1 animal, in which ovarian tissue was available and examined, had cystic rete ovarii. In a previous study, 17 cystic rete ovarii were found at autopsy in 76% of female guinea pigs between 1.5 and 5 years of age, and suggested compromised reproductive performance. In that same study, cystic rete ovarii were concurrent with CEH, mucometra, endometritis, and uterine fibroleiomyomas, similar to our findings. The fact that cystic rete ovarii are concurrent to many other neoplastic and non-neoplastic reproductive lesions could either be by chance given their high prevalence in guinea pigs, caused by unregulated hormone issues, or indicate that cystic rete ovarii could be a risk factor for the development of uterine lesions, especially if or when hormonally stimulated.
Little is known about the pathogenesis of uterine neoplasia in domestic guinea pigs. In other species, ER and PR expression have been identified in uterine tumors. 9 In the present study, nuclear expression of PR was present in all smooth muscle tumors except 1 leiomyoma and 2 of 3 adenomas. That same adenoma that was negative for nuclear expression of PR was negative for nuclear expression of ER, and 1 of the anaplastic tumors was similarly characterized. The intensity and distribution of ER and PR expression were variable between all neoplasms, but most of them were stronger for PR than for ER. Both hormones have been shown to contribute to the development of smooth muscle tumors in other species, such as the dog, human, and potbellied pig.6,15,24 In women, PR and ER expression have been observed in epithelial and spindle cell tumors; however, data are contradictory. Although some groups have shown in women that leiomyomas have higher expression of ER and PR than normal myometrium,6,16 other studies have demonstrated that leiomyosarcomas have lower PR and ER expression when compared with leiomyomas. 24 Moreover, in women with endometrial proliferations, loss of PR and ER expression has been associated with more aggressive behavior. 10
Footnotes
Acknowledgements
We thank Misty Bailey for technical editing.
Authors’ contributions
T Veiga-Parga and SJ Newman contributed to conception and design of the study, and contributed to acquisition, analysis, and interpretation of data. KMD La Perle contributed to analysis and interpretation of data. All authors drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Dako North America Inc., Carpinteria, CA.
b.
Monoclonal rabbit antihuman EPR9686, Abcam PLC, San Francisco, CA.
c.
Santa Cruz Biotechnology Inc., Santa Cruz, CA.
d.
Immunotech-Beckman Coulter, Fullerton, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
