Abstract
Differences in reported testosterone concentrations in male sea turtle blood samples are common in the veterinary literature, but may be accounted for by differences in sample handling and processing prior to assay. Therefore, our study was performed to determine best practices for testosterone analysis in male sea turtles (Caretta caretta and Chelonia mydas). Blood samples were collected into 5 collection tube types, and assay validation and measured testosterone concentrations were compared across different sample storage (fresh, refrigerated 1 week, or frozen), extraction (unextracted or ether-extracted), and processing treatment (untreated, homogenized, or dissociation reagent) conditions. Ether-extracted and dissociation reagent–treated samples validated in all conditions tested and are recommended for use, as unextracted samples validated only if assayed fresh. Dissociation reagent treatment was simpler to perform than ether extraction and resulted in total testosterone concentrations ~2.7–3.5 times greater than free testosterone measured in ether-extracted samples. Sample homogenization did not affect measured testosterone concentrations, and could be used to increase volume in gelled samples. An annual seasonal testosterone increase was observed in both species when ether extraction or dissociation reagent treatment was used. Annual deslorelin implant treatments in a Chelonia mydas male resulted in suppression of seasonal testosterone following the fourth treatment. Seasonal testosterone patterns resumed following discontinuation of deslorelin. Comparison of in-house and commercially available enzyme immunoassay kits revealed similar patterns of seasonal testosterone increases and deslorelin-induced suppression. Our study highlights the importance of methodological validation and provides laboratorians with best practices for testosterone enzyme immunoassay in sea turtles.
Introduction
A common practice for veterinary and aquarium staff is to utilize commercial laboratories for routine hormone analysis, as most zoos and aquariums do not have an in-house endocrinology laboratory, or the laboratory does not routinely analyze samples on all species. Most commercial laboratories perform analyses of biological samples using generalized in-house methods or using commercial kits that may not be validated specifically for each species being tested. The accuracy, repeatability, and validity of measured testosterone (T5) values can be critical because T5 measurements are utilized for a variety of research applications, including sex determination,4,6,16 or to determine the efficacy of treatments to reduce circulating T5 for population management purposes or aggression control. A review of the literature on sea turtles shows that there have been a number of studies of sex steroid hormones in male sea turtles. The majority of analyses have been performed using in-house or commercial radioimmunoassay (RIA) kits. Reported sample treatment methods and measured T5 ranges have been variable.3–6,9,12–14,16,17,20,22,25 While some of these studies have included a limited number of biological, chemical, or physiological validations performed on RIA or enzyme immunoassay (EIA) systems to measure circulating T5 in sea turtle species,3,4,6,16 a full, comparative, systematic sample-processing method validation7,23 has not been published to our knowledge. Currently, there are no standardized sample collection and treatment methods for the analysis of T5 in male Chelonia mydas or Caretta caretta. Therefore, we hypothesized that differences in the RIA or EIA methods, antisera used, or sample treatments such as ether extraction3,6,16,20,22,25 or other processing, may account for the variation in T5 measurements reported between studies and research groups.3,4,6,16
The objective of our study was to perform a series of validation tests to determine the best blood sample collection tube types, storage conditions, sample processing techniques, and extraction methods for T5 analysis in male sea turtles (Chelonia mydas and Caretta caretta) using an established in-house EIA. Two commercially available EIA kitsa,b were also tested as alternative options to in-house analysis. Validated methods were then applied to monitor seasonal changes in T5, and evaluate the efficacy of deslorelin c contraception treatment in 1 male (C. mydas) to suppress seasonal testosterone increases associated with aggression. Results provide best practices for laboratory diagnosticians when measuring T5 concentrations using enzyme immunoassays in sea turtles.
Materials and methods
Animals
Our study examined 2 adult male sea turtles housed in a 22,000-m3 mixed-species salt water aquarium environment at Walt Disney World’s EPCOT “The Seas with Nemo and Friends” (Lake Buena Vista, Florida). The sea turtles in this study were non-releasable and held under authorization from the Florida Fish and Wildlife Conservation Commission (Marine Turtle permits 167 and 083). One male (C. mydas) was ~30 years old and weighed 114.7 ± 3.3 kg, and the second male (C. caretta) was ~80 years old and weighed 140.9 ± 5.3 kg during the course of the study. Water temperature of the tank was held at a constant 25 ± 0.5°C. Lighting for the tank was provided by 4 banks of lights supplemented with natural light provided by solar tubes and skylights positioned above the tank. The light banks were automated with timers set to turn on in a gradual sequence over 1.5 hr in the morning and turn off over a period of 3.5 hr in the evening, producing a light:dark cycle of 16:8 hr.
Blood collection
All blood samples were collected during regular health assessments. Serum and plasma samples (~12–24 mL per sample) were collected from the supravertebral sinus using single-use 21-gauge needles with twist-on syringes.15,18 Blood separation beads d were added to all serum samples. Samples were allowed to clot at room temperature before centrifuging at 791 × g for 15 min. The serum or plasma fractions were partitioned into 2 or more separate aliquots (1.2-mL storage vials) prior to sample storage or extraction testing as indicated below.
Storage temperature and ether extraction validation tests
For these tests, blood samples (C. caretta only) from each time point were collected in 5 commonly used blood collection tube types (serum with no additives e or with thrombin f ; and plasma with lithium heparin, g with sodium citrate, h or with ethylenediamine tetra-acetic acid i [EDTA]) to test the effects of blood collection tube type on T5 measurement. Aliquots from each sample collected in each collection tube type were then tested under several sample storage conditions (temperature, time) and sample extraction (unextracted, ethyl ether extraction) methods. The 3 storage conditions included fresh (immediately assayed within 1–2 hr following centrifugation, never refrigerated or frozen), refrigerated for 1 week (4°C, never frozen), or frozen (–50°C) for at least 1 week. Sample extraction validation tests were performed by extracting samples in ethyl ether j prior to serial dilution validation tests. A range of volumes and processing times were tested. In short, 20, 30, 50, 80, or 100 μL of serum or plasma was diluted up to 1 mL in double-distilled water in glass test tubes, and vortexed for 15 or 30 sec to mix. Next, 2 mL of ethyl ether was added to the test tube, and each sample was vortexed 15 or 30 sec. Water and ether phases were allowed to separate. The aqueous phase was flash frozen in a dry ice and ethanol bath. The ether phase was then decanted into a clean glass test tube and evaporated. Samples were reconstituted in phosphate buffer before serial dilution validation tests. Based on the results from these tests, only validated blood sample collection tube types, sample volumes, and extraction methods were used for the remaining tests of sample-processing treatment and analysis of seasonal T5 patterns.
Sample-processing treatment validation tests
Two sample-processing treatments (homogenization or dissociation reagent k ) were tested on aliquots of samples from 3 collection tube types (serum with no additives, serum with thrombin, and plasma with lithium heparin samples) following frozen-only storage conditions. Following centrifugation and storage, many serum and plasma samples formed gelled clots, therefore a test of homogenization was performed to determine if T5 was uniformly distributed in the gelled and non-gel fractions. A subset of split samples (C. caretta only) were homogenized for ~30 sec using a variable-speed homogenizer l set on medium–high speed, or left untreated. Samples (20-μL volume) were ether-extracted using the previously described protocol prior to assay.
As an alternative to ether-extraction methods, a commercially available dissociation reagent k was tested. Dissociation reagent treatment should allow for measurement of the total T5 in a sample, including any unbound, as well as protein-bound, T5. 1 Total T5 concentrations were compared with those from ether-extracted methods (a measure of unbound, free T5) described above. To validate the use of the dissociation reagent treatment, parallelism to the standard curve was assessed using a pooled blood sample. As per reagent kit instructions, 1 part pooled sample (20-μL volume of previously frozen; serum with no additives, serum with thrombin, or plasma with lithium heparin) was added to 1 part dissociation reagent and vortexed for 15 sec. The treated sample (now a 1:2 dilution) was then serially diluted in phosphate buffer in stepwise halving-dilutions (1:2) up to a final dilution of 1:1,024. To determine the optimal dilution factor of those that demonstrated parallelism, a subset of serum with no additives samples representative of seasonally low and high T5 concentrations were tested for differences in measured T5 across validated dilutions.
Seasonality and biological validation in ether-extracted versus dissociation reagent–treated samples
Blood samples for the main study period of November 2011 through June 2013 were collected from both turtles approximately once per month in 3 blood collection tube types (serum with no additives, serum with thrombin, or plasma with lithium heparin), partitioned into multiple aliquots, and stored frozen (–50°C). Aliquots from each blood collection tube type at each time point were thawed and homogenized prior to ether extraction or dissociation reagent treatment. Ether extraction was performed on 20-μL of serum or plasma as previously described. Ether-extracted samples were reconstituted in phosphate buffer and assayed at dilutions of 1:30 and 1:120 to measure free T5 content. For dissociation reagent treatment, a 20-μL sample and 20 μL of dissociation reagent were combined, vortexed, and then further diluted to 1:200 in phosphate buffer to measure total T5 content. Concentrations of free versus total T5 were compared.
As part of a biological validation of the EIAs, samples from a deslorelin c -treated male (C. mydas) were compared with a nontreated male (C. caretta) through 3 seasons of expected seasonal T5 increase (November 2011–October 2014). Banked C. mydas samples from 2009, 2010, and early 2011 were also included in the analysis to monitor for changes in seasonal testosterone patterns pre– and post–deslorelin treatment. These analyses monitored 1.5 years following the fourth and final deslorelin treatment to assess efficacy of suppression and recovery of seasonal T5. The treated male received a set of four 4.7-mg implants (the Association of Zoos and Aquariums Wildlife Contraception Center’s recommended dose for mammalian species is one 4.7-mg implant for 6 months of treatment), intramuscularly, on 4 separate dates that corresponded with periods of observed seasonal behavioral aggression (June 2010, February 2011, December 2011, and December 2012). Previous implants could not be removed as they had become embedded into the surrounding tissue.
In-house EIA system validation and hormone analysis
For the previously described tests, serum and plasma samples were analyzed for T5 on an in-house EIA system using antisera R156/7. m Cross-reactivity of the antisera was 100% against T5, 57.37% against dihydrotestosterone, 0.27% against androstenedione, and <0.05% for androsterone, dehydroepiandrosterone, cholesterol, and β-estradiol. 19
A 96-well microtiter plate n was coated with 50 μL of the R156/7 antisera at a concentration of 1:10,000. After the antibody bound overnight at 4°C, the plate was washed 3 times with a wash solution containing Tween 20 and then tapped dry. Next, 50 μL of phosphate buffer was added to each well. Following this, 50 μL of serially diluted testosterone standards o (range: 0.975–1,000 pg/50 µL) or diluted samples were added to the plate, followed immediately by 50 μL of T5 conjugated to horseradish peroxidase (1:120,000). The plate was then shaken for 5 min and placed in a dark cupboard at room temperature overnight to incubate. Following incubation, plates were washed and tapped dry. A color reaction solution consisting of ABTS (2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)) and hydrogen peroxide was then added to each well (100 µL/well). The plate was shaken until the reaction produced a green solution with an optical density between 0.7 and 0.8. Plates were read using a microplate reader p using a 405-nm filter with 650 nm as a reference. Values were calculated using microplate analysis software. q All samples were assayed in duplicate and values reported in ng/mL.
Samples used to determine seasonal profiles were assayed after frozen storage conditions and ether extraction using the in-house EIA system. This was considered the standard methodology, and other sample treatments and EIA systems were compared to this method. In order to provide an additional validation for the standard method using the in-house assay system, a spike–recovery test was performed on a known low concentration sample using the ether extraction technique. The sample was spiked with a known quantity of each standard prior to ether extraction. The average percent recovery of T5 in spiked samples was determined to be 96.3% for the readable range of the assay.
A total of 68 assays were performed for the in-house system. Two measured testosterone standard controls of low (5 pg/50 µL) and high (120 pg/50 µL) concentration were run on all assays. The intra-assay percent coefficients of variation (CVs) for low and high T5 concentration controls were 4.5% and 3.5% respectively (n = 68). Interassay CVs were 16.7% and 11.2% for low and high controls, respectively (n = 68). Assay sensitivity for the in-house system (defined at the 80% binding of the assay) averaged 2.5 pg/50 µL.
Commercial EIA kit comparison
Using validated sample types and treatments determined through the previously described tests, 2 commercially available T5 EIA kit options (rabbit a and sheep b antisera) were validated by assessing parallelism of a serially diluted dissociation reagent–treated pooled sample to the standard curve and tested against the R156/7 in-house EIA system. For this comparison, the single antibody in-house assay was adapted to a double antibody system with a 1.5-hr room temperature incubation period equivalent to a commercially available kit. r Previously frozen serum with no additives samples were treated with dissociation reagent following the validated methods outlined above. Cross-reactivity of the rabbit a antisera kit was 100% against T5, 35.4% against dihydrotestosterone, 0.024% against progesterone, and <0.004% against cortisol, cortisone, corticosterone, and estradiol; cross-reactivity of the sheep b antisera kit was 100% against T5, 58.85% against dihydrotestosterone, and <0.01% against progesterone, cortisol, cortisone, corticosterone, and estradiol (Arbor Assays, personal communication, 2014).
Statistical analysis
This case study had a small sample size with several validation tests, thus primarily descriptive statistics are presented. Where appropriate, T5 values are presented as mean ± standard deviation. Although T5 may be present in measureable quantities in a sample, interferences inherent to the sample or from sample treatment may prevent T5 from binding with the antibody in a predictable manner, resulting in non-parallelism. Statistical software s was used to perform an F-test (GLM) to determine parallelism of serial dilution validation curves to the T5 standard curve, where a significant F-test would indicate that the slopes of the curves were not parallel, and therefore the sample collection tube type, storage, or treatment method did not validate in the assay. As a secondary test, calculated T5 values were compared across sample serial dilutions to determine if the dilution factor affected the final calculated T5 result (if the dilution provided a value within the readable range of the assay [conservatively defined as 20–80% binding]). Both test results were used to determine successful validation of each tested sample collection tube type, storage condition, and treatment method in the T5 assay.
An F-test was also used to compare T5 profiles from the 3 blood collection tube types in the main study period (serum with no additives, serum with thrombin, and plasma with lithium heparin). A student t-test n was performed to determine the effect of homogenization on measured T5 concentrations. A value of p < 0.05 was used to determine significance for all statistical tests.
Results
Storage temperature and ether extraction validation tests
Serial dilutions of C. caretta samples collected using the 5 different blood collection tube types across the 3 temperature conditions were tested for parallelism to the T5 standard curve. All fresh, unextracted (excluding plasma with EDTA) and fresh, ether-extracted samples validated. However, unextracted samples stored refrigerated or frozen did not validate (p < 0.05). Refrigerated and frozen samples only validated when ether-extracted (Table 1). All samples were stored frozen following collection and centrifugation in all but the initial storage treatment experiment, thus ether extraction was required and utilized for the remainder of the study. Following initial storage temperature tests, samples were only collected in serum with no additives, serum with thrombin, and plasma with lithium heparin collection tubes, as they validated under most conditions and showed similar serial dilution validation curves in the parallelism tests.
Results of parallelism validation tests for sample extraction and storage treatment by blood sample collection tube type.*
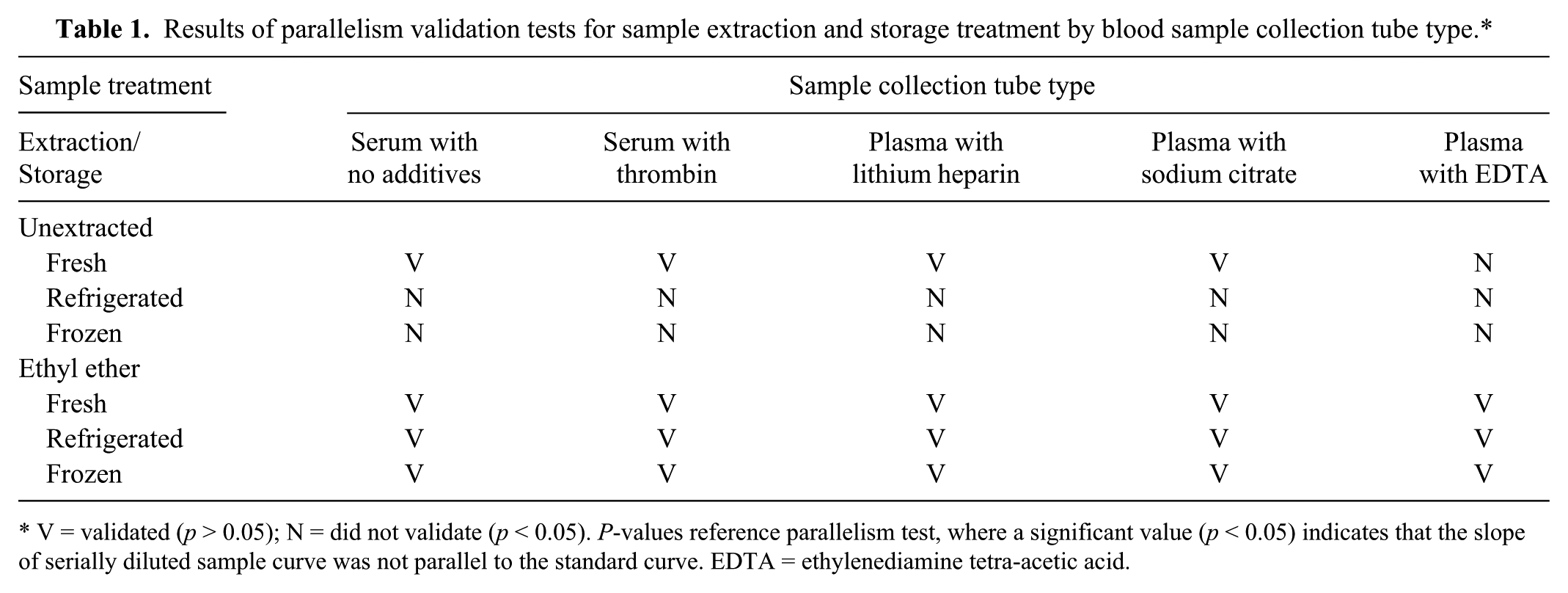
V = validated (p > 0.05); N = did not validate (p < 0.05). P-values reference parallelism test, where a significant value (p < 0.05) indicates that the slope of serially diluted sample curve was not parallel to the standard curve. EDTA = ethylenediamine tetra-acetic acid.
During ether extraction testing, large volumes of serum or plasma (≥50 µL) and extended vortex times (>15 sec) produced an additional gel-like layer between the water and ether phases that appeared to be comprised of some of the ether component volume. This layer did not freeze or partition consistently in the tests, thus large sample volumes and extended vortex times did not validate and could not be used. All remaining tests used a maximum of 20 μL of sample volume and a vortex time of 15 sec.
Sample-processing treatment validation tests
Serum and plasma samples were observed to partially gel following centrifugation and separation in all blood collection tube types and storage conditions. This produced a pipettable liquid fraction and a non-pipettable gel fraction in the serum or plasma, which limited available volume for hormone analysis. Sample homogenization increased the available pipettable volume in the tested samples. Measured T5 concentrations were not significantly different in homogenized and nonhomogenized samples (p > 0.05).
Serial dilutions of dissociation reagent–treated samples were parallel (p > 0.05) with the standard curve between dilutions of 1:128 and 1:1,024 in low and high T5 pool samples (serum with no additives, serum with thrombin, and plasma with lithium heparin). A range of final sample dilutions of 1:100–1:1,400 tested in serum samples through the seasonal change in T5 produced similar patterns but different amplitudes of T5 concentrations. A dilution of 1:200 was chosen for all further analysis because it gave the highest measured T5 concentration (total T5) for seasonal low and high samples, while remaining within the readable range of the assay for all samples tested.
Seasonality and biological validation in ether-extracted versus dissociation reagent–treated samples
Testosterone pattern and concentrations in previously frozen, homogenized, ether-extracted samples (January 2011–June 2013) did not differ between serum with no additives, serum with thrombin, or plasma with lithium heparin (p > 0.05). Seasonal elevations in T5 were observed each year from approximately late December through June in males of both species. When averaged across the 3 blood collection tube types, testosterone concentrations ranged from 0.46 to 37.7 ng/mL for C. mydas and 6.4 to 73.8 ng/mL for C. caretta. Seasonal T5 peak concentrations for C. caretta averaged 67.0 ± 17.1 ng/mL and were 9.6 times higher than seasonal lows. Peaks occurred in February and March in 2012 and 2013, respectively, while the lowest observed T5 values occurred in July 2012. The average peak T5 concentrations in C. caretta were 1.8 times greater than the average peak concentrations detected in C. mydas in January 2011 and March 2012 (Fig. 1).
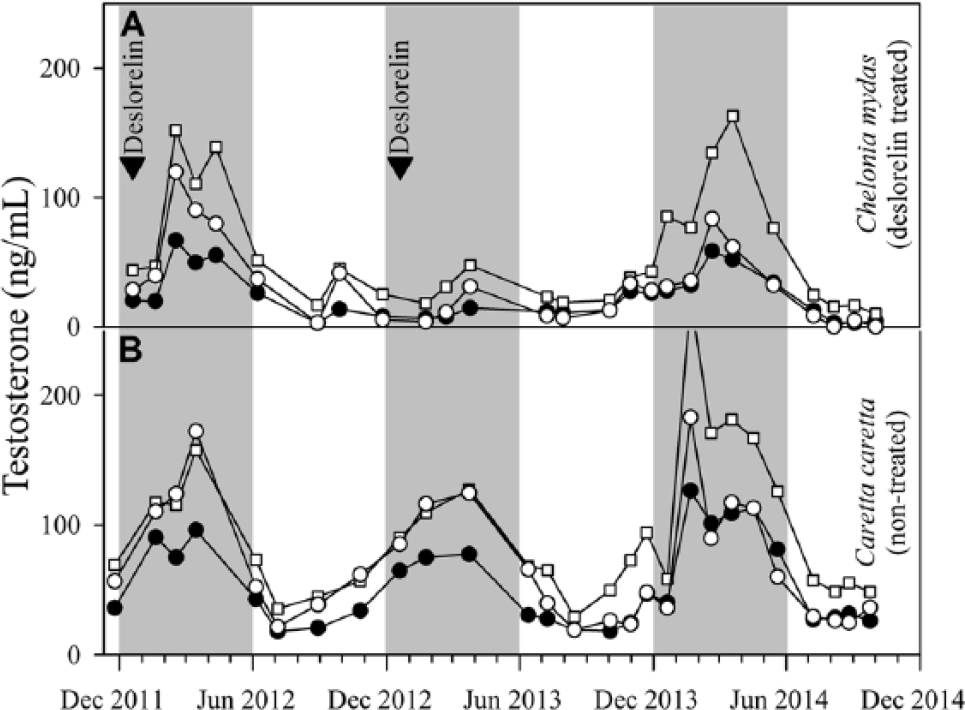
Comparison of seasonal changes in total T5 concentrations measured in dissociation reagent–treated serum with no additives samples in 3 tested enzyme immunoassay (EIA) systems in the deslorelin-treated male (Chelonia mydas; panel
Deslorelin treatment was not effective in preventing, interrupting, or reducing seasonal T5 concentrations in the treated C. mydas male for the first 3 treatments, which were administered either at the start of the annual seasonal T5 increase (December 2011) or following the start of the seasonal T5 increase (June 2010 and February 2011). However, suppression of the seasonal T5 increase was observed in 2013 following the fourth annual deslorelin treatment (December 2012). In the following year (2014), deslorelin treatment was not administered and seasonal T5 increase was observed again.
Because it was determined that measured T5 was not significantly different (p > 0.05) in serum with no additives, serum with thrombin, and plasma with lithium heparin samples, a single sample collection type (serum with no additives) was chosen to compare seasonal T5 profiles measured by dissociation reagent versus ether-extracted sample values. Serum with no additives samples treated with dissociation reagent provided a measure of total T5, and produced similar seasonal T5 and deslorelin suppression profile patterns to that seen in ether-extracted samples (free T5) for both species. Total T5 concentrations averaged 2.7 times greater than free T5 in C. mydas and 3.5 times greater than free T5 in C. caretta.
Commercial EIA kit comparison
All assay systems tested successfully validated and showed comparable seasonal T5 profiles using dissociation reagent–treated serum with no additives samples. The commercial sheep antibody EIA kit averaged total T5 concentrations 2.3 times greater than the in-house (rabbit antibody) and 1.8 times greater than the commercial rabbit antibody EIA kit. The commercial rabbit antibody EIA kit averaged total free T5 concentrations 1.3 times greater than the in-house system (Fig. 1). The same pattern of seasonal T5 changes and deslorelin suppression detected by the standard ether-extraction method was observed across all kits.
Discussion
Sea turtle serum and plasma T5 values have been analyzed by researchers for many years, but a wide range of T5 values and sample treatment methods have been reported.3–6,9,12–14,16,17,20,22,25 These differences could be the result of differences between sea turtle species or populations. 24 However, as our study demonstrates, some of the differences in T5 values between studies may also be a result of differences in sample collections, treatments, and processing between laboratories, or the assay system used to analyze the samples. 24
Our study tested the effects of sample collection, storage, processing, and extraction methods on measured T5 concentrations in C. mydas and C. caretta using an in-house T5 EIA with antisera R156/7. While the results demonstrated that sea turtle serum and plasma samples (excluding plasma samples with EDTA) could be assayed fresh (within hours of collection) without requiring ether extraction on this T5 assay system, this may not be feasible for many institutions or clinical laboratories that may store samples under refrigerated (4°C) or frozen (–20 to –80°C) conditions. If the researcher or clinical veterinarian must send samples to a commercial laboratory for analysis, it is imperative that they request either immediate testing of freshly collected samples, or that ether extraction or a dissociation reagent is used for previously refrigerated or frozen samples in order to receive valid T5 measurements.
Results also indicated that sample collection of plasma with EDTA-treated tubes validated in the fewest conditions and is therefore not recommended for use when performing T5 measurements on EIA systems in male sea turtles. This is consistent with previously published work that states that the anticoagulant EDTA causes hemolysis in the blood and should be avoided. 15 Lithium heparin and sodium heparin have been recommended 15 ; however, our study demonstrated that, for the EIAs and antisera tested, serum with no additives or serum with thrombin were also suitable for collection. Measurements of T5 from serum with additives, serum with thrombin, and plasma with lithium heparin demonstrated similar patterns of T5 and can be used interchangeably without resulting in significant (p > 0.05) differences in measured T5 concentrations.
Although traditional ether-extraction methods3,6,16,20,22,25 were used throughout most of the study and validated under all tested storage conditions, this method is labor intensive and requires the handling of hazardous chemicals. Our study has shown that dissociation reagent is a safer and faster alternative to ether-extraction methods, and may be useful in laboratories that do not have the supplies or facilities necessary to perform ether extraction. Dissociation reagent provided a measure of total T5 that was on average 2.7 times higher than free T5 in C. mydas samples and 3.5 times higher than free T5 in C. caretta samples, while maintaining the seasonal profile pattern observed in ether-extracted samples. Free T5 (ether-extracted) and total T5 (dissociation reagent–treated) showed the same seasonal patterns. This suggests that either technique can be used to monitor for changes in hormone concentration. It remains unclear if the amount of total T5 (bound and unbound hormone) in a sample is biologically relevant, or if only free T5, which is readily available for attachment to a receptor, is of clinical importance. 1 Regardless, because the seasonal patterns are consistent across the 2 measurements, it appears either can be used to assess the seasonal pattern of T5 in male sea turtles. However, if a quantitative measure of T5 is needed, the researcher must decide if a measure of free or total T5 is desired and choose the proper technique accordingly.
The use of this dissociation reagent was validated for measurement of steroids in blood samples from Dasyatis rays (C Adams and L Penfold, personal communication, 2013), suggesting that it may be a useful technique to measure hormone concentrations of other marine species as well. However, it is important to note that the measured hormone concentration (total T5) varied depending on the sample dilution. Therefore, if a dissociation reagent is to be used, results from our study suggest that a range of dilutions should be tested in samples from several individuals with various hormone concentrations, and a single dilution that will measure both low and high T5 concentrations in the readable range of assay should be chosen and used for all samples in the species tested.
Sea turtle serum and plasma samples were challenging to process for EIA. Serum and plasma samples formed non-pipettable gel clots regardless of collection tube type and storage condition, and only limited sample volumes and vortex times could be used during ether extraction. Although the composition of these clots is not known, T5 concentrations did not differ in the pipettable liquid fraction versus homogenized samples containing the liquid plus gelled fractions of the same sample. Therefore, sample homogenization can be used to increase available sample volume for T5 measurements in samples that have insufficient pipettable volumes, without affecting the measured T5 concentrations.
Seasonal T5 increases were observed in both males throughout the winter and spring months (approximately January–June) across multiple years. As this study had a limited number of subjects (n = 2), used 2 different species of differing ages, and was conducted in an aquarium setting with controlled lighting and water temperature, specific conclusions cannot be made about T5 concentrations or seasonality for these species as a whole. More testing is needed to effectively determine seasonal T5 profiles and average concentrations in males of these species.
Deslorelin c implants have been successfully used to suppress reproduction and reduce hormone concentrations in a variety of species.2,10,11 It was hypothesized that deslorelin treatment would reduce or eliminate the seasonal T5 increase commonly seen in sea turtles.3,4,9,13,14,20,25 Testosterone suppression was observed in C. mydas following the fourth deslorelin treatment (December 2012). There was no known habitat, husbandry, or social changes during this timeframe that could account for the observed seasonal suppression. Therefore, we speculate that deslorelin may have worked through a latent or additive effect to reduce circulating T5. Further study is needed to determine the effective dose and mechanism of deslorelin efficacy in male C. mydas and other sea turtle species.
The validation of 2 commercial EIA kits along with the in-house system provides options for measuring T5 in sea turtles. Although there is slight variation in measured sample values between the EIA kits, the similarity in pattern of seasonal changes in T5 across the kits suggests that either kit may be used to measure seasonal changes in T5 with similar results.
We propose several recommendations for analyzing testosterone in blood samples from male sea turtles. Blood samples should be collected in serum with no additives, serum with thrombin, or plasma with lithium heparin collection tubes. Serum and plasma sample homogenization is only recommended if an increased sample volume is needed. If testosterone testing cannot be performed within 1–2 hr post-collection, samples should be frozen, and ether extraction or dissociation reagent treatment must be utilized prior to hormone assay. Dissociation reagent treatment may be preferred for its increased efficiency of sample processing.
Data from our study has implications for care and management of sea turtles in captive settings, but also important conservation implications. Measurement of sex steroid concentrations is frequently cited as a tool to determine the reproductive status of free-ranging threatened and endangered sea turtles, or for determining the sex of juvenile turtles or hatchlings.4–6,8,16,24 Accurate measurement of T5 is necessary in these situations to draw clear conclusions about sea turtle reproduction and populations. Ultimately, the results of this study regarding sample collection, extraction, and treatment provides clinicians and laboratory diagnosticians with a set of guidelines for accurately measuring T5 in sea turtles. Similar validation tests should be performed for all other hormones measured on EIA and RIA systems in sea turtle species. 15 On a broader scale, this study further highlights the necessity of thorough methodological validation for any species or biological sample type. 21
Footnotes
Authors’ contributions
KM Graham contributed to design of the study; contributed to analysis and interpretation of data; and drafted the manuscript. ND Mylniczenko contributed to conception and design of the study, and contributed to acquisition, analysis, and interpretation of data. CM Burns contributed to acquisition of data. TL Bettinger contributed to interpretation of data. CJ Wheaton contributed to conception and design of the study; contributed to analysis and interpretation of data; and drafted the manuscript. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Testosterone EIA kit (goat anti-rabbit T5 antisera; catalog no. K032-H5R), Arbor Assays, Ann Arbor, MI.
b.
Testosterone EIA kit (goat anti-sheep T5 antisera; catalog no. K032-H5S), Arbor Assays, Ann Arbor, MI.
c.
Suprelorin (gonadotropin-releasing hormone agonist, 4.7-mg implants), Virbac Animal Health Inc., Milperra, New South Wales, Australia.
d.
10 beads per tube; Spin-Quik, Oti Specialties, Santa Monica, CA.
e.
BD Vacutainer plastic blood collection tubes for trace element testing: serum clot activator (catalog no. 368380), BD Diagnostics, Franklin Lakes, NJ.
f.
BD Vacutainer rapid serum tube with thrombin-based clot activator (catalog no. 368774), BD Diagnostics, Franklin Lakes, NJ.
g.
BD Vacutainer plastic blood collection tubes with lithium heparin: Hemogard (catalog no. 367885), BD Diagnostics, Franklin Lakes, NJ.
h.
BD Vacutainer glass blood collection tubes: citrate solution (catalog no. 366415), BD Diagnostics, Franklin Lakes, NJ.
i.
BD Vacutainer glass blood collection tubes with K3 EDTA (catalog no. 366450), BD Diagnostics, Franklin Lakes, NJ.
j.
Ethyl ether (anhydrous, ACS grade ≥99%; catalog no. E138-1), Fisher Scientific, Fair Lawn, NJ.
k.
Dissociation reagent (catalog no. X017), Arbor Assays, Ann Arbor, MI.
l.
“Tissue-Tearor” (rotor speed: 5,000–35,000 rpm; model no. 985370-395), BioSpec Products, Bartlesville, OK.
m.
Testosterone antisera R156/7 (prepared in rabbit), C. Munro, University of California–Davis, Davis, CA.
n.
Nunc 96 micro well polystyrene plate (catalog no. 12-565-135), Thermo Scientific, Waltham, MA.
o.
Testosterone ≥98% (catalog no. T1500), Sigma-Aldrich, St. Louis, MO.
p.
EMax endpoint ELISA microplate reader, Molecular Devices, Sunnyvale, CA.
q.
SoftMax Pro 4.8, Molecular Devices, Sunnyvale, CA.
r.
Goat anti-rabbit IgG (catalog no. A009), Testosterone EIA kit (goat anti-rabbit T5 antisera; catalog no. ISWE001); Arbor Assays, Ann Arbor, MI
s.
SYSTAT 13, Systat Software Inc., Chicago, IL.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
