Abstract
Betaretrovirus-induced transmissible respiratory tumors in sheep arise at 2 distinct anatomic locations, either deep in the lung tissue caused by jaagsiekte sheep retrovirus (JSRV) or in the nasal cavity induced by ovine enzootic nasal tumor virus (ENTV-1). JSRV and ENTV-1 are found in many countries worldwide and have a significant economic and animal health impact. Although JSRV is endemic in sheep in the British Isles, ENTV-1 has not been reported. We report herein a nasal adenocarcinoma in a cull 8-y-old Belclare ewe from Ireland. The gross and microscopic features and immunohistochemistry results were consistent with an ENTV-1–associated tumor. However, differential PCR, using primers specific to regions of divergent sequence between the viruses, was performed on different parts of the adenocarcinoma and produced consistent results: positive for JSRV and negative for ENTV-1. An association of JSRV with nasal adenocarcinoma in sheep has not been reported previously, to our knowledge. Our case shows the necessity of using PCR in combination with immunohistochemistry to reach an accurate etiologic diagnosis, which is of importance in countries currently free of ENTV-1.
Nasal adenocarcinomas in sheep are most commonly caused by a betaretrovirus, known as species Enzootic nasal tumor virus 1 (ENTV-1; Retroviridae, Betaretrovirus). 17 ENTV-1 and the associated disease enzootic nasal adenocarcinoma (ENA) has never been reported in the UK or Ireland, although it is not uncommon in many European countries and naturally occurs on the other continents, excluding Australia and New Zealand. 3 ENTV-1 is closely related to species Jaagsiekte sheep retrovirus (JSRV; Retroviridae, Betaretrovirus), the causative agent of ovine pulmonary adenocarcinoma (OPA) 9 which is endemic in Ireland 4 and to ENTV-2 the causative agent of ENA in goats. 7 There is 90–98% homology between the genomes of JSRV, ENTV-1, ENTV-2 and also with enJSRV, an endogenous virus found in multiple copies in the sheep genome. 10 enJSRV is not associated with any lesions, but is proposed to be the reason why detectable immune responses are not raised against the exogenous viruses. 18 Despite their homology and the sharing of the same surface receptor for virus attachment, hyaluronidase 2 (HYAL-2), 6 JSRV is exclusively associated with neoplastic transformation of type II pneumocytes and club cells in the lung,9,12 whereas ENTV transforms secretory epithelial cells of the ethmoid turbinate in the nasal cavity. 13 Herein, we report a case of nasal adenocarcinoma in an adult sheep associated with JSRV infection.
An 8-y-old Belclare ewe was culled because of a hernia in the ventrolateral abdomen. The animal originated from a flock of 360 ewes in County Kildare, Ireland. Caseous lymphadenitis and OPA were endemic on the farm, and Heptavac P (MSD Animal Health, Dublin, Ireland) vaccination was routinely used. Postmortem examination conducted for student training revealed a large firm, gritty, pale-yellow to red mottled multilobulated mass (3.5 × 3 × 8 cm) bilaterally in the caudal nasal cavity, infiltrating the ethmoid bone, and effacing the nasal turbinates (Fig. 1). The tumor further extended through the vomer into the ventral nasal meatus and nasopharyngeal meatus. In addition to a large hernia (10 cm diameter) observed in the left ventrolateral abdominal wall, the ewe was in poor body condition with very little subcutaneous and visceral fat, the cervix was thickened by fibrosis, and the uterus contained small amounts of yellow thick mucoid material. A few small abscesses surrounded by extensive fibrosis were observed throughout the lung.
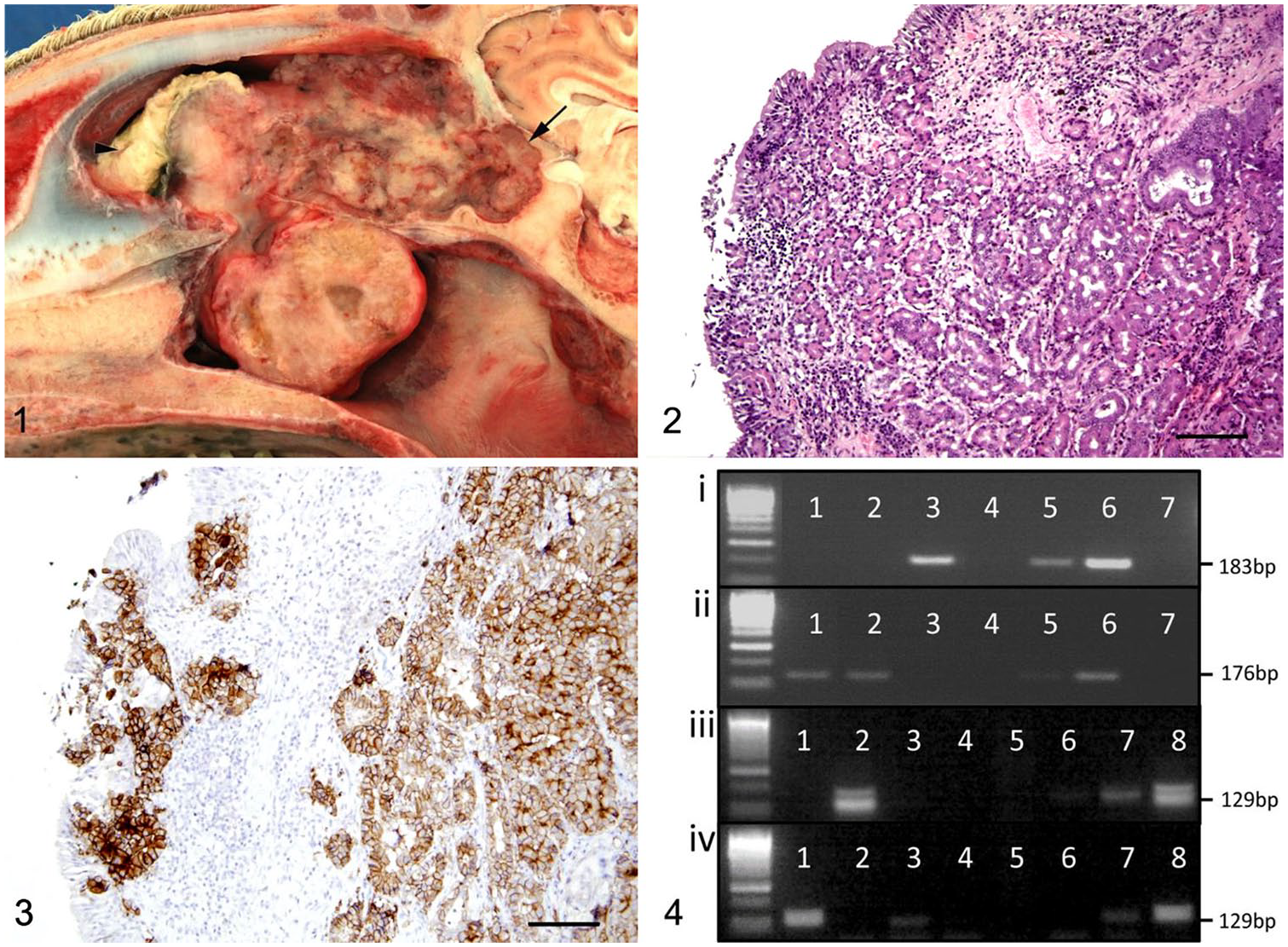
Nasal adenocarcinoma in a sheep.
Samples from 3 different areas in the nasal mass were fixed in 10% neutral-buffered formalin and processed routinely for microscopic examination. Histopathology revealed an infiltrative mass effacing the nasal turbinates composed of cuboidal cells forming acini, tubules, and papillary projections either densely arranged in lobules in a fine fibrovascular stroma or seen singly in a thick fibrous tissue stroma (Fig. 2). Nuclei were small, round, and centrally located. There was mild anisokaryosis and anisocytosis and ~2 mitotic figures in ten 400× fields. Mild accumulations of mucinous material covered the neoplastic cells. Marked infiltration of macrophages, lymphocytes, and plasma cells was seen. Infiltration of bone by the neoplasm and bone remodeling were present. A diagnosis of well-differentiated nasal adenocarcinoma was made.
Immunohistochemistry (IHC) for sheep betaretrovirus was performed on sections of the tumor using mouse monoclonal antibody raised against the SU subunit of the envelope protein of JSRV, as described previously. 19 This antibody cross-reacts with the other ovine betaretroviruses and has been used in the diagnosis of ENTV-1–associated nasal tumors. 16 Brown membranous labeling was seen in all of the neoplastic cells (Fig. 3), consistent with ENA in sheep caused by ENTV-1.
Given that this virus has never been reported in the British Isles, a PCR specific for ENTV-1 was conducted on frozen nasal tumor tissue to confirm the results. DNA was extracted (Tissue DNA extraction kit; Qiagen, Manchester, UK) according to the manufacturer’s instructions. The primers used for ENTV-1 were ENTV1tmCC3F (CTCACTCGAGRTTTAATTAAAGATTTCC) and ENTV1tmCC6r (GACAGGGAGCTTAGGTACTTGC). JSRV PCR employed primers P1 and P3 as described previously. 11 The reagents and cycle conditions were the same for both PCRs: 1 U Qiagen HotStarTaq polymerase, 1× Qiagen PCR mix, 0.5 mM of MgCl2, 0.2 µM of each primer, 0.2 mM dNTPs (Roche, Hertfordshire, UK), and ~100 ng of DNA, in a total volume of 40 µL; 15 min at 94°C, then 30 cycles of 15 s 94°C, 15 s 59°C, 15 s 72°C. The controls used were DNA from a previous confirmed ovine ENA case, normal sheep DNA (lung), DNA from a confirmed OPA case, dilution series of plasmids pJSRV21-VRU3 2 and ENTV-1 pTNO38eL, 1 and water.
The PCR results for the control samples were as follows: ovine ENA was positive for the ENTV-1 PCR and negative for the JSRV PCR, OPA tissue was positive for JSRV and negative for ENTV-1, and normal sheep lung was negative for both PCRs showing that enJSRV was not amplified. Surprisingly the nasal tumor sample of our case was negative for ENTV-1 and positive for JSRV by PCR (Fig. 4).
Because this result was unexpected, we repeated the PCR on DNA extracted from a different piece of tumor tissue from the same animal, this time from formalin-fixed, paraffin-embedded (FFPE) tissue from the center of the neoplasm. To exclude the possibility of cross-contamination, no OPA sample was extracted, and the procedure was conducted in a laboratory that does not work on OPA or JSRV.
In order to obtain a signal from the FFPE-extracted DNA, nested PCRs for JSRV or ENTV-1 were conducted. The first-round PCRs were as described above, then 5 µL of the product was used as template for the second (nested or hemi-nested) round of PCR. Reagents and cycling conditions were as described above. Nested primers were ENTV1tmCC4f (CCTACAAATGAGARTTGAATTAATACATA) and ENTV1thCC5r (TCCCAGGACTTAACCCTCCG) for ENTV-1 and P1 and P6 11 for JSRV. The results showed that this second sample of nasal adenocarcinoma from the same animal was also positive for JSRV and negative for ENTV-1 (Fig. 4).
From these results, we conclude that the nasal adenocarcinoma was associated with JSRV and not ENTV-1 infection. This is an unusual manifestation of JSRV that has not been reported previously, to our knowledge. However, diagnosis of ENA is often based on histopathology, 14 IHC, western blot, or electron microscopy 15 without further differentiation by PCR, and therefore may have been missed. Given the homology of the viruses, the anti-SU monoclonal antibodies that have been used widely for IHC of this family of viruses do not differentiate between ENTV-1, ENTV-2, JSRV, or enJSRV.16,19 For differential PCR, it is necessary to choose primers from regions of divergent sequence between the viruses, and then to confirm their specificity using appropriate controls. In our case, we repeated the PCR on a central part of the tumor to rule out possible cross-contamination from the nasal mucosa, which may carry JSRV in affected flocks without causing a tumor. 8 Coinfections of ENTV-1 and JSRV have been detected in sheep previously, but ENTV-1 was always found to be present in the nasal tumors. 8 The long terminal repeat (LTR) region of the virus has been suggested to restrict JSRV expression to particular cells in the lung that are transformed and develop into OPA. 10 Although JSRV is endemic in the flock in our study, the ewe had no gross evidence of OPA. Although sheep retroviruses are highly conserved,16,20 our findings do not rule out virus recombination or mutation of JSRV. The PCR primers used would not detect a recombinant between ENTV-1 and JSRV 5′ of the env gene given that the ENTV-1 primers are specific for ENTV-1 env gene whereas the JSRV PCR primers amplify the region from the 3′-end of env to U3 of the 3′-LTR. Therefore, we cannot exclude a mutant virus with an ENTV-1 5′-LTR. Additional PCR or complete sequencing of the virus could be used to investigate this possibility. It would be interesting to compare the sequence of the virus detected in this nasal tumor with JSRV detected in lung tissue of sheep on the same farm and to investigate whether any other cases of nasal adenocarcinoma are present in the flock.
The ewe did not have clinical signs suggestive of a nasal tumor and without the splitting of the head at autopsy the tumor would have gone undetected. The age of the ewe was much higher than generally reported for ENTV-associated ENA, which is generally < 4 y.5,15 Histopathologic changes were similar to those described for ENA.5,15,17
Fortunately, Ireland and the UK remain free of ENTV for the time being. However introduction is very possible and would be of animal welfare and economic importance to the sheep industry given that a prevalence of up to 15% has been reported in some countries. 3 In the absence of reliable preclinical antemortem tests and any treatment or protective vaccine, the presence of widespread subclinical infection would be difficult to control. 3 Our report highlights that little is known about ENA, even though this disease is common in other parts of the world, and emphasizes the importance of PCR validation of IHC or electron microscopy results.
Footnotes
Acknowledgements
We thank Brian Cloak, Alex Fawcett, and Susan Peters in the Pathobiology Section, School of Veterinary Medicine, University College Dublin, and Helen Todd and Val Forbes at the Moredun Research Institute for their technical assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
H. Jahns received no financial support for the research, authorship, and/or publication of this article. C. Cousens was funded by the Scottish Government’s Rural and Environment Science and Analytical Services Division (RESAS).
