Abstract
A novel herpesvirus was detected in a 17-day-old Chilean flamingo (Phoenicopterus chilensis) with pneumonia, hepatopathy, and severe anemia that was housed in California. Postmortem examination identified a pale, enlarged liver, mildly increased fluid in the lungs, and red foci in the spleen. Histologic examination revealed marked hepatic necrosis with syncytia, splenic necrosis, and interstitial pneumonia with eosinophilic intranuclear inclusions within hepatocytes and in unidentified cells of the lung. Transmission electron microscopy identified virions consistent with a herpesvirus in the nucleus and cytoplasm of degenerative hepatocytes. Nested consensus PCR, sequencing, and phylogenetic analysis identified a novel herpesvirus within the genus Iltovirus in the subfamily Alphaherpesvirinae.
Keywords
Introduction
Flamingos (family Phoenicopteridae) consist of 6 extant species, 3 of which are near threatened (Phoenicopterus chilensis, P. minor, and Phoenicoparrus jamesi) and 1 species (Phoenicoparrus andinus) that is classified by the International Union for Conservation of Nature as vulnerable as a result of a rapid decline in the wild population over the previous 3 generations (http://goo.gl/V6rWDU). The near threatened Chilean flamingo (P. chilensis) is a New World species of flamingo, native to southwestern South America, predominantly Chile, Argentina, and Bolivia. Part of the “shallow-keeled” subgroup of flamingos, 23 the Chilean flamingo has a highly specialized ecology and thrives in estuaries, lagoons, salt lakes, and other brackish water environments. A census in 2010 tallied 283,000 individuals, and a total population estimate was made at 300,000 (http://goo.gl/V6rWDU). Highly gregarious, this species can be found in extremely large flocks in the wild, numbering in the tens of thousands. Flamingos are sporadic breeders, reproduction seems highly dependent on environmental conditions, and flocks can go many years without successful reproduction. 4 Chilean flamingos are predicted to undergo a moderate to rapid population decline over the next 3 generations. Factors affecting this predicted decline include egg harvesting, disturbance and degradation of appropriate habitat caused by tourism and farming, and hunting (http://goo.gl/V6rWDU).
Flamingos in captive collections have few viral diseases, the most common of which are West Nile virus 25 and cutaneous poxvirus.16,26,34 However, captive flamingos have been shown to be susceptible to other viral diseases of importance, such as avian influenza 5 and Newcastle disease. 10 Although herpesvirus outbreaks have been reported in 1 population of captive American flamingos within the United States, 17 no sequence data or phylogenetic analyses of fatal herpesvirus infections in captive or wild flamingos is currently available. It has been reported that herpesvirus sequences were identified in pododermatitis lesions in greater flamingos (Phoenicopterus roseus). 33 Herpesvirus DNA was seen in only a few cases, and it was concluded that the viruses were not the cause of the lesions.
Herpesviruses are large, linear, double-stranded DNA viruses, with genome ranges from 125 to 295 kbp in size. 19 Herpesviruses exhibit a high degree of host specificity and sequence conservation, and are thought to coevolve with their host species. Herpesviruses are an ancient viral lineage that predate the reptilian–mammalian divergence, and genetic analyses suggest herpesviruses may have existed for over 400 million years. 15 The DNA-dependent DNA polymerase gene of herpesviruses is one of roughly 40 highly conserved genes 15 and has been shown to be an excellent target for PCR detection. 30
Described herein is a novel herpesvirus that caused fatal disease in a neonatal captive Chilean flamingo. The virus was identified in affected tissues by PCR, then sequenced and subjected to phylogenetic analysis to confirm its identity.
Materials and methods
Case and pathology
A parent-reared 15-day-old Chilean flamingo chick housed in an open exhibit was observed to be lethargic on the nest, and although the parent birds had been attentive to the chick, no feeding had been observed during the previous 2 days. The chick was hospitalized, and an examination revealed a low body weight (240 g), weakness with labored respiration, in addition to intermittent mild gurgling sounds and a sporadic cough or sneeze. During the pinioning at 1 day old, unusually excessive bleeding occurred at the pinion site, which required the placement of sutures for hemostasis. The chick was also noted to have a low birth weight (90 g) compared with other parent-reared conspecifics born at this facility, which average 121 g. Blood was collected from the jugular vein for hematology and biochemistry. Respiratory infection was suspected, and the chick was hospitalized in an incubator. Antibiotic therapy (clarithromycin 10 mg/kg orally, once a day, and ceftiofur 20 mg/kg subcutaneously), antifungal treatment (itraconazole 10 mg/kg orally, twice a day), subcutaneous fluids (lactated Ringer solution containing 5% dextrose), and tube feeding were initiated. The hemogram identified a very low packed cell volume of 10% with rare polychromasia. The white blood cell count and differential were unremarkable. Abnormal chemistry analytes included an elevated aspartate aminotransferase concentration of 1,797 U/L (normal range: 0–318 U/L), mild hyponatremia of 135 meq/L (normal: 136–158 meq/L), and a mild hypoalbuminemia of 0.8 g/dL (normal: 0.9–2.2 g/dL). 8 There was no noted change in the chick’s condition the following day. Intermittent gurgling sounds and sneezing continued, and the crop appeared to be emptying slowly after tube feeding. The chick was found dead (at 17 days of age) the following day, and a postmortem examination was performed. Tissues were fixed in 10% neutral buffered formalin, processed routinely, sectioned at 5 μm thickness, mounted on frosted glass slides, and stained with hematoxylin and eosin (HE). Select tissues were frozen for future reference.
For the transmission electron microscopy images, formalin-fixed sections of liver were post-fixed in 2.5% glutaraldehyde, then postfixed in 2% osmium tetroxide, and embedded in epoxy resin. a Thick sections were mounted on glass slides, stained with toluidine blue, and examined by light microscopy. Nine hundred angstrom thin sections were mounted on a 200 mesh copper grid and stained with uranyl acetate and lead citrate. Examination was conducted using a 120-kv transmission electron microscope. a Digital images were obtained using a fiber optic charge coupled device digital camera. b
PCR and sequencing
DNA was extracted from a frozen liver sample, c and blank extractions were used as negative controls. A nested PCR amplification was performed using consensus primers for the DNA-dependent DNA polymerase gene found in herpesviruses, as previously described. 30 The second round of amplification was modified, using DFA as the forward primer instead of the TGV primer as previously described. The PCR amplicon was resolved in 1% agarose gels, excised, and purified using a commercial kit. d The PCR amplicon was then sequenced via the Sanger method with a commercial kit e and analyzed on an automated DNA sequencer. e Amplicons were sequenced twice in each direction.
Phylogenetic analysis
The viral sequence obtained was compared with sequences in the databases of GenBank (National Center for Biotechnology Information, Bethesda, Maryland), EMBL (Cambridge, United Kingdom), and the Data Bank of Japan (Mishima, Shizuoka, Japan) using BLASTX. 2 The predicted homologous 153–166 amino acid partial sequences of herpesvirus DNA-dependent DNA polymerase were aligned using MAFFT. 12 Iguanid herpesvirus 2 (IgHV-2) was selected as the outgroup because of its early divergence from other herpesviruses. 13 Bayesian analyses of each alignment were conducted using Mr. Bayes 3.2.2 on the CIPRES server with mixed amino acid substitution models, gamma distributed rate variation, and a proportion of invariable sites. 22 A total of 4 chains were run, and statistical convergence was assessed via the average standard deviation of split frequencies and potential scale reduction factors of parameters. The initial 25% of 2,000,000 iterations were discarded as burn in.
Maximum likelihood (ML) analyses of each alignment was performed using RAxML-HPC2 version 8.0.24 on the CIPRES server, 24 with a gamma distributed rate variation and proportion of invariable sites. IgHV-2 was selected as the outgroup. ProtTest server version 2.4 was used for model selection. 1 Bootstrap analysis was used to test the strength of the tree topology; 1,000 re-samplings were performed. 6
Results
Pathology
Postmortem examination findings included generalized pallor, a diffusely, mildly enlarged liver, a reddened spleen, scant fluid in the lungs, moderate hematoma formation in the right jugular region, and the previously fed slurry in the crop. Histologic examination identified coalescing zones of mild to moderate hepatocellular lytic necrosis. Hepatocellular syncytial and low numbers of granulocytes were detected at the periphery of the necrotic foci, and occasional hepatocytes contained single, variably sized eosinophilic to amphophilic intranuclear inclusions that peripherally displaced of effaced nuclear chromatin (Fig. 1). Hemorrhage, fibrin deposition, and necrosis were present throughout the red pulp of the spleen, with fibrinoid necrosis in the ellipsoids and erythrophagocytosis in the red pulp. Within the lung, the interstitium was infiltrated by low numbers of lymphocytes and granulocytes, endothelial cells were mildly hypertrophied, and rare unidentified cells (likely endothelium) contained intranuclear inclusions. Respiratory epithelium was histologically within normal limits. Kidney, proventriculus, ventriculus, ganglia, intestine, and pancreas had no lesions identified histologically.
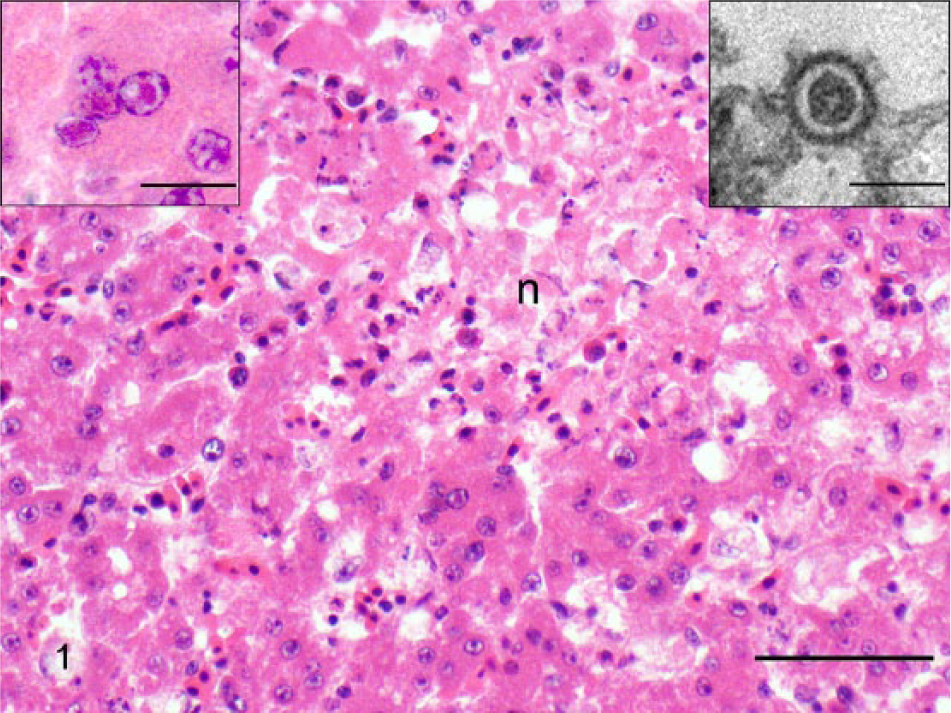
Liver of a Chilean flamingo (Phoenicopterus chilensis). Note large focus of lytic necrosis (n) surrounded by degenerative hepatocytes and hepatocellular syncytia. Hematoxylin and eosin (HE). Bar = 100 µm. Inset left: higher magnification of hepatocellular syncytia showing intranuclear eosinophilic inclusions in 3 of the nuclei. HE. Bar = 15 µm. Inset right: enveloped herpesvirus particle in cytoplasm of hepatocyte. Uranyl acetate and lead citrate. Bar = 130 nm.
Electron microscopy showed icosahedral viral particles ~100 nm in size in the cytoplasm of affected hepatocytes (Fig. 1). Morphologic features of the virus were consistent with a herpesvirus. 11
PCR and sequencing
A 496-bp sequence of herpesviral DNA-dependent DNA polymerase was amplified from the liver tissue of the affected flamingo chick. The sequence was submitted to GenBank under accession KP244360. Using the guidelines set by the International Committee on the Taxonomy of Viruses, this herpesvirus is hereafter referred to as phoenicopterid herpesvirus 1 (PhoeHV-1). 19
Phylogenetic analysis
A 165 amino acid sequence was translated from the 496-bp fragment of herpesvirus polymerase gene. BLASTX sequence comparison showed closest sequence homology with gaviid herpesvirus 1 (GavHV-1), which is a herpesvirus of loons within the subfamily Alphaherpesvirinae, clustered into the genus Iltovirus. The phoenicopterid and gaviid sequences had an identity score of 76%. Bayesian model jumping found that the WAG model of amino acid substitution was most probable, with a posterior probability of 100%. The LG amino acid substitution model had the lowest Akaike Information Criterion (AIC) using the ProtTest Server version 2.4. 1 The LG model is not implemented in Mr. Bayes 3.2.2. The LG model was used for ML analysis. Bootstrap values from the analysis are shown on the Bayesian tree (Fig. 2). Both phylogenetic analyses showed that PhoeHV-1 clustered within the genus Iltovirus in the subfamily Alphaherpesvirinae (Fig. 2). The Bayesian analysis found a 97% posterior probability, and the ML analysis found 89% bootstrap support for the monophyly of Iltovirus. PhoeHV-1 clustered significantly with GavHV-1, with 100% posterior probability in the Bayesian analysis and an 88% bootstrap value in the ML analysis.
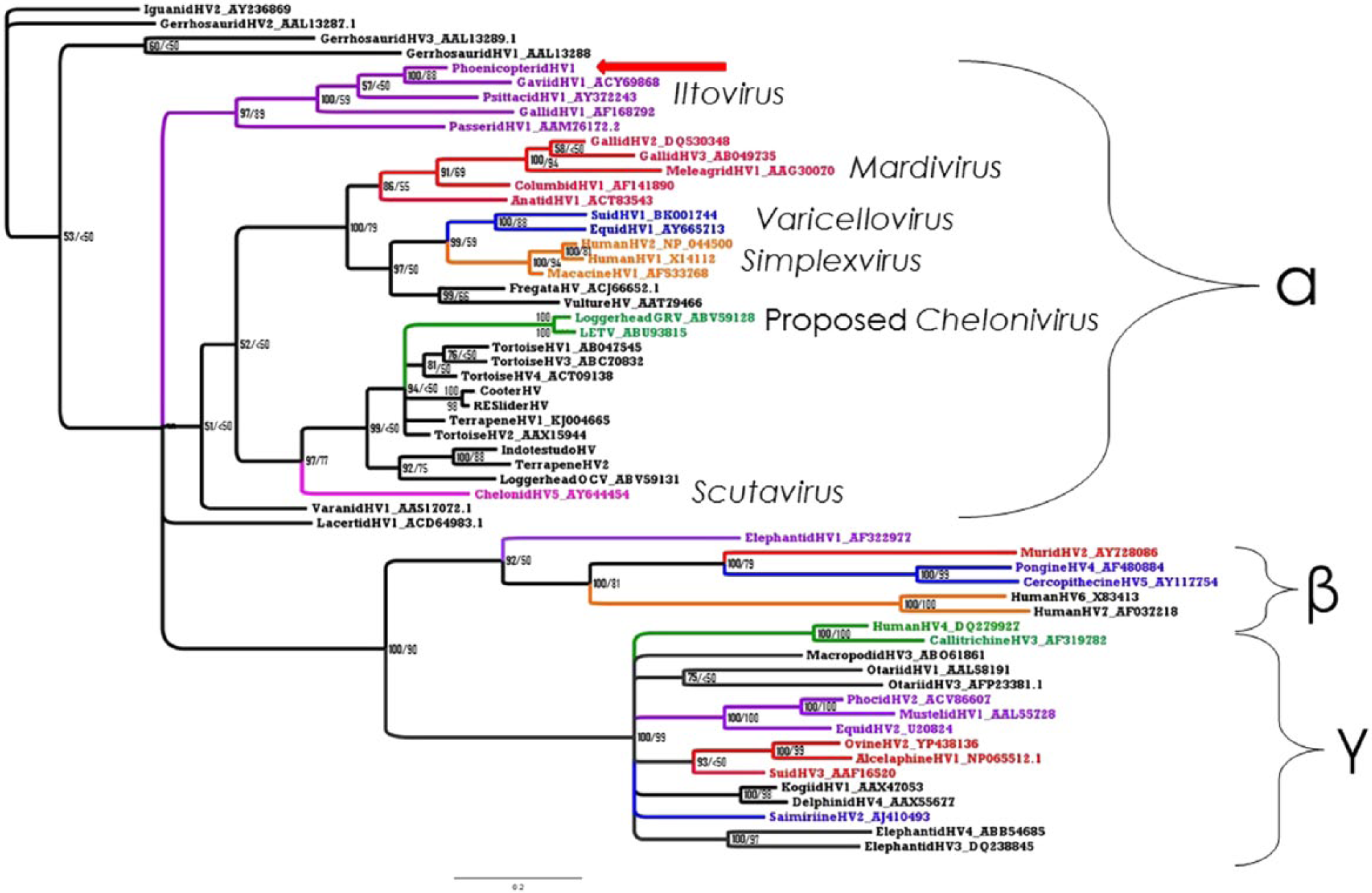
Bayesian tree for partial herpesvirus DNA polymerase. The first number (in bold) represents the posterior probability. The second number is the maximum likelihood bootstrap value given as a percentage. Numbers <50 are considered insignificant. Phoenicopterid herpesvirus 1 is indicated by a red arrow.
Discussion
Herpesvirus infection was initially suspected in this neonatal Chilean flamingo because the gross and histologic presentation was similar to an outbreak previously reported in American flamingos. 17 Phylogenetic analyses suggest that it clusters within the genus Iltovirus, most closely related to herpesvirus of loons, GavHV-1.
Iltoviruses have been associated with respiratory and hepatic disease in their host species. GavHV-1 has been associated with ulcerative tracheal lesions in common loons. 21 Likewise, passerid HV-1 (PaHV-1) has been recovered from Gouldian finches with necrotic tracheitis and intranuclear inclusions. 32 However, Psittacid herpesvirus 1 (PsHV-1), the cause of Pacheco’s disease in parrots, often presents with acute hepatic necrosis, with intranuclear inclusions as the most consistent lesion. Other variable lesion presentations include tracheitis, air sacculitis, and splenic necrosis. 28 The lesions seen in our case appear much more similar to those of PsHV-1 than the more closely related GavHV-1 and Gallid herpesvirus 1, 2, and 3 (GaHV-1, -2, -3, respectively).
The origin of PhoeHV-1 within this collection is not definitively known at this time. The source in our case may be from the other Chilean flamingos within the collection. The chick may have had health concerns at the time of hatching, as it had a lower birth weight than conspecifics hatched around the same time, and demonstrated unusual hemorrhage during pinioning. The bursa of Fabricius was not among tissues evaluated histologically, and may have been useful in assessing immunologic status. There have been no other specimens exhibiting signs of herpesvirus infection. Further investigation via postmortem examination, histology, and PCR on samples from appropriate individuals in the collection may detect the presence of endemic PhoeHV-1.
A previous study 26 described a novel flamingo poxvirus that was very closely related to Pigeonpox virus, suggesting a host jump from feral pigeons to captive collection animals. In contrast to the typical mild to moderate morbidity in the host-adapted species, herpesvirus infections in aberrant hosts tend to have more severe pathology. 3 Poxviruses, unlike herpesviruses, replicate within the cytoplasm of host cells. A previous study 20 found that the factor most strongly correlated with a virus’s tendency to host jump is the ability to replicate in the cytoplasm. Because of the complex pathways involved in nuclear entry within a cell and the utilization of host cell machinery for replication, viruses with intranuclear replication tend to be highly host-adapted. This makes the origin of most herpesvirus infections more likely to be from conspecifics, and we have found no evidence to strongly indicate otherwise in this case.
Numerous phylogenetic studies have indicated that herpesviruses coevolve with their adapted host species. 14 Herpesviruses have a relatively stable genome and low nucleotide substitution rate, 27 and estimates of the overall rate of the evolutionary “molecular clock” of herpesviruses put the rate of change in the genome at 1 to 2 orders of magnitude greater than their hosts.14,27
Although flamingo fossils date back to the early Oligocene, more than 28 million years ago (MYA), the most recent common ancestor of the extant phoenicopterids is thought to have diverged 4.37 MYA. 29 The Chilean flamingo represents the earliest divergence within the genus Phoenicopterus, diverging ~2.29 MYA. 29 There appears to be good genetic mixing within the Chilean flamingo; the species appears to be composed of a single metapopulation, 29 and this is expected for their herpesviruses. Sequence comparison of viruses isolated from other flamingo species could provide useful information regarding not only the evolution of the phoenicopterids, but perhaps that of the genus Iltovirus as well. Although specific sequence information was not available for comparison in public databases, a previous report determined that the herpesvirus sequences obtained from greater flamingos was 69% identical to GavHV-1. 33
Although our phylogenetic tree shows the genus Iltovirus as the earliest diverging named genus within the subfamily Alphaherpesvirinae, both Bayesian and ML support for this is poor, and further data is needed. The rest of the overall tree topology in our study is consistent with previously published data.3,13,31 The overall tree topology of squamate, chelonian, and avian herpesviruses, and the mammalian betaherpesviruses and gammaherpesviruses in this study are consistent with the branching orders of their respective host groups. 7 The mammal alphaherpesviruses are a notable exception to this. Additionally, within the genus Iltovirus, the first divergence is that of PaHV-1, followed by GaHV-1, PsHV-1, GavHV-1, and lastly PhoeHV-1. This topology is congruent to the pattern of divergence of the respective avian hosts found in 2011, 18 with the exception of the early divergence of PaHV-1. The early divergence of PaHV-1 may represent a host jump from another species rather than a coevolutionary origin. Recent whole-genome analyses have changed the topology of the relationships between taxa within the clade Neoaves. This includes the finding that order Phoenicopteriformes clusters with order Columbiformes within the clade Columbea, not the previously believed Passerea. 9 Findings in a 2014 study 9 also clearly demonstrate a rapid explosion of diversification within Neoaves in a short period of time immediately after the Cretaceous–Paleogene boundary. This rapid expansion of taxa likely contributes to the difficulty in resolving deep phylogeny branches of birds and their viruses.
Our study provides strong evidence of a novel herpesvirus of flamingos in the genus Iltovirus. Sequencing and analysis of the full virus genome would be helpful to further resolve the deeper level phylogeny. Although similar herpesvirus infections have been suspected in at least 1 captive collection previously, the prevalence of herpesviruses in both captive and wild populations of flamingos is unknown. The discoveries of many novel herpesvirus species in various avian taxa suggest that other bird species likely harbor yet undiscovered species of herpesviruses. As a result of the coevolutionary nature of these viruses with their host species, more detailed characterization of other novel herpesviruses may aid in clarifying both viral and host evolution.
Footnotes
Acknowledgements
We thank Roy Brown of Histology Consulting Service for histology slide preparation, and Elizabeth Post and Cathy Minogue of Northwest ZooPath for data retrieval.
Authors’ note
The views expressed in this article are those of the authors and do not reflect the official policy of the Department of the Army/Navy/Air Force, Department of Defense, or U.S. Government. The identification of specific products or scientific instrumentation does not constitute endorsement or implied endorsement on the part of the author, Department of Defense, or any component agency.
Authors’ contributions
CC Coverdill contributed to conception and design of the study, and drafted the manuscript. MM Garner and JFX Wellehan Jr contributed to conception and design of the study. All authors contributed to acquisition, analysis, and interpretation of data; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Poly-Bed, Polysciences Inc., Warrington, PA.
b.
JEM 1400 transmission electron microscope scope, JEOL, Peabody, MA.
c.
Orius camera model 832, Gatan, Pleasanton, CA.
d.
DNeasy kit, Qiagen, Valencia, CA.
e.
QIAquick gel extraction kit, Qiagen, Valencia, CA.
f.
Big Dye terminator kit, Applied Biosystems, Foster City, CA.
g.
ABI 3130 sequencer, Applied Biosystems, Foster City, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Partial support for avian herpesvirus discovery in this project was provided by the California Department of Fish and Game’s Oil Spill Response Trust Fund through the Oiled Wildlife Care Network at the Wildlife Health Center, School of Veterinary Medicine, University of California, Davis.
