Abstract
Our study aimed to identify the levels of various contaminants in both wet and dry commercial pet foods in Egypt. A total of 20 local and imported pet food products (3 samples each) were screened for heavy metals by atomic absorption spectroscopy, for mycotoxins by enzyme-linked immunosorbent assay, and for nitrate and nitrite levels by nitrate–nitrite spectrophotometry. Cat food, on average, had greater concentrations of the metals cadmium, chromium, lead, and tin than dog food. Of the investigated metals, only tin concentration exceeded the safe level compared with the standards of the National Research Council and the European Commission for the dog and cat. According to the guidelines of the Association of American Feed Control Officials for canned pet foods, the nitrate and nitrite contents of examined foods greatly exceeded the recommended level. No total aflatoxins were detected in the surveyed samples. None of the samples analyzed had levels above international limits established by the Food and Agriculture Organization (FAO) of the United Nations for ochratoxin, and only 1 sample exceeded the level for aflatoxin B1. Of the 20 samples analyzed for zearalenone, 4 samples had higher levels than the FAO maximum tolerable levels. These results indicate that pet foods marketed in Egypt, especially cat foods, occasionally contain contaminants that could result in adverse effects in pets.
Most pet owners prefer commercial pet foods as convenient and economical ways to meet the nutrient needs of their animals. 19 Unfortunately, occasional pet illness outbreaks have been traced back to either microbiological or chemical contamination of pet food (U.S. Food and Drug Administration, Melamine pet food recall of 2007. Available at: http://www.fda.gov/AnimalVeterinary/SafetyHealth/RecallsWithdrawals/ucm129575.htm).15,17
Levels of various trace metals, including copper (Cu), nickel (Ni), lead (Pb), iron (Fe), manganese (Mn), chromium (Cr), and cadmium (Cd) were determined in commercially available pet foods in Turkey, and found to be within acceptable limits. 7 Nitrates and nitrites are routinely added during the meat curing process for canned products to enhance flavor and color and to prevent spoilage and the growth of toxigenic microorganisms, especially Clostridium botulinum. 4 There have been several reports of mycotoxin outbreaks in pets resulting from the consumption of contaminated commercial foods.15,17 These outbreaks were associated with cases of renal failure in cats and dogs, and resulted in a significant number of deaths.
In most countries, pet food is regulated by the maximum mycotoxin contamination levels for all foodstuffs rather than by pet-specific legislation. 12 For example, in the European Union, the level of total aflatoxin and aflatoxin B1 in feed ingredients is set at 50 and 10 µg/kg of food respectively, according to Food and Agriculture Organization (FAO) of the United Nations regulations. 9 For zearalenone and ochratoxin A, the FAO maximum tolerable levels (MTLs) range from 50 to 100 µg/kg of food and 100 to 200 µg/kg of food, respectively. 9
There is a paucity of information regarding the contamination of commercially prepared pet foods in Egypt. Therefore, the purpose of our study was to investigate the concentrations of heavy metals, nitrate, nitrite, and some types of mycotoxins in the pet foods that are commercially available in Egypt.
During the summer of 2014, examples of 20 local and imported pet food brands of both wet and dry type (3 replicates of each sample) were collected from supermarkets, retail shops, veterinary clinics, and pet food shops in Cairo and Zagazig cities, Egypt (Table 1). The moisture contents of the samples were also determined.
Characteristics of the pet foods examined in the current study.*
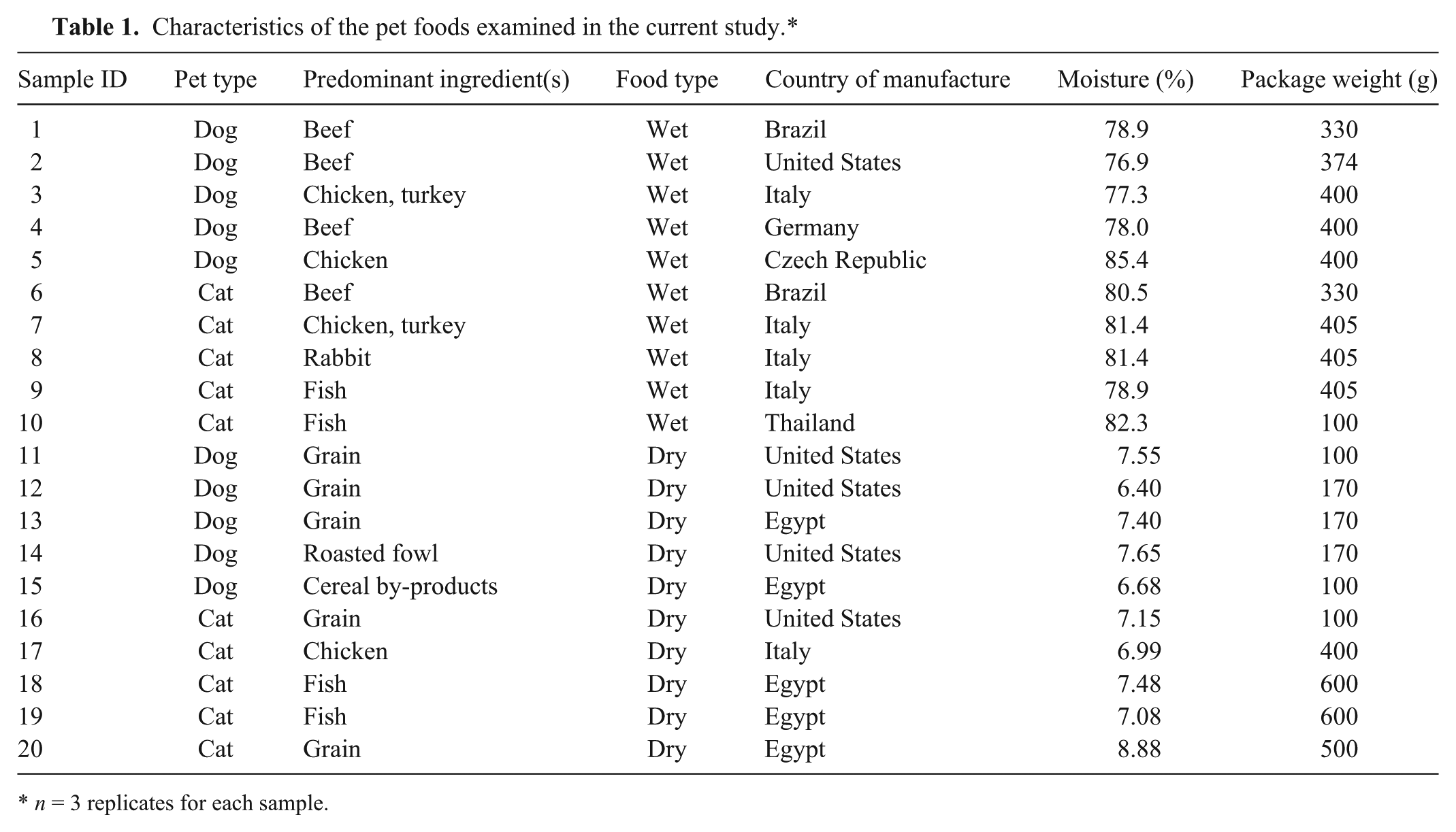
n = 3 replicates for each sample.
For the heavy metal analyses, the contents of the canned samples were immediately homogenized and dried overnight at 65°C and subsequently heated for 2 hr at 105°C. The dry foods were directly dried at 105°C for 3 hr. The dried samples were stored in polyethylene bags at room temperature away from sunlight and moisture until analyzed. For the nitrate, nitrite, and mycotoxin analyses, samples were kept frozen without prior drying.
Using pure certified metals, nitric acid 65% (HNO3) and hydrogen peroxide 30% (H2O2), a standard solutions were prepared at different concentrations to construct a calibration curve. All plastic ware and glassware was cleaned by rinsing several times with distilled water, soaking in 10% HNO3, and rinsing in deionized water prior to use. All reagents were analytical grade. All working solutions were prepared with deionized water.
The samples were digested using the wet method. 7 One gram of the sample was transferred to a clean screw-capped glass bottle and digested with 8 mL of digesting solution (65% HNO3and 30% H2O2, 3:1). The mixture was heated to 150°C until complete solubilization, which was followed by the addition of 5 mL of distilled water to each sample and remixing. The residue was filtered through 0.45-μm filter paper and diluted to 20 mL with deionized water. The filtrate was refrigerated until the analyses were performed. The same procedure was applied to the blanks.
A standard reference material (NBS bovine liver, 1577a) b was used to monitor the precision and accuracy of the analytical procedure. Comparisons of the certified values for each metal with the analyses of the reference material revealed recovery rates of 98%, 94%, 97%, and 95% for Cd, Cr, Pb, and tin (Sn), respectively.
Quantitative determination of metal residues was conducted at the central laboratory, Faculty of Veterinary Medicine, Zagazig University, Egypt, using a flame atomic absorption spectrophotometer equipped with a digital absorbance and concentration readout. c A double-beam ultraviolet visible spectrophotometer, d equipped with quartz cells with 1-cm light paths, was used to estimate the nitrate and nitrite contents in the wet pet food samples.
All chemicals were analytical reagent grade. e Bidistilled water was used in the preparation of all solutions. Stock solutions of 0.02 M sodium nitrite (NaNO2) and potassium nitrate (KNO3) were prepared by dissolving 0.15 and 0.72 g of the salts in water and diluting to 100 mL. Then, working standard solutions were prepared by appropriate dilution. Sulfanilamide (0.5 g in 150 mL of 15% acetic acid) and NED (0.02 g N-(1-naphthyl) ethylenediamine dihydrochloride in 150 mL of 15% acetic acid) were used.
Nitrate and nitrite were extracted from the canned food samples using the hot water method. 18 Exactly 25 g of sample content was weighed, finely homogenized, and transferred to a 250-mL volumetric flask. Next, 50 mL of water was added, the mixture was heated to 80°C for 15 min, transferred into a 250-mL volumetric flask, and then the volume was brought to ~200 mL with hot water. The flask was transferred to a steam bath for 2 hr with occasional shaking. The solution was cooled to room temperature, the volume was adjusted to 250 mL with water, and the sample was then filtered and centrifuged to obtain the supernatant.
The clear supernatant was added to 2.5 mL of sulfanilamide reagent, and aliquots of stock solution containing 5–50 µL of NaNO2 were transferred into a series of 50-mL calibrated flasks and mixed. NED reagent (2.5 mL) was added to each flask, and the solutions were thoroughly shaken for 15 min to allow the diazotization reaction to proceed to completion. A portion of the solution was transferred to a photometer cell and measured at 490 nm against the corresponding reagent blank, and the calibration graph was set. The concentrations of nitrate and nitrite were determined similarly, but nitrate was determined after the reduction of the nitrate to nitrite by the addition of a concentrated HCl and Zn–NaCl granular mixture for 30 min. The difference between the nitrate and nitrite values is a measure of the nitrate concentration.
A representative sample of each pet food was ground, thoroughly mixed, and weighed prior to extraction with 70:30 (v/v) methanol f and distilled water to determine the total aflatoxin, aflatoxin B1 (AFB1), and zearalenone. Sodium hydrogen carbonate buffer was used to extract ochratoxin A. The extracted samples were then filtered. g Enzyme-linked immunosorbent assay (ELISA) kits h were used for the quantitative analyses of total aflatoxin, AFB1, ochratoxin A, and zearalenone in the filtrates. A microplate reader i with an absorbance filter of 450 nm was used to optically measure the microwells. The detection limits of the methods for total aflatoxin, AFB1, ochratoxin A, and zearalenone were 5, 1, 2.5, and 1.75 ppb, respectively.
Data was statistically analyzed with a computer program. j The statistical method involved one-way analysis of variance tests followed by Duncan multiple range tests. 6 The data is presented as the means ± standard errors. The level of significance was set at p ≤ 0.05.
Our study recorded wide variations in the contents of the investigated heavy metals in the examined pet food samples (Table 2).The abundant levels of these elements might be caused by several factors, such as incorrect formulation of the food, insufficiencies in analytical testing of food for toxicants, mixing errors during the production process, or incorporation of contaminated raw materials (e.g., grains, meats, or other feed components) into the product. 16
Heavy metal, nitrate, and nitrite data in the examined pet food samples.*
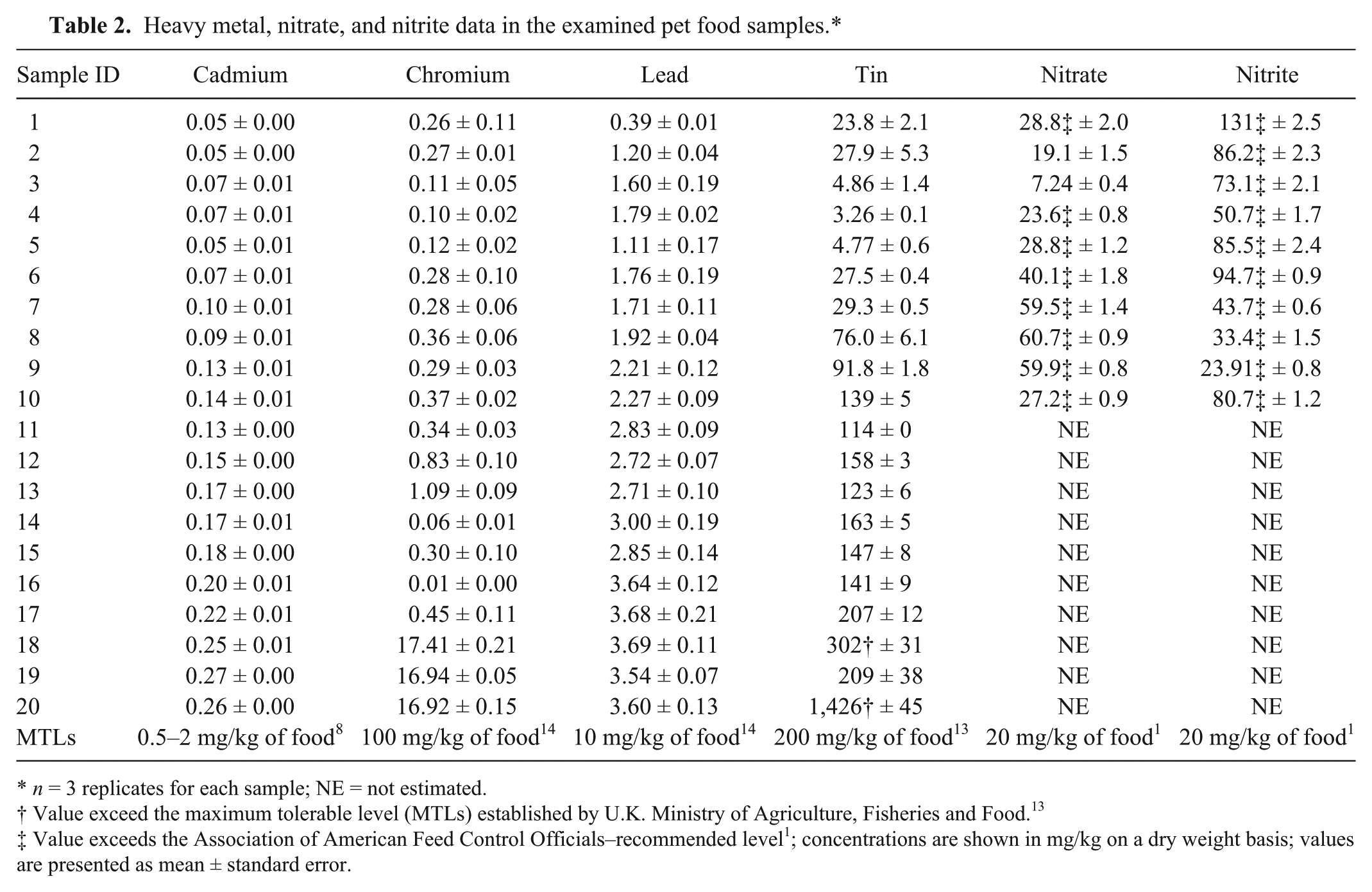
n = 3 replicates for each sample; NE = not estimated.
Value exceed the maximum tolerable level (MTLs) established by U.K. Ministry of Agriculture, Fisheries and Food. 13
Value exceeds the Association of American Feed Control Officials–recommended level 1 ; concentrations are shown in mg/kg on a dry weight basis; values are presented as mean ± standard error.
There are no guidelines set by regulatory agencies for the levels of Pb, Cd, Ni, or Cr in pet foods.2,3 Hence, in our study, the detected concentration of these heavy metals were compared with the standards of the National Research Council (NRC) and the directives of the European Commission for the dog and cat.8,14
The analyses of the examined pet food samples revealed that the mean levels of Cd, Cr, Pb, and Sn in the dry foods were 0.20 ± 0.01, 5.44 ± 1.42, 3.23 ± 0.08, and 299 ± 71 mg/kg of food, respectively; these levels were significantly higher than those observed in the wet foods (0.08 ± 0.01, 0.24 ± 0.02, 1.59 ± 0.10, and 42.9 ± 8.0 mg/kg of food, respectively). Cereal grains and their by-products in dry commercial pet foods might be responsible for these high elemental contents as a result of environmental contamination of resources including fertilizers and irrigation water containing waste disposal products and municipal sewage. 11
The cat food had greater amounts of all of the metals compared to the dog food (Table 2).This difference might be related to the fact that most of the cat food samples contained seafood products as the predominant ingredients, and such products are typically heavily laden with bioaccumulated metals; it has been estimated that ~13.5% of the worldwide catch of fish is used by the cat food industry. 5 Supporting this finding, pet food samples 18 and 19 that contained fish as the main ingredient contained the highest concentrations of Cd, Cr, and Pb.
In the present study, Cd was detected in all of the investigated samples, and the levels ranged from 0.05 to 0.27 mg/kg of food (Table 2). According to the directives of the European Union, the MTLs of Cd for all animal species are 0.5 and 2 mg/kg of food for dogs and cats, respectively. 8 Herein, the greatest recorded concentration of Cd (sample 19) was only 13.5% of the MTLs. Thus, no adverse effects due to Cd are expected to result from the consumption of these pet foods.
In the current study, the levels of Cr in pet foods ranged from 0.01 to 17.4 mg/kg of food, and the highest concentration was observed in the dry fish–containing cat food samples. The NRC standards state that the MTL for Cr for all animal species is 100 mg/kg of food. 14 Hence, no risk in terms of Cr level is expected to result from the consumption of these pet foods.
Pb was detected in all of the investigated samples, and the levels ranged from 0.39 to 3.69 mg/kg of food (Table 2). The MTL for Pb in complete feed has been set by the NRC at 10 mg/kg of food for all animal species. 14 The highest observed concentration of Pb (sample 18) was 36.9% of the MTLs. Therefore, no adverse effects caused by Pb are expected from the consumption of the examined pet foods.
Cd, Cr, and Pb concentrations detected in the analyzed pet food samples included in the current survey were generally low and unlikely to exert toxic effects. Nevertheless, prolonged exposure leading to a bioaccumulation of metals in animal tissues could be expected.
The Sn concentrations of the analyzed samples were in the range of 3.26 to 1,426 mg/kg of food (Table 2).The highest level of Sn was recorded in the grain-containing dry cat food sample 20 (Fig. 1A, 1B). In the United Kingdom, maximum permissible levels of Sn in food are typically 200 mg/kg according to the regulation of the Ministry of Agriculture, Fisheries and Food. 13 Hence, substantial risks are associated with the consumption of pet food samples 18 and 20 because of elevated Sn concentrations.
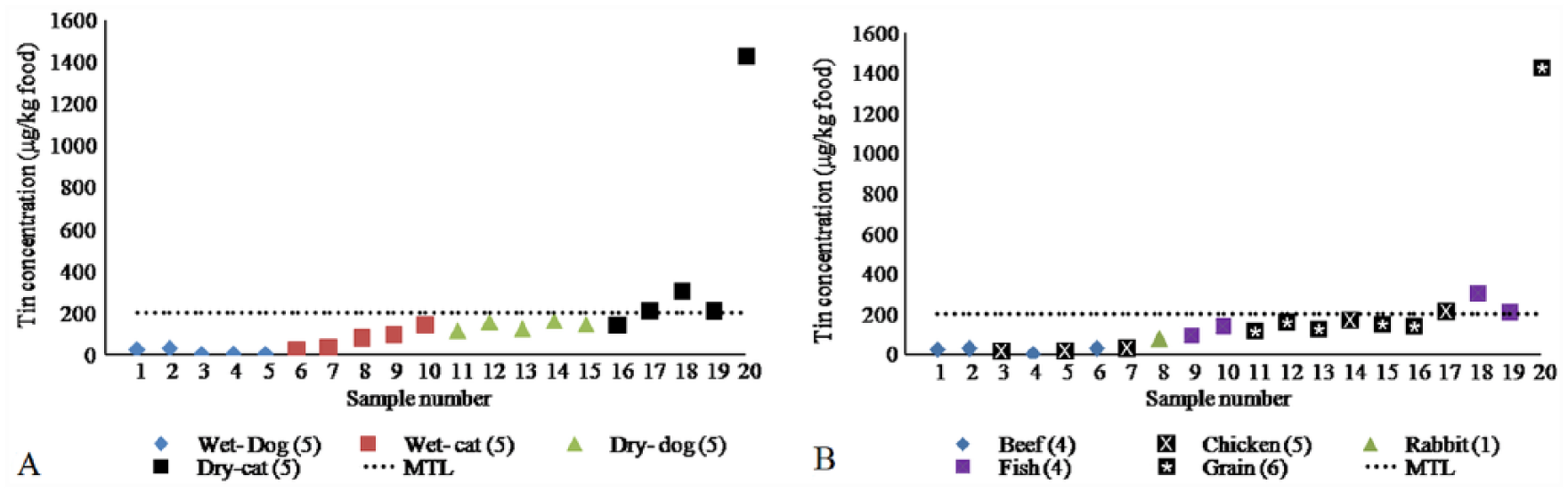
Plot of tin concentrations in the examined pet food products according to type of food (
The interrelationships between the metal contents in the investigated pet food samples were evaluated using correlation tests. The correlation coefficients revealed significant positive correlations between all of the tested metals (Table 3). The correlation between Pb and Sn was weak, whereas the Cr–Pb, Cr–Sn, Cd–Cr, and Cd–Sn correlations were moderate. The only strong correlation was between Cd and Pb.
Correlation coefficients between the metal concentrations in pet food samples.*
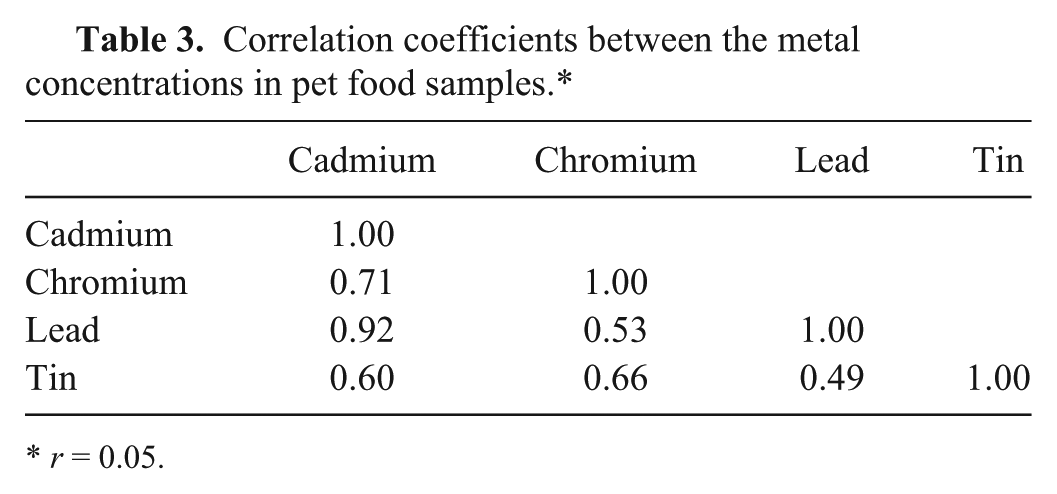
r = 0.05.
The values of nitrates and nitrites in the analyzed wet dog and cat food samples varied between 7.24–60.7 mg/kg of food and 23.9–131 mg/kg of food, respectively (Table 2). The cat food samples contained the highest concentrations of nitrates (samples 7–9), and the maximum nitrite levels were recorded in dog food sample 1 (Fig. 2A, 2B). The NRC did not establish nitrate and nitrite MTLs for non-ruminant species because of the wide array of diets and other conditions that affect nitrate toxicity. 14 However, the Association of American Feed Control Officials recommended a maximum concentration of 20 mg of sodium nitrite/kg of food. 1
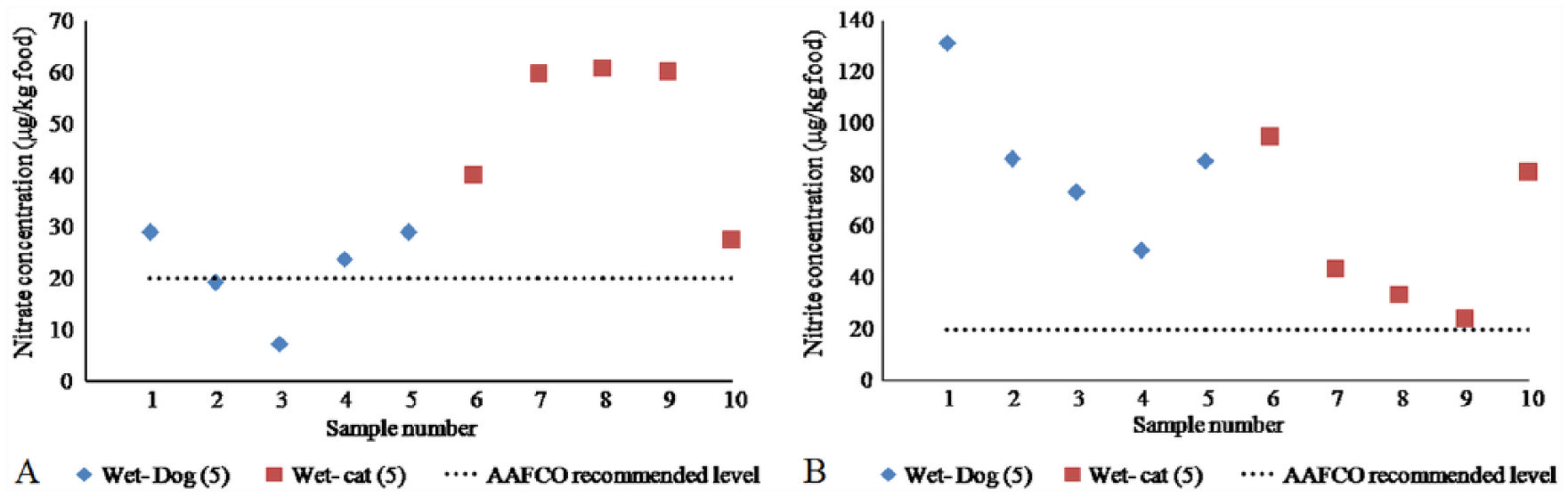
Plot of nitrate (
No total aflatoxins were detected in the surveyed pet feed samples (Table 4). Of the 20 samples analyzed, only 3 samples (1, 17, 19) were contaminated with aflatoxin B1. However, the aflatoxin B1 level in the dry cat food (sample 17) was over the safe FAO level of 10 µg/kg of food (http://www.fda.gov/AnimalVeterinary/SafetyHealth/RecallsWithdrawals/ucm129575.htm). Ochratoxin A was detected in most samples analyzed, and the levels ranged from 2.83 to 3.38 µg/kg of food. Nevertheless, none of these samples exceed the FAO MTLs of 100–200 µg/kg of food. 9 Only 1 dog food sample and 3 cat food samples were positive for zearalenone. However, the values of these 4 samples were high, ranging from 148 to 1,170 µg/kg of food, and all of them exceeded the FAO MTLs of 50–100 µg/kg of food. 9 Zearalenone, an estrogenic Fusarium mycotoxin, has been reported to produce pathologic changes in the dam’s reproductive system, including apoptotic-like changes in granule cells, atresia of follicular cells, and edema and hyperplasia of the oviductal and uterine cells when fed at a dose of 200 µg/kg body weight for 7 days. 10
Level of mycotoxins detected by enzyme-linked immunosorbent assay in pet food samples.*
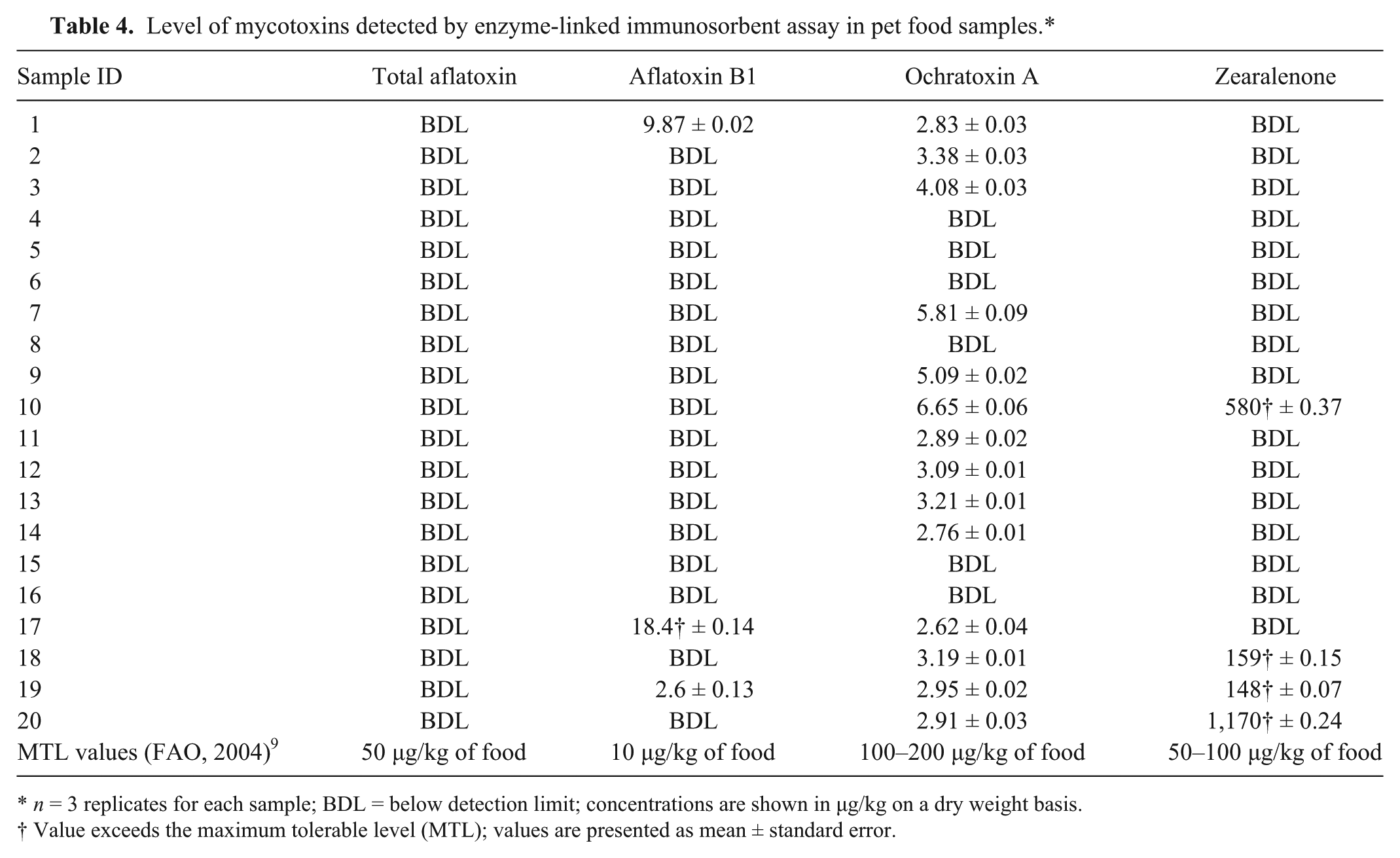
n = 3 replicates for each sample; BDL = below detection limit; concentrations are shown in µg/kg on a dry weight basis.
Value exceeds the maximum tolerable level (MTL); values are presented as mean ± standard error.
Based on our analyses of the ready-made pet foods marketed in Egypt, it can be concluded that while manufactured pet foods are safe regarding levels of some contaminants, such as Cd, Cr, Pb, and total aflatoxin, some products contain concerning levels of other contaminants, including Sn, nitrate, nitrite, and zearalenone, which are capable of causing chronic toxicity to pets. Notably, substantial risks are associated with marketed cat foods because of tin. Therefore, it is concluded that periodic monitoring of commercial pet food products is strongly needed.
Footnotes
Authors’ contributions
YM Abd-Elhakim contributed to conception and design of the study and drafted the manuscript. NI El Sharkawy contributed to acquisition, analysis, and interpretation of data and critically revised the manuscript. GG Moustafa contributed to conception and design of the study and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
HNO3 and H2O2 (HPLC grade), Merck KGaA, Darmstadt, Germany.
b.
National Institute of Standards and Technology (NIST), Gaithersburg, MD.
c.
Atomic absorption spectrophotometer (210VG), Buck Scientific Instrument Manufacturing Co., Norwalk, CT.
d.
UV-VIS spectrophotometer, Specord250, Analytika-Jena, Jena, Germany.
e.
Sigma-Aldrich, St. Louis, MO.
f.
Methanol (HPLC grade), Merck KGaA, Darmstadt, Germany.
g.
Filter papers (Whatman No.1), Sigma-Aldrich, St. Louis, MO.
h.
ELISA Ridascreen 30/15 kits (art. no. R4701, R1211, R1311, and R1401), R-Biopharm AG, Darmstadt, Germany.
i.
Micro plate reader (PR 4100), Bio-Rad Laboratories, Hercules, CA.
j.
SPSS/PC+2001, IBM Corp., Armonk, NY.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
