Abstract
Two solitary and minute tumors of 1 and 1.5 mm diameter were identified by microscopy in the left fourth mammary gland of a 13-year-old female Labrador Retriever dog, in addition to multiple mammary gland tumors. The former tumors were well circumscribed and were composed of small-to-large polyhedral neoplastic oncocytes with finely granular eosinophilic cytoplasm, and were arranged in solid nests separated by fine fibrovascular septa. Scattered lumina of variable sizes containing eosinophilic secretory material were evident. Cellular atypia was minimal, and no mitotic figures were visible. One tumor had several oncocytic cellular foci revealing cellular transition, with perivascular pseudorosettes consisting of columnar epithelial cells surrounding the fine vasculature. Scattered foci of mammary acinar cell hyperplasia showing oncocytic metaplasia were also observed. Immunohistochemically, the cytoplasm of neoplastic cells of the 2 microtumors showed diffuse immunoreactivity to anti-cytokeratin antibody AE1/AE3, and finely granular immunoreactivity for 60-kDa heat shock protein, mitochondrial membrane ATP synthase complex V beta subunit, and chromogranin A. One tumor also had oncocytic cellular foci forming perivascular pseudorosettes showing cellular membrane immunoreactivity for neural cell adhesion molecule. The tumors were negative for smooth muscle actin, neuron-specific enolase, vimentin, desmin, S100, and synaptophysin. Ultrastructural observation confirmed the abundant mitochondria in the cytoplasm of both neoplastic and hyperplastic cells, the former cells also having neuroendocrine granule–like electron-dense bodies. From these results, our case was diagnosed with mammary oncocytomas accompanied by neuroendocrine differentiation. Scattered foci of mammary oncocytosis might be related to the multicentric occurrence of these oncocytomas.
Mammary gland tumors are frequent in dogs and cats, but appear extremely sporadically in other domestic animals.
21
However, oncocytic tumors, neoplastic proliferations of oncocytes, are rare in both domestic animals and humans.4,16 The term “oncocyte” has been designated from the Greek language (
Neuroendocrine tumors are a group of biologically and clinically heterogeneous neoplasms that most commonly originate in the lungs, gastrointestinal tract, and pancreas. 18 Although occurrence is rare, neuroendocrine breast tumors have been reported in humans. The majority of them are a special type of invasive breast carcinoma and represent <1% of breast carcinomas in which >50% of the tumor cells express neuroendocrine markers. 26 Neuroendocrine tumors arising primarily in the mammary gland had not been reported in domestic animals until the 2015 report of a mammary neuroendocrine carcinoma in a bitch. 22
Our report presents a canine case of mammary oncocytoma with neuroendocrine differentiation. It is also unique in that the oncocytoma developed in association with multiple occurrences of acinar cell hyperplastic foci demonstrating oncocytic metaplasia.
A 13-year-old female Labrador Retriever dog with multiple mammary gland tumor nodules was referred to a veterinary clinic in Mie Prefecture, Japan. Tumor tissues with adjacent mammary gland tissues at the right first and second to third mammary glands and left second, third, and fourth mammary glands were surgically removed and subjected to histopathologic analysis.
For histopathology, all tumor tissue samples were fixed in neutral phosphate-buffered 10% (v/v) formalin, then processed in a routine manner, embedded in paraffin, and stained with hematoxylin and eosin. Replicate sections of all tumor samples were used for immunohistochemistry and periodic acid–Schiff (PAS) staining.
Histologically, macroscopic mammary gland tumor nodules were diagnosed as 2 benign mixed tumors, 2 simple adenomas, 2 complex adenomas, and 1 simple carcinoma. Two solitary and minute tumors of 1 and 1.5 mm diameter were found adjacent to 1 simple adenoma at the left fourth mammary gland. Microscopic examination of these minute tumors revealed well-circumscribed, nonencapsulated, noninfiltrative expansive neoplastic masses consisting of polyhedral neoplastic cells arranged in solid nests lined with small spindle cells and supported by a scant fibrovascular stroma (Fig. 1A, 1B). Individual neoplastic cells exhibited “oncocytic metaplasia,” having moderate to abundant finely granular, eosinophilic, and occasionally vacuolated cytoplasm with distinct cell boundaries and round to oval centrally located nuclei containing 1 or 2 nucleoli (Fig. 1B). Scattered lumina of variable sizes containing eosinophilic secretory material were evident (Fig. 1B). Cellular atypia was minimal, and no mitotic figures were visible in high magnification fields. There was moderate anisocytosis and anisokaryosis. A few small foci of lymphocytic infiltration were noted in the interstitium of tumor tissues. No local and vascular invasions were noted in the 2 minute tumors. In one of the tumors, there were several foci of perivascular pseudorosettes consisting of columnar epithelial cells surrounding fine vascular interstitium showing cellular transition with polyhedral neoplastic cells (Fig. 1C). Cytoplasmic granules of neoplastic cells were strongly positive by PAS reaction. Apart from the minute tumors, there were sparsely distributed mammary gland hyperplastic foci showing oncocytic metaplasia consisting of polyhedral acinar cells with lightly eosinophilic cytoplasm demonstrating fine cytoplasmic PAS-positive granules (Fig. 1D).
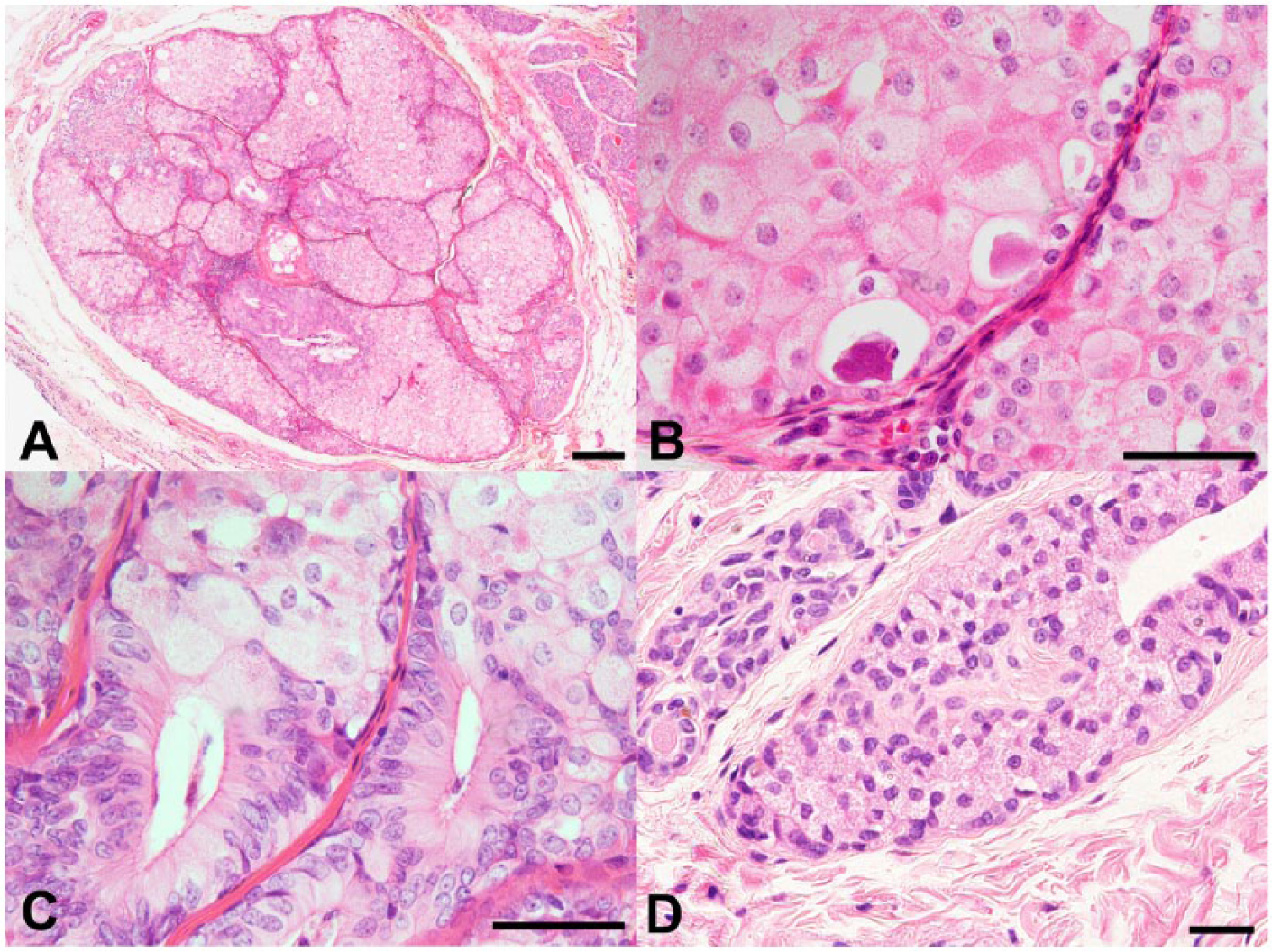
Light micrographs of a minute tumor nodule and foci of acinar cell hyperplasia showing oncocytic metaplasia in the mammary gland of a dog.
For immunohistochemistry, the avidin–biotin peroxidase complex technique was performed a with primary antibodies against cytokeratins (CKs) with AE1/AE3 (1:50; clones AE1 and AE3), b vimentin (1:200), c desmin (ready-to-use; clone D33), b smooth muscle actin (SMA; 1:100; clone 1A4), b synaptophysin (1:100; clone SY38), b chromogranin A (1:3,000), d neuron-specific enolase (NSE; 1:100; clone BSS/NC/VI-H14), b neural cell adhesion molecule (NCAM; 1:500), e S100 (ready-to-use), b 60-kDa heat shock protein (HSP60; 1:500; clone LK1), f mitochondrial membrane ATP synthase complex V beta subunit (ATPB; 1:500; clone 3D5), f and proliferating cell nuclear antigen (PCNA; 1:200; clone PC10). b Before endogenous peroxidase blocking, sections were microwaved at 90°C for 10 min with target retrieval solution b (pH 6.0) for vimentin, NSE, NCAM, and PCNA staining, autoclaved at 121°C for 10 min with target retrieval solution b (pH 6.0) for AE1/AE3 staining, microwaved at 90°C for 10 min with citrate buffer (pH 9.0) for SMA staining, or autoclaved at 121°C for 10 min with citrate buffer (pH 9.0) for synaptophysin, HSP60, and ATPB staining. No antigen retrieval methods were performed for desmin, chromogranin A, and S100 staining. The chromogen was 3,3′-diaminobenzidine, and the counterstain was hematoxylin. Positive controls for immunohistochemistry were all prepared from canine tissues (i.e., stomach tissue for HSP60, ATPB, and PCNA; adrenal gland for synaptophysin and chromogranin A; cerebrum for NSE, NCAM, and S100; skin for AE1/AE3, vimentin, and SMA; and heart tissue for desmin). Non-immunized sera were substituted for the primary antibody as negative controls for immunoreactivity.
In the 2 minute tumors, finely granular cytoplasmic immunoreactivity was observed in the majority of neoplastic cells exhibiting oncocytic metaplasia with HSP60 and ATPB (Fig. 2A, 2B). Diffuse cytoplasmic immunoreactivity was observed in all neoplastic cells with CKs using AE1/AE3 antibody, which was associated with strong immunoreactivity at the cellular periphery (Fig. 2C). Finely granular cytoplasmic immunoreactivity was also observed with chromogranin A in a variable number of neoplastic cells exhibiting oncocytic metaplasia, particularly at the periphery of neoplastic cell nests (Fig. 2D). Neoplastic cell foci forming perivascular pseudorosettes consisted of columnar epithelial cells that showed membranous immunoreactivity with NCAM (Fig. 2E). Immunoreactivity was lacking in the neoplastic cells with SMA, NSE, vimentin, desmin, S100, and synaptophysin, whereas spindle cells that lined the margins of neoplastic cellular nests were immunoreactive for SMA. Sparse nuclear immunoreactivity with PCNA was observed, showing a mean PCNA-positive cell ratio of 1.7% in the 2 tumors, in contrast to the mean ratio of 5.1% in ten 400× magnification fields of 1 simple adenoma adjacent to the minute tumors.
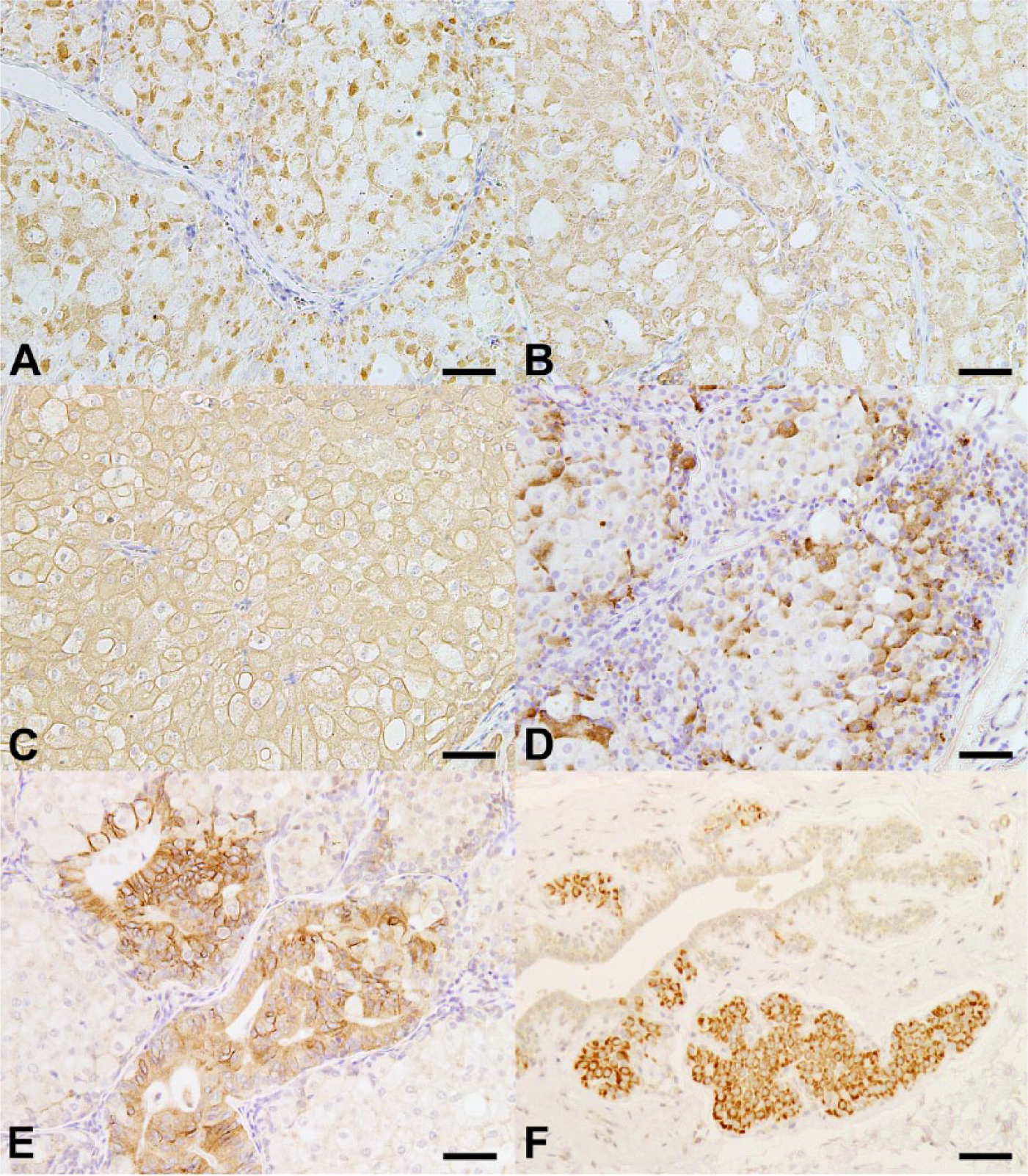
Immunohistochemical analyses of a minute tumor nodule and foci of acinar cell hyperplasia showing oncocytic metaplasia in the mammary gland of a dog.
Foci of acinar cell hyperplasia showing oncocytic metaplasia revealed strong immunoreactivity with HSP60 (Fig. 2F), ATPB, and AE1/AE3. These oncocytic acinar hyperplasias lacked cells immunoreactive for NCAM, but some foci contained a few cells that were weakly positive for chromogranin A.
For transmission electron microscopy, deparaffinized 4-μm-thick tissue sections were fixed in 2.5% glutaraldehyde and post-fixed in 1% OsO4. Fixed specimens were then dehydrated through ascending grades of alcohol and embedded in epoxy resin. After confirming the presence of neoplastic or hyperplastic cells exhibiting oncocytic metaplasia using light microscopy in toluidine blue–stained semithin sections, ultrathin sections were prepared, stained with uranyl acetate and lead citrate, and observed using a transmission electron microscope g at 80 kV.
The ultrastructural appearance of the neoplastic cells exhibiting oncocytic metaplasia contained many variously sized mitochondria within the swollen cytoplasm (Fig. 3A, 3B). Neoplastic cells also contained various numbers of neuroendocrine granule–like electron-dense bodies within the cytoplasm (Fig. 3C). Hyperplastic acinar cells showing oncocytic metaplasia also contained many variously sized mitochondria within the cytoplasm (Fig. 3D).
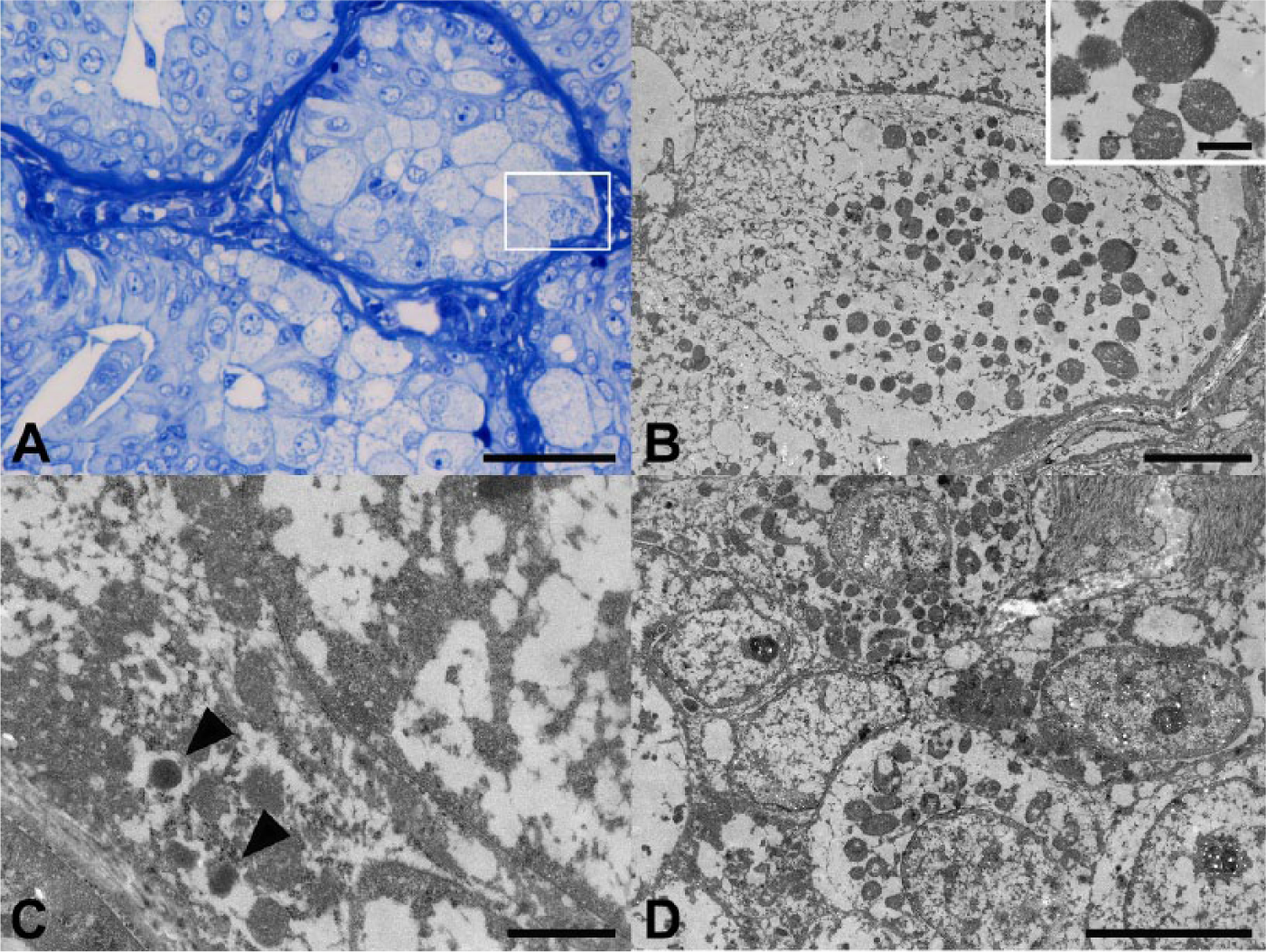
Transmission electron microscopic analysis of a minute tumor nodule and a focus of acinar cell hyperplasia showing oncocytic metaplasia in the mammary gland of a dog.
Histopathologically, differential diagnoses of oncocytomas, neuroendocrine tumors, and granular cell tumors were considered in these minute tumors. Although the number of PCNA-immunoreactive cells was low as compared with adjacent adenomas, these minute tumors showed apparent proliferative activity. Considering the monotonous cellular arrangement with minimal cellular atypia, as well as sparse distribution of PCNA-immunoreactive proliferating cells, these tumors were judged to be benign. Immunohistochemically, these tumors were diffusely immunoreactive with AE1/AE3 antibody recognizing a broad spectrum of CKs, but were consistently negative for vimentin, desmin, and SMA, suggesting an epithelial cell origin of these tumors. The tumors also were negative for S100. Because granular cell tumors are negative for CKs and positive for vimentin, S100, and NSE, 23 the possibility of these minute tumors as granular cell tumors could be excluded. On the other hand, these tumors revealed abundant finely granular cytoplasmic immunoreactivity with HSP60 and ATPB, and similar positivity by PAS reaction, reflecting the numerous mitochondria in the cytoplasm. The presence of many mitochondria was further ultrastructurally confirmed in the swollen cytoplasm. Considering that oncocytomas specifically express CKs, 1 the obtained results suggested that the minute tumors examined herein could be diagnosed as oncocytomas. In humans, most oncocytomas are benign; however, there have been reports of malignant cases in the literature.5,14,15 Except for a case of locally invasive bilateral renal oncocytomas in a dog, 1 all canine oncocytomas reported to date have also been pathologically and biologically benign,10,19,29 similar to the minute oncocytomas in the present case.
Although the precise histogenesis of oncocytes remains unclear, they have been found in the ducts and acini of various endocrine and exocrine glands. 4 Oncocytogenesis has been considered to be a result of the compensatory cellular response against mitochondrial dysfunction associated with mitochondrial DNA mutations. 3 Such mitochondrial DNA mutations might result from aging or oxidative stress. 32 In the present case, we found foci of acinar cell hyperplastic foci consisting of oncocytes that were confirmed by both immunohistochemical and ultrastructural approaches, in addition to 2 minute oncocytomas, in the mammary gland, suggestive of oncocytic metaplasia of acinar epithelial cells. Whether oncocytomas arise from already established oncocytes or transform directly from “normal” epithelial cells is not known. In the present case, portions of oncocytoma cells formed luminal structures containing eosinophilic material, suggesting that these oncocytomas might be generated by oncocytic metaplasia of acinar cells of the mammary gland, although it was unclear whether acinar cells undergoing such metaplasia were normal or hyperplastic.
Although the incidence rate is rather low, human cases of oncocytic neuroendocrine tumors occur in several organs, such as the adrenal medulla, exocrine pancreas, and lungs.17,27,31 In an extensive previous study, which analyzed a number of human oncocytic breast carcinomas, 24 there were a few cases showing neuroendocrine differentiation accompanied by chromogranin A expression. In the present study, a subpopulation of neoplastic cells consisting of minute tumors revealed finely granular immunoreactivity for chromogranin A, despite the lack of immunoreactivity for synaptophysin and NSE. Among neuroendocrine cellular markers, chromogranin A and synaptophysin are frequently and reliably used, and NSE is considered to be a nonspecific marker. 7 Similar to the minute tumors in our case, human neuroendocrine carcinomas may be positive for chromogranin A but negative for synaptophysin. 13 In the present study, one of the tumors further revealed neoplastic cellular foci showing membranous NCAM reactivity, which is a neuroendocrine marker. 12 Moreover, neoplastic cells were shown to contain neuroendocrine granule–like electron-dense bodies within the cytoplasm under electron microscopic observation. Therefore, on the basis of the histopathologic, immunohistochemical, and ultrastructural profiles of the neoplastic cells, together with PAS staining, a diagnosis of oncocytomas with neuroendocrine differentiation of the mammary gland was made.
The histogenesis of mammary neuroendocrine tumors has not been fully clarified in humans. An initial hypothesis suggested that neuroendocrine neoplastic cells derived from argyrophilic cells originate from neural crest cells that have migrated to the mammary ducts. 8 Another hypothesis supports in situ development from neuroendocrine cells naturally found in the breast, but these cells have not been consistently found in the mammary gland.2,30 A novel hypothesis suggests that mammary neuroendocrine tumors result from early divergent differentiation of neoplastic stem cells in the process of carcinogenesis to differentiate into both epithelial and neuroendocrine cell lineages.11,20 In the present study, a few foci of oncocytic acinar cell hyperplasia were revealed to show chromogranin A immunoreactivity, and immunoreactive cells were few in number compared with the minute oncocytic tumors. These results suggest neuroendocrine differentiation during the course of neoplastic transformation of hyperplastic oncocytes.
Oncocytomas in animals are rare tumors of unknown etiology, similar to those in humans. Because of the limited number of cases, associations with age, breed, or sex have not been established. The current report describes mammary oncocytomas with neuroendocrine differentiation in companion animals. Scattered foci of mammary oncocytosis might be related to the multicentric occurrence of such oncocytomas.
Footnotes
Acknowledgements
We thank Mrs. Shigeko Suzuki for her technical assistance in preparing the histologic specimens.
Authors’ note
Megu Itahashi is currently at the Division of Pathology, New Histo Science Laboratory Co. Ltd., 2-979-2, Kurosawa, Ome-shi, Tokyo 198-0005, Japan.
Authors’ contributions
R Nagahara and M Shibutani contributed to conception and design of the study, and contributed to acquisition, analysis, and interpretation of data. M Kimura contributed to conception of the study and to interpretation of data. M Itahashi and G Sugahara contributed to conception of the study and to acquisition and interpretation of data. M Kawashima and H Murayama contributed to design of the study and to analysis of data. T Yoshida contributed to conception of the study and to acquisition and analysis of data. All authors drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
VECTASTAIN Elite ABC kit, Vector Laboratories Inc., Burlingame, CA.
b.
Dako Denmark A/S, Glostrup, Denmark.
c.
Santa Cruz Biotechnology Inc., Dallas, TX.
d.
Yanaihara Institute Inc., Fujinomiya, Japan.
e.
Millipore Corp., Billerica, MA.
f.
Abcam plc, Cambridge, United Kingdom.
g.
JEOL 1210, JEOL Ltd., Tokyo, Japan.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
