Abstract
The current study was designed to validate an immunofluorimetric method to measure free triiodothyronine (fT3) and free thyroxine (fT4) concentrations in the serum of buffalo and to develop a better understanding of thyroid metabolism in buffalo (Bubalus bubalis) of different ages and reproductive statuses. Free triiodothyronine and fT4 were determined in sera from 4 groups of 10 animals, each representing 5–10-year-old lactating buffalo, 2–3–year-old first calf heifers, 1–1.5-year-old virgin heifers, and 5–10–year-old dry buffalo. The average sensitivity of the immunofluorimetric assay was 5.9 pmol/l for fT3 and 14.7 pmol/l for fT4. Intra-assay and interassay coefficients of variation were 5.6% and 5.1% for fT3 and 1.5% and 4.3% for fT4, respectively. Virgin heifers had higher average values for fT3 and fT4, while lactating buffalo had the lowest average values. Dry buffalo and first calf heifers had intermediate fT3 and fT4 values. The findings of this study suggested that lactating buffalo were in a state of low thyroid activity, while virgin heifers had increased thyroid activity.
Introduction
Thyroid hormones in buffalo (Bubalus bubalis), as well as in other species, influence reproductive efficiency. A previous study observed a correlation between plasma protein-bound iodine and follicular development interval, postpartum estrus interval, service period, and number of inseminations for conception. 4 Thyroid hormones are also important for the growth and development of the mammary gland and for increasing milk production. 5,13 However, the importance of thyroid hormones in postpartum metabolism of buffalo requires further investigation.
Amino acid composition of buffalo thyroglobulin is generally similar to that of other species. However, buffalo thyroglobulin has 2% more proline, and 5 glutamic acid is the sole N-terminal amino acid. In contrast, several other animal species have aspartic acid or asparagine as the N-terminal amino acid. 9 Despite a slightly different amino acid structure of thyroglobulins in various animals, the concentration of thyroid hormones has been measured with high efficiency using a radioimmunoassay with rabbit antithyroglobulin antibody. 7
The current study was designed to validate an immunofluorimetric analytical method to investigate the concentration of free triiodothyronine (fT3) and free thyroxine (fT4) of buffalo under various physiological and reproductive conditions. The delayed enhanced lanthanide fluorescence immunoassay (DELFIA) is an immunofluorimetric, time-resolved, solid-phase technique, based on the principles of titration and differential separation of thyroid hormones. This analytical technique uses a secondary antibody labeled with Europium, a metal chelate lanthanoid. These lanthanoid chelates have a fluorescence decay time of microseconds, compared with other organic molecules classically used as fluorophores that have a decay time of nanoseconds. The shorter decay times of lanthanoids allows excellent separation of the marker emission from background emission, provided a long enough time of decay is present to allow an adequate reading of the emission spectra. In addition, the difference between the wavelengths of emission and excitation (stokes shift) for lanthanoids allows for more accurate and easier measurements. The weak fluorescence of the lanthanoid chelate becomes more intense with the addition of an acidic amplification solution (enhancement solution) that dissociates the Europium ion from the lanthanoid chelate-antibody bond. The Europium ion subsequently is incorporated into micelles that protect the metal lanthanoid from the decay due to the presence of water molecules. The emission signal is detected at a wavelength of 613 nm using a fluorimeter with a xenon lamp. Under standard conditions, the fluorescence emitted is inversely proportional to the concentration of the antigen in the sample.
Furthermore, a validated fluorimetric assay could be useful to detect alterations in thyroid hormone concentrations of buffalo during the postpartum period or during the development of certain metabolic diseases such as hypothyroidism, hyperthyroidism, milk fever, muscle rigidity, and uterine and/or vaginal prolapse. Hypothyroidism, previously described in cattle, has recently been described in African buffalo. 1 If hypothyroidism occurs during pregnancy in buffalo, it could possibly explain abnormalities in placental detachment, low birth weight of progeny, stillbirths, and postpartum hemorrhage.
Materials and methods
Experimental animals
Forty clinically healthy buffalo were divided into 4 groups of 10 animals each related to age and physiological condition. These groups included: 1) 5–10–year-old lactating buffalo (pregnant about 6 months), 2) 2–3–year-old first calf heifers (pregnant about 6 months), 3) 1–1.5–year-old virgin heifers, and 4) 5–10–year-old dry buffalo (pregnant about 9 months).
Feeding
Animals were fed early in the morning, between 7:30 and 9:00 am. The pregnant buffalo and virgin heifers received a diet of maize silage (10 kg), feed concentrates (2 kg), and straw (7 kg). The diet was modified after calving by increasing the quantity of maize silage (18 kg), feed concentrates (5 kg), and straw (5 kg).
Blood samples
Blood samples (40 ml) were obtained between the end of October and early November. Animals were bled in the morning between 7:00 and 7:30 am to minimize the effect of diurnal variation. Blood samples were collected by jugular venipuncture into vacutainers. The blood samples were allowed to clot. Following centrifugation, the serum was harvested, placed in a new container, and stored at −20°C until analyzed.
Laboratory methods
A previously validated DELFIA method was used to determine fT3 and fT4 in serum. This method was based on the principles of titration and separation of thyroid
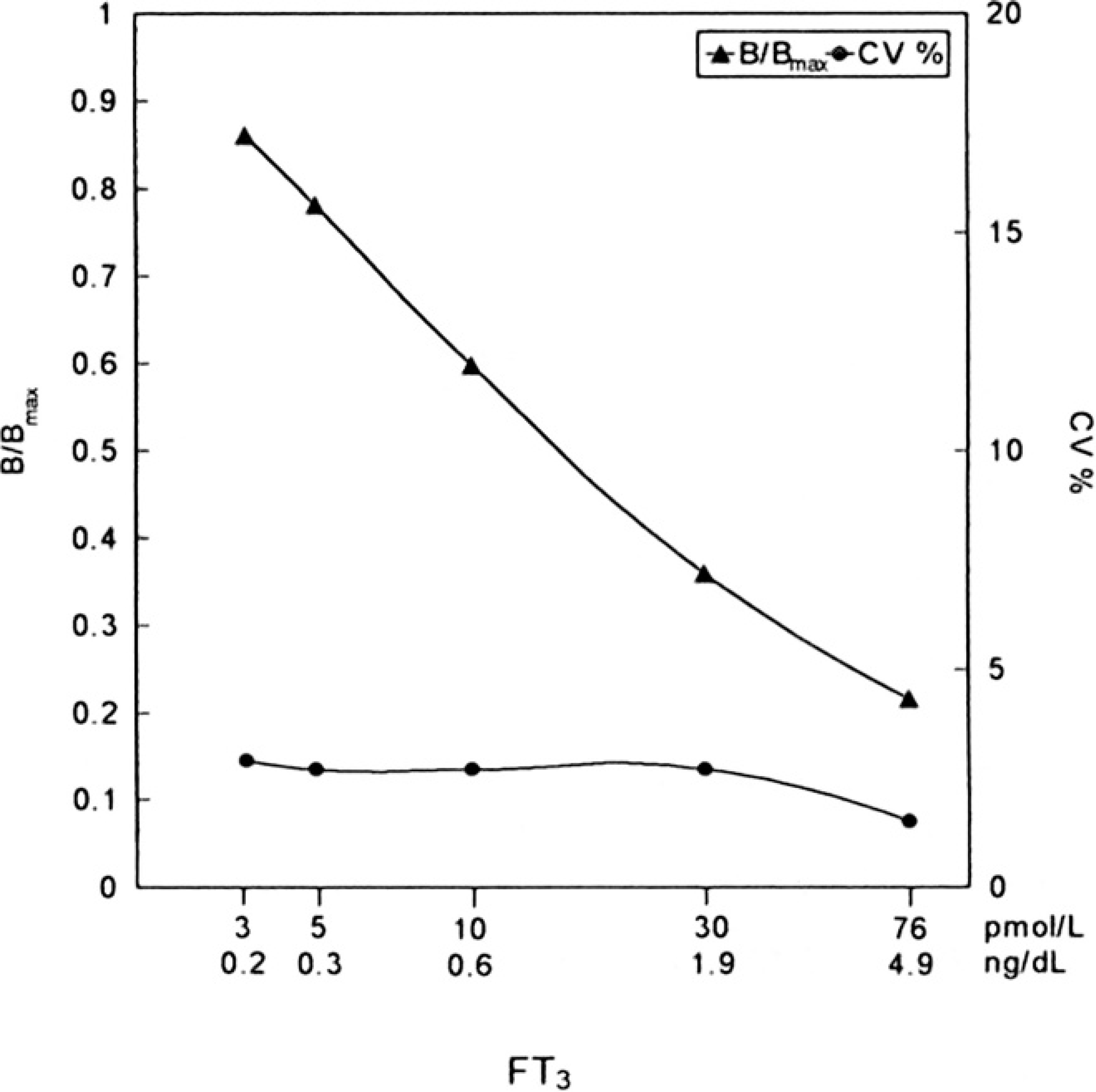
Delayed enhanced lanthanide fluorescence immunoassay (DELFIA) free triiodothyronine (fT3) calibration curve.
hormones using a secondary antibody labeled with a lanthanoid chelate containing Europium. The anti-triiodothyronine/thyroxine monoclonal antibody reacted with anti-mouse IgG immobilized on a solid phase (I a incubation). In a second incubation step, the anti-triiodothyronine/thyroxine monoclonal antibody bound the antigenic sites of fT3 and fT4. Buffer and extraneous serum were removed by washing. In a third incubation step, anti-triiodothyronine/thyroxine antibody was marked with Europium. Addition of the enhancement solution allowed the separation of Europium ions from the chelated, bound antibody. The fluorescence emitted was inversely proportional to the fT3 and fT4 concentrations.
Statistical analyses
The mean and standard deviation were calculated mathematically. Significant differences in the data were detected by analysis of variance for repeated measures using a level of significance of P 0.01. Regression analysis was used to compare the 2 DELFIA test kits.
Results
Analytical characteristics of the DELFIA fT3 method
The buffalo serum samples were used to construct a calibration curve for fT3, as presented in Figure 1. The imprecision of the analytical method was calculated through 139 double measurements of standards, controls, and serum samples using the MultiCalc data management program. a
Precision. The precision of the DELFIA fT3 analytical method was determined using a serum sample with low fT3 concentration (repeatability at 1 concentration level). This sample was evaluated in
Results of a standard curve constructed from serial dilutions of a known specimen. *
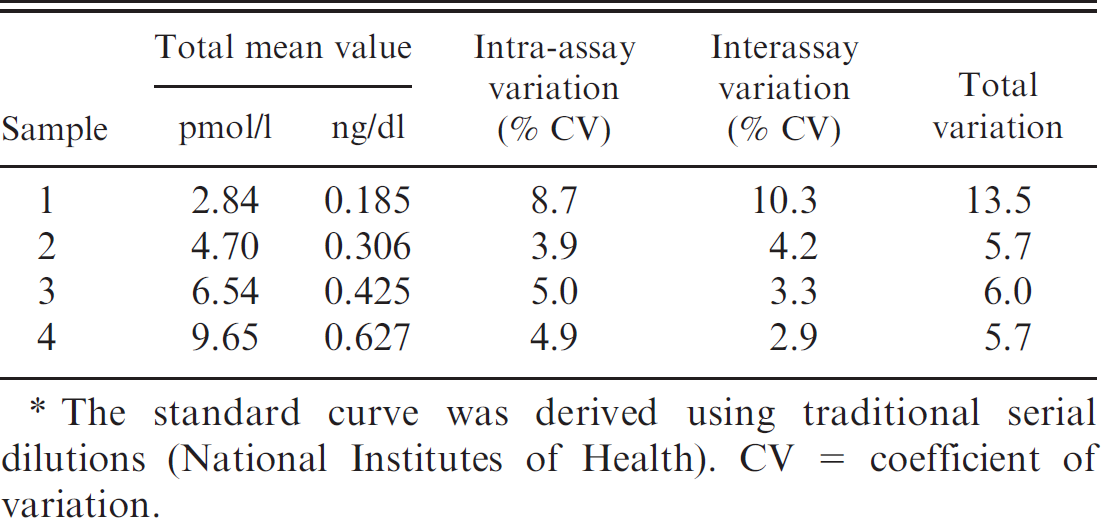
The standard curve was derived using traditional serial dilutions (National Institutes of Health). CV = coefficient of variation.
quadruplicate at 27 different times. The percentage coefficient of variation was subsequently calculated and is presented in Tables 1 and 2.
Accuracy. Three sets of 5 experimental measurements were done on a sample of known fT3 concentration, which resulted in a mean and standard deviation of 5.1 + 0.71 pg/ml. The concentration of fT3 in the samples from the field buffalo was 5.30 + 0.57 pg/ml.
Analytical sensitivity. The analytical sensitivity of the DELFIA fT3 was 1.5 pmol/l (0.10 ng/dl). For the purposes of this study, the analytical sensitivity was defined as the difference between the mean value and 2 standard deviations based on 8 measurements (average value–2 SD; n = 8).
Correlation. The DELFIA fT3 kit B063–201 (y) was compared with the DELFIA fT3 kit B063–101 (3-step) (–) by regression analysis using samples with an fT3 concentration ranging from 3.58 to 41.1 pmol/l (0.233–2.67 ng/dl). The correlation was determined to evaluate the different analytical sensitivity of the 2 kits. The correlation was y = 0.97– + 0.90 (r = 0.995; n = 311).
Cross-reactivity. The cross-reactivity (50% level of divergence) with other substances was determined by measuring total triiodothyronine (T3) using the DELFIA fT3 antibody. The results are presented in Table 3. A higher average fT3 concentration (6.60 pg/ml) was found in 1–1.5–year-old heifers (group 3), while the lowest fT3 concentration (4.43 pg/ml) was
Results using the reference curve and calibrators. *
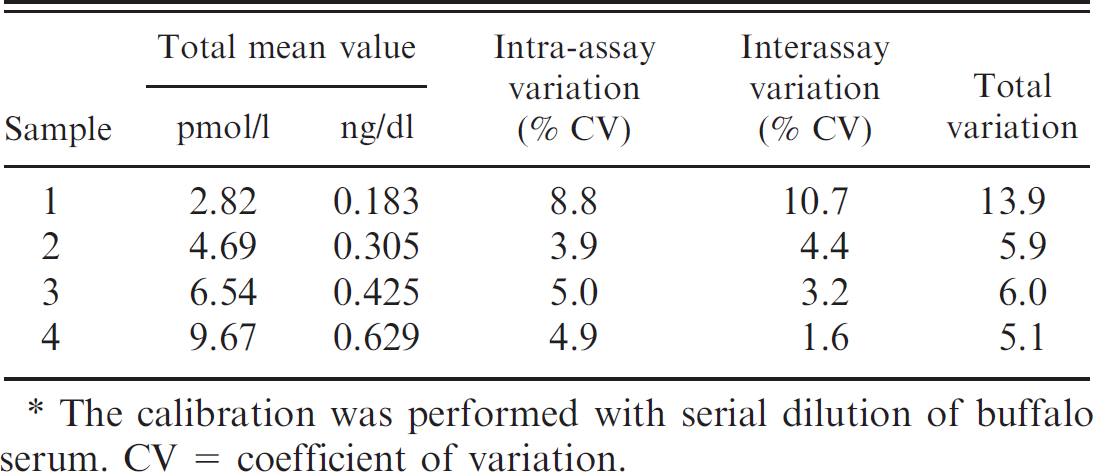
The calibration was performed with serial dilution of buffalo serum. CV = coefficient of variation.
Cross-reactivity of the antibody used in the delayed enhanced lanthanide fluorescence immunoassay free triiodothyronine test for various thyroid metabolites.
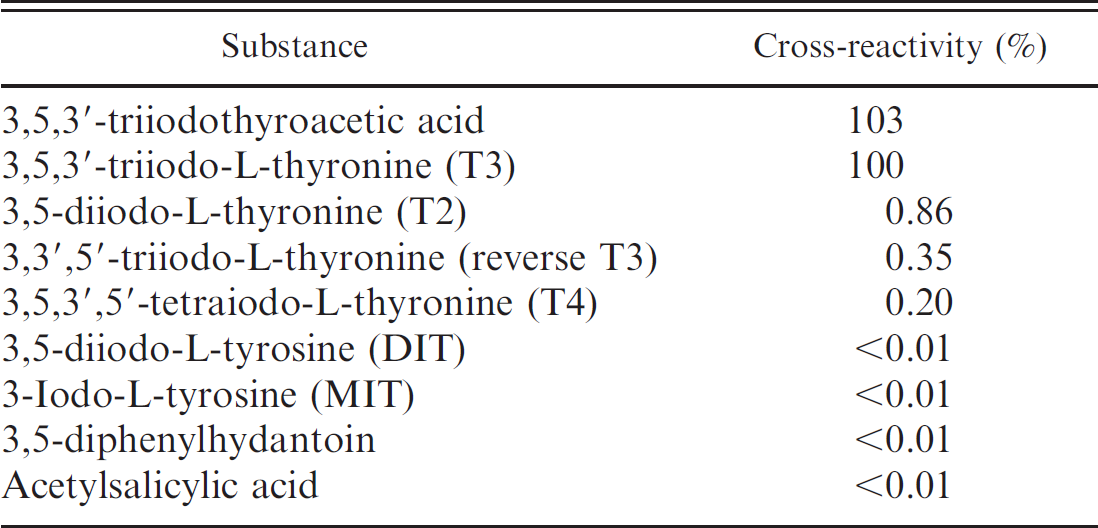
found in 5–10–year-old lactating buffalo (group 1). Intermediate fT3 concentrations were present in 5–10–year-old dry buffalo (fT3 = 4.57 pg/ml; group 4) and in first calf heifers (fT3 = 5.63 pg/ml; group 2).
Analytical characteristics of the DELFIA fT4 method
The buffalo serum samples were again used to construct a calibration curve for fT4, as presented in Figure 2. The imprecision of the method was calculated through 336 double measurements of standards, controls, and serum samples using the MultiCalc data management program.
Precision. The precision of the DELFIA fT4 method was determined using a serum sample of low fT4 concentration (repeatability at 1 concentration level). The specimen was analyzed in triplicate at 54 different times. The percentage coefficient of variation was calculated and is presented in Tables 4 and 5.
Accuracy. Four sets of 5 experimental measurements were done on a sample of known fT4
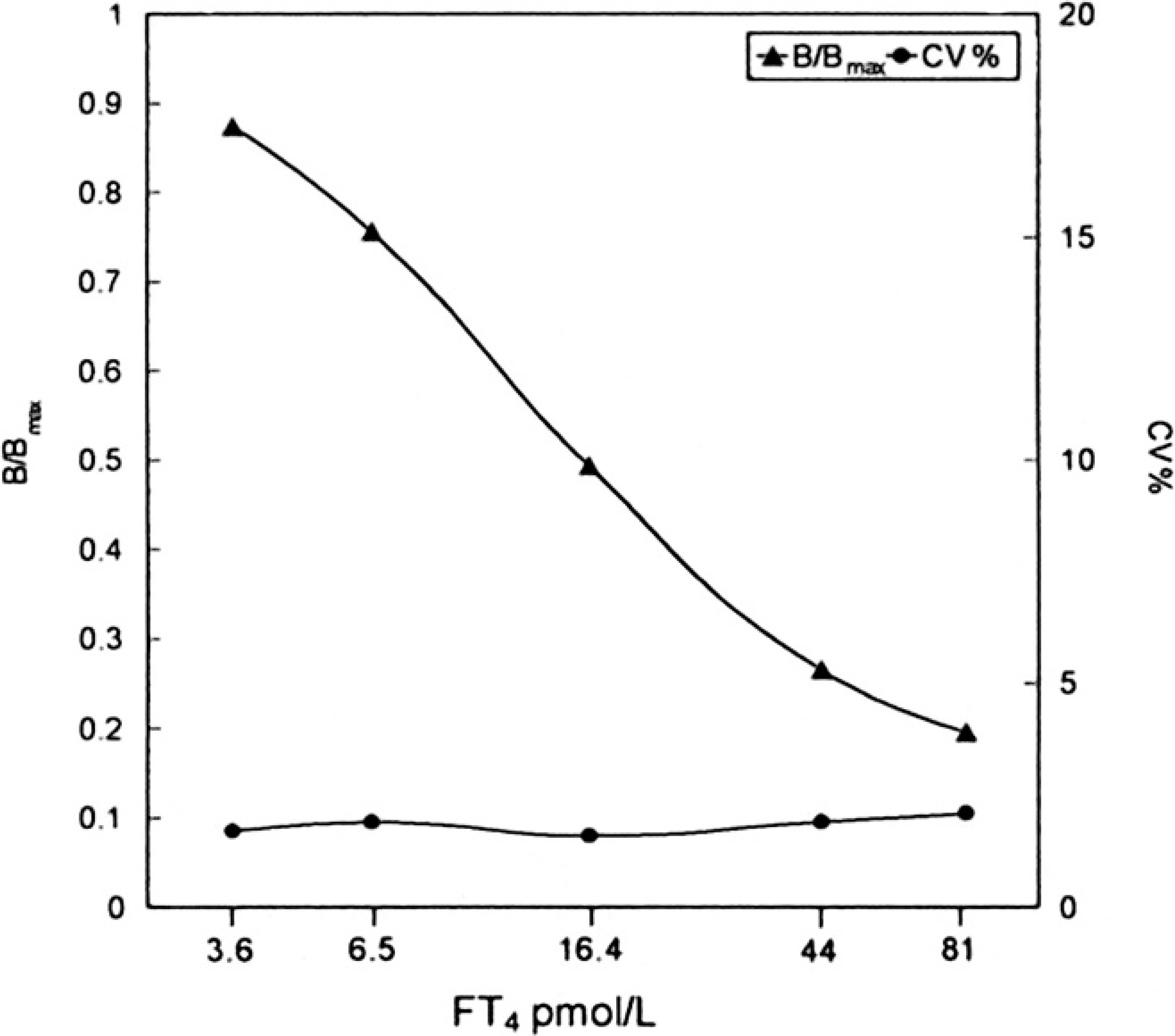
Delayed enhanced lanthanide fluorescence immunoassay (DELFIA) free thyroxine (fT4) calibration curve.
Intra- and interassay variation of the delayed enhanced lanthanide fluorescence immunoassay in the measurement of free triiodothyronine. *
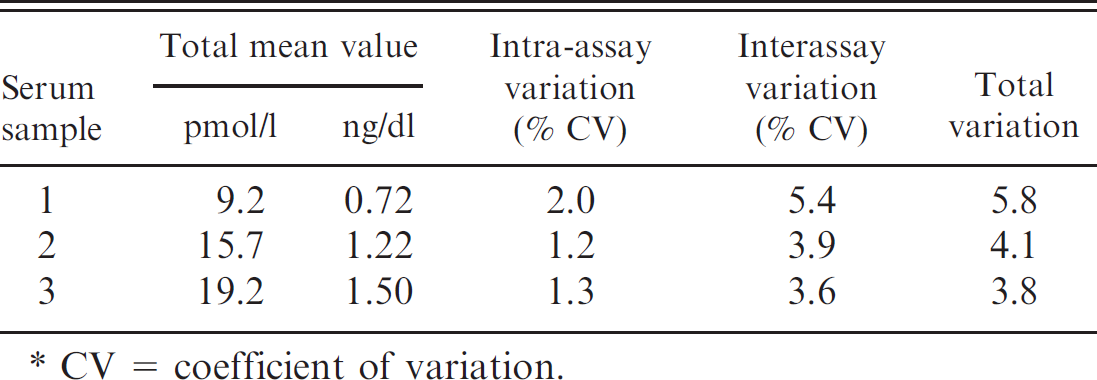
CV = coefficient of variation.
concentration, resulting in a mean and standard deviation of 14.90 + 2.80 pg/ml. The concentration of fT4 in the buffalo sera examined was 14.83 + 2.77 pg/ml and was within the experimental range of values.
Analytical sensitivity. Analytical sensitivity of the DELFIA fT4 was 2 pmol/l (0.16 ng/dl). In the current study, the analytical sensitivity was defined as the difference between the mean and 2 standard deviations for 72 measurements (average value–2 SD; n = 72).
Correlation. The DELFIA fT4 kit B061–201 (y) was compared with the DELFIA fT4 kit A061–201 (x) using samples with fT4 concentrations ranging from 4.5 to 84.3 pmol/l (0.35–6.60 ng/dl). The correlation was y = 0.99x + 0.39 (r = 1.00; n = 322). The DELFIA fT4 kit B061–201 (y) was compared with the DELFIA fT4 kit B061–101 (x) using samples with an fT4 concentration ranging from 5.1 to 83.1 pmol/l (0.40–6.50 ng/dl). The correlation was y = 1.03x–0.13 (r = 0.99; n = 322).
Cross-reactivity. Cross-reactivity (50% divergence level) with other substances was determined by measuring the total thyroxine (T4) concentration using the DELFIA fT4 antibody. The results are presented in Table 6. The values obtained by DELFIA in the 4 groups of animals are shown in Table 7, while the mean and standard deviation are reported in Table 8. A higher mean fT4 concentration was found in the 1–1.5-year-old virgin heifers (fT4 = 18.74 pg/ml; group 3). Conversely, the lowest average fT4 concentration was found in the 5–10-year-old lactating
Intra- and interassay variation of the delayed enhanced lanthanide fluorescence immunoassay in the measurement of free thyroxine. *
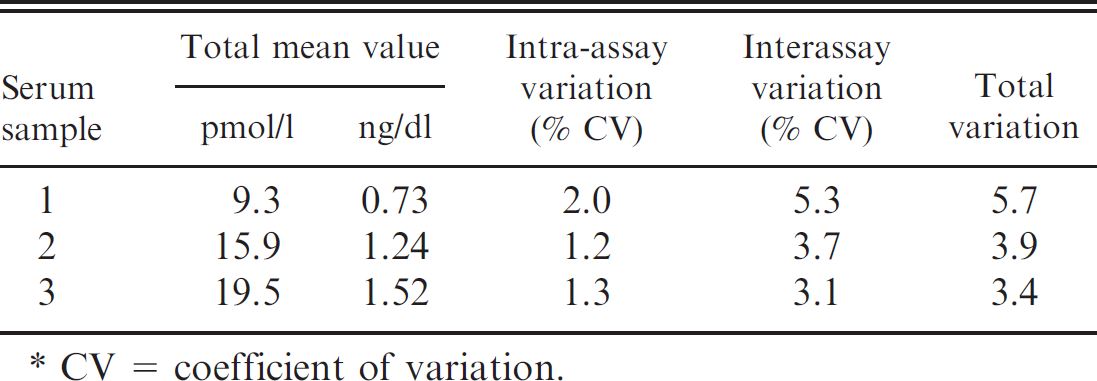
CV = coefficient of variation.
Cross-reactivity of the antibody used in the delayed enhanced lanthanide fluorescence immunoassay free thyroxine test for various thyroid metabolites.
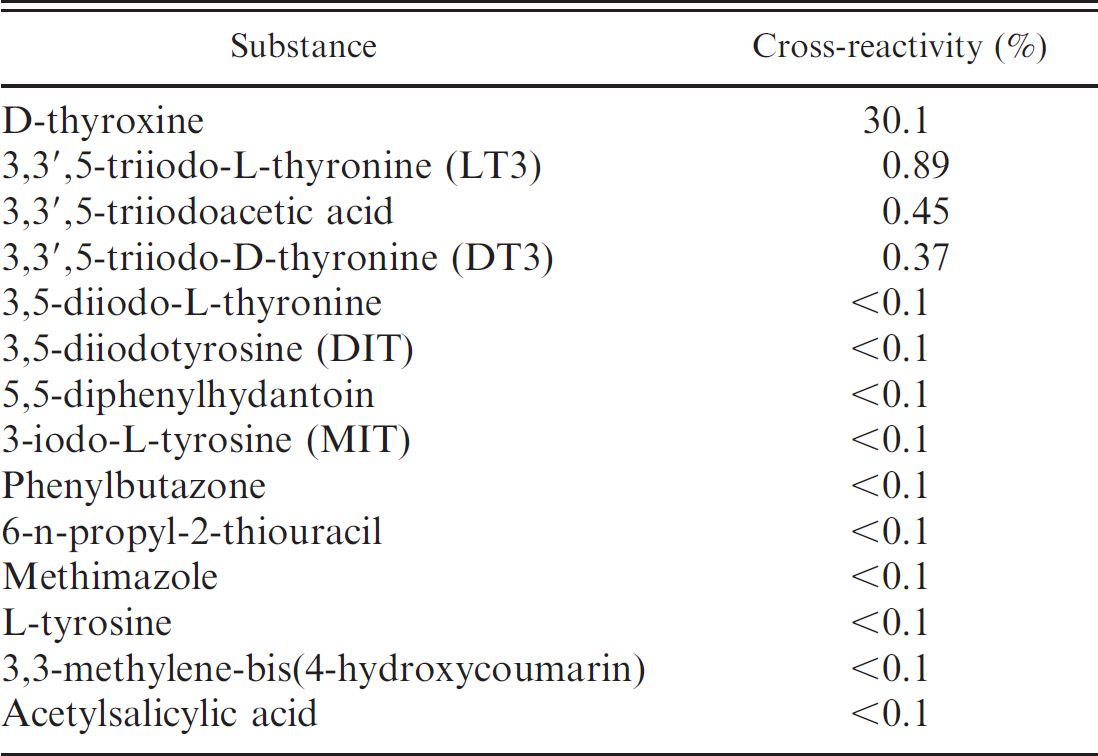
buffalo (fT4 = 12.39 pg/ml; group 1). Intermediate fT4 concentrations were present in the dry buffalo (fT4 = 13.43 pg/ml; group 4) and first calf heifers at (fT4 = 14.79 pg/ml; group 2)
General comments regarding fT3 and fT4 test results. With regard to fT3 (Fig. 3), group 1 had a mean of 4.43 pg/ml with a range of 3.48–6.12 pg/ml, group 2 had a mean of 5.63 pg/ml with a range of 4.83–7.04 pg/ml, group 3 had a mean of 6.61 pg/ml with a range of 5.38–7.58 pg/ml, and group 4 had a mean of 4.56 pg/ml with a range of 3.62–5.10 pg/ml. For fT4 (Fig. 4) analysis, group 1 had a mean of 12.39 pg/ml with a range of 9.90–17.60 pg/ml, group 2 had an average of 14.79 pg/ml with a range of 11.70–19.00 pg/ml, group 3 had a mean of 18.74 pg/ml with a range of 13.30–24.70 pg/ml, and group 4 had a mean of 13.43 pg/ml with a range of 10.00–19.30 pg/ml.
A significant difference was found for fT3 between group 1, which had lower values, compared with groups 2 and 3. Groups 2 and 3 had higher fT3 concentrations than group 4. No significant difference was observed in serum fT3 concentrations between lactating (group 1) and dry (group 4) buffalo or between first calf heifers (group 2) and virgin heifers (group 3). The fT4 concentrations were significantly higher in virgin heifers (group 3) compared with the other groups.
Discussion
The present study confirms that the DELFIA method is highly reliable and specific in the determination of fT3 and fT4 concentrations in buffalo sera. The values obtained by the DELFIA in the different groups of animals indicate that lactating buffalo have a low concentration of thyroid hormones. 13,14 The low fT3 and fT4 concentrations could be linked to a
Free triiodothyronine (fT3) and free thyroxine (fT4) concentrations in the serum of individual animals determined by the delayed enhanced lanthanide fluorescence immunoassay.
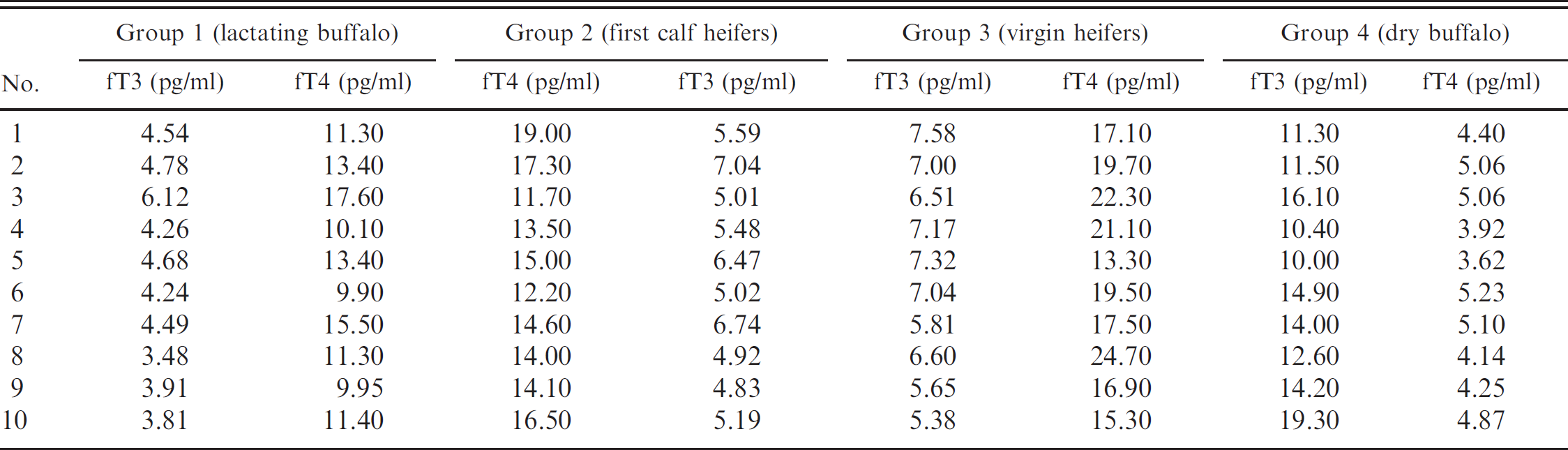
Free triiodothyronine (fT3) and free thyroxine (fT4) concentrations (– + SD) in buffalo serum during different physiological conditions. *
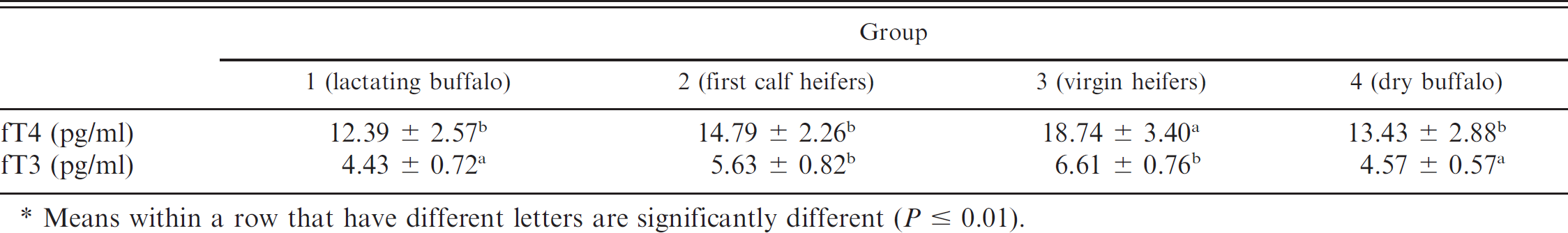
Means within a row that have different letters are significantly different (P 0.01).
secretion of these hormones into milk, as confirmed by previous studies in cattle. 2,3,11,17 However, other studies have shown that total and fractionated thyroid hormones are present in milk as a result of 5’ deiodinase activity of liver and breast. 14 Several studies have also indicated that thyroid hormones are present in milk in low concentrations, and their presence diminishes as milk production progresses. 13,14 Recent studies have attributed a significant role to 5’ deiodinase activity to cells of the mammary gland that transform T3 and T4 into inactive fragments. 6,10,14,16
The lack of a significant difference in fT3 and fT4 concentrations between lactating and dry buffalo
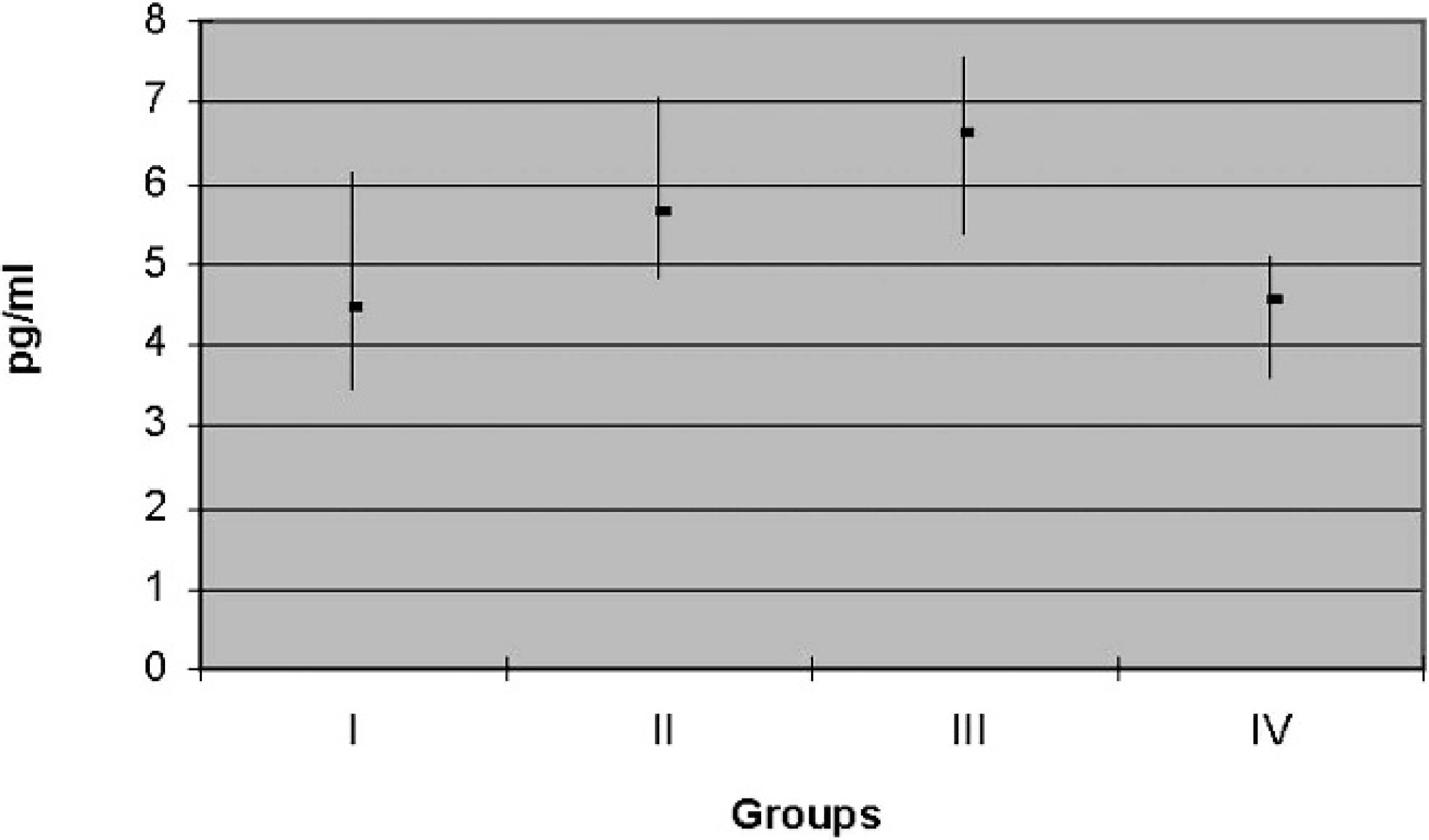
Free triiodothyronine (fT3) mean and range (pg/ml) for each group.
could be due to an increased need for hormonal iodates by the fetus. A marked increase in fetal fluids at the end of gestation could result in a greater distribution volume of circulating hormones. The above results are partially in agreement with lower serum fT314 concentrations of dry buffalo compared with those of animals at the beginning, middle, or end of lactation. 12,15 Some researchers have previously noted low concentrations of total thyroid hormones at the end of gestation and at the time of the parturition and have related these observations to altered bone turnover and reduction of serum osteocalcin. 7 The lack of a difference in fT3 and fT4
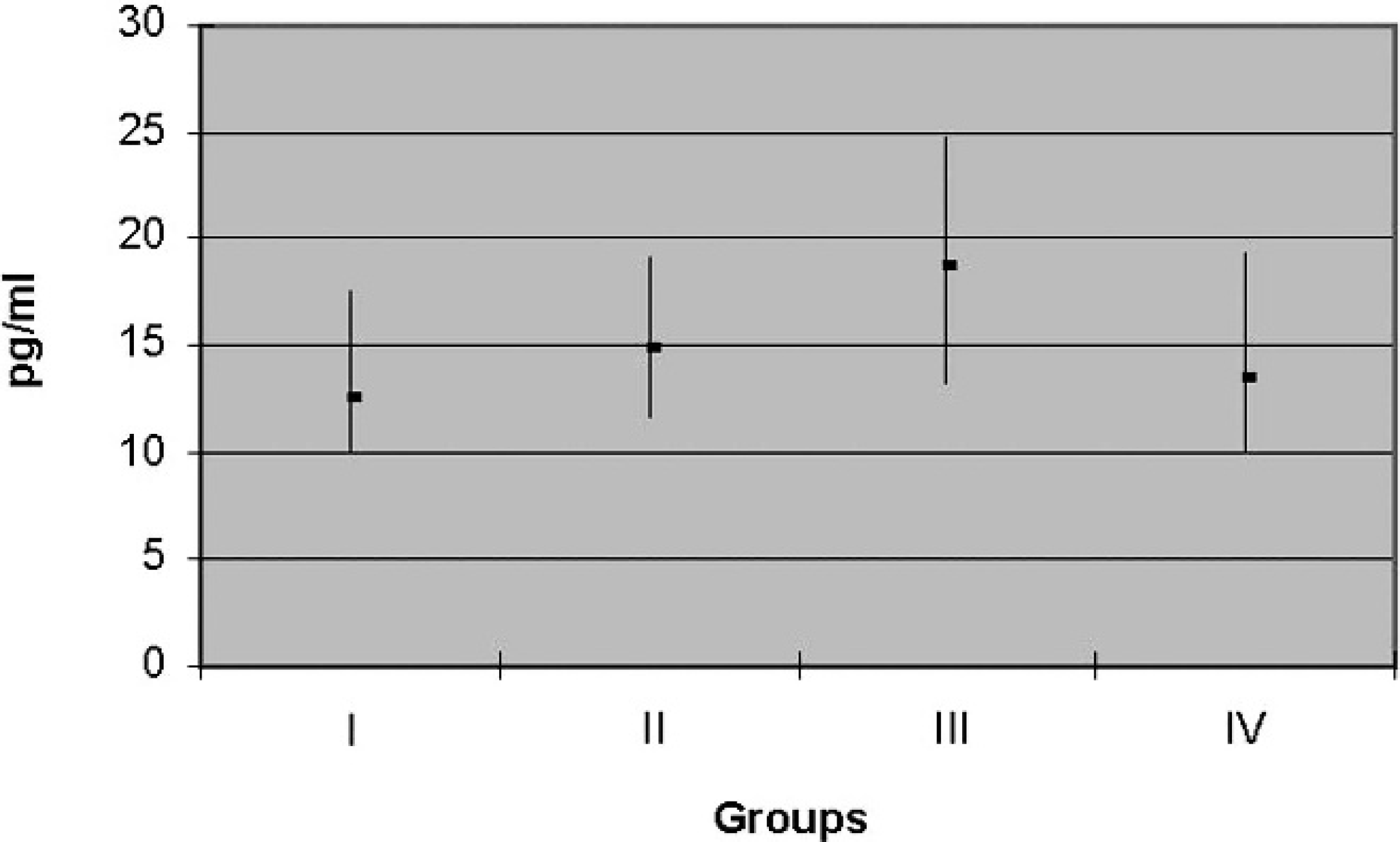
Free thyroxine (fT4) mean and range (pg/ml) for each group.
concentrations between the 2 groups of pregnant heifers is more difficult to explain. Pregnancy is commonly associated with increased thyroid activity due to an increase in the uptake of iodine that may be caused by competition for iodine between mother and fetus. The placenta also may produce a factor similar to TRH. 8 Perhaps plasma proteins have an increased affinity for binding to thyroid hormones that is mediated by progesterone during pregnancy. This might increase the amount of available total T3 and T4 while reducing fT3 and fT4 fractions. The concentrations of fT4 are significantly higher in 1.5–2–year-old heifers compared with first calf heifers as well as with the other groups. This difference could be age related because thyroid activity is greater in young animals during the growth period.
In conclusion, the current study demonstrates that the DELFIA assay can measure fT3 and fT4 accurately in buffalo serum. Furthermore, thyroid metabolism in buffalo changes remarkably in different physiological states. Lower thyroid hormone concentrations were associated with lactation, while higher hormone concentrations were associated with young virgin heifers.
Footnotes
a.
ConSoft GmBH, Hannover, Germany.
