Abstract
The aim of our study was to reveal the molecular basis of the serologic nontypeability of 2 Actinobacillus pleuropneumoniae field isolates. Nine field strains of A. pleuropneumoniae, the causative agent of porcine pleuropneumonia, were isolated from pigs raised on the same farm and sent to our diagnostic laboratory for serotyping. Seven of the 9 strains were identified as serovar 15 strains by immunodiffusion tests. However, 2 strains, designated FH24-2 and FH24-5, could not be serotyped with antiserum prepared against serovars 1–15. Strain FH24-5 showed positive results in 2 serovar 15–specific PCR tests, whereas strain FH24-2 was only positive in 1 of the 2 PCR tests. The nucleotide sequence analysis of gene clusters involved in capsular polysaccharide biosynthesis of the 2 nontypeable strains revealed that both had been rendered nontypeable by the action of ISApl1, a transposable element of A. pleuropneumoniae belonging to the IS30 family. The results showed that ISApl1 of A. pleuropneumoniae can interfere with both the serologic and molecular typing methods, and that nucleotide sequence analysis across the capsular gene clusters is the best means of determining the cause of serologic nontypeability in A. pleuropneumoniae.
Keywords
Introduction
Actinobacillus pleuropneumoniae is the etiologic agent of porcine pleuropneumonia, a severe respiratory disease that causes serious economic losses in the pig-rearing industry. 3 To date, 15 serovars have been recognized mainly based on the antigenic diversity of capsular polysaccharides (CPSs) in A. pleuropneumoniae, but in some cases also based on the antigenicity of lipopolysaccharide O-polysaccharides (O-PS).1,17 However, O-PS antigens are shared by multiple serovars. For example, O-PS molecules of essentially the same or similar structure are shared by A. pleuropneumoniae serovars 1, 9, and 11, serovars 4 and 7, and serovars 3, 6, 8, and 15; this results in cross-reactivity among the serovars in each of the 3 groups. 3 Because the virulence of strains differs depending on their serovars, 3 and bacterins for A. pleuropneumoniae confer only serovar-specific protection, 15 serotyping is important. Prevalent serovars differ from country to country. For example, the predominant virulent serovars causing the largest number of outbreaks are serovars 1 and 5 in North America, serovar 2 in most of Europe, serovar 15 in Australia,2,3,20 and serovars 1, 2, and 5 in Japan. 9 However, isolations of serovar 15 have recently increased in Japan. 9
We investigated the genetic characteristics of 9 A. pleuropneumoniae field isolates that were isolated from pigs raised on the same farm and sent to the National Institute of Animal Health, Tsukuba, Japan for serotyping. Immunodiffusion (ID) tests revealed that 7 of the 9 isolates were serovar 15 strains. However, 2 strains, designated FH24-2 and FH24-5, could not be serotyped with antiserum prepared against serovars 1–15, indicating that they are deficient in serovar-specific antigenic determinants or that they might represent a novel serovar. The aim of our study was to reveal the molecular basis of the serologic nontypeability of strains FH24-2 and FH24-5.
Materials and methods
Bacterial strains, growth conditions, and serotyping
Nine strains isolated from the lung lesions of 9 pigs raised on farm A and 3 serovar 15 field strains isolated in 3 different prefectures (at farms B–D) in Japan, which were all sent to the National Institute of Animal Health for serotyping, were used (Table 1). The following A. pleuropneumoniae serovar reference strains were used for ID tests: serovar 1, 4074; serovar 2, S1536; serovar 3, S1421; serovar 4, M62; serovar 5a, K17; serovar 5b, L20; serovar 6, Femø; serovar 7, WF83; serovar 8, 405; serovar 9, CVJ13261; serovar 10, D13039; serovar 11, 56153; serovar 12, 8329; serovar 13, N273; serovar 14, 3906; and serovar 15, HS143. The reference strains for serovars 1–13 were from the culture collection of the National Institute of Animal Health. The serovar 14 and 15 reference strains were kindly provided by Dr. Andresen (Denmark), and by Dr. Blackall (Australia) via Dr. Nagai (Japan), respectively. All bacterial strains were grown as previously described. 7 Serotyping of the 9 Japanese field isolates was carried out with serovar-specific rabbit antisera, prepared against the 15 serovar reference strains, by ID tests as described previously. 13
Strains used and accession numbers of the nucleotide sequences of the cps loci determined for the 12 Japanese isolates of Actinobacillus pleuropneumoniae examined in the current study.*
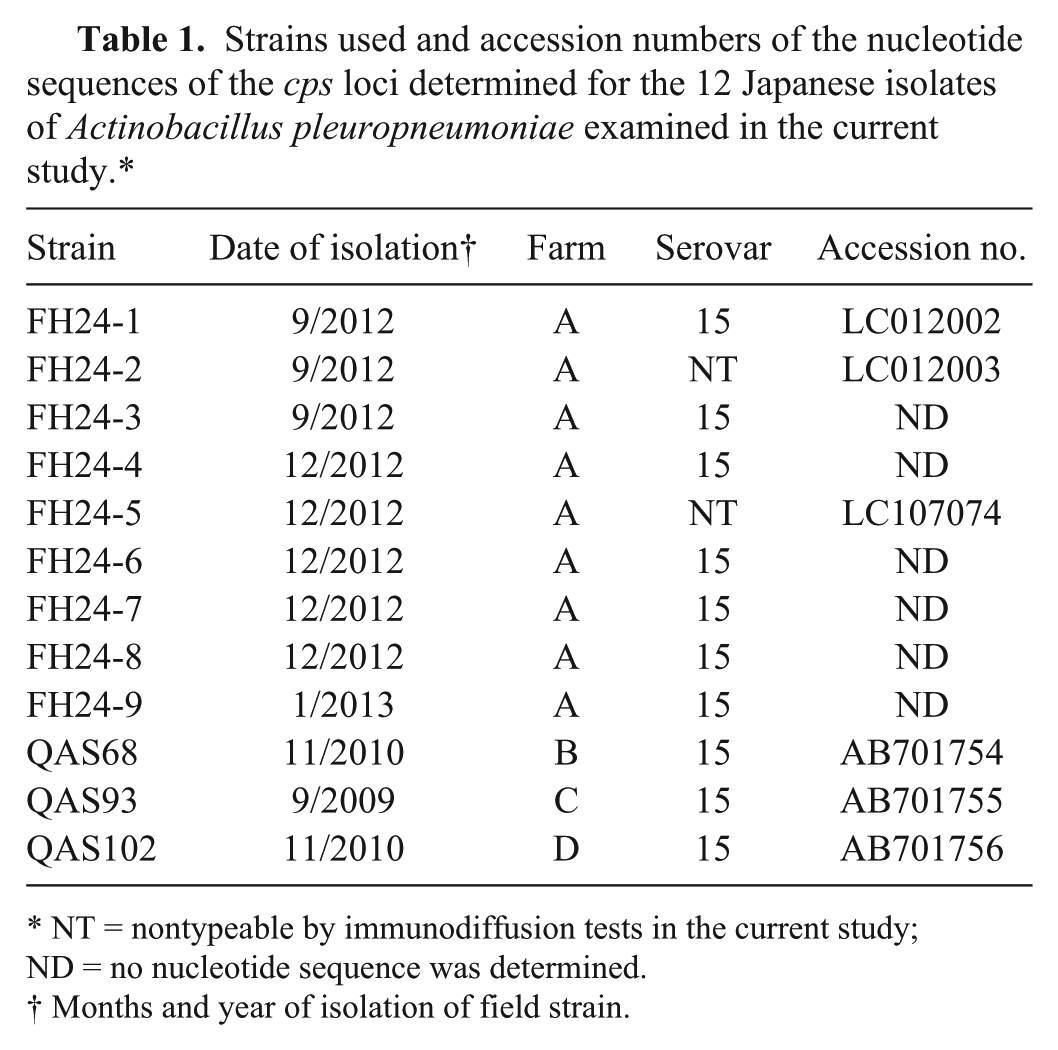
NT = nontypeable by immunodiffusion tests in the current study; ND = no nucleotide sequence was determined.
Months and year of isolation of field strain.
Polymerase chain reaction assays
The polymerase chain reaction (PCR) templates used in our study were prepared with a commercially available DNA extraction kit a as recommended by the supplier. Two serovar 15–specific PCR tests,7,20 having different target genes (Supplemental Table 1, available online at http://vdi.sagepub.com/content/by/supplemental-data), were used in order to characterize the 9 field strains isolated on farm A. Amplified PCR products were analyzed by agarose gel electrophoresis, stained with ethidium bromide (10 µg/mL), and visualized under ultraviolet light.
A toxin-profiling PCR and a omlA PCR typing assay, which both facilitate rapid serotyping or serogrouping, were performed as described previously. 4 The O-PS PCR assays targeting genes encoding the Wzz protein and VI polysaccharide biosynthesis protein, which are involved in biosynthesis of lipopolysaccharide O-PS, were performed as described previously. 21
Nucleotide sequencing
PCR primers for amplification of the CPS biosynthesis (cps) loci (Fig. 1; Supplemental Table 1) and those for the sequencing of the PCR amplicons were designed from previously published sequences.5,8,22 A touchdown PCR with a commercial DNA polymerase b was used for amplification of the cps loci as recommended by the supplier of the polymerase. The PCR conditions were the same as described previously 5 except that the annealing/extension time was 6 min. The amplified PCR products were purified and sequenced as described previously. 6
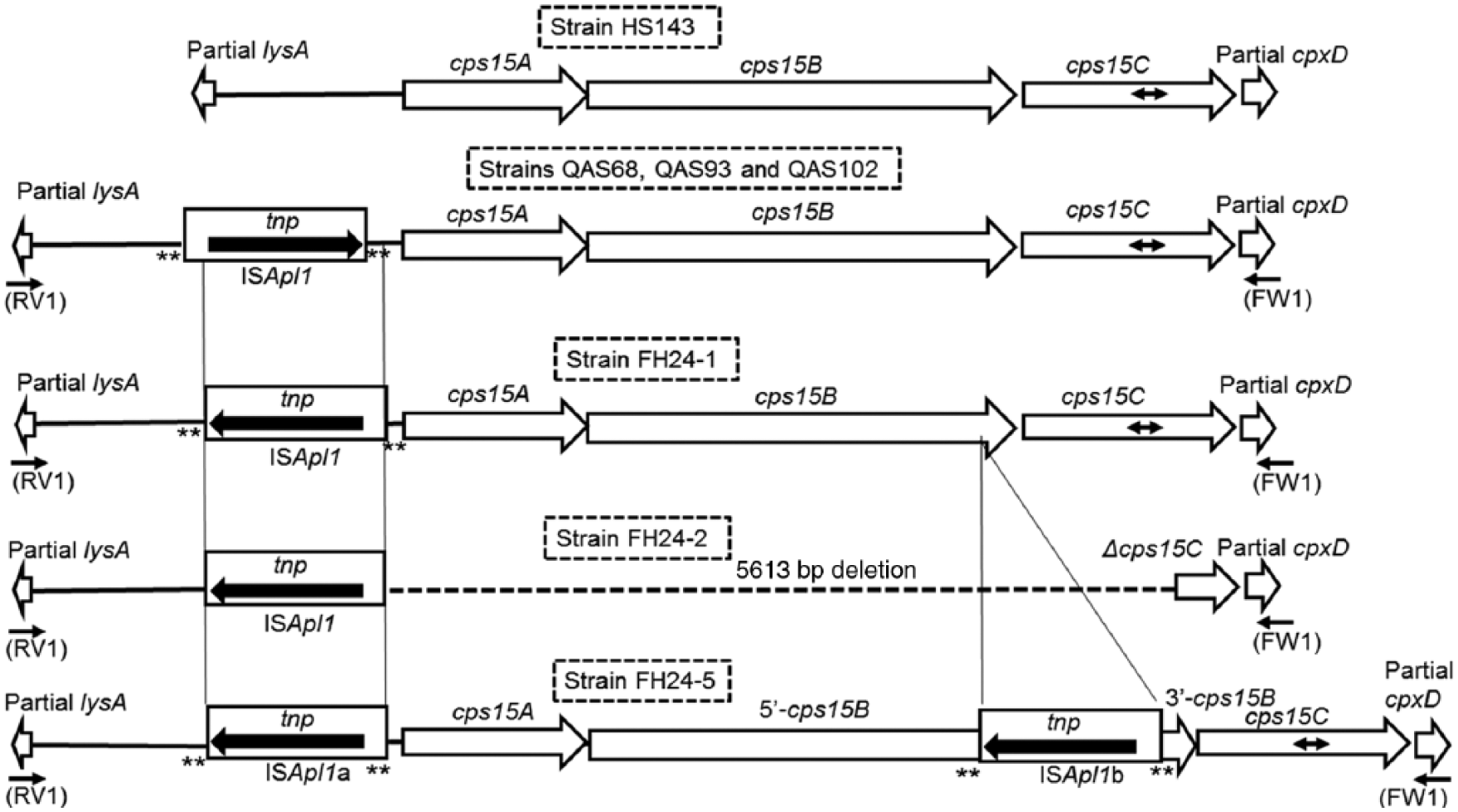
The genetic maps of Actinobacillus pleuropneumoniae serovar 15 reference and field strains as well as nontypeable strains. The inserted DNAs are indicated with solid line boxes (ISApl1) and large black arrows (transposase, tnp). The target site duplication of ISApl1 is shown with 2 asterisks flanked by ISApl1. Black arrows with 2 arrowheads indicate the target region of the serovar 15–specific polymerase chain reaction (PCR) test (cps15C PCR). The positions of PCR primer for amplification of the cps loci are shown below the genetic maps.
Homology searches of the DNA DataBank of Japan (DDBJ)/European Molecular Biology Laboratory (EMBL)/GenBank databases were performed using the BLAST server at the National Institute of Genetics, Japan. A commercially available genetic analysis software package c was also used for alignment of the nucleotide sequence. Nucleotide sequences of the cps loci of strains FH24-1, FH24-2, FH24-5, QAS68, QAS93, and QAS102 have been deposited in the DDBJ/EMBL/GenBank databases as shown in Table 1.
Pulsed-field gel electrophoresis
Using the restriction enzyme ApaI, d pulsed-field gel electrophoresis (PFGE) was performed as described previously. 14
Results
Serotyping
All strains except FH24-2 and FH24-5 (Table 1) were serotyped as serovar 15 by the ID test based on the observation of a serovar 15–specific precipitation line that seemed to be derived from the CPS (Fig. 2). However, FH24-2 and FH24-5 were nontypeable because no serovar 15–specific precipitation line was observed when using rabbit antisera prepared against the serovar 15 reference strain (Fig. 2) or when using those prepared against reference strains of serovars 1–14 (data not shown).

Immunodiffusion tests with antiserum prepared against Actinobacillus pleuropneumoniae serovar 15 reference strains. Antisera prepared against serovar 15 were placed in the center wells (labeled anti-15). Antigens of the field strains FH24-1 to FH24-9, isolated from farm A, were placed into the outer wells (labeled with the respective strain name). The antigen from the serovar 15 reference strain was placed in the outer well with the corresponding number (15).
Serovar 15–specific PCR tests
The results are shown in Supplemental Table 1. The 9 Japanese field strains isolated on farm A (Table 1) were positive by a serovar 15–specific PCR (cpxCD PCR; Supplemental Table 1) in which the target genes of the PCR primers were cpxC and cpxD. 20 The second serovar 15–specific PCR test, which targets the cps15C gene, 7 gave a positive reaction with 8 of 9 field isolates isolated on farm A (Table 1), with strain FH24-2 being negative (Supplemental Table 1).
Other PCR results
The PCR toxin gene assay for strains FH24-1, FH24-2, and FH24-5 was positive for apxIICA, apxIIICA, apxIBD, and apxIIIBD, but negative for apxICA, which is the profile seen in serovars 2, 4, 6, 8, and 15. 4 The omlA PCR type of strains FH24-1, FH24-2, and FH24-5 corresponded to that seen in serovars 3, 6–8, 13, and 15. 4 The PCR assays targeting genes for the Wzz protein and VI polysaccharide protein of strains FH24-1, FH24-2, and FH24-5 were positive, as previously seen in serovars 3, 6, and 15 21 and serovars 3, 8, and 15, 21 respectively.
We next attempted to amplify the cps loci with PCR primers RV1 and FW1 (Fig. 1; Supplemental Table 1) from the 2 nontypeable strains FH24-2 and FH24-5, serovar 15 strain FH24-1, and serovar 15 field strains QAS68, QAS93, and QAS102, respectively (Table 1). From the 4 serovar 15 field strains, a single PCR product with a size of 7.4 kb was amplified, which was larger than the size of the serovar 15 reference strain HS143 (6.4 kb 8 ; Supplemental Table 1). A single product (2.8 kb) and 2 PCR products (~8.4 kb and 4.0 kb) were amplified from the nontypeable strains FH24-2 and FH24-5, respectively (Supplemental Table 1).
Nucleotide sequencing
In order to reveal the molecular basis of the nontypeability of the 2 A. pleuropneumoniae strains and the size variations in capsule loci among the strains tested, the nucleotide sequence of the PCR products of Japanese serovar 15 strains FH24-1, QAS68, QAS93, and QAS102, as well as the nontypeable strain FH24-2, were determined (Table 1). The genetic map of the cps locus of these field strains as well as that of HS143, the serovar 15 reference strain, 8 is shown in Figure 1.
Three genes involved in CPS biosynthesis, cps15A, cps15B, and cps15C, were found in the Japanese serovar 15 field strains FH24-1, QAS68, QAS93, and QAS102 as well as the serovar 15 reference strain HS143 (Fig. 1). 8 An insertion sequence (IS), ISApl1, was found upstream of cps15A in the 4 Japanese serovar 15 field strains, but was not present in the Australian strain HS143 (Fig. 1). A previous study 18 has shown that insertion of ISApl1 results in a duplication at the target site of 2 bp, termed target site duplication (TSD). Sequences, which seem to be the TSD generated by insertion of ISApl1, were also found in serovar 15 field strains FH24-1, QAS68, QAS93, and QAS102 (Fig. 3). The location and orientation of the ISApl1 insertion of serovar 15 strain FH24-1 were distinct from those of the 3 other serovar 15 field strains QAS68, QAS93, and QAS102 (Fig. 1).
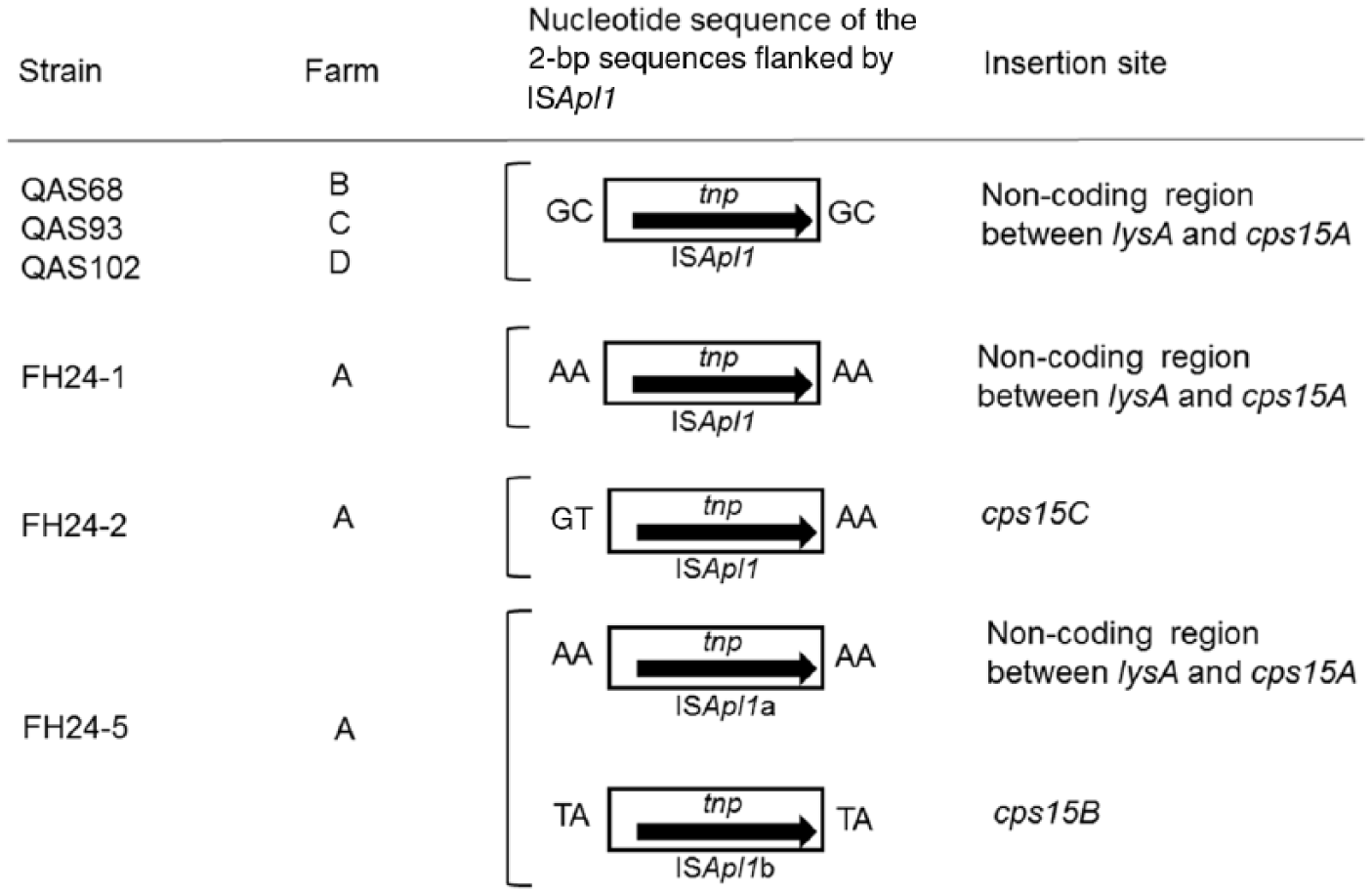
Nucleotide sequence of the 2-bp sequences flanked by ISApl1 in the serovar 15 field strains and nontypeable strains. The 2-bp duplicated sequences, the insertion site of target site duplication, was found from all strains except strain FH24-2.
Strain FH24-2 also contained ISApl1 (Fig. 1). The distance from ISApl1 to the start codon of lysA was identical between serovar 15 strain FH24-1 and nontypeable strain FH24-2 (Fig. 1). However, a 5,613-bp DNA region including the 5′-region of cps15C, which contains the sequences targeted by PCR primers in the serovar 15–specific PCR test, 7 as well as cps15AB was deleted in strain FH24-2 (Fig. 1). Each sequence of the 2 bp nucleotides flanked by ISApl1 was distinct in strain FH24-2 unlike in other strains used in a previous 18 and the present study (Fig. 3).
Strain FH24-5 also carried an intact ISApl1 (ISApl1a) in the same orientation and position as those of strains FH24-1 and FH24-2 (Fig. 1; Fig. 3). In addition, another copy of ISApl1 (ISApl1b) was identified within cps15B of the strain (Figs. 1, 3). However, the nucleotide sequence of the TSD flanked by ISApl1b (TA) was distinct from that flanked by ISApl1a (AA; Figs. 1, 3). Strain FH24-5 carried intact cps15A and cps15C as well as cps15B disrupted by ISApl1b.
Pulsed-field gel electrophoresis
The PFGE patterns of ApaI-digested genomic DNA are shown in Figure 4. The PFGE pattern of nontypeable strains FH24-2 and FH24-5 was very similar to that of serovar 15 strains QAS93 and FH24-1, with 2 or fewer band differences (Fig. 4).
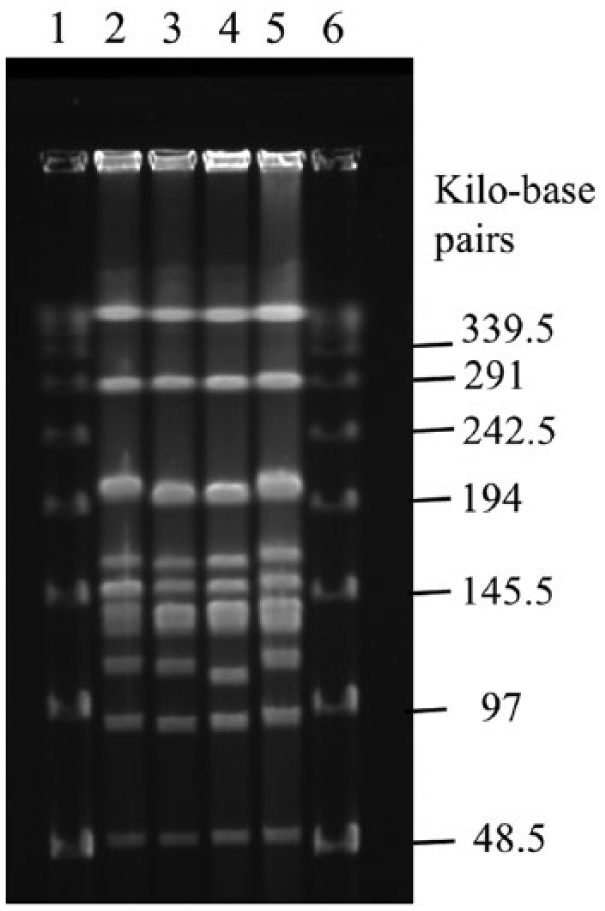
Pulsed-field gel electrophoresis. Lanes 1, 6: DNA size standards e ; lane 2: strain QAS93 (serovar 15); lane 3: strain FH24-1 (serovar 15); lane 4: strain FH24-2 (nontypeable); lane 5: strain FH24-5 (nontypeable).
Discussion
Our study revealed the molecular basis of the nontypeability of 2 strains isolated on a Japanese farm where serovar 15 strains have been isolated. The 2 nontypeable strains isolated on the farm could not be serotyped with ID tests (Fig. 2).
The nucleotide sequence analysis of the cps locus of strain FH24-2 revealed that the deletion of DNA segments in the cps locus was responsible for the nontypeability in strain FH24-2. Deletion of the DNA segment adjacent to the IS element is one of the frequently occurring genomic deletions caused by IS transposase in enterohemorrhagic Escherichia coli O157. 10 It is expected that deletion of the 5,613-bp DNA segment observed in A. pleuropneumoniae strain FH24-2 may also be a result of the IS-mediated genomic deletion. However, further studies would be needed to elucidate whether the ISApl1 was truly associated with deletion of the DNA segment.
The nucleotide sequence analysis of the cps locus of strain FH24-5 further revealed that strain FH24-5 was rendered nontypeable by the insertion of ISApl1b into the cps15B gene of the strain (Fig. 1). We concluded that the insertion of ISApl1b into the cps15B gene, as well as the deletion of the complete cps15AB and partial cps15C genes flanked by ISApl1, are responsible for the nontypeability of strains FH24-5 and FH24-2, respectively.
Interestingly, the results of the serologic typing method with ID tests were quite different from the results inferred from PCR (Supplemental Table 1). Such a discrepancy between phenotypic and genotypic results of isolates has been reported in Australian field strains. 20 However, our study further showed a discrepancy of results between 2 serovar 15–specific PCR tests, as the cpxCD PCR was positive in both strains FH24-2 and FH24-5, whereas cps15C PCR was positive only in strain FH24-5 (Supplemental Table 1). This finding, in conjunction with the results of the nucleotide sequence analysis of the 2 nontypeable strains, further elucidated why the serovar 15–specific PCR targeting cps15C was negative only in strain FH24-2. Thus, ISApl1 can interfere with a molecular typing method (PCR) as well as a serologic method such as an ID test.
Strain FH24-5 carried intact cps15A and cps15C, which are involved in biosynthesis of serovar 15 CPS, as well as cps15B disrupted by insertion of ISApl1 (Fig. 1), whereas strain FH24-2 carried partial sequence of cps15C, although cps15AB was not found in the strain (Fig. 1). In addition, the omlA PCR type and the apx toxin profile of the 2 nontypeable strains corresponded to those of serovars 6, 8, and 15. 4 Moreover, a positive reaction in both O-PS PCRs has only been reported for serovars 3 and 15. 21 Very similar PFGE patterns with 2 or fewer band differences were observed between the 2 nontypeable and serovar 15 strains (Fig. 4). The relatedness between the 2 nontypeable strains and serovar 15 strains can be categorized as “closely related” as described previouly. 19 From these genetic characteristics of the 2 nontypeable strains, in conjunction with the fact that only the serovar 15 strains and the 2 nontypeable strains have been isolated on farm A (Table 1), it is likely that the 2 nontypeable strains were originally serovar 15 strains that lost their serovar 15–specific antigenic determinants. However, further characterizations would be necessary to prove this hypothesis and to demonstrate whether the nontypeable strains originated from serovar 15 strains on the same farm.
To date, ISApl1 has been identified only in serovars 2, 3, 6–8, 12, and 13.11,12,16,18 The TSD nucleotide sequences, which were probably generated by insertion of ISApl1, differed between the 2 insertion types (i.e., the sequence was GC in strains QAS68, QAS93. and QAS102, and AA in strain FH24-1; Fig. 3). This variation in TSD suggested that ISApl1 was independently inserted in strains QAS68, QAS93, and QAS102, or strain FH24-1.
A previous study identified 4 kinds of TSD sequences (TG, TT, GG, and AA) for ISApl1, 18 which were distinct from the consensus sequence (RY) published for IS30 of E. coli. 18 Our study identified 2 new TSD sequences (GC and TA), as well as the previously identified TSD sequence (AA; Fig. 3), revealing a greater variety of target sites for ISApl1.
Our study revealed that all of the 4 serovar 15 strains in Japan, as shown in Figure 1, carried ISApl1. However, the difference of location and orientation against cps15A genes of the ISApl1 insertion between serovar 15 strain FH24-1 and the 3 other serovar 15 field strains QAS68, QAS93, and QAS102 (Fig. 1) indicates the presence of at least 2 genetically distinct groups in Japanese serovar 15 field strains. The location, orientation, and number of ISApl1 insertions into the cps loci may be useful for the epidemiologic study of serovar 15. Although A. pleuropneumoniae serovar 15 was originally isolated in Australia 1 more than 10 years ago, the serovar has now been recognized in North and South America as well as in Japan.3,9 It would be of interest to compare the presence or absence, along with the location, orientation, and number, of ISApl1 insertions among strains isolated on different continents (Fig. 1).
In conclusion, we have shown that the insertion of ISApl1 into the capsule locus was responsible for the nontypeability of the organisms investigated. A nucleotide sequence analysis of the cps loci would be the best means of determining the cause of nontypeability of A. pleuropneumoniae isolates.
Footnotes
Acknowledgements
We thank Drs. LO Andresen, PJ Blackall, and S Nagai for providing bacterial strains. The technical assistance of Ms. T Ishiga for PFGE analysis is acknowledged.
Authors’ contributions
H Ito designed the study, performed serotyping and PCR, determined the nucleotide sequence, performed the nucleotide sequence data analysis, and drafted the manuscript. T Ogawa, D Fukamizu, and Y Morinaga continuously isolated and identified A. pleuropneumoniae from farm A. M Kusumoto performed PFGE analysis, assisted in the writing of discussion passages related to IS-mediated deletion, and helped in revising the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
QuickExtract DNA extraction solution, Epicentre Biotechnologies, Madison, WI.
b.
KOD-FX Neo, Toyobo, Osaka, Japan.
c.
MegAlign and SeqMan Pro applications included in the Lazergene 12 Core Suite, DNASTAR Inc., Madison, WI.
d.
Takara Bio Inc., Otsu, Shiga, Japan.
e.
Lambda ladder, Bio-Rad Laboratories, Hercules, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
