Abstract
Herein we describe a glioblastoma partially occupying the telencephalic portion of the left cerebral hemisphere of a Sardinian (syn. Sarda) breed ewe. Microscopically, the mass consisted of a pleomorphic spindle-shaped cell component organized as bundles and numerous small areas of round cells displaying an oligodendroglioma-like aspect. A high number of mitotic figures, large areas of necrosis surrounded by pseudopalisading glial cells, and multiple foci of dystrophic mineralization were also observed. The neoplasm was highly vascularized with glomerular vascular proliferation. Immunohistochemically, neoplastic cells proved to be strongly positive for nestin, vimentin, and olig-2, whereas they were invariably negative for synaptophysin. Few neoplastic cells and reactive astrocytes, mainly located at the edge of necrotic foci, proved to be positive for glial fibrillary acidic protein, whereas glomerular vascular proliferation was clearly positive for factor VIII and vascular endothelial growth factor. Gene sequencing analysis demonstrated homozygous p53 tumor suppressor gene (TP53) point mutations in the DNA-binding domain located in exon 8. The presence of round cells immunoreactive for olig-2 demonstrated that this tumor is a glioblastoma with oligodendroglioma component. Our pathologic, immunohistochemical, and molecular findings largely overlap those previously reported in humans and dogs.
The occurrence of neoplastic disorders of the central nervous system (CNS) is considered quite common in humans, with an estimated overall incidence in the United States of 18.2 cases per 100,000 people. Approximately 50–75% of tumors are CNS primary tumors, whereas the remainder are metastatic. 12 In veterinary medicine, primary CNS neoplasms are most frequently reported in dogs and less commonly in cats, with an estimated annual incidence of 14.4 cases per 100,000 dogs and 3.5 cases per 100,000 cats. 9 The same CNS tumors are considered distinctly uncommon within other domestic animal species.6,9
Among CNS tumors, gliomas are of neuroepithelial origin and include astrocytomas, oligodendrogliomas, and ependymomas. The tumors usually consist of a mixture of glial cells and thus their nomenclature and classification is based on the predominant cell type. Although the real incidence of tumors in animals is evaluated in a range of ways and data are difficult to compare, astrocytomas have been reported to account for 17–28% of primary intracranial neoplasms in dogs and ~3% in cats.11,12
Glioblastoma (formerly glioblastoma multiforme) is considered the most malignant form of astrocytoma and is consequently classified as “high-grade” astrocytoma in animals, and grade IV malignancy in humans. 7 As described in dogs and humans, the gross appearance of glioblastoma is typically variable, whereas microscopically it closely resembles anaplastic astrocytoma with necrosis and vascular proliferation representing important diagnostic features. 12
In dogs, astrocytomas accounts for ~10% of all CNS tumors, and glioblastoma represents only ~5% of all astrocytomas. 6 Glioblastomas have only been occasionally documented in animal species other than dogs. 12
Regarding the genetic alteration associated with brain tumors, it is known that p53 tumor suppressor gene (TP53) mutations have been frequently reported in astrocytic tumors, whereas only 3.4% of canine brain tumors were found with exonic mutations. 16 Considering the low frequency, no data are available for brain tumors in other species.
In the current study, we describe the main gross, microscopic, immunohistochemical, and genetic features of a glioblastoma observed in a ewe. Finally, our findings are compared with those more frequently documented in humans and dogs.
A 6-year-old Sardinian (syn. Sarda) breed sheep showed severe and worsening sensorial depression with head pressing. Clinical signs lasted for ~8 months, and the sheep was then euthanized because of the severity of the clinical signs. At autopsy, the whole brain was collected, promptly fixed in 10% neutral buffered formalin, and routinely processed for histologic investigation (hematoxylin and eosin stain). Furthermore, in-depth immunohistochemical (IHC) investigations were carried out on selected tissue sections using primary antibodies specific for the following markers: vimentin, a synaptophysin, b nestin, c olig-2, d glial fibrillary acidic protein (GFAP), e vascular endothelial growth factor (VEGF), f factor VIII, g and p53 h (see Table 1 for further technical details). Primary antibodies were incubated overnight at 4°C, and immunoreactivity was detected via the biotin–avidin–peroxidase method, i by using 3,3′-diaminobenzidine j as a chromogen. Adequate positive control tissues were included in each IHC run, whereas negative controls were obtained by omitting the primary antibody.
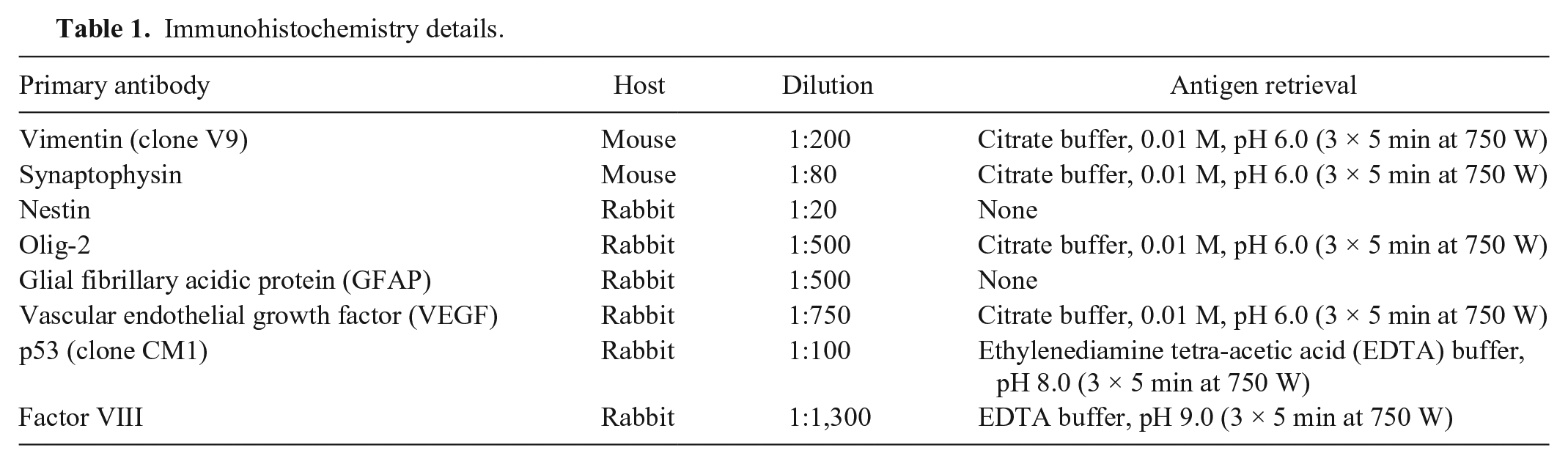
Immunohistochemistry details.
In addition, serial paraffin-embedded sections and formalin-fixed tissues sampled from the cerebral mass as well as from the histologically healthy medulla oblongata were submitted for DNA extraction. Genomic (g)DNA was extracted from paraffin-embedded sections by using a commercial kit, k whereas gDNA extraction from formalin-fixed tissues was performed following an in-house phenol-chloroform protocol. Exons 5–8 (DNA-binding domain) of TP53 were amplified by PCR using specific primers, as listed in Table 2. Finally, sequence analysis was carried out with a cycle sequencing commercial kit. l Sequencing products were separated and analyzed by using a genetic analyzer. m
Genomic DNA PCR primers for ovine TP53 exons.*
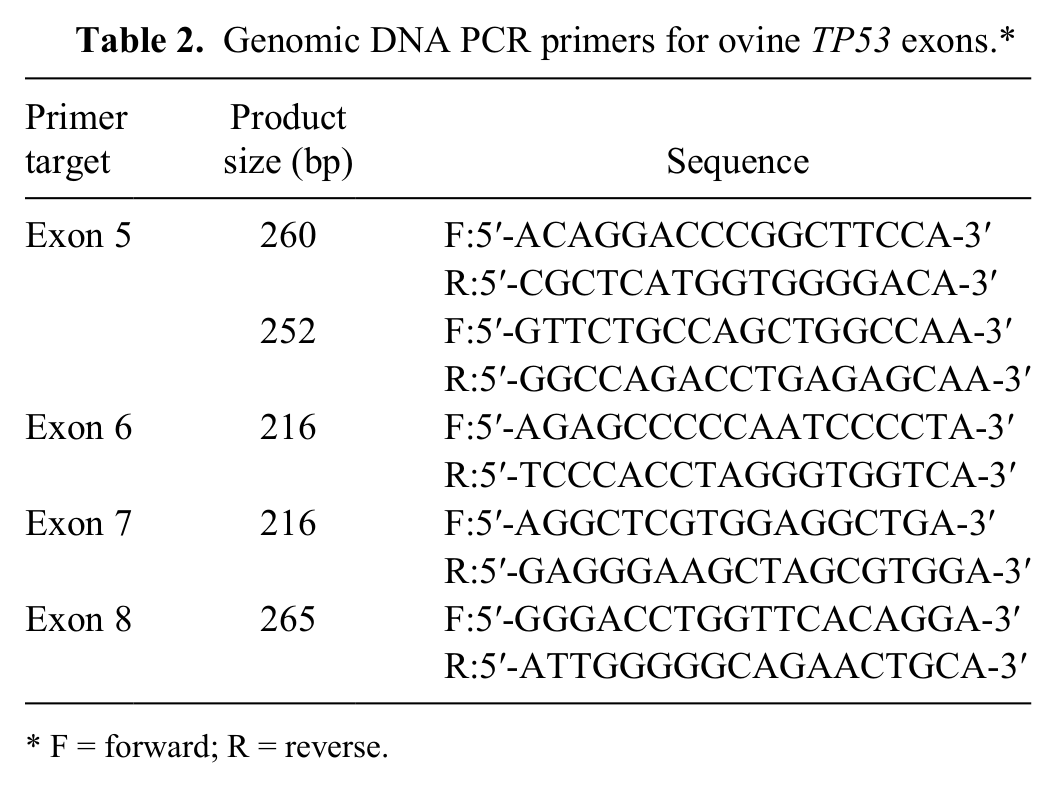
F = forward; R = reverse.
At gross examination of the brain, 1 large monolateral, 5 cm × 7 cm, white-gray mass was observed in the ventrolateral portion of the frontal lobe of the left hemisphere (Fig. 1A). Two lobes of the mass protruded externally onto the cortex, thus involving the meninges at the level of the sulcus presylvius and rhinalis lateralis. The external surface was soft, mucinous, and showed scattered hemorrhages together with yellow foci of necrosis. On cut section, the mass shared similar aspects with the surface; the mass was poorly bounded and replaced the basal nuclei. The medial side of the neoplasm invaded the lumen of the left lateral ventricle and caused partial compression of the contralateral one (Fig. 1B).
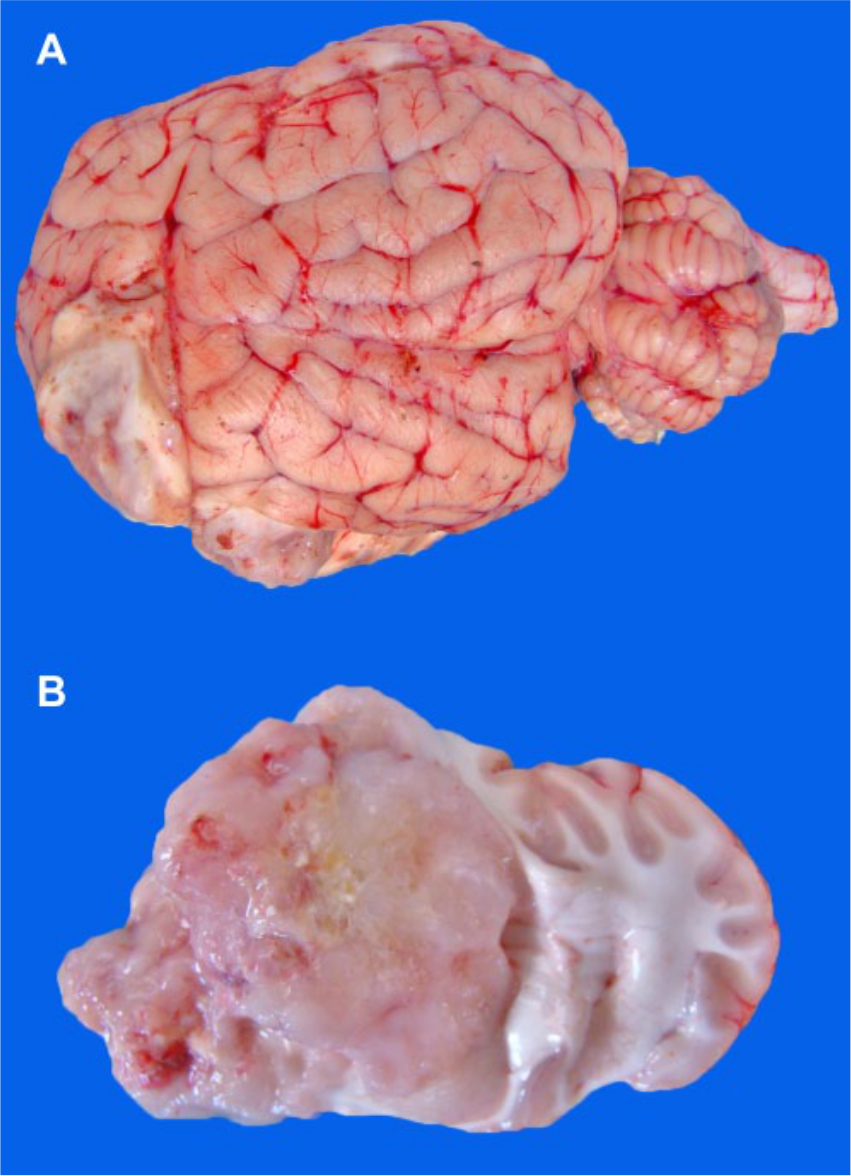
Glioblastoma in sheep brain.
Microscopically, the cerebral mass consisted of a pleomorphic spindle-shaped cell component organized as intermingled bundles defining a coarse meshwork (Fig. 2A, 2B). Occasionally, multinucleated neoplastic cells were observed. The number of mitoses was very high (up to 5 mitoses per high power field) in the pleomorphic spindle-shaped cell component (Fig. 2B). Multifocal proliferation of topknots of newly formed blood vessels (glomerular vascular proliferation) was mainly observed at the edge of the tumor (Fig. 2C). A number of large areas of necrosis, dystrophic mineralization and occasional corpora amylacea were observed throughout the mass. Necrosis often showed a serpiginous pattern and was bordered by pseudopalisading glial cells (Fig. 2D). The neoplasm was not well circumscribed, with glomerular vascular proliferation and neoplastic cells infiltrating the surrounding parenchyma. Scarce inflammatory infiltrates were observed within and around the neoplastic tissue.
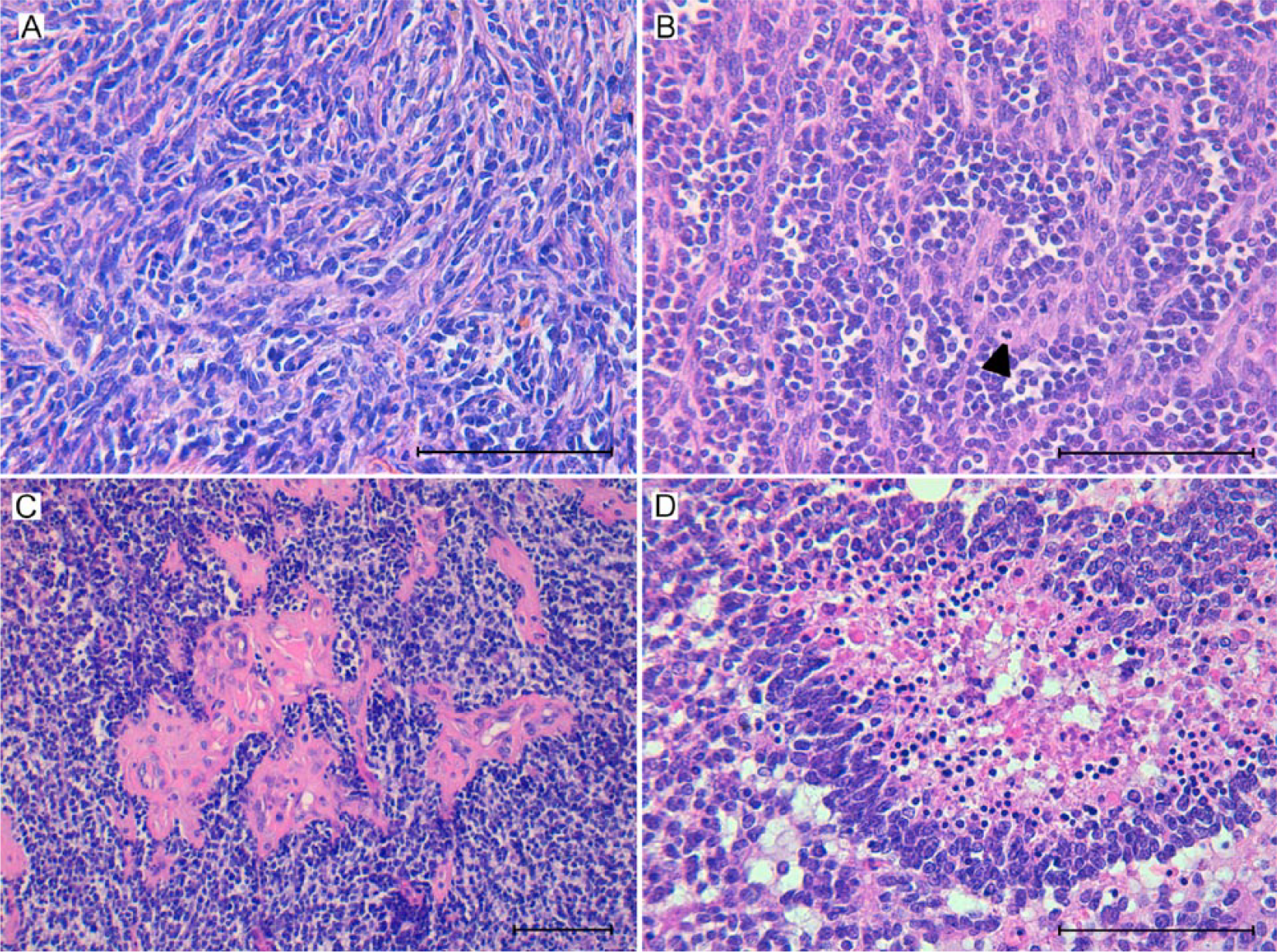
Glioblastoma in sheep brain.
Interestingly, interspersed with the bundles of the pleomorphic spindle-shaped cells were numerous areas of round cells that displayed an oligodendroglioma-like aspect characterized by a “fried-egg” appearance with hyperchromatic nuclei, scanty cytoplasm, and ill-defined borders (Fig. 2B). At IHC, sections of the tumor showed numerous spindle-shaped cells organized as bundles strongly cytoplasmic positive for nestin (Fig. 3A) and vimentin (Fig. 3B). Notably, nuclear olig-2–positivity was consistent with the round oligodendroglioma-like cells (Fig. 3C). Scattered foci of GFAP positivity were associated with a few neoplastic spindle-shaped cells and reactive astrocytes that surrounded the areas of necrosis (Fig. 3D). Glomerular vascular proliferation was clearly factor VIII positive (Fig. 3E), whereas VEGF-positivity was detected at the edge of necrosis in neoplastic cells, reactive astrocytes, and endothelial cells of the glomerular vascular proliferation (Fig. 3F). p53-positive neoplastic cells were only occasionally observed. Finally, no immunoreactivity was observed for synaptophysin throughout the mass.
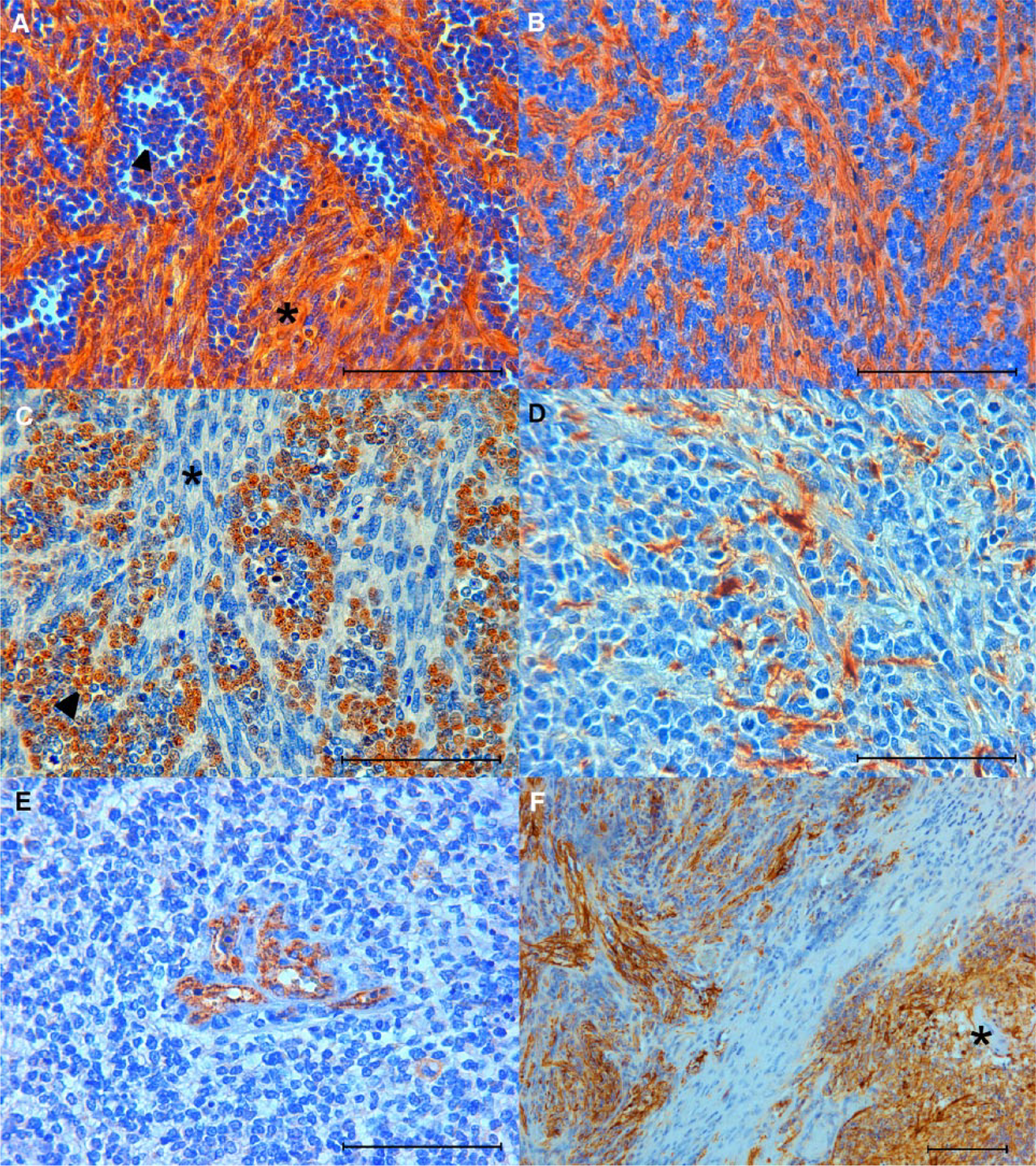
Glioblastoma in sheep brain.
Gene sequencing demonstrated TP53 point mutations in exon 8 in the glioblastoma, with a homozygous single-base substitution (G>A) at position 2 of codon 256. Such mutation determined the amino acid substitution of arginine with glutamine, and was absent in the autologous tissue sampled from the medulla oblongata.
On the basis of discriminating histopathologic aspects, namely glomerular vascular proliferation coupled with pseudopalisading glial cells around necrosis, as well as IHC for nestin and vimentin, glioblastoma was diagnosed. In fact, all of these histologic features have commonly been described in glioblastoma of other species.13,14 In addition, although vimentin positivity is not specifically found in astrocytes, it indicates cells of glial origin, 7 whereas nestin positivity is expressed in high-grade astrocytoma in humans 10 and dogs 5 as an immature marker of neuroblastic cells. However, the significant presence of round cells, which tested positive for olig-2, demonstrated that this was a glioblastoma with a large oligodendroglioma component. 14
Glioblastoma is considered the most common and lethal primary malignant brain tumor in humans and dogs. Although nonmetastatic, glioblastoma is highly invasive and tends to spread widely within the brain parenchyma. 12
In dogs, 7 different astrocytic tumors have been classified on the basis of microscopic features, including glioblastoma. 12 Canine glioblastoma occurs most frequently in brachycephalic breeds with the highest incidence being reported in the Boxer, which has been proposed as an excellent animal model of this brain tumor. 12
To date, very few cases of primary CNS tumors have been reported in sheep: medulloblastoma in lambs, 1 ependymoma in a Suffolk sheep aged <1 year, 4 and, more recently, oligodendroglioma in a 1-year-old male Iranian fat-tailed sheep. 2 Our investigation showed pathologic features that closely resemble those reported in humans and other animals, 6 including the age of onset. In fact, glioblastoma usually affects adult humans (50–60 years old) and dogs (>6 years of age) 12 ; in contrast, the previously mentioned reports of ovine CNS tumors were described in young sheep at the time of clinical onset.
The expression of cytoplasmic intermediate filaments modifies during CNS development in mammals, and such differences are useful tools for the diagnosis of neoplastic disorders. Herein, our IHC data are largely in agreement with those reported in other domestic animals and in humans. Notably, re-expression of nestin and down-regulation of GFAP have previously been reported in human cell lines derived from glioblastoma tumors. 15 In this respect, GFAP- positivity changes among the cases of canine glioblastoma, with low-grade astrocytoma having the higher expression. 5 In humans, high-grade astrocytomas have often been reported to be GFAP negative. 10 Collectively, these results suggest that the sheep glioblastoma described herein is a particularly malignant variant.
In our case, the IHC signal for olig-2–positivity was strong, confirming that oligodendroglial cells can be a significant component of glioblastoma in sheep, thus recognizing in this species a subtype of glioblastoma with an oligodendroglioma component, as classified in the 2007 World Health Organization (WHO) classification of tumors of the human central nervous system. 7 In this respect, it has been hypothesized that the presence of oligodendroglial cells in human glioblastoma is not related to its benign or malignant behavior. 3 A weak oligodendroglial component has also been described in dogs.6,13
VEGF-related angiogenic factors have been demonstrated in gliomas, and VEGF is considered the most important angiogenic factor in glioblastoma. 12 In this respect, a paracrine mechanism is likely with VEGF being secreted by tumor cells and bound by endothelial receptor. 8 In our report, the pattern of VEGF-positivity was similar to that described in canine glioblastomas.6,12 With regard to the pattern of necrosis, in human glioblastomas, localized hypoxia appears to upregulate migration-associated genes, leading to migration of tumor cells away from the central hypoxic core, the latter being more susceptible to necrosis. 8
Some TP53-inactivating acquired point mutations within the DNA-binding domain of the protein have been suggested to contribute to oncogenesis in every type of cancer, including brain tumors. In fact, in humans, TP53 mutations were among the first genetic mutations identified in astrocytic tumors and appear to be an early event, being present at first biopsy in most cases. They seem to be less common in primary or de novo glioblastomas, whereas secondary glioblastomas arising from prior lower-grade astrocytomas have a high incidence of TP53 mutations. In dogs, no TP53 mutations involving exons 3–9 were found in WHO grade II astrocytomas, in strong contrast to analogous human astrocytomas. 8 Our report suggests that TP53 exonic mutations can also occur in glioblastoma in sheep, and could likely contribute to the pathogenesis of such neoplastic disorders.
The present report documents the histologic and IHC aspects of glioblastoma in sheep. It also emphasizes the importance of careful postmortem examination of the CNS in order to reach accurate diagnoses of neurologic disorders and gain a better understanding of the prevalence and biological behavior of neoplastic disorders in sheep.
Footnotes
Acknowledgements
We thank Drs. Giovanni Antonio Carboni and Pierangela Campagnani for logistic and administrative assistance.
Authors’ contributions
D Pintus contributed to conception of the study; contributed to analysis and interpretation of data; drafted the manuscript; and critically revised the manuscript. G Marruchella contributed to design of the study; contributed to interpretation of data; drafted the manuscript; and critically revised the manuscript. M Masia contributed to acquisition and analysis of data. C Maestrale contributed to design of the study, and contributed to analysis and interpretation of data. MG Cancedda contributed to analysis of data, and critically revised the manuscript. C Contu and S Macciocu contributed to analysis of data. C Ligios contributed to conception and design of the study; contributed to interpretation of data; drafted the manuscript; and critically revised the manuscript. All authors gave final approval, and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Monoclonal anti-vimentin clone Vim V9, Dako North America Inc., Carpinteria, CA.
b.
Monoclonal anti-synaptophysin clone SY38, Dako North America Inc., Carpinteria, CA.
c.
Anti-nestin rabbit IgG affinity purify, IBL Immuno-Biological Laboratories, Minneapolis, MN.
d.
Polyclonal antibody anti-olig 2, EMD Millipore Corp., Billerica, MA.
e.
Anti-glial fibrillary acidic protein GFAP, BioGenex, Freemont, CA.
f.
Anti-VEGF polyclonal antibody, EMD Millipore Corp., Billerica, MA.
g.
Polyclonal Von Willebrand factor, Dako North America Inc., Carpinteria, CA.
h.
P53 CM1 polyclonal antibody, Covance Inc., Princeton, NJ.
i.
R.T.U. Vectastain Universal Elite ABC kit, Vector Laboratories, Burlingame, CA.
j.
Peroxidase substrate kit DAB SK-4100, Vector Laboratories, Burlingame, CA.
k.
QIAamp DNA FFPE tissue kit, Qiagen Inc., Valencia, CA.
l.
Big Dye Terminator cycle sequencing kit 1.1, Applied Biosystems, Foster City, CA.
m.
ABI 3500 genetic analyzer, Applied Biosystems, Foster City, CA.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Grant RC IZS 01/10 from Ministero della Salute (Italy).
