Abstract
Rabies has been an enigmatic disease because microscopic findings in central nervous system tissues do not always correlate well with the severity of the clinical illness. Immunohistochemical staining of the calcium-binding protein calbindin (specifically CbD28k) seems to be the technique most used to identify Purkinje neurons under normal and pathological conditions. In the present work, we evaluated CbD28k immunoreactivity in the cerebellar cortex of normal and natural Rabies virus (RABV)-infected cattle. We examined brains from 3 normal cows and from 6 crossbreed cattle with a histologic diagnosis of rabies. Samples were taken from the cerebral cortex, cerebellum, hippocampus, and brainstem. Immunohistochemistry was carried out using the following primary antibodies: anti-RABV, anti-GFAP, and anti-CbD28k. In the cerebellar cortex, RABV infection caused the loss of CbD28k immunostaining in Purkinje cells; some large interneurons in the granular layer maintained their positive CbD28k immunoreaction. The identification of this loss of CbD28k reactivity in cerebellar Purkinje cells of RABV-infected cattle presents a potentially valuable tool to explore the impairment of Ca2+ homeostasis. In addition, this may become a useful method to identify specific molecular alterations associated with the higher prevalence of Negri bodies in Purkinje cells of cattle. Furthermore, we detected the presence of rabies viral antigens in different regions of the central nervous system, accompanied by microglial proliferation and mild reactive astrogliosis.
Introduction
Calcium plays a fundamental role in cells as a second messenger, and it is principally regulated by calcium-binding proteins (CaBPs). 3 Such proteins are in turn regulated by intracellular calcium levels. In neurons, the absence of calcium-buffer proteins results in marked abnormalities in cell firing, with alterations in simple and complex spikes or the transformation of synapses exhibiting depression into synapses exhibiting facilitation. In the cerebellum, where rapid-firing neurons such as Purkinje cells experience high level of calcium influx, tight calcium regulation is essential to avoid rapid neuronal death because of excessive excitatory stimulation. 3 The CaBP calbindin D28kd (CbD28k), a neuronal marker of great interest in neuroanatomy and neuropathology,10,40 is highly expressed in bovine Purkinje cells 41 and has been suggested as a marker of Purkinje cells in normal and degenerative cerebellar tissue in different species.12,18,32,36,41 Its expression could be affected during aging in dogs, 36 and it is specifically expressed early in cerebellar development in Purkinje cells in fetuses and newborn kittens during neuronal migration. 32 In humans, differential CbD28k immunoreactivity is seen between 2 other large inhibitory interneurons of the cerebellar granular layer: Golgi cells, which are immunopositive to CbD28k, and Lugaro cells, which can be immunonegative 3 or immunopositive, 10 although differences in these immunostaining patterns are known to occur in other vertebrate species. 3 In cattle, there is no previous work specifically describing CbD28k immunoreactivity and the responses in both of the large GABAergic interneurons of the cerebellar granular layer our knowledge.
In the cerebellar cortex, neural circuits rely on inhibition mediated by γ-aminobutyric acid (GABA) to shape the spatiotemporal patterns of electrical signaling in different species. 14 The cellular and molecular mechanisms that specify and modify these inhibitory innervation patterns are only just beginning to be understood. 14 Purkinje cells are the sole output neurons of the cerebellum, interacting with other GABAergic interneurons of the cerebellar cortex to define their inhibitory output. 13
Previous reports of various neurodegenerative diseases postulate a relationship among dysfunctions in GABAergic neurotransmission and changes in CbD28k immunoreactivity in different regions of the central nervous system (CNS).36,40 In rabies, some clinical signs and experimental trials in cell cultures and mice support the possible involvement of the GABAergic systems during viral infection.17,20,21,40 In addition, a possible relationship among changes in CbD28k immunoreactivity and dysfunction in GABAergic neurons in the telencephalic cortex, corpus striatum, and spinal cord in experimental Rabies virus (RABV)-infected mice has been suggested.24,31,39,40
In other viral encephalitides, the use of CbD28k as a molecular marker of intracellular calcium homeostasis, or to detect early neurodegeneration, has been very rare. In Human immunodeficiency virus (HIV) encephalitis, decreased CbD28k immunoreactivity in cerebral cortex is detected. 30 In rats, experimental neonatal Borna disease viral infection induces a loss of cerebellar Purkinje cells that is easily detected using CbD28k as a specific marker of these neurons. 9 In scrapie-affected adult sheep, the assessment of CbD28k, parvalbumin, and calretinin also demonstrate that the use of these CaBPs could be a valuable diagnostic tool to identify specific alterations in this encephalopathy, showing that the cerebellar cortex—mainly the Purkinje cell layer—is the sole CNS region with positive CbD28k immunolabeling, although this immunoreactivity is not affected by scrapie.38,42
The purpose of the present work was to describe the distribution of CbD28k immunoreactivity in the cerebellar cortex of RABV-infected cattle, and to characterize this antibody as a specific tool to detect alterations in this CaBP and therefore likely changes in calcium homeostasis in the cerebellar cortex during RABV-induced neurodegeneration. Samples from different regions of the nervous system were also evaluated with immunohistochemistry (IHC) to confirm the presence of both lyssaviral ribonucleoprotein in the nonsuppurative meningoencephalitis caused by RABV, and the degree of reactive astrogliosis, using glial fibrillary acidic protein (GFAP).
Materials and methods
Cases
Six cases of natural RABV-infected crossbreed cattle, 1–3 years of age, confirmed previously by fluorescent antibody test (FAT) during 2010–2011 by the Sanitary Defense Service of Goiás, Brazil, and 3 normal cows of similar age, used as controls, were obtained from the archives of the Laboratory of Veterinary Pathology (LPV), Federal University of Goiás, Jataí, Brazil. All formalin-fixed, paraffin-embedded (FFPE) samples from cerebellum, cerebral cortex (frontal, parietal, and occipital), hippocampus, and brainstem (midbrain, pons, and medulla oblongata) were sectioned at 5 µm thickness. At least 1 slice from each region was stained with hematoxylin and eosin (HE) for histologic characterization of viral nonsuppurative encephalitis to identify intracytoplasmic Negri bodies in neurons.
Immunohistochemistry
All tissues were examined by IHC using the following protocol to detect lyssaviral ribonucleoprotein, CbD28k, and GFAP. After deparaffinization, tissue sections were quenched with hydrogen peroxide for 5 min a and then rinsed with water. The antigenic sites were exposed with citrate buffer solution for 10 min (two 5-min intervals) in a steam cooker, followed by blocking in casein solution for 10 min. b Primary antibodies were incubated overnight at 4°C followed by two 15-min room temperature incubations in biotinylated link antibody and horseradish peroxidase–conjugated streptavidin, c respectively. Sections were incubated with diaminobenzidine for a maximum of 5 min, c giving a brown reaction at sites of antibody binding. The slides were counterstained with Mayer hematoxylin and then coverslipped. d Pertinent data of primary antibodies used are detailed in Table 1.
Immunohistochemistry antibodies used to evaluate calbindin (Cb)D28k distribution to detect lyssaviral ribonucleoprotein and reactive astrogliosis in brains of Rabies virus (RABV)-infected cattle.*
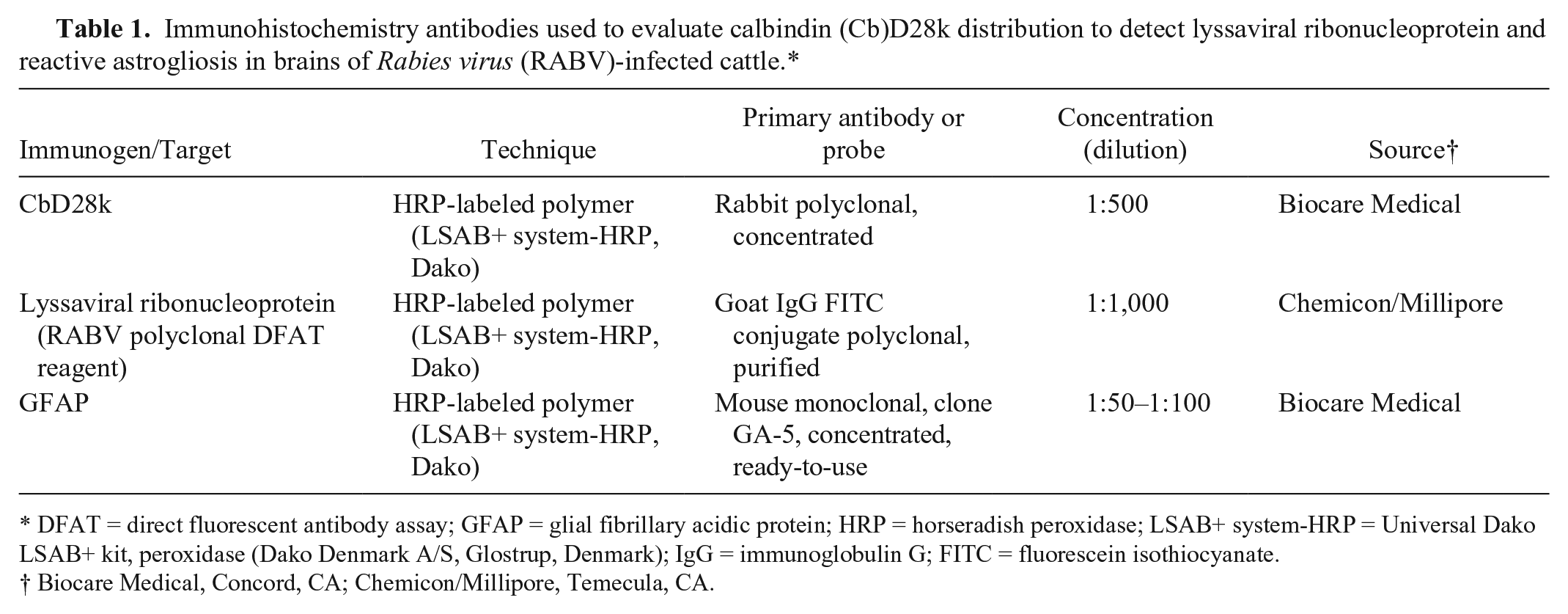
DFAT = direct fluorescent antibody assay; GFAP = glial fibrillary acidic protein; HRP = horseradish peroxidase; LSAB+ system-HRP = Universal Dako LSAB+ kit, peroxidase (Dako Denmark A/S, Glostrup, Denmark); IgG = immunoglobulin G; FITC = fluorescein isothiocyanate.
Biocare Medical, Concord, CA; Chemicon/Millipore, Temecula, CA.
Western blotting
The specificity of bovine CNS tissue identification using the previously mentioned primary antibodies against CbD28k and GFAP was tested in samples obtained from several regions of the CNS (frontal cortex, cerebellar cortex and peduncles, and medulla oblongata) of a normal 2-year-old Hereford cow. Frozen samples were homogenized in lysis buffer (20 mM Tris pH 7.4, 100 mM NaCl, 1 mM EDTA, and 0.5% NP-40 e ) containing protease inhibitor cocktail f and centrifuged at 10,000 × g for 15 min at 4°C. Protein concentration was determined in the resultant supernatants using the Bradford assay. A total of 20 µg of total protein per sample were used to perform Coomassie blue–stained gels and Western blots. To detect GFAP, we used a monoclonal antibody (mAb) raised against a purified GFAP from pig spinal cord. CbD28k protein was detected using a polyclonal antibody (pAb) raised against the whole molecule. Western blots were developed using chemiluminescence. g Whole mouse brain extract was used as a positive control, and proteins extracted from liver were used as a negative control.
To evaluate CbD28k immunoreactivity, 5 slides of the cerebellum from each case were prepared. Specific immunoreactivity of cerebellar Purkinje neurons and large granular layer interneurons from natural RABV-infected cattle were evaluated by counting positive and negative cells in 25 microscope fields from each slide at 100×.
Results
In the natural RABV-infected cattle, there was no apparent association between neurologic signs and CNS lesions. The presence of severe neurologic signs (i.e., mainly aggressiveness, drooling, and incoordination) did not consistently correlate with gross or histopathologic changes that could explain these neurologic signs, and vice versa.
In all 6 cases, the RABV-infected animals died 1–3 days after the onset of clinical signs, and were FAT-positive for RABV after death. The affected animals showed mild to severe multifocal lymphoplasmacytic encephalitis or meningoencephalitis, with lesions mainly in the cerebellum, midbrain, pons, medulla oblongata, and thalamus. In 1 case, lesions were also noted in the basal nuclei, hippocampus, and frontal, parietal, and occipital cortex. Eosinophilic intracytoplasmic inclusions (Negri bodies) were always observed in neurons from different CNS regions, but these inclusions were more abundant in the cerebellar cortex, mainly in Purkinje neurons (Fig. 1). In the cerebellar cortex, the histopathologic changes consisted of neuronal chromatolysis, Babès nodules, multifocal gliosis, and mild to moderate lymphoplasmacytic perivascular cuffing (Figs. 2, 4). The presence of Negri bodies in the cerebellar cortex was not associated with the severity of the observed inflammatory changes, with mild inflammation and moderate to high numbers of Negri bodies in the studied cases.
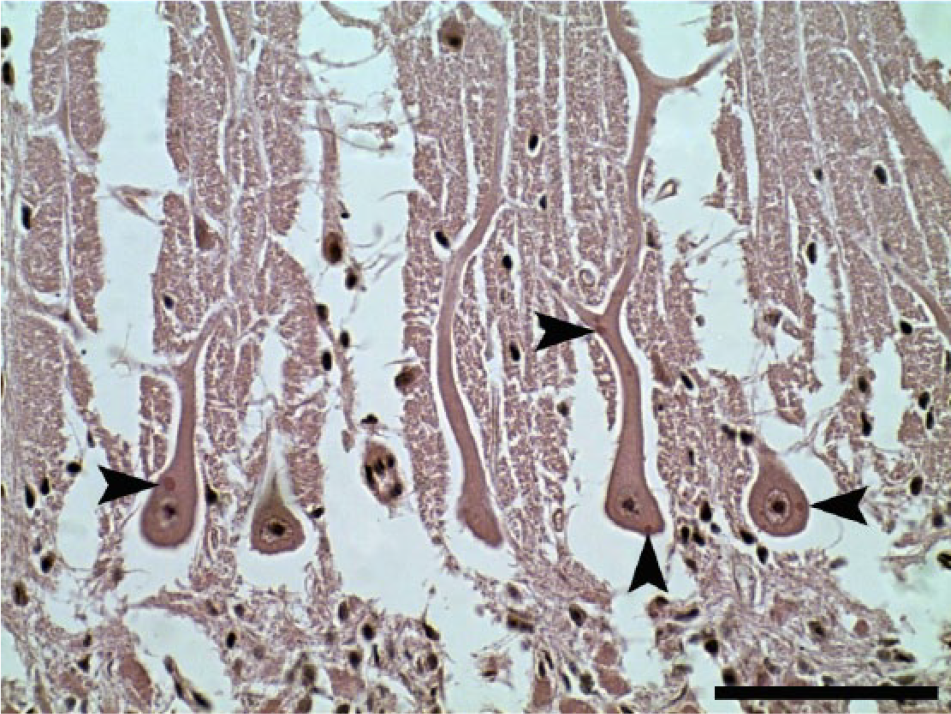
Cerebellum of Rabies virus–infected cow (case 4). Negri bodies in Purkinje cells (arrowheads). Hematoxylin and eosin. Bar = 75 µm.
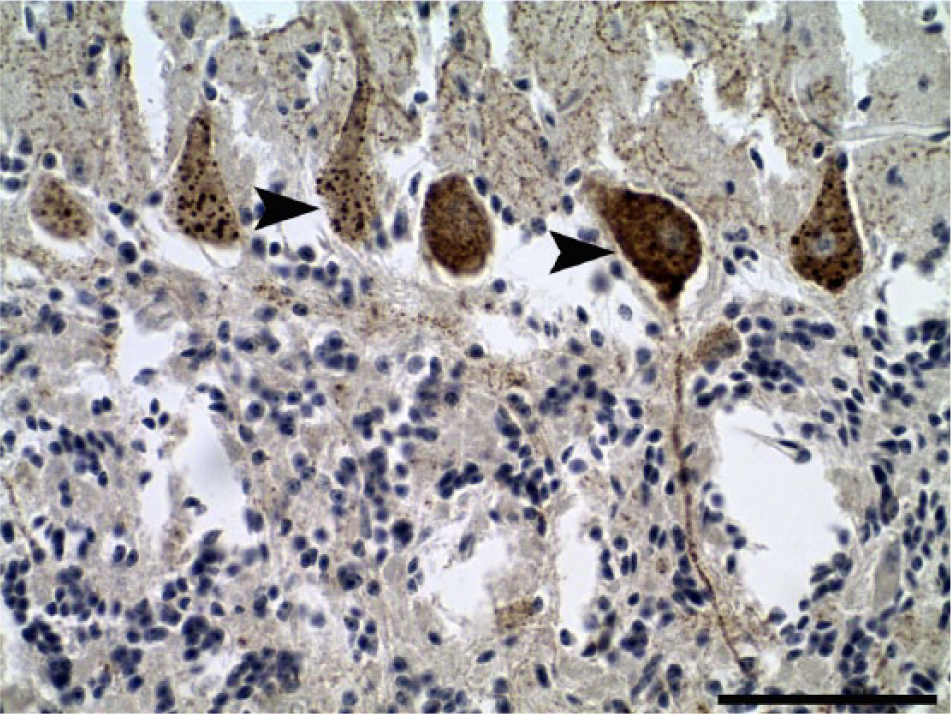
Cerebellum of Rabies virus–infected cow (case 6). Intracytoplasmic brown, oval, homogeneous structures within the perikaryon of Purkinje cells, and dendritic and axonal processes (arrowheads). Lyssaviral ribonucleoprotein immunohistochemistry. Bar = 75 µm.
Lyssaviral ribonucleoprotein (RABV) IHC showed positive immunoreaction in all samples of the 6 cases studied, mainly in the cerebellum and brainstem. In general, this finding was observed in gray matter and was characterized by sparse to diffuse granularity throughout the perikaryon of neurons and their axonal and dendritic processes. In some cases, inclusions appeared as brown, oval, homogeneous structures within the perikaryon of neurons, corresponding to the identification of Negri bodies using HE stain. In the cerebellar cortex, these immunostained intracytoplasmic inclusions were most frequent in the Purkinje neurons (Fig. 2), but were present in lower numbers in interneurons from the molecular and granular layers.
GFAP immunoreactivity revealed diffuse and mild reactive astrocytosis in the RABV-infected cases, with foci of reactive Bergmann glia in the cerebellar molecular layer (Fig. 3), accompanied by focal and diffuse reactive microglia with formation of microglial (Babès) nodules. The reactive astrogliosis detected by GFAP in cerebellar cortex did not have any evident association with the abundance of Negri bodies or RABV-positive immunoreaction in the affected Purkinje cells.
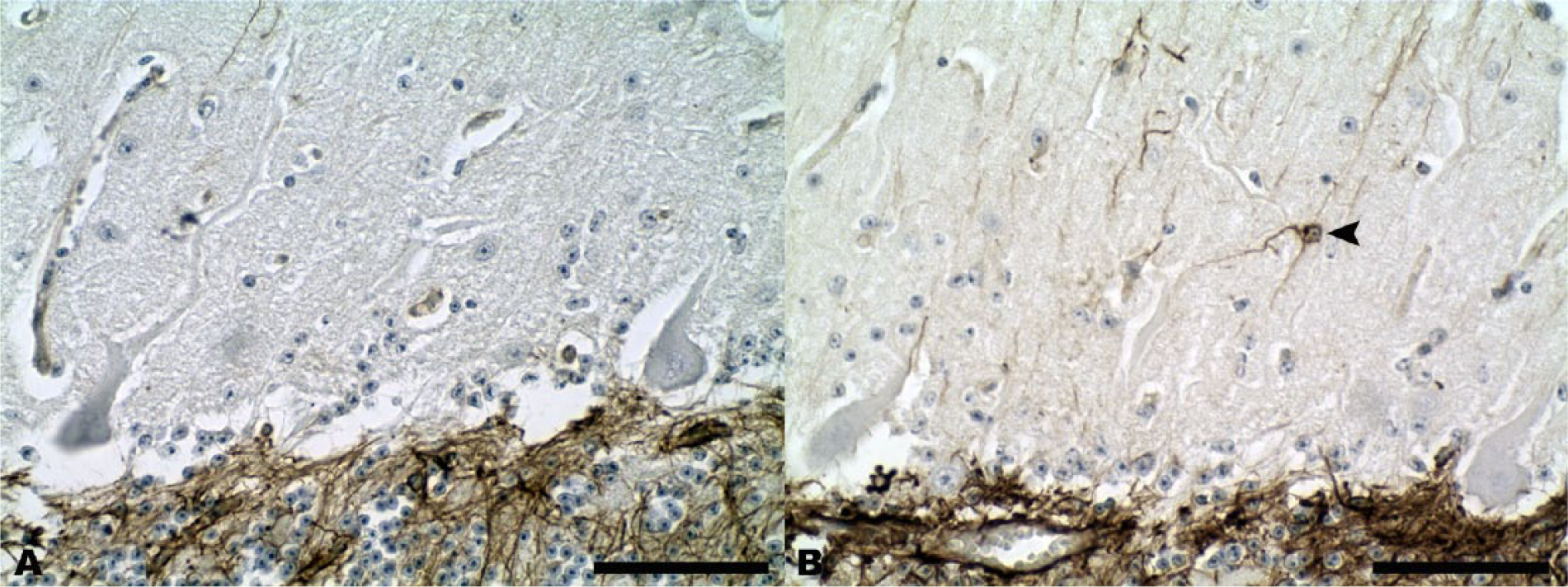
Cerebellar cortex.
In normal animals, CbD28k showed a specific and intense signal in the perikaryon, dendritic trees, and axonal processes in almost all normal cerebellar Purkinje neurons, and in groups of large interneurons from the granular layer (Fig. 4A, 4C). However, this specific immunoreactivity of Purkinje neurons was not found in RABV-infected cattle, with the maintenance of CbD28k immunolabeling of the large interneurons in the cerebellar granular layer (Fig. 4B, 4D). The specific loss of CbD28k immunoreactivity in Purkinje cells showed a clear relationship with the abundance of Negri bodies or RABV-positive immunoreaction in this neuronal type. The results of immunoreactions with CbD28k are summarized in Table 2.
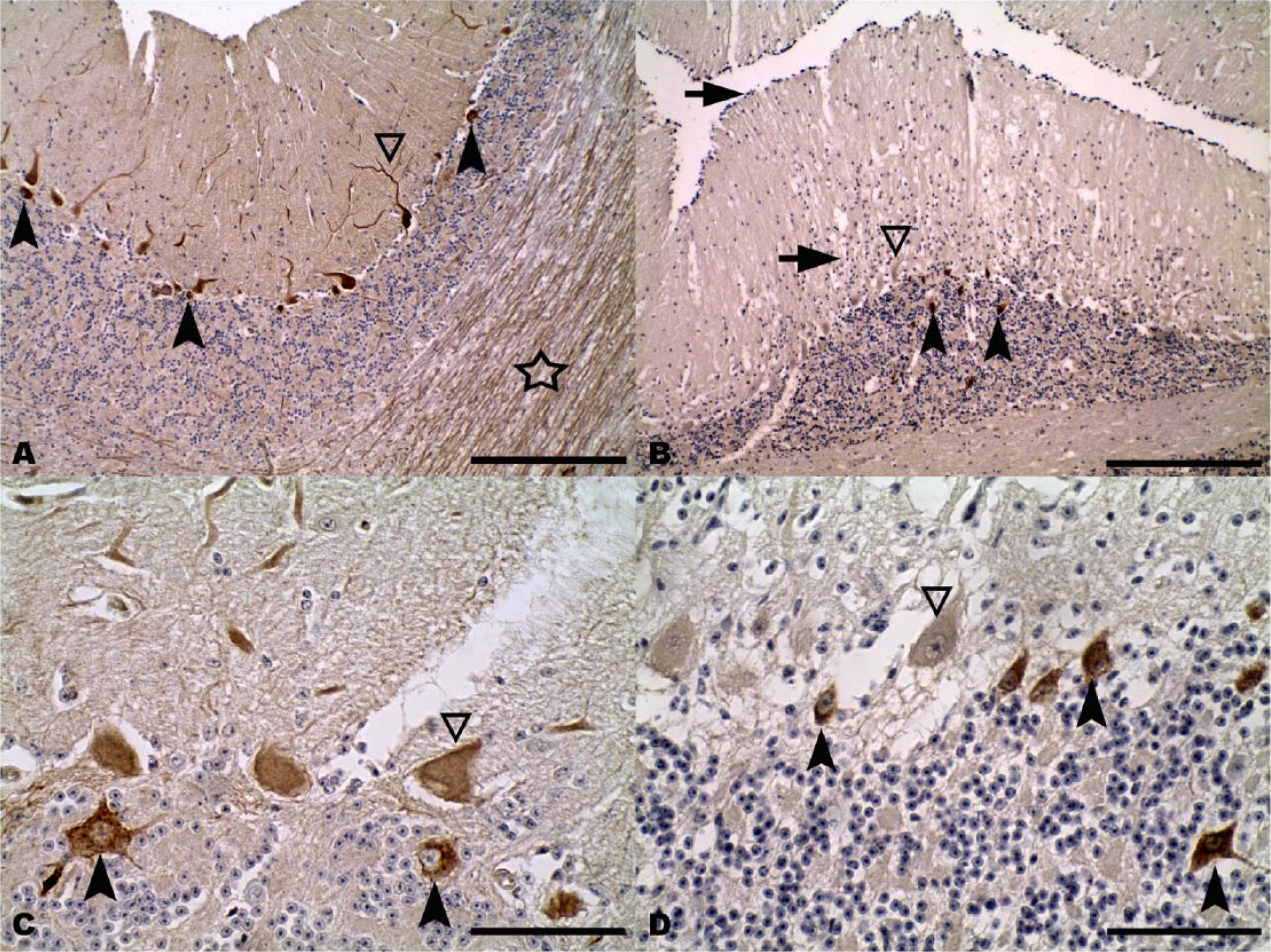
Cerebellar cortex.
Immunoreactivity of calbindin (Cb)D28k in Purkinje neurons and large interneurons in the granular layer of the cerebellar cortex of Rabies virus–infected cattle.*
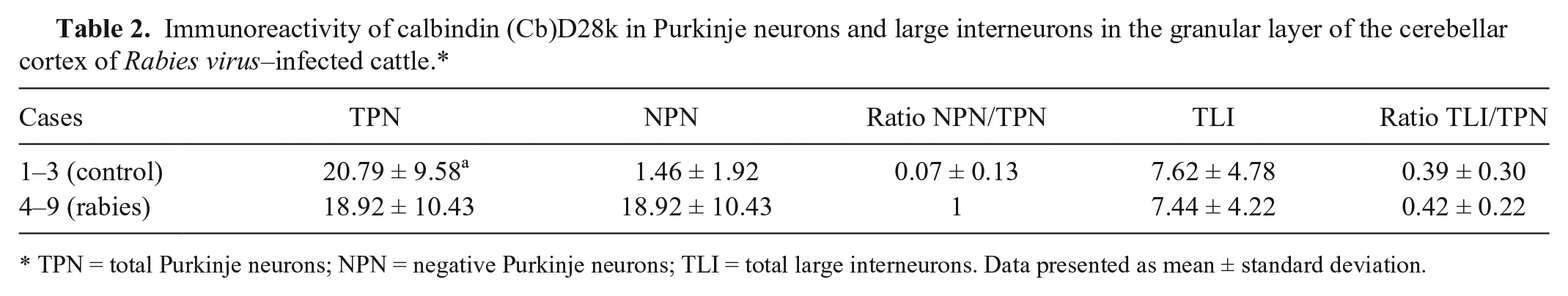
TPN = total Purkinje neurons; NPN = negative Purkinje neurons; TLI = total large interneurons. Data presented as mean ± standard deviation.
The specificity of CbD28k and GFAP protein identification in bovine CNS samples was demonstrated using the Western blot technique, showing in the case of CbD28k a strong reaction with a single band in the cerebellar cortex and cerebellar peduncle samples, with a subtle reaction in the medulla oblongata, and an absence of any reaction in the left and right regions of the forebrain cortex. In the case of GFAP, a strong reaction was detected mainly in the medulla oblongata, the cerebellar cortex, and the cerebellar peduncles, with a pattern of several bands corresponding to different GFAP isoforms present in these CNS regions; in the case of the left and right forebrain cortex, only 1 light band was detected by the GFAP antibody (Fig. 5).
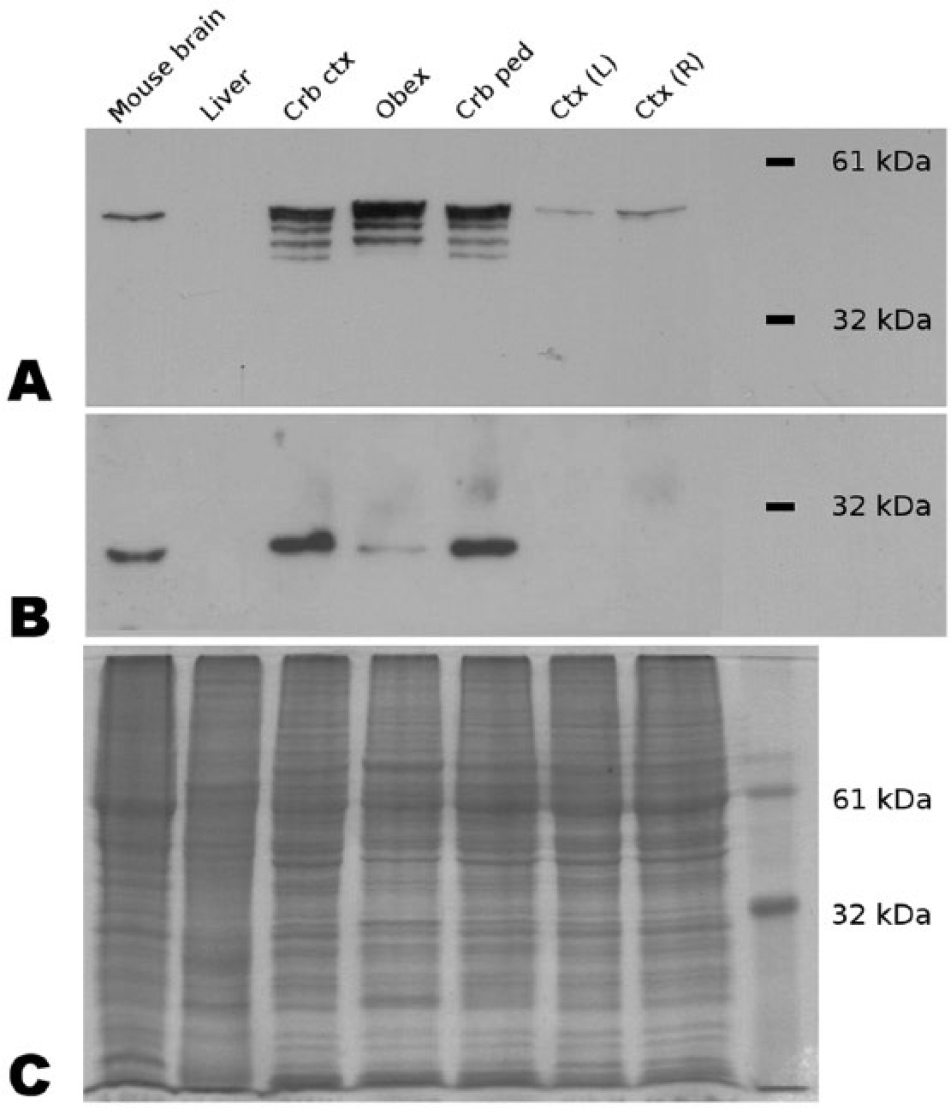
Bovine central nervous system tissue specificity of primary antibodies glial fibrillary acidic protein (GFAP) and calbindin (Cb)D28k. Anti-GFAP and anti-CbD28k antibodies specifically recognize bovine proteins. Western blots for GFAP (
Discussion
In South America, the paralytic form of rabies in herbivores occurs in cyclic outbreaks, with a periodic reappearance in 3–7 years, and is transmitted by the vampire bat Desmodus rotundus. Rabies has been considered an enigmatic neuropathologic disease, because the severity of its clinical signs were not associated with equivalent morphologic changes in the affected brain.1,2,7,15 In the present study, all of the animals died 1–3 days after the onset of clinical signs, and this early death, with the pathogenic properties and/or variations of RABV strains, could contribute to the absence of an association of neurologic signs and lesions in the CNS. Microscopic changes are subtle and inconspicuous in the nervous tissue when studied by conventional HE histologic methods.19,40
In our cases, each CNS sample was subjectively compared, and the histologic studies with HE confirmed the diagnosis of nonsuppurative meningoencephalitis with eosinophilic intracytoplasmic inclusions (Negri bodies) always present in the neurons, especially in the Purkinje neurons and their dendrites and axonal processes, confirming similar results previously reported in ruminants.27,43 The severity of inflammatory changes were not associated with the presence, size, or abundance of Negri bodies in the neurons, supporting previous reports on RABV-infected cattle under similar environmental conditions. 27
In accord with the RABV FAT results, IHC demonstrated the presence of RABV antigen in all of the studied cases, similar to previous reports in mammals in which the same polyclonal primary antibodies (pAbs) were used.29,33,37 In agreement with these reports, the pAbs accurately detected RABV in FFPE samples from the CNS of cattle, probably with more sensitivity than monoclonal primary antibodies, because pAbs would potentially detect a wider range of RABV strains, and could be useful to differentiate other non-rabid inclusion bodies in bovine CNS in cases where rabies was suspected. 37
In the cerebellum, Purkinje neurons were the best neuronal type to study Negri bodies, and also to evaluate RABV immunoreactivity. Furthermore, in the cerebellar cortex, other immunopositive large interneurons were identified in the granular layer, probably Golgi and/or Lugaro cells, in a pattern similar to that which was previously reported. 37 These findings agree with previous reports in cattle and other herbivores,4,25,27,43 which are unlike the immunoreactivity in the cerebellum of RABV-infected horses.5,37 The IHC protocol used here confirms its utility as a safe alternative to FAT, by preserving the tissue architecture and allowing histologic evaluation to reach a diagnosis.25,29,33,34,37
GFAP immunoreactivity showed a mild astroglial reaction in CNS tissue, thus confirming our results from HE histopathology and a previous report that used another type of GFAP antibody in RABV-infected cattle, 28 along with multifocal microglial proliferation. In the cerebellar cortex, patches of reactive Bergmann glia were evident occasionally in the cerebellar molecular layer with GFAP, accompanied by extensive multifocal proliferation of microglia organized as Babès nodules, which agrees with other reports.29,33
The specificity to bovine CNS tissues of the GFAP mAb used here was tested by Western blot, demonstrating that our antibody recognized a specific pattern of GFAP immunolabeling in different regions of the bovine CNS. Particularly in the medulla oblongata and cerebellum, the identification of several bands could represent the expression of different GFAP isoforms in these areas, in comparison with the less abundant expression of GFAP in the forebrain cortex.
The distribution of CbD28k immunoreactivity in the cerebellar cortex in normal cattle was similar to that reported in our previously published work using another mAb. 41 In the present report, we used a rabbit pAb that was tested by Western blot to demonstrate its specificity to bovine CNS tissues, confirming our previous results in the cerebellum of normal cattle 41 ; the results demonstrated intense and highly specific immunolabeling in the cerebellum, and poor immunolabeling in other regions of the bovine CNS. This high specificity to cerebellar tissues was confirmed in normal cattle, and similar results were reported in adult sheep brains. 42 In our cases, all of the animals were adults of similar age (1–3 years old), therefore we did not see differences in the typical IHC pattern related to age effects, as could occur in other species during aging. 36
The typical IHC pattern in normal bovine cerebellum consisted of strong immunoreactivity of Purkinje neurons and their dendrites and axonal processes, and also some other large GABAergic interneurons from the granular layer, which are probably Golgi or Lugaro cells. Although both cells have a specific shape and are localized in the granular layer,8,22,23 morphologically it is difficult to differentiate them. Both are GABAergic interneurons, although in other species Golgi cells are CbD28k immunopositive and Lugaro cells are immunonegative, probably determined by the expression of other specific CABPs. 3 The current study describes the loss of CbD28k immunoreactivity in cerebellar Purkinje neurons in RABV-infected cattle, thus confirming the alteration of calcium homeostasis in this inhibitory neuronal type. These findings may have resulted from an excitotoxic overload and the alteration of neurotransmission mediated by GABA in these neurons. 16 In agreement with this possibility, previous work in other regions of the CNS in experimental cases of rabies suggested a possible involvement of the GABAergic systems during RABV infection.17,21,40 In our cases, CbD28k immunoreactivity also showed that other types of GABAergic interneurons (probably Golgi and/or Lugaro cells) were CbD28k immunopositive, similar to findings in other species.3,6,10,11,26,35 Our results studying RABV-infected cattle indicate that CbD28k immunolabeling results in a useful diagnostic tool to identify a differential response in the modulation of their calcium internal buffering. In the case of the rapid-firing Purkinje neurons, loss of CbD28k immunoreactivity was observed, and in the case of the GABAergic interneurons from the granular layer, there was maintenance of CbD28k immunoreactivity, similar to the immunolabeling observed in normal cattle.
Our results demonstrate that Purkinje cells have altered calcium homeostasis that could affect inhibitory output from the cerebellar cortex mediated by GABA in other regions of the CNS—mainly the cerebellar modulation of descending motor systems—that could affect some neurologic signs, such as incoordination, which is frequently described in RABV-infected cattle. 27 Previous reports of different viral encephalitides from HIV encephalitis in humans 30 to neonatal Borna disease infection in rats, 9 and experimental infection of rabies in mice, 40 or aging in dogs, 36 postulate a relationship between loss of CbD28k immunoreactivity and dysfunctions in GABAergic neurotransmission.
Further studies using CbD28k and/or other CABPs are needed to better understand the specific pathogenic mechanisms of neurodegeneration and their putative relationship with GABAergic neurotransmission systems in RABV-infected cattle, and could provide a specific tool to confirm similar mechanisms in other RABV-infected animals, or in other viral encephalitides.
Footnotes
Acknowledgements
We thank Eduardo Aguirre (UdelaR, Uruguay), Claudio Borteiro (Museo Nacional de Historia Natural, Uruguay), and Jim Pfister (PPRL, USDA, Logan, Utah) for their assistance, constructive criticism, and proofreading.
Authors’ contributions
JM Verdes contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; and critically revised the manuscript. FJF Sant’Ana contributed to conception of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; and critically revised the manuscript. MJ Sabalsagaray and A Calliari contributed to design of the study and contributed to acquisition and analysis of data. K Okada contributed to design of the study; contributed to acquisition and analysis of data; and critically revised the manuscript. JA Moraña contributed to design of the study; contributed to acquisition of data; and critically revised the manuscript. CSL de Barros contributed to conception of the study; contributed to interpretation of data; and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Peroxidazed 1 blocking reagent, Biocare Medical, Concord, CA.
b.
Background Sniper blocking reagent, Biocare Medical, Concord, CA.
c.
LSAB+ system-HRP, Dako Denmark A/S, Glostrup, Denmark.
d.
Permount mounting medium, Thermo Fisher Scientific, Waltham, MA.
e.
Nonidet P-40, Sigma-Aldrich, Milwaukee, WI.
f.
Protease inhibitor cocktail (catalog no. M221), Amresco Inc., Solon, OH.
g.
Invitrogen Novex ECL chemiluminescent substrate reagent kit (catalog no. WP20005), Thermo Fisher Scientific, Waltham, MA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Covenant ProSul (National Council of Scientific and Technological Development [CNPq] grant 490389/2008-2), Brazil, and the Committee of Scientific Research of the University of the Republic (CSIC, UdelaR) and Basic Science Development Program (PEDECIBA), Uruguay. JM Verdes and A Calliari are Research Career Members of the National Research System (SNI-ANII), Uruguay. K Okada was Senior Volunteer of the Japan International Cooperation Agency, Japan. CSL de Barros is a recipient of CNPq fellowships.
