Abstract
A duplex rapid assay for detection of serum antibodies to canine parvovirus (CPV) was developed. Canine immunoglobulin (Ig)M or IgG were captured in immunotubes with anti-canine IgM or IgG and detected with parvovirus VP2 recombinant protein followed by an anti-VP2 monoclonal antibody. The assay was tested using a collection of sera from dogs that were vaccinated against CPV on arrival at an animal shelter in Madrid, Spain. Results were compared with those of 2 commercial enzyme-linked immunosorbent assays (ELISAs) considered as reference techniques. A high correlation was found between the duplex rapid assay and the ELISAs, presenting an accuracy of 98% and 100% for IgG and IgM, respectively. According to the IgG and IgM levels at days 0–3 postvaccination, the samples were divided into 2 groups. One group of dogs showed high IgG and low IgM values at the first sampling post-vaccination and during the following 14 days, indicating that they had previously been in contact with the virus, either by vaccination or infection before arrival at the animal shelter. A second group of dogs appeared to be unvaccinated or uninfected before arrival at the animal shelter because they had negative IgM and IgG values soon after vaccination. These animals responded to vaccination, as demonstrated by seroconversion of both isotypes of immunoglobulins. The developed assay appears to be useful in determining the unknown immune status of dogs to CPV, especially in kennels and shelters where the rate of infection by CPV is relatively high.
Canine parvovirus (CPV) is a nonenveloped virus with a capsid of ~25 nm in diameter that packages a single-stranded DNA genome of ~5,000 nucleotides. 4 The CPV genome contains 2 open reading frames (ORFs). ORF1 encodes for 2 nonstructural proteins (NS1 and NS2), and ORF2 encodes for 2 structural proteins (VP1 and VP2).12,17 VP2 is the major capsid protein containing the antigenic determinant sites.2,9,14 This antigen can be used for detection of CPV-specific antibodies in serum. CPV affects dogs of all ages, but severe infections occur predominantly in puppies after maternal antibodies have declined to low levels, generally between 2 and 4 months of age depending on the maternal antibody titer and level of transfer. 8 A diagnosis of CPV enteritis can be made based on clinical signs and virus detection using a detection assay. The humoral immune response correlates with the outcome of the disease, although the cellular immune response is also important in protection. 1 Positive results of CPV immunoglobulin (Ig)M and IgG antibodies in serum provide veterinarians with useful information about the immune status regarding previous vaccinations or infections in unvaccinated healthy dogs.7,15 Vaccination with a modified live viral vaccine is widely used and recommended at 6–8, 10–12, and 14–16 weeks of age, followed by a booster administered 1 year later and then every 3 years. 5 Dogs vaccinated against CPV have serum antibody titers that persist for 12–48 months and often require revaccination. 13 Maternal antibodies are passed to the newborn by intestinal absorption from colostrum. However, maternally derived immunity is the primary cause of failure of CPV vaccination.6,16 Therefore, IgG antibodies can be used for assessment of immune status of puppies postvaccination.
Previous serologic studies showed that high levels of IgM antibodies to CPV were first detected 7 days after puppies were vaccinated and, after 9 days, all of the puppies had high titers of IgG antibodies to CPV.11,15 Knowing the immune status of dogs to CPV is especially relevant in the case of animal populations in shelters and kennels. The adult animals should be protected by the vaccine, and CPV infection usually causes a mild disease; nevertheless, if there is a suspicious case in the population, it will be very important to know the levels of IgG and IgM of the dogs in order to take appropriate action. Several commercial assays are available for CPV IgM and IgG detection. 15 However, these techniques can only be performed in diagnostic laboratories by employing specialized equipment not commonly available in veterinary practices. A rapid and simple in-clinic assay would be very useful in CPV antibody detection. In the present study, we developed 2 easy and rapid assays for CPV IgG and IgM detection. Both assays are based on the CPV VP2 recombinant protein expressed in the baculovirus system as the target antigen for antibody detection. Initially, 2 indirect enzyme-linked immunosorbent assays (iELISAs) were developed by coating the VP2 protein onto 96-well plates to analyze dog sera. IgM and IgG levels against CPV in canine serum samples were detected with anti-canine IgM or anti-canine IgG monoclonal antibodies (mAbs;1AB4 and 1BH4, respectively).a,b However, the detection of IgM by iELISA resulted in a high background signal in the analyzed sera. To solve this, an ELISA based on an antibody-capture format was developed in which anti-canine IgG or anti-canine IgM mAbs were used to coat plates. After adding serum samples, the VP2 recombinant protein followed by a specific mAb (4EA8) c to VP2 protein labeled with peroxidase was used to develop the reaction3,10 (Fig. 1). In order to improve and adapt the assay to a rapid format, the 96-well plate support was substituted by 11-mm polyethylene immunotubes, d which are easier to handle given their size, and dilution of samples can be done directly in the immunotubes without predilution. As control samples, well-characterized pools of sera from CPV experimental infections in dogs were used. After testing different dilutions of the control samples, a 1:100 serum dilution was chosen as the optimal dilution for the assay. Using small fixed-volume disposable pipettes, 5 μL of serum and 500 μL of dilution buffer (0.33 M NaCl, 0.05% Tween-20 in phosphate buffered saline, pH 7.4) were added to the immunotubes. Incubation periods were 15 min at room temperature; washing was performed manually with washing buffer. Bound antibodies were detected by incubation with VP2 recombinant protein followed by peroxidase-labeled 4EA8 mAb and subsequent addition of the substrate tetramethylbenzidine (TMB). e The reaction was stopped with 0.002% sodium dodecyl sulfate solution converting the substrate to blue-green, which enabled the readout of the test to be done visually (Fig. 2). These features avoid the use of laboratory instruments, thus making the assay easy to perform and appropriate to be used in veterinary practices.
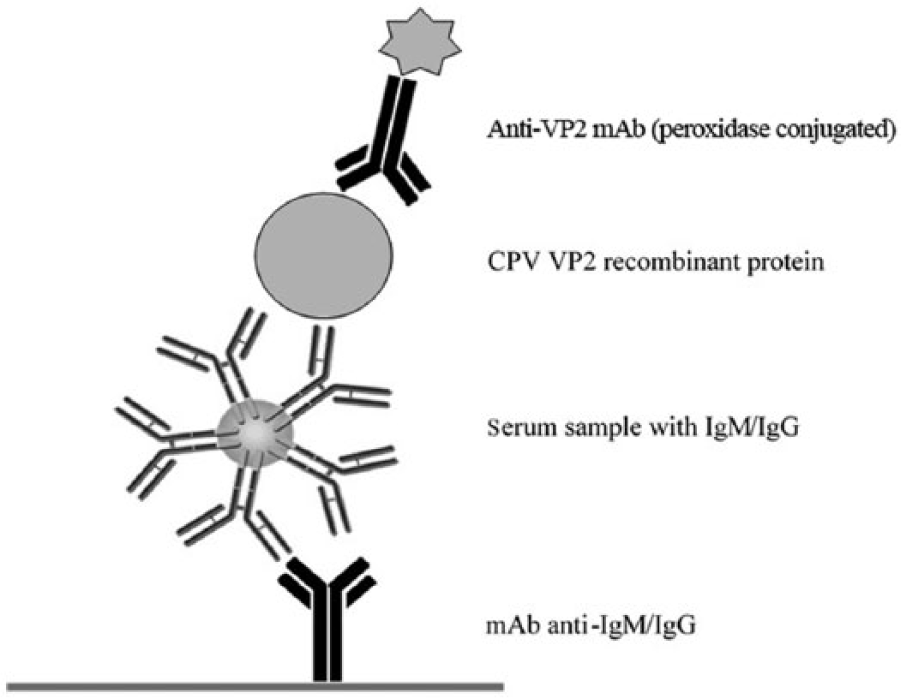
Rapid assays for canine immunoglobulin (Ig)M or IgG detection. Canine IgM or IgG were captured in 2 separate immunotubes with anti-canine IgM or IgG monoclonal antibodies (mAbs) and then detected with canine parvovirus (CPV) VP2 recombinant protein and anti-VP2 mAbs conjugated with horseradish peroxidase.

Immunotubes with positive and negative canine parvovirus (CPV) antibody samples. The rapid assays were carried out in polyethylene immunotubes. 1: positive CPV immunoglobulin (Ig)M sample; 2: negative CPV IgM sample; 3: positive CPV IgG sample; and 4: negative CPV IgG sample.
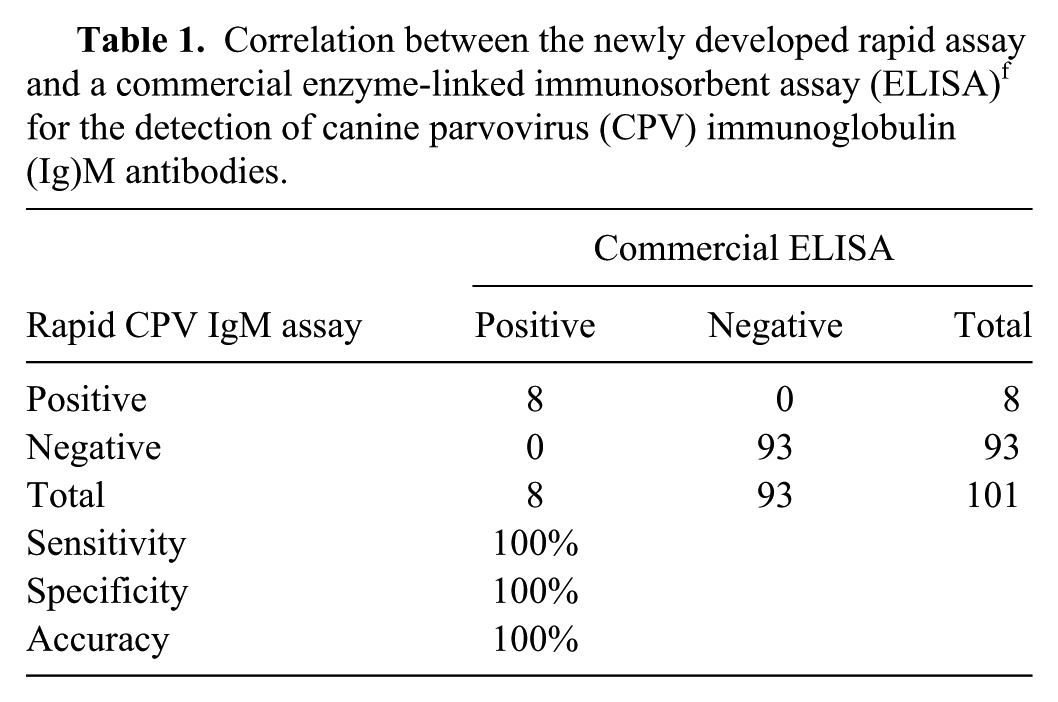
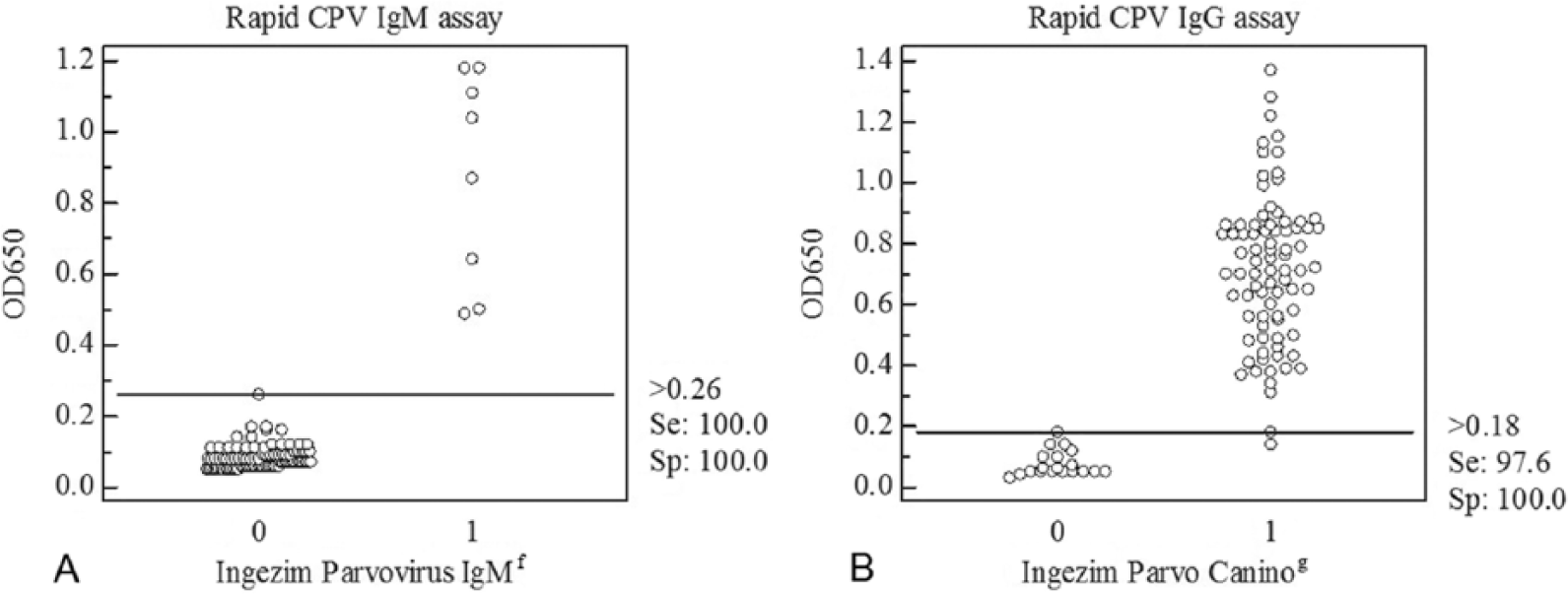
The conditions for the CPV IgM and IgG rapid assays were optimized to be done in parallel as a duplex assay. In addition to visual interpretation, absorbance at 650 nm was measured in an ELISA reader in order to carry out statistical comparisons with the results obtained with 2 commercially available ELISA kits used as reference techniques.f,g Serum samples from 41 stray dogs, of various breeds and ages, were provided by the Animal Protection Center (CPA) in Madrid. Most of the animals were vaccinated h against common canine diseases on arrival to the animal shelter. This vaccine h offers protection against parvovirus, hepatitis, parainfluenza, distemper, and leptospirosis. Serum samples from all dogs were collected between days 0–3 postvaccination. Then 1 or 2 additional samples from each animal were collected at different days and grouped into 3 sets as follows: 7–9, 10–13, and 14–17 days postvaccination. Serum samples were stored at −20°C until tested. A group of 14 samples from dogs not vaccinated at the CPA was also included in the study, and sera were collected at the same time points as the previous group. In total, 101 serum samples were analyzed and considered as individual samples for statistical purposes. Initially, the sera were analyzed by the commercial CPV IgM and IgG ELISAs, following the manufacturers’ instructions. Eight samples were IgM positive and 93 IgM negative by CPV IgM ELISA (Table 1; Fig. 3A). In the CPV IgG ELISA, 82 samples were positive and 19 negative for CPV IgG (Table 2; Fig. 3B). The same sera were tested by the rapid assay developed in immunotubes. Eight samples were positive for IgM (Table 1; Fig. 3A) and 80 for IgG (Table 2; Fig. 3B). The results obtained by the rapid assays were compared with the results obtained by the commercial ELISAs using statistical analysis software i (Fig. 3). The overall actual agreement between the rapid tests and the ELISA kits was 100% for the IgM assay and 98.0% for the IgG assay, and the kappa agreements were 1.0 for the IgM (Table 1) and 0.94 for the IgG assay (Table 2). The sensitivity was 100% for the IgM and 97.6% for IgG rapid assays. The specificity was 100% for the IgM and the IgG rapid assays.
Correlation between the newly developed rapid assay and a commercial enzyme-linked immunosorbent assay (ELISA) f for the detection of canine parvovirus (CPV) immunoglobulin (Ig)M antibodies.
Scatter plot of canine parvovirus (CPV) immunoglobulin (Ig)M and IgG levels. One hundred and one serum samples were analyzed with the duplex rapid assay, with commercial ELISA kitsf,g used as reference tests. Each dot represents an individual subject. Validation of the (
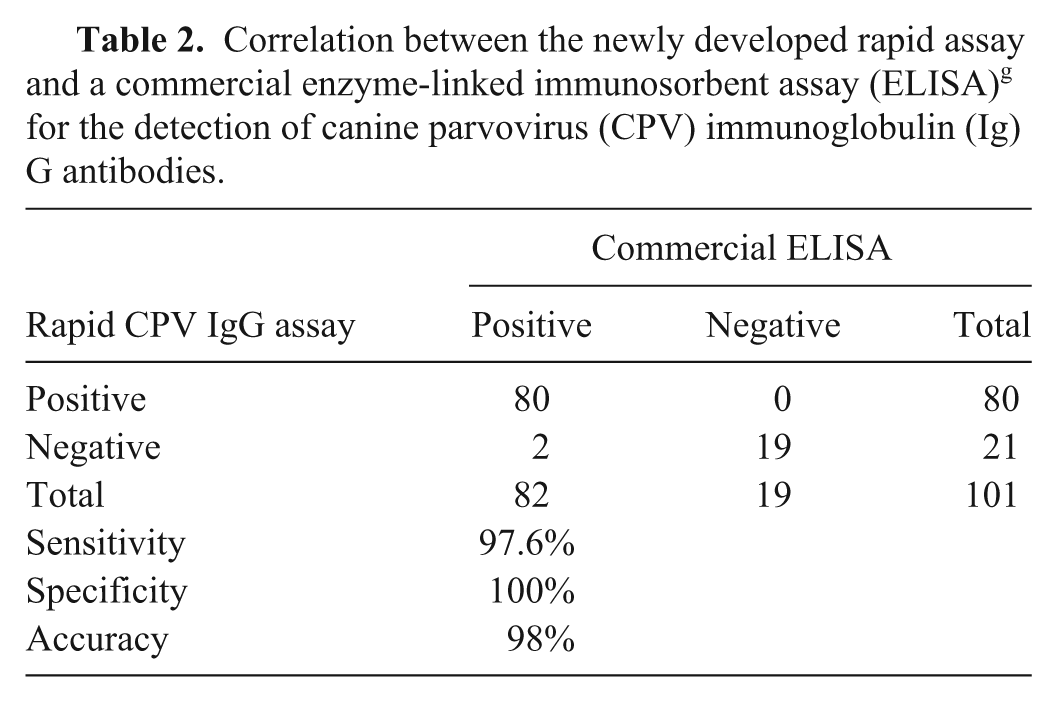
Correlation between the newly developed rapid assay and a commercial enzyme-linked immunosorbent assay (ELISA) g for the detection of canine parvovirus (CPV) immunoglobulin (Ig)G antibodies.
Seventy of the 101 samples that had been collected from CPV-vaccinated dogs at CPA were analyzed further. Because the information on their vaccination status and the time of sample collection was available, the samples from CPV-vaccinated dogs were classified based on their IgM and IgG levels and grouped by collection date postvaccination. Thirty of 70 samples were positive for IgG and negative for IgM antibodies against CPV at the first sampling at days 0–3 postvaccination, and the status remained unchanged during the 14 days thereafter (Fig. 4A). The results indicated that these dogs were CPV vaccinated or had acquired a CPV infection at some point before they arrived at the animal shelter; thus, no IgM seroconversion was observed. Most of the stray dogs in the animal shelter were most likely raised as pets previously, and therefore they were expected to be seropositive against CPV by vaccination. The other group that received the vaccine on arrival at the CPA had undetectable levels of IgM and IgG at days 0–3 postvaccination, and their IgM and IgG values increased during the 14 days thereafter (Fig. 4B). These results suggested that these dogs had either not been vaccinated or acquired a CPV infection before they arrived at the CPA and responded to the CPV vaccine by producing IgM and IgG. In the same way, samples from healthy dogs that were not vaccinated at the animal shelter were classified according to IgG and IgM levels. Of 14 sera analyzed, 9 had high IgG levels with undetectable levels of IgM, indicating that they came from dogs that were CPV vaccinated or had a CPV infection before they arrived at the shelter. In addition, 5 samples yielded undetectable IgM and IgG values (data not shown). These results indicated that these were CPV-naïve dogs (no prior vaccination or infection) and therefore these dogs would require a CPV vaccine to avoid CPV infection.
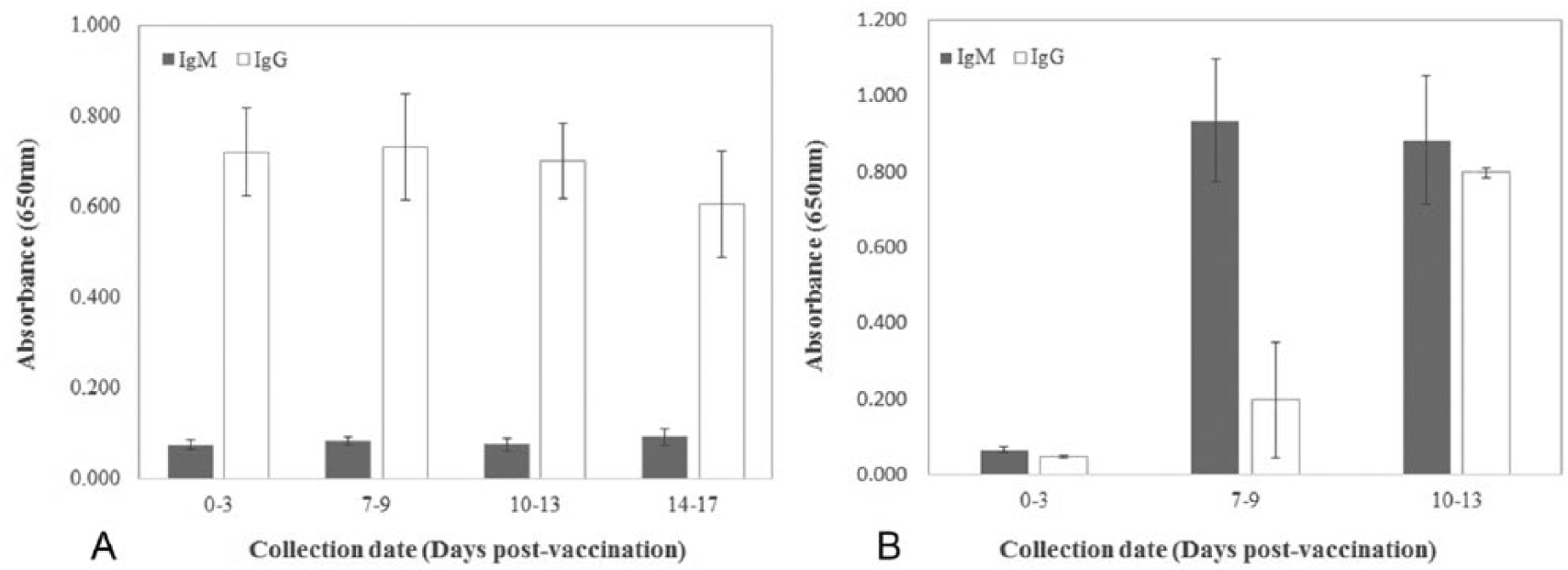
Graph of immunoglobulin (Ig)M and IgG levels in serum samples from dogs vaccinated with canine parvovirus vaccine.
The immune status of dogs to CPV could be determined using this newly developed CPV IgM–IgG duplex rapid assay in immunotubes. This assay can be easily performed without the need for laboratory instrumentation, and is intended to be used in the field. This test may be especially useful in determining the immune status of dogs to CPV in animal shelters.
Footnotes
Authors’ contributions
M Palma contributed to design of the study; contributed to analysis of data; and drafted the manuscript. M Montón, S Ramírez, and B Barreiro contributed to acquisition of data. P Sastre contributed to analysis and interpretation of data, and critically revised the manuscript. A Venteo contributed to design of the study, and contributed to analysis and interpretation of data. P Rueda contributed to conception and design of the study; contributed to analysis and interpretation of data; and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Anti-canine IgG monoclonal antibody (1BH4, reference M.15.000.I1BH4), Inmunología y Genética Aplicada SA, Madrid, Spain.
b.
Anti-canine IgM monoclonal antibody (1AB4, reference M.15.000.I1AB4), Inmunología y Genética Aplicada SA, Madrid, Spain.
c.
Peroxidase conjugated mAb 4EA8 against VP2, Inmunología y Genética Aplicada SA, Madrid, Spain.
d.
11-mm polyethylene immunotubes, Thermo Scientific, Waltham, MA.
e.
TMB, Novagen, Madison, WI.
f.
Commercial ELISA assay INgezim Parvovirus IgM (reference 15.CPM.K.2) was used for canine CPV IgM detection, Inmunología y Genética Aplicada SA, Madrid, Spain.
g.
Commercial ELISA assay INgezim Parvo Canino (reference 15.CPV.K.1) was used for canine CPV IgG detection, Inmunología y Genética Aplicada SA, Madrid, Spain.
h.
Canigen 7, Virbac, Carros Cedex, France.
i.
Statistical analyses were performed using MedCalc for Windows, version 10.1.7.0, MedCalc Software, Mariakerke, Belgium.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results received funding from the Seventh Framework Programme FP7-KBBE-2011-5 under grant agreement 289364 (RAPIDIA-FIELD).
