Abstract
Ten cattle and 10 buffalo were divided into 2 groups (control [n = 8] and experimental [n = 12]) that received daily administration of copper. Three hepatic biopsies and blood samples were performed on days 0, 45, and 105. The concentration of hepatic copper was determined by spectrophotometric atomic absorption, and the activities of aspartate aminotransferase (AST) and gamma-glutamyl transferase (GGT) were analyzed. Regression analyses were done to verify the possible existing relationship between enzymatic activity and concentration of hepatic copper. Sensitivity, specificity, accuracy, and positive and negative predictive values were determined. The serum activities of AST and GGT had coefficients of determination that were excellent predictive indicators of hepatic copper accumulation in cattle, while only GGT serum activity was predictive of hepatic copper accumulation in buffalo. Elevated serum GGT activity may be indicative of increased concentrations of hepatic copper even in cattle and buffalo that appear to be clinically healthy. Thus, prophylactic measures can be implemented to prevent the onset of a hemolytic crisis that is characteristic of copper intoxication.
Copper (Cu) toxicosis is considered an important disease associated with the death of sheep in Brazil, resulting in great economic losses to the sheep industry. 11 Although the frequency of this intoxication is lower in other species of ruminants, Cu intoxication in cattle has recently evolved as an emerging disease, with descriptions of cases of acute and chronic intoxication in several countries, including Brazil. 2–4,6,12,16 Recent research 1 has shown that there is an increase in the number of cases in cattle and has described 14 outbreaks within 6 months. However, in buffalo, there is only 1 description of this intoxication, during which young animals that received reconstituted Cu-enriched powdered milk for a few months demonstrated signs indicative of chronic Cu poisoning and died suddenly. 18
In cases of chronic Cu poisoning, clinical manifestations are only observed during the hemolytic crisis, or a few days before, so it is very difficult to establish an early diagnosis of this disease based only on clinical signs. 6,11 Consequently, several researchers have tried to establish parameters of clinical diagnosis based on laboratory analyses. 9,11,13 Serum Cu and other traditional biochemical variables have limited diagnostic value in terms of Cu accumulation, 9,13 and the best results were observed in sheep in association with alterations of hepatic enzymes. 7
Some researchers 13 have concluded that the most reliable factor in diagnosing early accumulation of Cu is determination of the serum activities of aspartate aminotransferase (AST) and gamma-glutamyl transferase (GGT), not the determination of serum Cu concentration, acid phosphatase, and sorbitol dehydrogenase serum activities. Consequently, it is necessary to determine the usefulness of measuring hepatic enzyme activities in cases of Cu accumulation, in which animals are close to an intoxication episode. Therefore, the current study evaluated the alterations in the serum activities of 2 hepatic enzymes to determine if these values can be used as early reliable diagnostic indicators of Cu poisoning in cattle and buffalo.
The present study was carried out in accordance with appropriate ethical standards of animal care, as approved by the Bioethics Committee of the College of Veterinary Medicine, University of São Paulo (São Paulo, Brazil; protocol 746/2005). Twenty male animals were used in the current study: 10 mixed-breed cattle and 10 Murrah breed of buffalo. All animals were 8 months old, approximately 170 kg in body weight, and were divided into 2 groups. The treatment group (n = 12; 6 cattle and 6 buffalo) received daily increasing doses of Cu via ruminal fistula. The initial dose was 2 mg/kg body weight (BW) of Cu (in an aqueous solution) administered during the first week; this was increased by 2 mg weekly until the 14th week, when the final dose was 28 mg/kg BW. The control group (n = 8; 4 cattle and 4 buffalo) received only normal rations and water by ruminal fistula. Three serial hepatic biopsies were obtained in all animals to determine the concentration of Cu on the following days: day 0 (baseline), day 45 after administration of Cu, and day 105 (at the end of the experiment).
To determine the serum activities of AST and GGT, blood was obtained from the jugular vein using vacuum collecting tubes a few hours before the hepatic biopsies. Serum activities of AST, GGT, and creatine kinase (CK) were determined using a biochemical automatic analyzer a with a commercial kit. b Enzymatic activity was determined at 30°C, as recommended. 8
A total of 60 correlation points (30 for each species) between hepatic Cu concentration and serum enzyme activities were obtained. Individual regression analyses and respective coefficients of determination were done for each species to verify the relationship between the variables that were being evaluated. The data were analyzed for linear regression by the F-test. 14 Sensitivity, specificity, accuracy, and positive and negative predictive values were calculated to obtain the best discrimination value to determine the relationship between hepatic enzymatic activity and Cu accumulation. 10,15
The correlations between the GGT serum activities and hepatic Cu concentrations observed in cattle and buffalo are illustrated in Figures 1 and 2, respectively. For cattle, a potential regression found that the coefficient of determination (R 2 ) = 0.5838, while for buffalo, the exponential regression was R 2 = 0.5786.
Considering 1,000 ppm to be indicative of excessive hepatic accumulation of Cu in both cattle and buffalo, the best discrimination value for the serum GGT activity in both species was sought and used as a diagnostic indicator. Consequently, the positive predictive values and negative predictive values were used. The discrimination values indicative of excessive Cu accumulation within the liver and the elevation of serum GGT activity for cattle and buffalo are shown in Figures 3 and 4, respectively. For cattle, these positive and negative predictive values were highest (100%) when the activity of GGT was 24.6 U/l. Since no values were observed in either the first quadrant (false-positive result) or fourth quadrant (false-negative result), the sensitivity, specificity, and accuracy were 100%. For buffalo, the discrimination value for GGT serum activity was 20 U/l, which was similar to that observed in cattle.
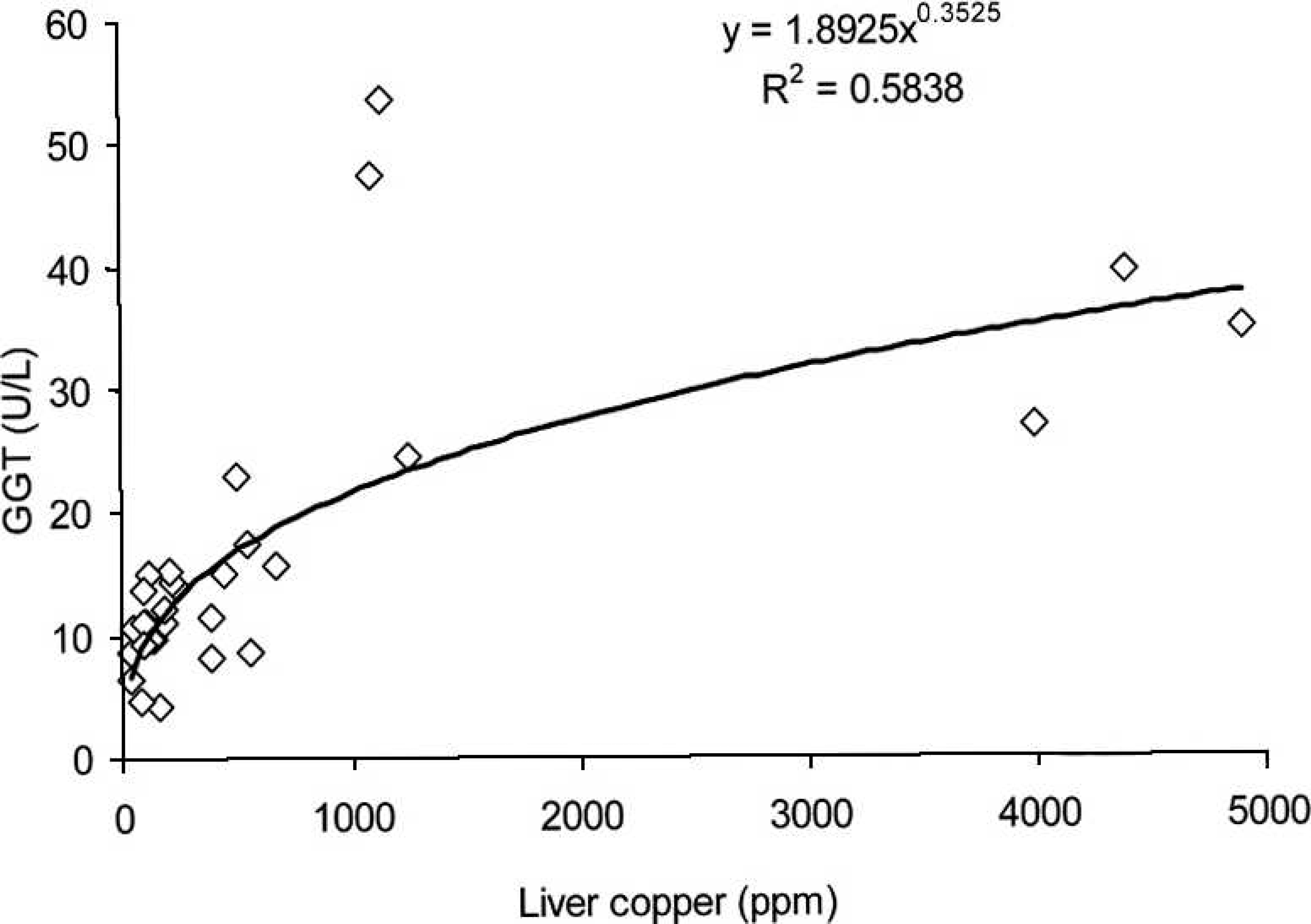
Relationship between serum gamma-glutamyl transferase (GGT) and hepatic concentration of copper (Cu) in cattle from Cu-treated and control groups during the experiment.
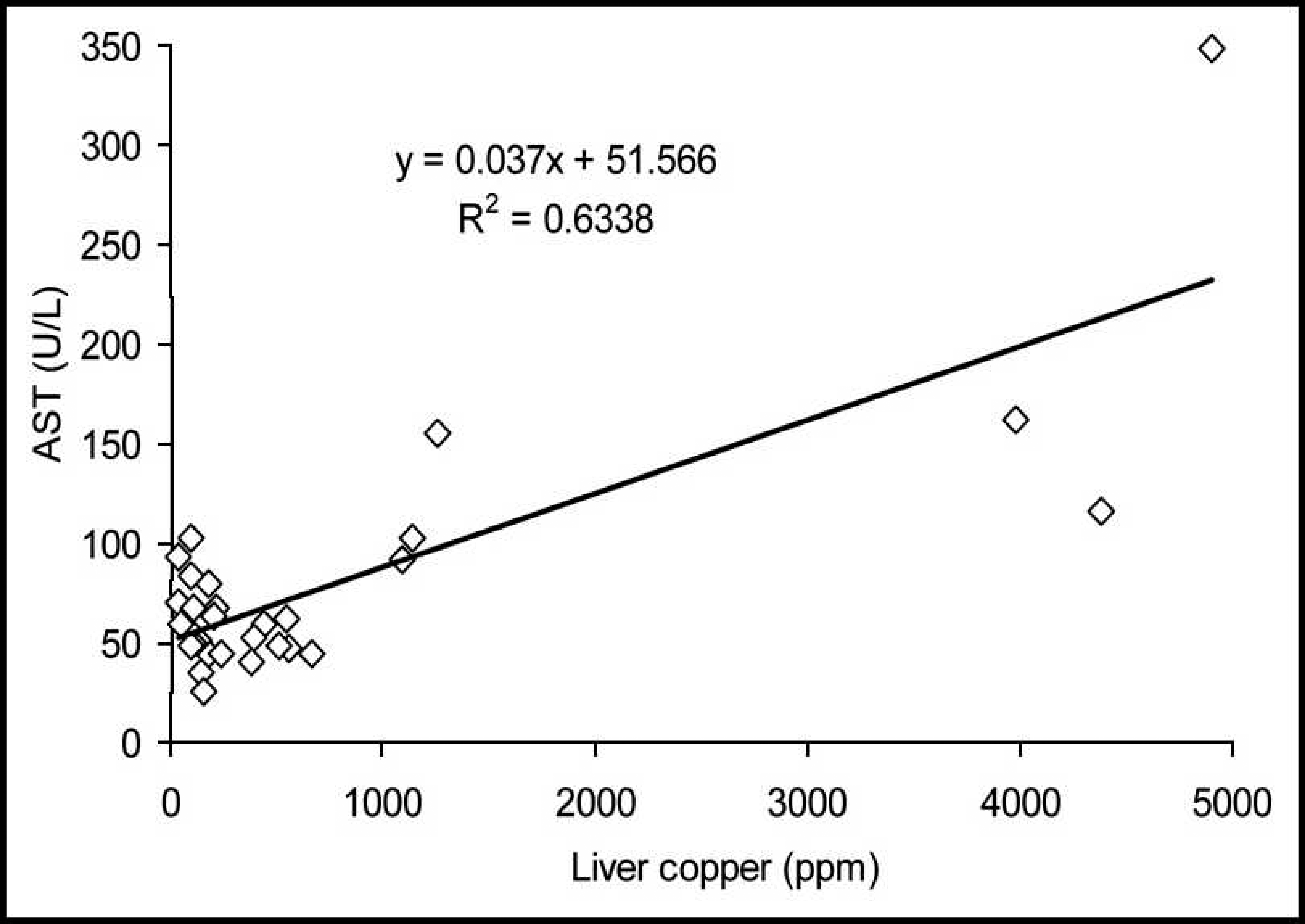
The correlations between the serum activities of AST and the hepatic accumulation of Cu in cattle and buffalo are demonstrated in Figures 5 and 6, respectively. In cattle, an elevated coefficient of determination (R 2 = 0.6338) and a first-degree regression equation were observed. In buffalo, the coefficient of determination was poor (R 2 = 0.2625), and the regression equation was linear.
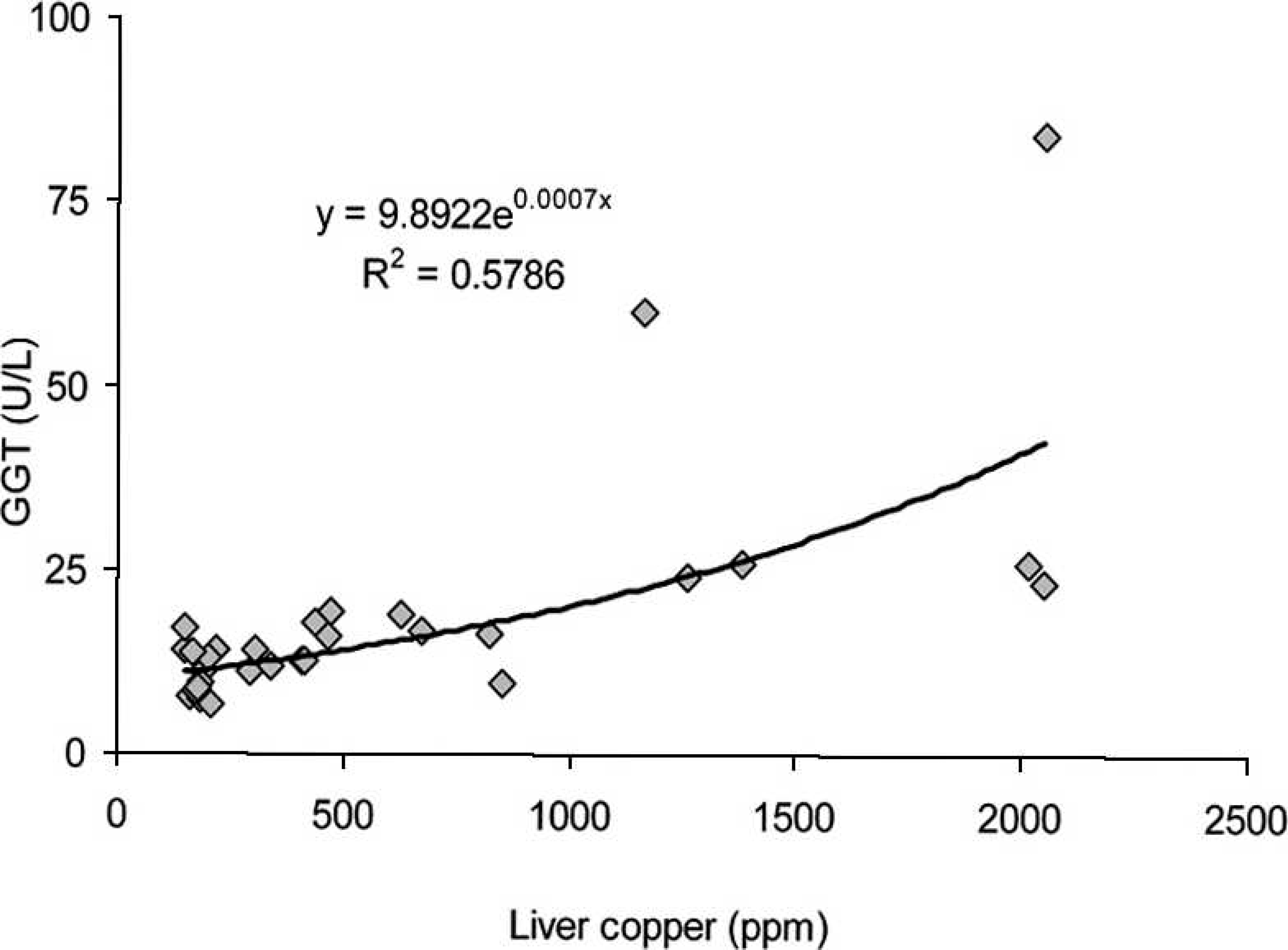
Relationship between serum gamma-glutamyl transferase (GGT) and hepatic concentration of copper (Cu) in buffalo from Cu-treated and control groups during the experiment.
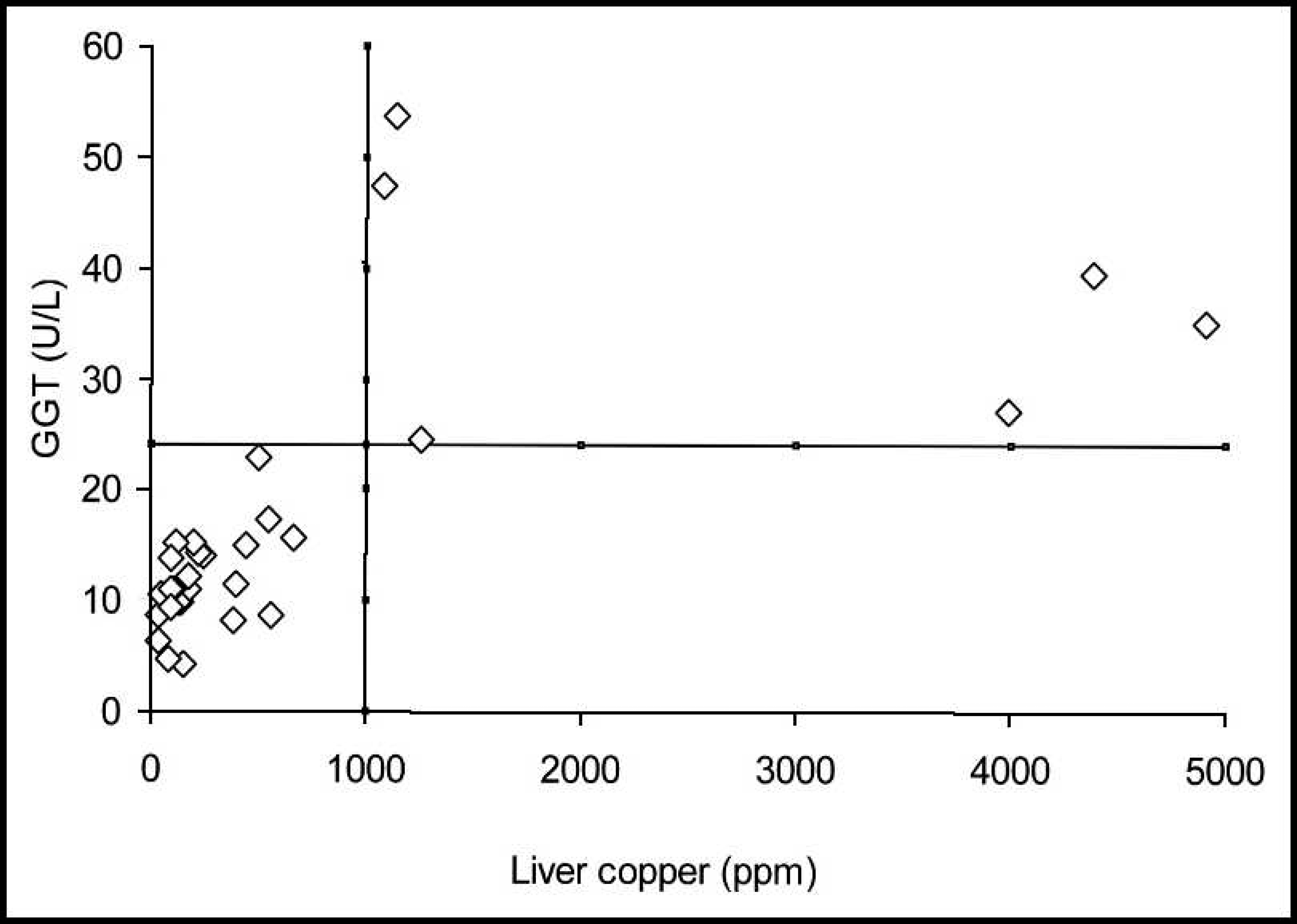
Cut-off line for the accumulation of liver copper and the elevation of serum gamma-glutamyl transferase (GGT) in cattle.
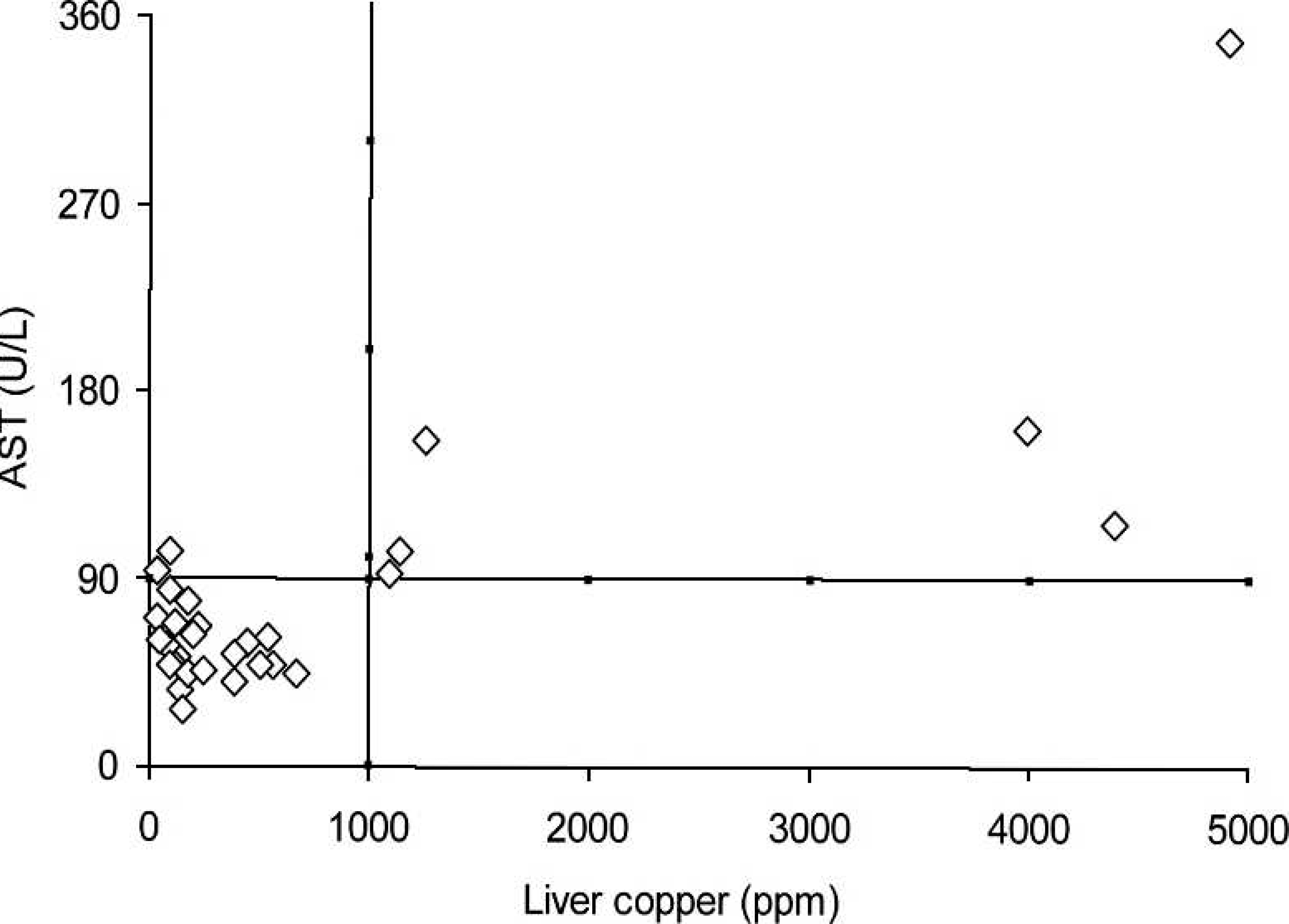
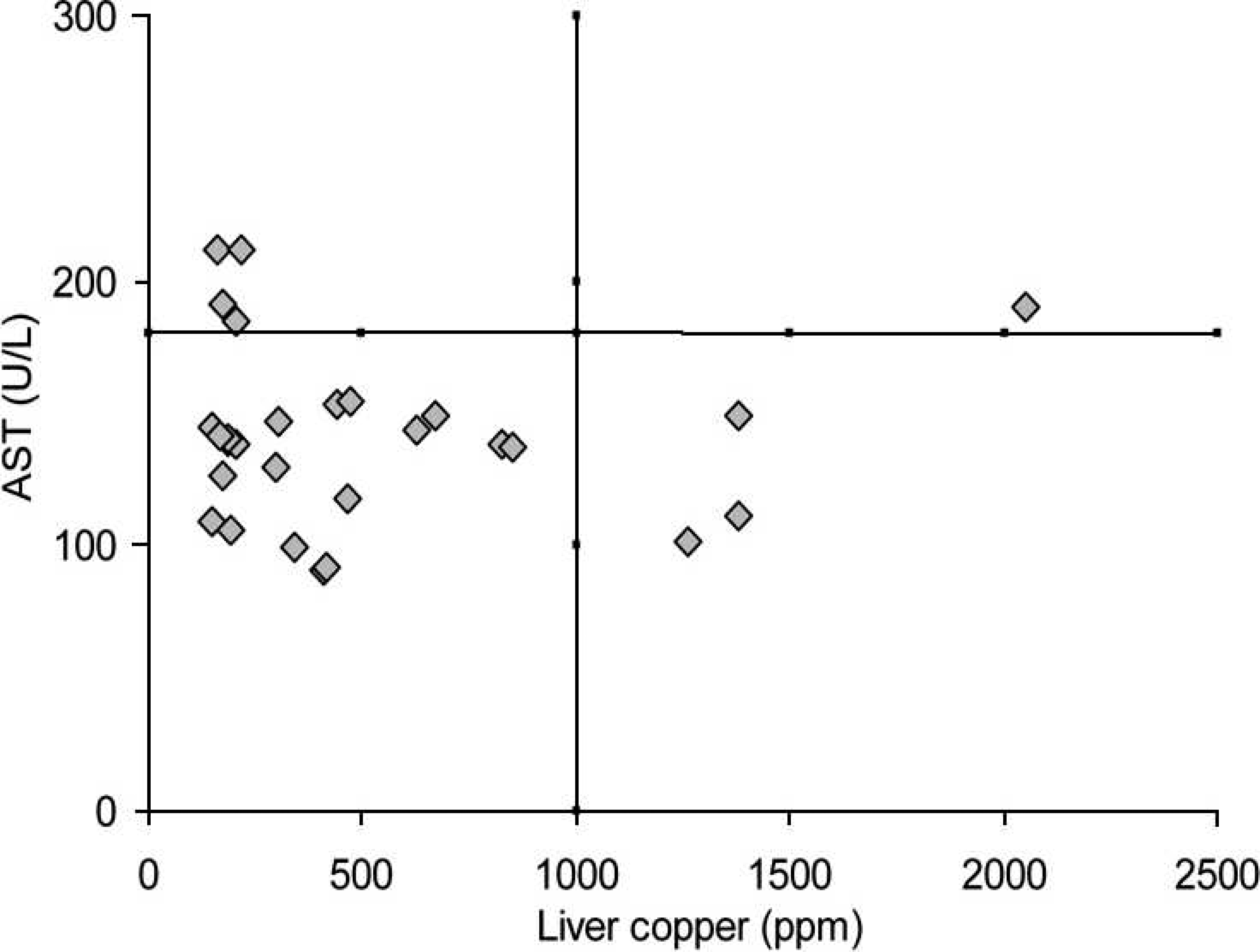
The discrimination values for the elevated serum activity of AST in cattle and buffalo indicative of hepatic Cu accumulation are presented in Figures 7 and 8, respectively. When hepatic concentrations (1,000 ppm) indicative of chronic Cu poisoning were used to evaluate serum AST activity, the discrimination value was obtained at 90 U/l. With these limits, the positive (75%) and negative (100%) predictive values, sensitivity (100%), specificity (91.7%), and accuracy (93.3%) were determined. For buffalo, the best discrimination value for serum AST activity was 180 U/l. This resulted in lower positive (46%) and negative (86%) predictive values, sensitivity (50%), specificity (83%), and accuracy (76.7%).
Although the number of animals used during the present study was relatively low, the results obtained were consistent and indicate that determination of serum GGT activity is a better indicator of hepatic Cu accumulation than serum AST activity. However, both enzymes can be
Cut-off line for the accumulation of liver copper and the elevation of serum gamma-glutamyl transferase (GGT) in buffalo. Relationship between serum aspartate amino-transferase (AST) and the hepatic concentration of copper (Cu) in cattle from Cu-treated and control groups during the experiment.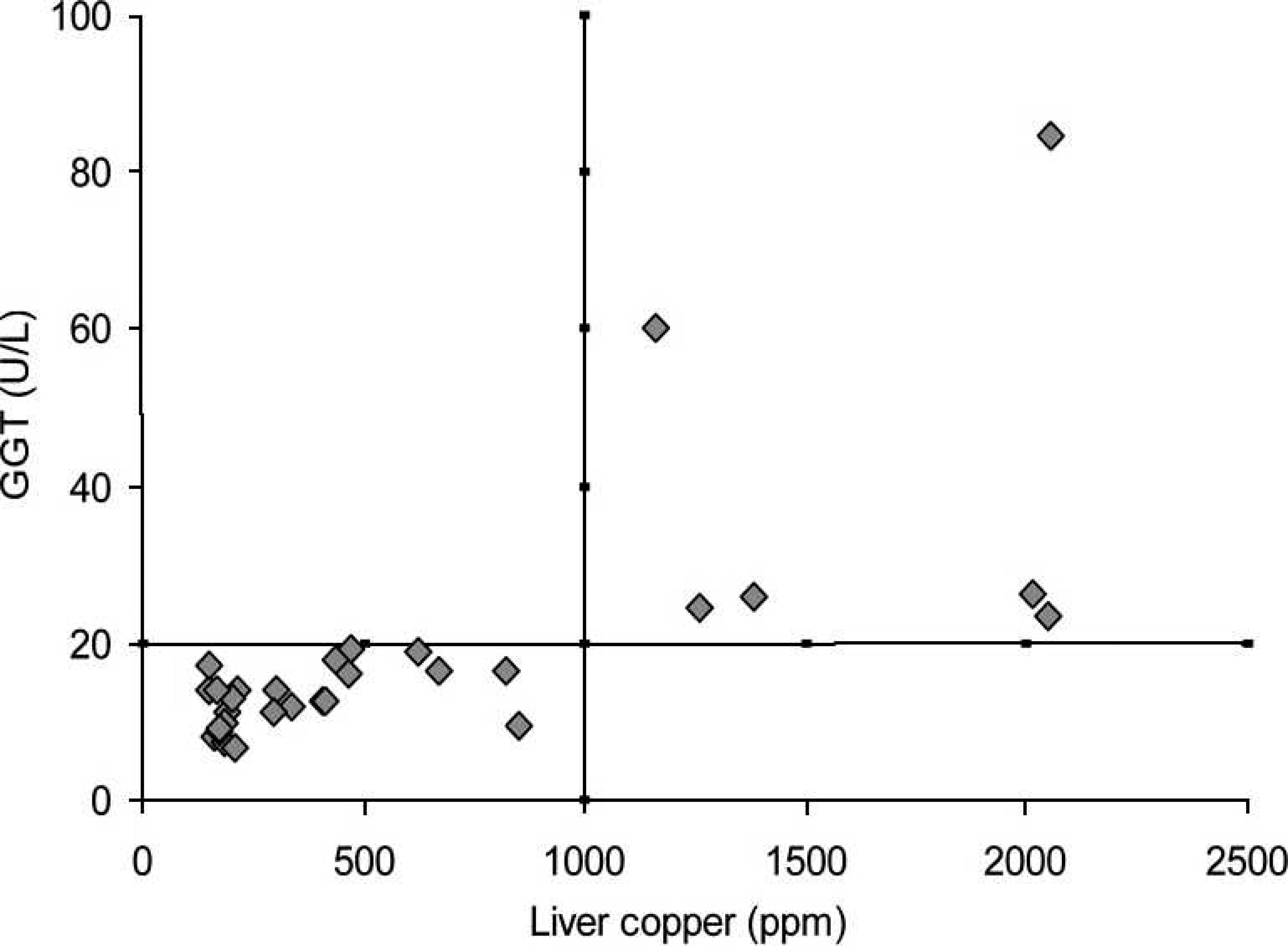
Another fact that must be considered when evaluating AST serum activity is that its basal activity is higher in
Relationship between serum aspartate amino-transferase (AST) and hepatic concentration of copper (Cu) in buffalo from Cu-treated and control groups during the experiment. Cut-off line for the accumulation of hepatic copper and the elevation of serum aspartate aminotransferase (AST) in cattle. Cut-off line for the accumulation of hepatic copper and the elevation of serum aspartate aminotransferase (AST) in buffalo.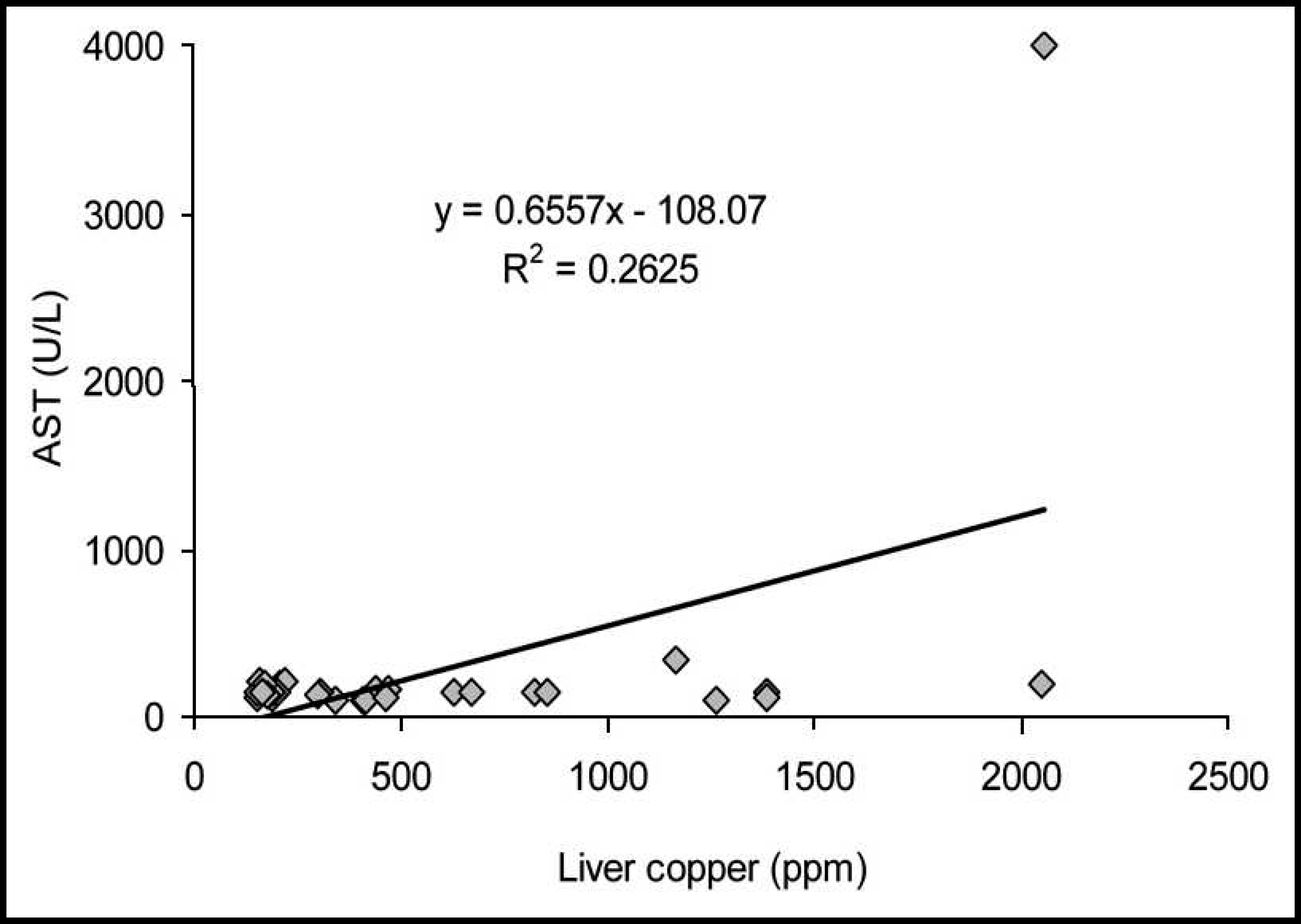
Both AST and GGT serum activities had increased coefficients of determination and were almost as equally efficient in determining elevated concentrations of hepatic Cu in cattle. However, determination of AST activity was not as good at predicting elevated concentrations of hepatic Cu in buffalo, since a low coefficient of determination was verified (R 2 = 0.26) and false-positive and false-negative test results were observed.
Contrary to the results of previous publications, 5,7,9 GGT serum activity was considered as a possible indicator for the diagnosis of excessive hepatic accumulation of Cu in cattle and buffalo in the current study. Considering the established discrimination value for GGT serum activity in cattle (24.6 U/l), a research study 5 involving a Cu-overload diet (20 mg/kg BW of Cu) has shown that bulls had values indicative of excessive liver Cu accumulation. In contrast, the heifers did not have serum values of GGT activity that were indicative of elevated hepatic Cu concentration. 5 Although AST serum activity had reduced accuracy and predictive values when compared with GGT activity, increased AST activity was relatively efficient in the diagnosis of excessive accumulation of hepatic Cu in cattle, as demonstrated by the high coefficients of determination. However, contradictory results have also been described for AST serum activity. 5
Under normal field conditions, liver dysfunctions, such as fatty liver, can elevate the serum activity of liver enzymes and could induce false-positive results. Nevertheless, the discrimination values obtained during the present study may have clinical significance as a superior and alternative method for the early diagnosis of Cu accumulation, since it is comparatively inexpensive, less invasive, and safer for animal health than conventional liver biopsy.
The serum activities of sorbitol dehydrogenase and acid phosphatase might have low levels of accuracy to predict Cu accumulation in sheep, 13 while alkaline phosphatase has a weak correlation with hepatic Cu in cattle. 9 Additionally, glutamate dehydrogenase has elevated plasma activity even after the period of Cu overload and presents better results as an indicator of hepatic Cu accumulation than do AST and GGT in sheep. 7 However, because of their availability in Brazil and as a result of importation restrictions, these enzymatic evaluations were not performed as part of the current study. To evaluate these results, further studies are necessary with a larger number of animals using different enzymatic combinations in cattle and buffalo with high Cu intake that leads to a hemolytic crisis over a short period.
Elevated serum GGT activity may be indicative of increased concentrations of hepatic Cu even in cattle and buffalo that appear to be clinically healthy. Thus, prophylactic measures can be implemented to prevent the onset of a hemolytic crisis that is characteristic of Cu intoxication.
Acknowledgements. E. L. Ortolani and R. A. Barrêto Júnior thank CNPq (Brazil) for their fellowships. A. H. H. Minervino received a FAPESP fellowship (São Paulo State, Brazil). This research was supported by FAPESP (process 2005/03204-0). The authors are grateful to Marly E. F. de Castro and Clara S. Mori for laboratory assistance.
Footnotes
a.
LIASYS® (Latest Innovative Automation SYStem), Analyzer Medical System Srl, Rome, Italy.
b.
BioSystems SA, Barcelona, Spain.
