Abstract
The current report describes the use of a molecular technique to identify immature Fascioloides magna. An 18-month-old Brangus heifer was found dead in the field without any prior clinical signs. The cause of death was exsanguination into the thoracic cavity associated with pulmonary embolization and infection by immature Fascioloides magna resulting in 2 large foci of pulmonary necrosis and focal arteriolar and lung rupture. The liver had a few random migratory tracts with typical iron and porphyrin fluke exhaust, but no identified fluke larvae. A single immature fluke was found in the lungs, and species level identification as F. magna was confirmed by DNA sequence analysis of the ribosomal internal transcribed spacer regions (ITS1 region, 5.8S rRNA gene, and ITS2) and of partial 28S rRNA gene sequence. This is one of only a few pulmonary fascioloidiasis cases associated with hemothorax in the veterinary literature.
Fascioloidiasis is an important parasitic infection of wild and domestic ungulates caused by the giant liver fluke Fascioloides magna Bassi 1875. 11 The life cycle consists of adult trematodes encapsulated often in pairs in the liver of the definitive natural hosts, white-tailed and mule deer. Eggs are liberated in the feces where they embryonate under ideal conditions, and miracidia develop within the eggs. Mature miracidia hatch in an aquatic environment and seek the first intermediate host, an aquatic snail. Suitable lymnaeid snail hosts become infected after the miracidia penetrate, and asexual reproduction produces larval cercariae.11,13 Cercariae emerge from infected snails and encyst on vegetation as metacercariae until ingested by the host. The larval trematodes leave the intestine and migrate in the abdominal cavity to the liver where they eventually become encapsulated and reproduce. Often of little consequence to the definitive cervid host, infections can cause clinical disease or death in aberrant or dead-end hosts when immature flukes undergo ectopic migration through non-hepatobiliary sites such as the abdominal cavity and lungs.13,16 Geographically, F. magna is native and restricted largely to North America11,14; however, several geographic foci exist in central Europe. 8 The white-tailed deer (Odocoileus virginianus) is a common definitive host for F. magna in eastern North America. The red deer (Cervus elaphus) and the fallow deer (Dama dama) serve as definitive hosts in Europe.11,13 The overlap of areas where domestic ungulates, infected definitive hosts, and suitable snail hosts are found allows for successful transmission of this parasite.11,13
Sheep and goats are aberrant hosts, and infections with F. magna can cause excessive hepatic damage without encapsulation of adult trematodes, sometimes resulting in death, often caused by germination of clostridial spores at sites of fluke-induced tissue damage creating anaerobic conditions favorable for these bacteria. 13 Cattle and other large ungulates are considered dead-end hosts for F. magna. Infected livers often have fibrosis secondary to trematode migration and are darkly pigmented resulting in condemnation at slaughter.11,13 In dead-end hosts, adults may be encapsulated in pseudocysts, but eggs are usually not passed into the environment. However, it may be possible to detect eggs in the feces of dead-end hosts with heavy or severe infections if the pseudocyst ruptures.7,11 Pulmonary infections by immature F. magna have been described experimentally in cattle, sheep, and guinea pigs, and severe pulmonary thrombosis and hemorrhage have been reported in sheep and guinea pigs.4,7 In addition, a 2014 article reported fatal pulmonary hemorrhage caused by F. magna in a steer in Canada. 16 Our report documents a lethal case of pulmonary embolization and infection of immature F. magna in a domestic Brangus heifer in the southern United States and molecular identification of the immature fluke.
On April 8, 2015, an 18-month-old Brangus heifer weighing 430 kg with a body condition score of 6 of 9 (Mississippi State University extension, 2008, Body condition scoring beef cattle. Available at: https://goo.gl/95wdbi) was found dead in the field of a cattle farm in Oktibbeha County, Mississippi. The animal was eating well, had no previous signs of disease, and was in a pasture with 100 other cattle. The pasture had been fertilized with ammonia a week prior to the death of the animal. High magnesium minerals had been added as a supplement only a short time previously, and the producer suspected grass tetany.
At autopsy, routine tissue samples were collected, including sections of lung with the trematode. The sections were fixed in 10% neutral buffered formalin, processed routinely, sectioned at 5 μm, and stained with hematoxylin and eosin for histologic examination.
On gross examination, the right hemithorax was massively filled with ~8–10 L of clotted and unclotted blood (Fig. 1A). Additional blood was present in the left hemithorax. The lungs were generally pink to light red and palpably soft. Two distinct nodular foci were identified in the lungs. A 4 cm diameter firm expansile red nodule with centrally disrupted visceral pleura was on the cranioventral aspect of the right caudal lung lobe (Fig. 1B). This nodule contained red-black friable tissue and appeared to merge with a large vessel wall. Further dissection revealed multiple small cavities containing black friable necrotic material, a 1.5 cm diameter thrombus, and thin irregular black migratory tracts with a 22 × 1 × 0.3 cm leaf-shaped, unsegmented, dorsoventrally flattened trematode (Fig. 1C, 1D). The other 6 × 6 × 4 cm roughly round discrete firm nodule was identified on the caudodorsal aspect of the left cranial lobe and had intact overlying visceral pleura. On cut surface, there were multiple variably sized dark-brown to black thrombi and necrotic debris surrounded by fibrous tissue (Fig. 1E). The liver was diffusely pale tan with scattered thin black tracts that coursed through the parenchyma.
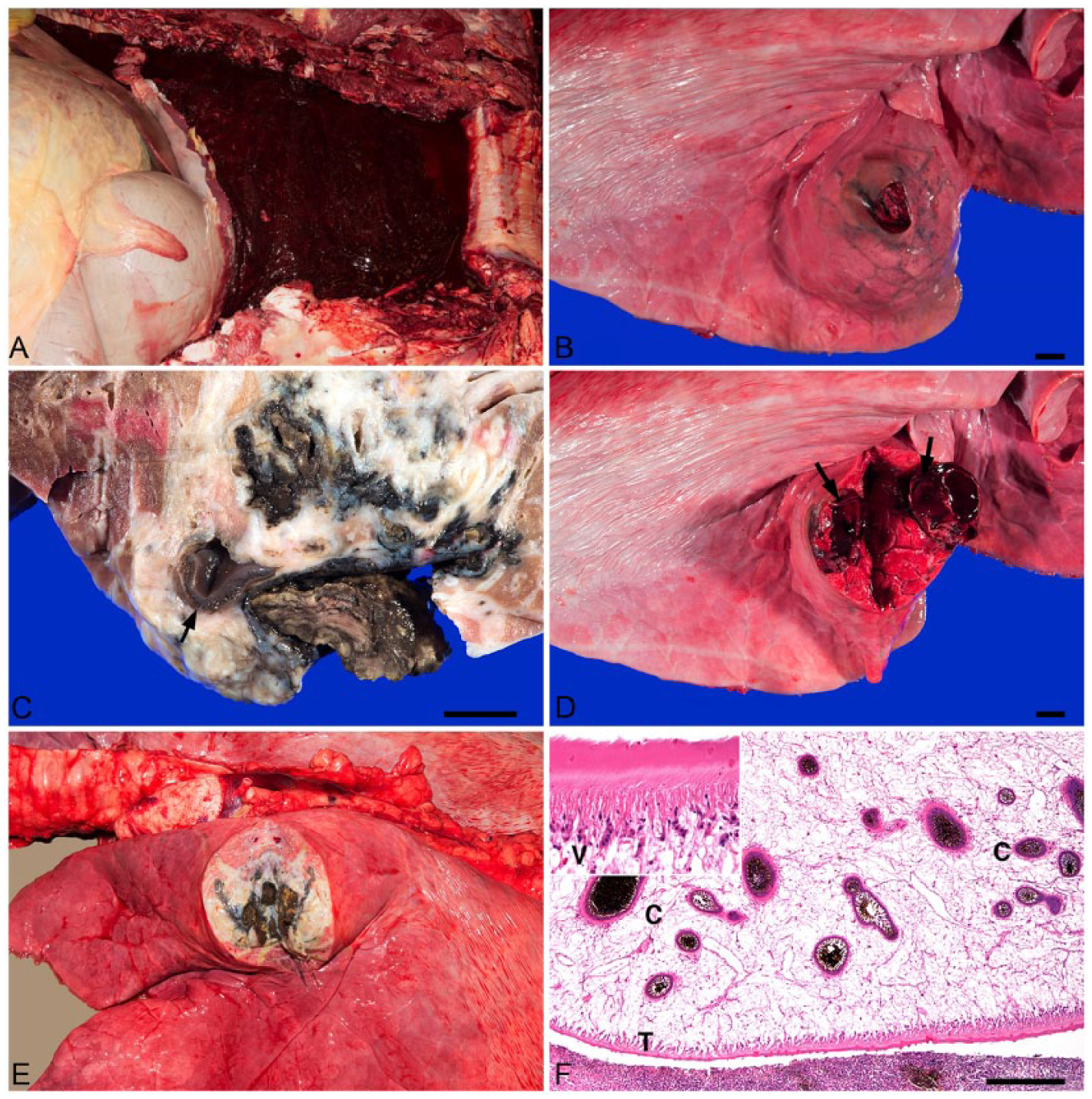
Brangus heifer. Photos of right hemithorax with rib cage removed (A), right (B–D) and left (E) lungs, and histologic section of lung and fluke (F). Hematoxylin and eosin (H&E).
On histologic examination, the 2 firm pulmonary nodules had multiple irregularly arranged prominent arteries and arterioles within heavily inflamed fibrovascular tissue that was interspersed with large numbers of eosinophils, lymphocytes, macrophages, degenerate leukocytes, karyorrhectic debris, intracellular and extracellular dark-brown to black pigment (iron and porphyrin), fibrin, hemorrhage, and multiple mineralized foci. These arteries and arterioles adjacent to a massive hemorrhage were disrupted, even ruptured, and associated with large numbers of eosinophils, lymphocytes, and fewer neutrophils. Mild to moderate proliferations of tunica intima, intimal edema, and vascular necrosis with lymphocytic perivascular infiltrates occurred in the nodular foci in regions of hemorrhage. One of the cavitated foci contained a cross-section of a trematode without visible reproductive structures, indicating immaturity (Fig. 1F). 13 Pulmonary interlobular septa were frequently necrotic and thickened by similar inflammatory infiltrates and fibrous cords extending into adjacent parenchyma. More recent migratory tracts and cavities contained massive hemorrhage and blood clots with similar marginating inflammation and pigment. A tracheobronchial lymph node had large numbers of scattered pigmented macrophages in the cortical and medullary sinuses.
The hepatic parenchyma had multiple random typical fluke migratory tracts containing large numbers of macrophages with black cytoplasmic pigment, eosinophils, lymphocytes, karyorrhectic debris, and dark-brown to black granular pigment associated with necrotic cellular debris. Hepatic lobules were occasionally disrupted with mild incomplete portal bridging fibrosis and biliary ductular hyperplasia. However, trematode eggs or adult trematodes were not identified.
Because of the immaturity of the fluke, genomic (g)DNA was extracted from a section (20–30 mg) of fresh trematode tissue. a Molecular confirmation of the trematode was conducted by sequencing of the internal transcribed spacer 1 (ITS1) region, 5.8S ribosomal (r)RNA gene, and ITS2 region, and partial sequence from the 28S rRNA gene. Polymerase chain reaction (PCR) b assays were performed using the BD1 and BD2 and the LSU5 and 1500R primers for amplification of the ITS regions and 28S rRNA gene, respectively. 15 Briefly, the 25-µL reactions contained 22 µL of PCR supermix, b 10 pmol of each primer, and 1 µL of gDNA (10 ng/µL). The cycling profile consisted of 94°C for 5 min followed by 35 cycles of 94°C for 30 s, 54°C for 30 s, and 72°C for 1 min. Amplicons were visualized in 1.2% agarose gels stained with ethidium bromide (0.5 mg/mL), examined under ultraviolet light, and compared to a concurrently run molecular weight marker. Both amplicons were purified c and commercially sequenced bidirectionally with both forward and reverse primers. d Full sequences were aligned to check for ambiguous bases. e A basic local alignment search tool (BLASTn) search was performed to compare the sequences to others deposited in the National Center for Biotechnology nonredundant nucleotide database. 1
On molecular examination, the 974-bp sequence obtained for the ITS1 region, 5.8S rRNA gene, and ITS2 region (KU232369) shared 99.9% (970/971 bp) sequence identity with an unpublished sequence of F. magna (EF051080) in fallow deer from Oregon and 100% (571/571 bp) sequence identity with F. magna (EF534992) in red deer from Slovakia. 8 The 1,164-bp sequence obtained for the 28S rRNA gene (KU232370) shared a 99.3% (1129/1137 bp) sequence identity with the only F. magna 28S rRNA gene sequence (EU025872) in the NCBI nr/nt database, from a domestic pig from the United States. 10 The high percent similarity supports the identification of the isolate as F. magna, especially when considering the ITS regions, which have been previously shown to have low intraspecific variability among F. magna isolates from definitive hosts in the United States and Europe, but increased interspecific variability for differentiation from other trematodes that infect domestic ruminants. 2 Additionally, sequence similarity for both regions was <94.2% and <98.2%, when compared with ITS1, 5.8S rRNA gene, and ITS2 regions, and the 28S rRNA gene sequence of isolates of the liver fluke, Fasciola hepatica, respectively.
The cause of death in this heifer was exsanguination into the thoracic cavity that was associated with a 4 cm diameter ruptured nodule in the cranioventral aspect of the right caudal lung lobe. This ruptured nodule had a heavy inflammatory reaction, large areas of necrosis, pulmonary thrombi, and dark brown pigment with multiple prominent arteries. The intralesional parasite within the nodule was identified as a fluke based on morphology and, specifically, F. magna based on DNA sequence results for the ITS1, 5.8S rRNA gene, and ITS2 regions and the 28S rRNA gene. Identification and speciation were confirmed using molecular techniques because the trematode was at an immature stage. The massive hemothorax was a result of thrombosis, necrosis, and rupture of large pulmonary arteries and arterioles. The other pulmonary nodule was larger but was not associated with the visceral pleura. The liver also had evidence of typical trematode migratory tracts, but no eggs or adult trematode was present. Although only a single F. magna was identified in the lungs, a larval stage likely penetrated into a hepatic vein during the aimless hepatic migration and, subsequently, embolized via the caudal vena cava and right heart to the lungs. Distal arteriolar thromboembolism followed by lung migration of the fluke larva resulted in severe local necrosis, vascular and lung rupture, and hemothorax resulting in the death of the heifer.
In the southeastern United States, the common definitive host of F. magna is the white-tailed deer. F. magna infections in white-tailed deer usually do not cause clinical disease except in heavy infections.11,13 Infections in domestic cattle with F. magna likely occur in areas where infected white-tailed deer and suitable snail hosts reside in close proximity. The Gulf Coast and lower Mississippi region is considered enzootic for F. magna. Previously reported prevalence rates for white-tailed deer are sporadic for the southern United States: 69.8% in Texas and 33.9% in South Carolina.6,12 Domestic cattle become infected by ingesting metacercariae from the environment and these excyst in the intestine and migrate into the liver. F. magna infections in cattle are considered minimally pathogenic, and extensive migratory tracts are uncommon. 13 However, an experimental study showed pulmonary infection in cattle but none died or exhibited clinical signs. 7 In dead-end hosts, F. magna usually remains immature and infrequently migrates to non-hepatobiliary organs. 13 A 2014 report was described where F. magna infection caused aberrant pulmonary thrombosis and sudden death in a steer in Canada. 16 Similarly, our case also describes pulmonary embolism of F. magna resulting in pulmonary thrombosis, necrosis, massive thoracic hemorrhage, and death. This case further emphasizes the risk of cattle feeding in areas where excessive moisture encourages suitable habitats for snail hosts that may be releasing cercariae. Because domestic cattle are considered dead-end hosts, the heifer in our study likely acquired F. magna as a result of parasite spillover from infected white-tailed deer in the area.
The life cycle of F. magna mirrors that of F. hepatica, and both species are often present in the same geographic areas. Furthermore, some snail intermediate hosts can be suitable for both species of trematodes.5,9 Eggs of both species and of other trematodes that infect ruminants can be difficult to identify morphologically on examination of host feces. 13 However, there are morphologic differences between the adults of these 2 trematodes. The mature size of F. magna is up to 8 cm long, whereas that of F. hepatica is smaller, averaging 3 cm long. Additionally, F. hepatica has an anterior cone, whereas this structure is absent in F. magna.11,13 However, the morphologic identification of immature trematodes can be difficult if they share similar sites in the host, are improperly fixed, or are immature and lack developed organs for necessary morphologic identification. Therefore, molecular techniques (sequencing of multiple gene targets) can aid in obtaining confirmation of the species and have been used to confirm previous cases of F. magna in a nondomestic species, namely elk. 3
The occurrence of F. magna in Mississippi is well-recognized, but largely undocumented in the literature and warrants further investigation, especially in regard to cases of suspected spillover infections from wild cervids to domestic cattle and small ruminants. Additionally, aberrant pulmonary migration of F. magna should be included as a differential diagnosis for pulmonary parasitic thrombosis and hemothorax in cattle and other animals where F. magna is found.
Footnotes
Authors’ note
JK Lee and TG Rosser contributed equally to this work.
Authors’ contributions
JK Lee and J Cooley contributed to acquisition, analysis, and interpretation of data. TG Rosser contributed to acquisition and analysis of data. All authors contributed to conception and design of the study; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
DNeasy blood and tissue kit, Qiagen Inc., Valencia, CA.
b.
Platinum PCR supermix, Invitrogen Corp., Carlsbad, CA.
c.
QIAquick PCR purification kit, Qiagen Inc., Valencia, CA.
d.
Eurofins, MWG Operon LLC, Huntsville, AL.
e.
SeqMan of Lasergene Genomics Suite, DNASTAR Inc., Madison, WI.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the College of Veterinary Medicine, Mississippi State University, Mississippi State, Mississippi, for funding support.
