Abstract
Organisms previously classified as Streptococcus bovis (i.e., the S. bovis/S. equinus complex) are common in cattle feces, but may also act as opportunistic pathogens. In the current work, Streptococcus infantarius subsp. coli, a member of this complex, was associated of a cluster of calves that died within hours of injection with a modified live viral vaccine. Within 12 h of vaccination of 46 calves at a cow/calf operation, 4 calves had died, 3 calves were ill, and 1 unvaccinated cow was dead. Autopsies were performed on the cow, 2 dead calves, and 1 affected surviving calf, which was euthanized ~24 h after vaccine administration. The animals had similar gross anatomic and microscopic lesions, including subcutaneous and intramuscular dark hemorrhage on the caudal neck, multiorgan ecchymosis and petechiation, and alveolitis to interstitial pneumonia. Gram-positive cocci were in the vasculature of the lung and skeletal muscle, and S. infantarius subsp. coli was cultured from tissues and from the vaccines used on affected animals, but not in vials used on unaffected animals. Together, these findings suggest death caused by streptococcal septicemia and toxemia as a result of contamination.
Vaccination of food animals plays a critical role in both animal and public health; therefore, it is important that vaccines and their use are safe and effective. Surveillance of adverse vaccine reactions in the United States is based on voluntary reporting to the manufacturer or the U.S. Department of Agriculture Center for Veterinary Biologics (Ames, Iowa). This likely facilitates underreporting, 15 which can be supplemented by published reports.
There are few published scientific reports of systemic adverse reactions following vaccination in food animals.3,7,18 Most reports are anecdotal and do not evaluate the underlying cause of the reactions, which can be difficult to determine. Very few cases document vaccine reactions in cattle, and even fewer highlight cases in calves. Previous studies have reported systemic vaccine reactions in calves after in-advertent intravenous vaccination against respiratory and clostridial diseases 18 and following administration of a combination vaccine. 7
Adverse vaccine reactions in veterinary patients are often local reactions, which include edema, erythema, swelling, and urticaria at the injection site. 4 Signs of systemic vaccine reactions are most often attributed to either anaphylaxis or sepsis and typically include fever, lethargy, anorexia, edema, urticaria, vomiting, diarrhea, dyspnea, excitement, collapse, convulsions, and death. 4 Systemic anaphylaxis due to vaccination is uncommon in veterinary patients. 4 It is estimated that anaphylaxis after vaccine and antibiotic injection occurs with a frequency of <1 event per 10,000 animals. 18 Anaphylaxis secondary to vaccination has been observed in cattle vaccinated against foot-and-mouth disease 3 and in cattle due to parenteral administration of penicillin–streptomycin. 16 Anaphylactic reactions are type I hypersensitivity reactions that initiate within minutes of exposure to a presensitized antigen. 18 Clinical signs of anaphylaxis are species dependent, 17 with the lung being the major anaphylactic shock organ in cattle. 9 Anaphylaxis in cattle is characterized by rapid onset of respiratory distress, 16 marked systemic hypotension, and hypertension in the pulmonary arteries and abdominal vena cava. 9 Calves that underwent experimental induction of anaphylaxis collapsed within minutes and then either recovered or died within an hour with pulmonary edema and emphysema. 13
Another likely cause of an aberrant, systemic reaction associated with vaccination is septicemia due to either gram-negative and/or gram-positive bacteria. 26 Gram-negative bacteria contain lipopolysaccharide, which can cause endotoxemia and cytokine-mediated injury. 5 Calves administered a combination vaccine containing endotoxins showed clinical signs 8–10 h after vaccination. 5 Gram-positive bacteria contain a number of species- and strain-specific immunogenic cell wall components and often exotoxins. Exotoxins may act as bacterial superantigens, which can induce toxic shock syndrome and cause multiple organ failure. 24 Features of sepsis include fever, endothelial damage, disseminated intravascular coagulation, tachypnea, hypovolemia, and death. 2 Pathogenicity from aberrant injection of bacteria depends on dose, route of entry, age, genetics, immune status, and immunological response to bacterial structures. 5
The current report documents septicemia in calves that was associated with group D streptococcal (Streptococcus bovis/Streptococcus equinus complex) contamination. S. bovis/S. equinus complex organisms are gram-positive coccoid bacteria, a common component of intestinal and fecal flora of cattle,6,10,20 and can be opportunistic pathogens contributing to ruminal acidosis and mastitis.1,20 The classification of S. bovis and related organisms has undergone several changes since their original identification. Although S. bovis, S. equinus, and other species categories were originally used, these related organisms have been divided into biotypes 11 and DNA clusters. 22 For this report, we will refer to these related organisms as the S. bovis/S. equinus complex. The cases described here present reported cases of vaccine reaction associated with S. bovis/S. equinus complex vaccine contamination and demonstrate a peracute disease progression, which is considered atypical for bacterial sepsis. 21
In early October 2014, a cow/calf operation in Georgia vaccinated 46 Aberdeen–Angus calves with a modified live vaccine (protecting against Bovine herpesvirus 1, Bovine parainfluenza virus 3, Bovine respiratory syncytial virus, and Mannheimia haemolytica) on the left neck and a 7-way blackleg (without Histophilus somni) vaccine on the right neck. Cows were run through a separate chute and were checked for pregnancy and received a topical deworming agent. Several hours later that same day, the producer discovered 4 dead calves, 3 down calves, and 1 down cow. The body temperature of down calves was 42–43°C. Down calves had signs of respiratory distress and hematochezia. Seven hours postvaccination, 2 additional calves were dead, and 2 were in critical condition. The down cow had died.
Two of the dead calves (calves A and C) and the deceased cow were submitted for postmortem examination at the College of Veterinary Medicine at the University of Georgia (Athens, Georgia). The college also received one of the surviving calves that originally presented with a fever of 42°C and rectal bleeding (calf B). This animal was euthanized (~24 h after vaccination) and immediately submitted for autopsy. The calves were 6–9 months old, and the cow was a young adult. All animal were in good body condition.
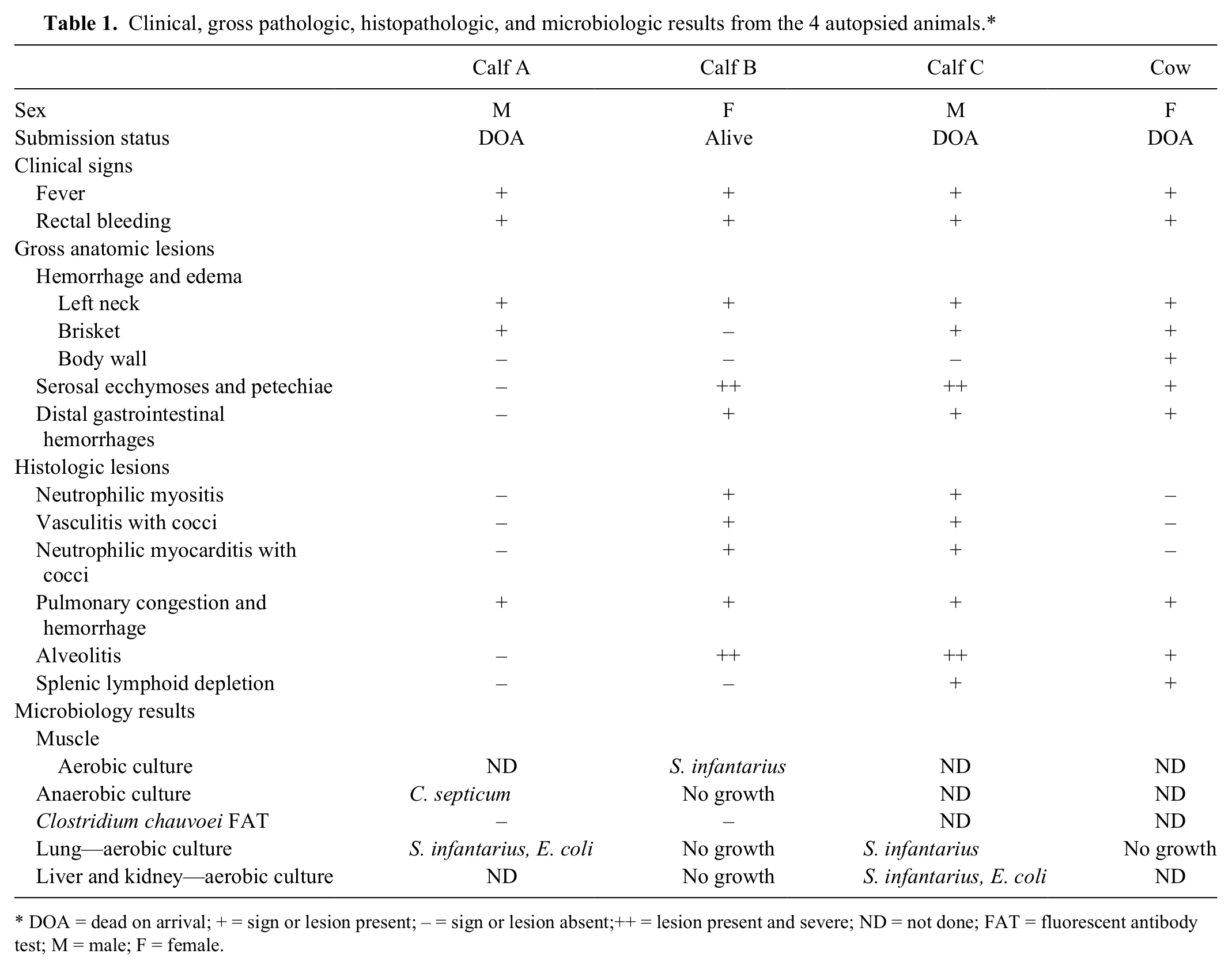
All 3 calves and the cow showed similar gross anatomic lesions. Subcutaneous and intramuscular hemorrhage and edema expanded the left neck of all animals, and, in calves A and C, hemorrhage and edema extended ventrocaudally into the brisket area. In the cow, the left thoracic body wall was also edematous and hemorrhagic. In calf B, the lesions were confined to a well-demarcated 2 cm × 3 cm area in the left caudal neck/craniodorsal shoulder. In all animals, there were ecchymoses and petechiae on numerous serosal and mucosal surfaces, including the epicardium, endocardium, omentum, mesentery, visceral pleura, renal pelvises; serosal surfaces of the ureter, esophagus, small intestine, cecum; and mucosal surfaces of the trachea, nasopharynx, rumen, abomasum, small intestinal, cecum, gallbladder, and urinary bladder. These lesions were most evident in calves B and C. The cecal and spiral colon lumens of calf C were filled with melena and clotted blood. In the cow, there was severe intraluminal hemorrhage within the rectum, colon, and cecum.
Multiple tissues were processed for routine histology. Skeletal muscle from the reported site of the modified-live vaccine injection was disrupted by neutrophilic myositis and vasculitis with hemorrhage and intravascular gram-positive diplococci (Fig. 1; Table 1). Autolysis in calf A and the cow precluded detection of skeletal muscle histologic changes. Cardiac lesions included varying degrees of acute hemorrhage, intramuscular edema, coccoid bacteria, and mild multifocal neutrophilic myocarditis. There was pulmonary congestion and hemorrhage in lung tissue from calf A. In calves B and C, there were scattered foci of alveoli containing small aggregates of neutrophils mixed with fibrin. Associated pneumocytes were sloughed or hypereosinophilic; the interalveolar walls were focally effaced by fibrin. In rare vessels, the endothelium of pulmonary vessels was denuded with exposure of the basement membrane (Fig. 2) and occasional areas of small fibrin aggregates. Gram-positive diplococci were in several pulmonary vessels. In less affected areas, neutrophils expanded the intra-alveolar septa. Lung from the cow showed a mild fibrinous pneumonitis. A Gram stain of the lungs of the cow was negative for gram-positive diplococci. In the cow and calf C, splenic lymphoid nodules were depleted, and there were perifollicular neutrophils and interfollicular hemosiderophages.
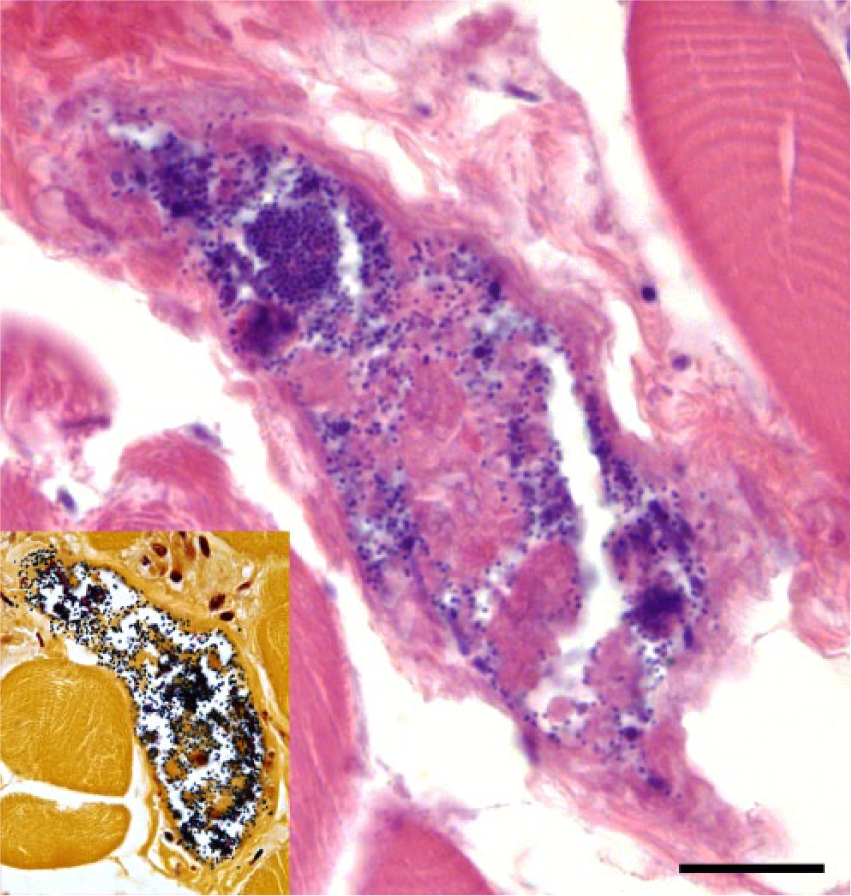
Skeletal muscle from calf B with intravascular gram-positive diplococci. 1,000×. Hematoxylin and eosin. Bar = 20 μm. Inset: Gram stain.
Clinical, gross pathologic, histopathologic, and microbiologic results from the 4 autopsied animals.*
DOA = dead on arrival; + = sign or lesion present; – = sign or lesion absent;++ = lesion present and severe; ND = not done; FAT = fluorescent antibody test; M = male; F = female.
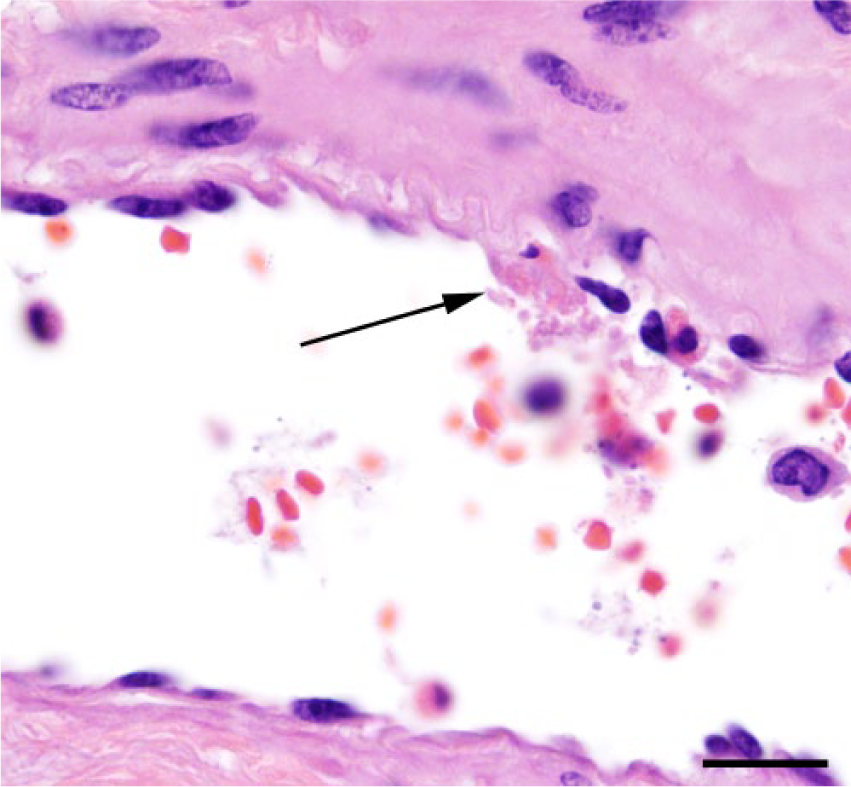
Pulmonary artery from calf C with denuded wall (arrow) and early neutrophilic vasculitis. 1,000×. Hematoxylin and eosin. Bar = 20 μm.
Tissue samples from liver, kidney, lung, and muscle collected during autopsy were cultured for aerobic and anaerobic bacteria. At least 1 tissue from all 3 calves resulted in growth of alpha-hemolytic, gram-positive, catalase-negative cocci identified as Lancefield group D streptococcal organisms, with a similarity index of 0.826, as determined by a commercial system. a These results are consistent with S. bovis/S. equinus complex (Table 1). Following biochemical assays, isolates were subjected to matrix-assisted laser desorption ionization–time of flight mass spectrometry (MALDI-TOF MS) b to validate this identification. Results ranged from 100% confidence for S. bovis/S. equinus complex DNA cluster III (50% S. infantarius subsp. coli, 50% S. infantarius subsp. infantarius) to 75% confidence for S. bovis/S. equinus complex DNA cluster III (50% S. infantarius subsp. coli, 25% S. infantarius subsp. infantarius) and 25% confidence for DNA cluster II (S. gallolyticus). 22 There was inconsistent growth of other bacteria (e.g., Escherichia coli), which were considered clinically insignificant based on the lack of concordance between animals and the histopathology. There was no bacterial growth after aerobic culture of lung tissue from the cow, which was not injected with the modified-live product that day. Anaerobic cultures of skeletal muscle from 2 calves only resulted in moderate growth of Clostridium septicum from 1 animal (calf A; Table 1). Skeletal muscle from the same 2 animals was tested for Clostridium chauvoei by fluorescent antibody staining, and both were negative (Table 1).
Portions of remaining vaccine material were saved by the referring veterinarian pending autopsy results. Approximately 7 days after the death of these animals, residual vaccine material from the bottle of respiratory vaccine used for the affected calves (reportedly injected on the left side of the neck) and residual vaccine from 3 bottles used on unaffected calves were cultured along with the clostridial vaccine used in all animals. One sample from the bottle of respiratory vaccine used in affected calves grew alpha-hemolytic, Lancefield group D streptococcal organisms, whereas the second sample from the same bottle yielded very light growth (single colony) of Corynebacterium spp. and similar very light growth (single colony) of unidentifiable gram-positive cocci under aerobic conditions. The group D streptococcal isolate was subjected to MALDI-TOF MS with identical results to that of the tissue-based isolate (highest confidence for DNA cluster III of the S. bovis/S. equinus complex). No significant bacteria were isolated from 3 bottles (representing 2 different lots) used in unaffected animals or from the clostridial vaccine, which was reportedly given in the right side of the neck in affected animals. To further characterize the 2 S. bovis/S. equinus complex isolates, partial 16S ribosomal RNA (rRNA) sequencing was performed as previously described.19,22 The partial 16S rRNA for both isolates was 99% similar to S. infantarius subsp. coli JB1 (GenBank accession AF104109), which is consistent with the biochemical and MALDI-based categorizations.
Growth of S. infantarius subsp. coli from the vaccine used in affected animals, from the lung of calves A and C, from the liver and kidney pool of calf A, and from the skeletal muscle (injection site) of calf B, implicates S. infantarius subsp. coli in the death of the calves. Further supporting the role of S. infantarius subsp. coli in these deaths is the presence of cocci in the lungs of 2 of the affected calves. Death of the calves was most likely due to streptococcal septicemia stemming from contamination of the multidose vaccine vial. Most changes described in calves were indicative of a septic process and correlated with the gross anatomic lesions of widespread hemorrhages and the alveolitis to interstitial pneumonia observed in calves B and C. The lack of histologic changes in calf A is likely due to a more peracute clinical presentation prior to death.
Although the similarities in clinical presentation and gross anatomic lesions for the cow were suggestive of an etiology similar to that observed in the calves, the cow was reportedly not vaccinated. Furthermore, the cow lacked significant histologic changes associated with sepsis, identifiable cocci in tissues, and no bacteria were isolated from the lungs. Thus, it is possible the cow died from other, currently unknown, causes.
Changes in calf B were not as severe as those in some of the other submitted animals. This may explain why it was still alive on presentation. The lack of readily apparent cocci in this calf suggests a lower infectious dose or more successful clearance of the bacteria.
Clostridium septicum was isolated from the injection site muscle of calf A, but this is most likely due to postmortem overgrowth. Evidence supporting this notion includes the time period before the postmortem of calf A (estimated 20 h between time of death and autopsy) and the lack of C. septicum isolation from the fresh calf (calf B).
Streptococcus bovis/S. equinus complex is a part of normal cattle gastrointestinal flora. The ruminal population of S. bovis/S. equinus complex in cattle remains low under normal feeding (e.g., roughage diet), but increases with introduction of concentrate or high-quality forage.10,14 The bacterium is a main lactic acid–producing bacterium in cattle. 10 Increased levels of S. bovis/S. equinus complex are associated with ruminal acidosis and subsequent rumen dysfunction. 20 In humans, S. bovis/S. equinus complex is present in the intestinal flora of 10% of the healthy population and up to 55% of patients with inflammatory bowel diseases or colon cancer. 12 Streptococcus bovis/S. equinus complex sepsis in people is associated with malignant gastrointestinal diseases 23 and endocarditis. 25
How the contaminating bacteria got into the vaccines is beyond the scope of this investigation and would require an in-depth look at vaccination practices at the facility and quality control of the vaccine itself. The most likely scenario is user contamination of a multidose vial. Further investigation into the history did result in identifying multiple needle entries into the vial, and possibly overnight storage of a partially used vial.
Although S. infantarius subsp. coli is highly suspected as the cause of the deaths in this case, it is recognized that the pathology in these animals is relatively mild and not pathognomonic. For instance, the reason for the rapid clinical progression is unknown, especially considering the relatively light growth of organisms from the residual vaccine vials. Anaphylaxis was initially considered on presentation; however, it is considered unlikely for 2 reasons: type 1 hypersensitivity reactions usually commence within minutes after administration of an allergen, and it would be unusual for multiple animals to be presensitized to the same antigens.8,9,16
Another possibility is that the calves described in this report experienced a type of streptococcal toxic shock syndrome (TSS), which has been described in human medical literature. 24 In humans, streptococcal TSS is caused by group A Streptococcus spp., which produce pyrogenic exotoxins A and/or B. 24 These exotoxins then elicit a peracute to acute widespread inflammatory response and ultimately shock and death. 26 These exotoxins are not reported to be associated with organisms from the S. bovis/S. equinus complex. Additional research into the pathogenesis of S. bovis/S. equinus complex organisms may be warranted, particularly if these bacteria are implicated in future incidents of vaccine contamination and adverse reactions. It is important to explore the potential synergistic interaction of bacteria with vaccine components, including adjuvants.
The route of administration may be another contributing factor to the disease presentation. Inadvertent intravascular administration of a modified-live vaccine has been previously implicated in a series of calf deaths. 18 Although intravascular injection could not be confirmed in this case, it is possible that any small needle puncture was not visible grossly. Intravascular injection may explain the excessive hemorrhage at the site of vaccination, and may help explain the systemic response to the contaminated vaccine, instead of development of the more common postvaccination abscess.
These cases highlight the need for proper vaccination protocols to ensure proper safety and efficacy of vaccinations. Cases of adverse vaccine reactions should be investigated for evidence of anaphylaxis, improper route of administration (e.g., intravenous), and evidence of abnormal vaccine contents (e.g., bacterial contamination). Producers, veterinarians, and vaccine manufacturers should monitor and report adverse vaccine reactions so that factors underlying adverse reactions can be addressed, as well as maintaining on-farm hygiene during vaccination procedures.
Footnotes
Acknowledgements
We thank the histology and microbiology laboratory staff at the Athens Veterinary Diagnostic Laboratory for their contributions to this case.
Authors’ contributions
LL Clarke and JB Stanton contributed to conception and design of the study, and contributed to acquisition, analysis, and interpretation of data. RL Fathke contributed to conception of the study and drafted the manuscript. S Sanchez contributed to analysis and interpretation of data. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Biolog GEN III microbial identification system, Biolog Inc., Hayward, CA.
b.
VITEK-MS by bioMérieux Inc. and SARAMIS database software, Durham, NC.
Declaration of conflicting interests
The author(s) declared that they have no conflict of interest with respect to their research, authorship, and/or the publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
