Abstract
Malignant catarrhal fever (MCF)-like disease was diagnosed at postmortem in 2 Stone's sheep (Ovis dalli stonei). On gross examination, the predominant abnormality in both sheep was severe perirenal hemorrhage and multiple renal infarcts. Microscopically, there was severe, multisystemic lymphocytic arteritis. Both sheep were positive for Ovine herpesvirus 2 (OvHV-2) on polymerase chain reaction, and partial sequencing of the viral DNA polymerase genes from the 2 sheep revealed >99% homology, with 96% similarity to the reference GenBank OvHV-2 viral sequence. Based on the histological lesions, polymerase chain reaction results, and viral DNA polymerase gene sequencing, a diagnosis of OvHV-2-mediated MCF was made. Massive perirenal hemorrhage has not been described previously as a manifestation of MCF.
Malignant catarrhal fever (MCF) is generally considered to be a sporadic disease of low morbidity and high mortality that affects members of the order Artiodactyla, particularly the families Bovidae, Cervidae, and Giraffidae. 4,10 Malignant catarrhal fever is caused by a group of viruses belonging to a subgroup of rhadinoviruses, which are a genus of the subfamily Gammaherpesvirinae. 5 There are at least 4 MCF viruses that have been clearly associated with MCF, including Alcelaphine herpesvirus 1 (AlHV-1) found in wildebeest (Connochaetes spp.), Ovine herpesvirus 2 (OvHV-2) found in domestic sheep, Caprine herpesvirus 2 found in domestic goats, and malignant catarrhal fever virus (MCFV) of white-tailed deer (Odocoileus virginianus), which is of unknown origin. 5 These viruses are asymptomatic in their natural hosts but may cause a MCF-like disease when transmitted to a different ungulate species. 10 Animals that develop MCF, however, are not thought to further transmit the virus. 10
Malignant catarrhal fever virus transmission and MCF development is complex, because there are dramatic differences in susceptibility to the various MCF viruses among species and even within a group of conspecifics, as reflected by sporadic development of clinical disease within a susceptible herd. 10 Domestic sheep, as natural hosts of OvHV-2, are thought to be resistant to the development of OvHV-2-induced MCF. The susceptibility of wild sheep species, however, has not been well characterized. The current report describes 2 Stone's sheep (Ovis dalli stonei) that developed MCF-like disease with the addition of bilateral perirenal hemorrhage that, to the authors' knowledge, has not been previously described.
Stone's sheepare considered a subspecies of thinhorn sheep, distinguished from Dall's sheep (Ovis dalli dalli) on the basis of coat color, but the validity of subspecies status is debatable. 9,13 Thinhorn sheep are found in remote mountainous areas of northwestern North America, where there have been few introductions of domestic livestock.
On August 30, 2007, a 6-year-old male Stone's sheep at a zoological park was found dead with no prior clinical signs. Eleven days later (September 10, 2007), a 10-year-old female Stone's sheep from the same park presented to the Western College of Veterinary Medicine Large Animal Clinic (Saskatoon, Saskatchewan, Canada) with a history of hind limb lameness beginning 2 weeks prior to presentation. Clinically, weakness of the hind limbs, depression, and aggression toward humans was observed. Hematology revealed marked leukocytosis (white blood cells [WBC] = 30.1 × 109/l, reference [ref.] range: 4.0–12.0) with neutrophilia, 2+ toxic change, left shift (neutrophils = 18.66 × 109/l, ref. range: 0.7–6.0; bands = 6.9 × 109/l, ref. range: 0.0–0.1), and mild, nonregenerative anemia (red blood cells [RBC] = 5.6 × 1012/l, ref. range: 8.0–16.0). There were increases in total bilirubin (54 μmol/l, ref. range: 1.0–11.0), gamma-glutamyl transferase (GGT = 613 U/l, ref. range: 9–61), aspartate aminotransferase (AST = 2180 U/l, ref. range: 62–260), sorbitol dehydrogenase (SDH = 320 U/l, ref. range: 5–39), urea (73.8 mmol/l, ref. range: 4.5–12.1), and creatinine (612 umol/l, ref. range: 55–107). Metabolic acidosis (anion gap = 43 mmol/l, ref. range: 17–33; bicarbonate = 6 mmol/l, ref. range: 17–29) was also diagnosed. The ewe had a seizure and died shortly after presentation.
Both sheep were necropsied. On gross examination, the ram had massive (20 × 10 × 8 cm) oval, perirenal, retroperitoneal, dark-red blood clots that completely encompassed each kidney. The ewe had 2 retroperitoneal, perirenal masses, each approximately 15 × 10 × 9 cm, composed of clotted blood and fibrinous to fibrous material (Fig. 1), as well as copious amounts of serosanguineous fluid. These masses also entirely encompassed both kidneys. In both animals, there were numerous multifocal renal cortical infarcts of varying size and chronicity, and the clotted blood surrounding the kidneys was intimately adherent to 1 or more areas of infarction (Fig. 1). Both sheep had petechial hemorrhage in multiple tissues, including the myo- and epicardium, splenic capsule, serosal surface of the rumen and abomasum, and vena caval intima. Additionally, there was serosanguineous to fibrinous effusion in 1 or more body cavities. Samples from multiple organs taken at necropsy were fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin for histological examination.
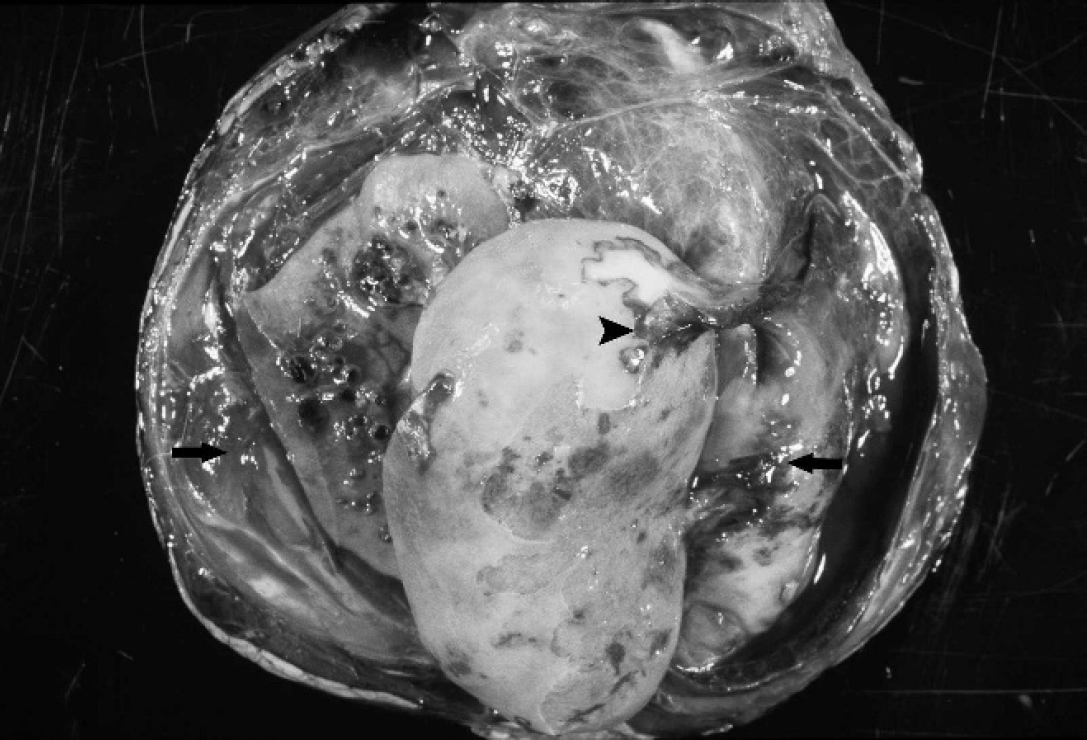
External surface of a kidney from a female Stone's sheep with malignant catarrhal fever-like disease. There is a large perirenal hematoma filled with clotted blood and fibrin (arrows) that is attached to a large cortical infarct (arrowhead).
Microscopic renal lesions were similar in both animals. Lymphocytic vasculitis, fibrinoid degeneration, and thrombosis were present in multiple arcuate and intralobular arteries (Fig. 2). There were multiple renal cortical infarcts of varying chronicity, numerous sclerotic glomeruli, and moderate interstitial fibrosis. Arteritis was limited to the kidney in the ram but was present in many organs in the ewe. Both animals had multifocal lymphocytic infiltrates in the hepatic periportal area, with the ewe having similar infiltrates scattered throughout the renal interstitium, adrenal capsule, and cortex. On the basis of the presence of fibrinoid arteritis with lymphocytic infiltration and accumulation of lymphocytes in nonlymphoid organs, a tentative diagnosis of MCF was made.
Fresh kidney tissue from the ram was examined by polymerase chain reaction (PCR) for OvHV-2. The tissue (50 μg) was homogenized in 500 μl of lysis buffer (l00 mmol NaCl, 500 mmol Tris pH 8.0, 10% w/v sodium dodecyl sulfate) with proteinase K (0.2 mg/ml final concentration) and then mixed by vortexing. Tissue homogenates were incubated for 2 hr at 65°C and then extracted twice with phenol:chloroform (1:1). Nucleic acids were precipitated with 2.5 volumes of cold ethanol, and pellets were washed twice in 80% ethanol before resuspension in 50 μl of sterile water. Initial screening of the tissue sample from the ram for the presence of OvHV-2 was done using a seminested diagnostic, modified PCR protocol, 2,8 in which a region of a gene encoding a tegument protein of OvHV-2 is amplified, producing a 238-bp PCR product. Polymerase chain reaction products were detected on 1.25% agarose gel stained with ethidium bromide.
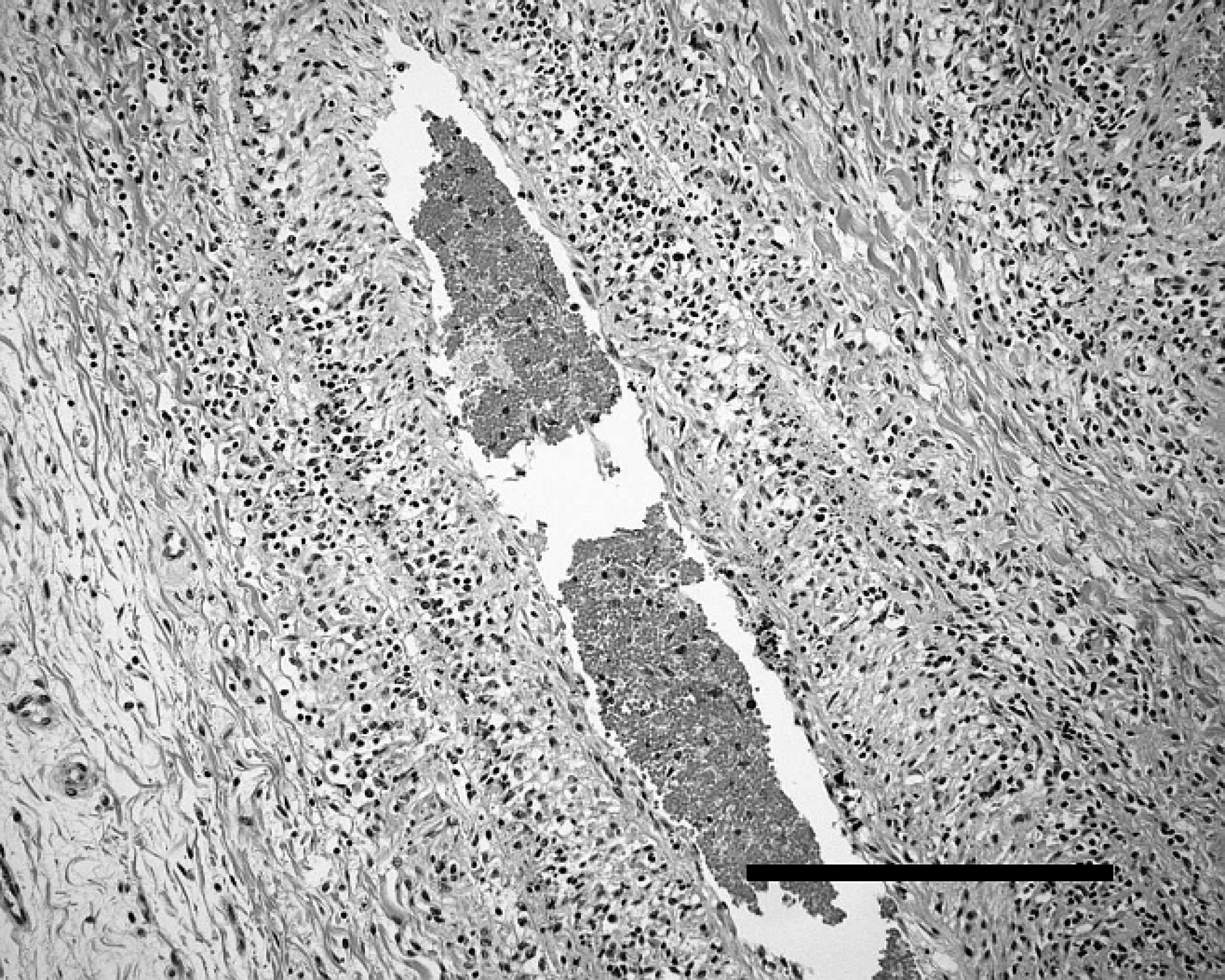
Lymphocytic arteritis in the adrenal capsule of a female Stone's sheep with malignant catarrhal fever-like disease. Hematoxylin and eosin. Bar = 250 μm.
To confirm the presence of OvHV-2 and generate viral sequence data for comparison with other strains, nested consensus PCR of viral DNA polymerase gene sequences was performed on kidney samples from both animals. 12 Briefly, 5 μl of extracted DNA was used as template in a 50μl PCR reaction containing 5 U of Taq DNA polymerase, 2 mmol of MgCl2, 50 mmol of KCl, 10 mmol of Tris/HCl (pH 8.3), 250 μmol each of dNTP, and 20 pmol each of primers DFA (5′-GAYTTYGCNAGYYTNTAYCC-3′), ILK (5′-TCCTGGACAAGCAGCARNYSGCNMTNAA-3′), and KG1 (5′-GTCTTGCTCACCAGNTCNACNCCYTT-3′). 12 Reactions were incubated at 94°C for 5 min followed by 45 cycles of 30 sec at 94°C, 60 sec at 46°C, and 60 sec at 72°C, with a final extension of 10 min at 72°C. 12 Five microliters of primary PCR product was used as template in a secondary PCR reaction with primers TGV (5′-TGTAACTCGGTGTAYGGNTTYACNGGNGT-3′) and IYG (5′-CACAGAGTCCGTRTCNCCRTADAT-3′), with the same thermocycling protocol as the primary PCR reaction. Polymerase chain reaction products were detected on a 1.25% agarose gel stained with ethidium bromide. 12
DNA sequencing was performed at the National Research Council Plant Biotechnology Institute (Saskatoon, Saskatchewan, Canada). Raw data were assembled and edited using programs pregap4 and gap4 of the Staden Package. 11 Finished sequence data were compared with the NCBI GenBank nucleotide database using nucleotide BLAST (BLASTn; http://www.ncbi.nlm.nih.gov/blast/Blast.cgi). 1
Both tissue samples were positive on PCR assays targeting the viral tegument gene. Sequencing of secondary PCR products produced with consensus herpesvirus DNA polymerase primers TGV and IYG yielded 228 nucleotides of polymerase gene high-quality sequence data (deposited in GenBank; accession numbers EU309722 and EU309723). Sequences determined for the herpesviruses identified in the 2 Stone's sheep were >99% identical and 95–96% identical to the 2 OvHV-2 strains for which complete genome sequences have been determined (GenBank accession numbers DQ198083 and AY839756). The Stone's sheep-derived sequences were only 80% identical to white-tailed deer MCFV (AF387516) and 74% identical to Caprine herpesvirus 2 (AF283477). On the basis of PCR and genomic sequencing results, OvHV-2-induced MCF was diagnosed.
Making a definitive diagnosis of MCF is somewhat problematic in this species. Although the prevalence and characteristics of MCFV in wild sheep have not been extensively investigated, antibody to MCFV was detected in 95% of a group of free-ranging Dall's sheep in Alaska. 15 Fragments of OvHV-2 DNA were detected by PCR in 6 of 14 of these sheep. 15 This might suggest that some wild sheep are asymptomatic carriers. However, unlike domestic sheep, OvHV-2–naive populations of wild sheep do occur. In a serologic study of Bighorn sheep (Ovis canadensis), none of the 28 sheep from 1 population had antibodies against OvHV-2. 7
There is evidence of OvHV-2 transmission at the zoological park from which the sheep in the current case originated. Four crossbred Stone's × Dall's sheep introduced to the premises in January 2006 were negative for OvHV-2 using PCR on blood samples collected just prior to entering the park. On July 24, 2006, a 3-year-old ewe died of a MCF-like disease and was positive for OvHV-2 using PCR on tissue samples. The remaining 3 sheep were subsequently tested for OvHV-2 using PCR on blood and were negative, indicating transmission of OvHV-2 at the park and its possible association with the development of an MCF-like disease. This park also contained a number of other hoof stock of the subfamily Caprinae, including Dall's sheep, Bighorn sheep, Mouflon sheep (Ovis aries musimon), and Rocky Mountain goats (Oreamnos americanus). There were no domestic sheep on the premises. The Stone's, Dall's, Stone's × Dall's, and Bighorn sheep had direct contact with one another, either through comingling or fence line contact; however, all of the aforementioned species may have experienced indirect contact through shared husbandry equipment and/or public feeding stations.
Malignant catarrhal fever-like disease has been experimentally induced in domestic sheep through the aerosol administration of OvHV-2 to naive animals. 6 Because some wild sheep may be naive, it is reasonable to infer that the sheep in the current case died from MCF. Malignant catarrhal fever has been reported in a Barbary sheep (Ammotragus lervia) kept at a zoological park, and OvHV-2 was detected in this animal. 14 Sequence analysis of the viral genome revealed the source of the virus to be a nearby domestic sheep herd. 14
In the present case, the disseminated lymphocytic arteritis, particularly involving the kidneys of the 2 sheep, is characteristic for MCF. 10 Although thrombosis of inflamed arteries is rare elsewhere in the body in MCF, it has been reported to occur in the renal arcuate and interlobular arteries in other cases of MCF and may lead to infarction, 10 as was seen in the present case. However, to the authors' knowledge, massive perirenal hemorrhage has not been previously reported in cases of MCF in any species or in any other diseases affecting wild or domestic sheep. It is suspected that the source of hemorrhage in the present case is the marked vasculitis in the kidneys, because spontaneous perirenal hemorrhage has been associated with renal vascular injury in humans. 3 In both sheep, the adherence of clotted blood to the renal cortical infarcts likely represents progression of this lesion, with initial vasculitis and hemorrhage leading to thrombosis and infarction.
