Abstract
Serotonin regulates many intestinal motor and sensory functions. Altered serotonergic metabolism has been described in human gastrointestinal diseases. The objective of our study was to compare expression of several components of the serotonergic system [serotonin (5-HT), serotonin reuptake transporter protein (SERT), tryptophan hydroxylase-1 (TPH-1), 5-HT receptor2B (5-HT2B)] and the enterochromaffin cell marker chromogranin-A (CgA) in the intestinal mucosa between dogs with chronic enteropathy and healthy controls. Serotonin and CgA expression were determined by immunohistochemistry using banked and prospectively obtained, paraffin-embedded canine gastrointestinal biopsies (
Keywords
Introduction
The gastrointestinal (GI) tract is the only organ that manifests complex integrative behaviors and reflexes in the absence of input from the central nervous system, mainly because of the enteric nervous system. 16 The enteric nervous system includes both myenteric and submucosal neuronal plexi, spinal, and vagal afferent neurons, and the production of both amine and peptide hormones by mucosal enterochromaffin cells (ECCs). The role of the enteric nervous and neuroendocrine system in both normal gastrointestinal motility and dysmotility states, such as irritable bowel syndrome (IBS) in humans, has been extensively reviewed.7,39 This neuroendocrine system of the gut plays a significant role in regulating intestinal motor and sensory functions, as well as modulating inflammation. A major mediator for this system is serotonin (5-HT), a monoamine neurotransmitter produced both in the central nervous system and peripherally in ECCs. In the context of GI disease, changes in serotonergic metabolism have received relatively little attention in veterinary species. This is in marked contrast to human medicine, where 5-HT metabolism in the GI tract has been an area of active interest for more than half a century. 32 The intestinal mucosa is the dominant site of 5-HT synthesis in mammals, with >95% of the intestinal 5-HT present in the ECCs. 33 When released from ECCs, 5-HT acts locally to regulate intestinal motility, fluid secretion, and regional blood flow. 20 Serotonin excess is associated with autonomic hyperactivity of the intestinal tract. 32 Although the role of 5-HT varies in the pathophysiology of GI diseases, 2 of its most important roles are its influence on GI motility and modulation of the immune system. There is a 2-way communication between the immune system and ECCs; inflammation can influence 5-HT expression, while 5-HT can also influence the immune system, as many 5-HT receptors are found on various immune cells. 30
Canine patients may present with clinical signs of GI dysfunction, such as vomiting, diarrhea, inappetence, and weight loss, resulting from a broad variety of etiologies. A common feature of many of these conditions is the presence of GI inflammation, which may be the result of dietary intolerance (so-called “food-responsive diarrhea” 28 ), intestinal dysbioses, chronic colonization by bacterial pathogens (so-called “antibiotic-responsive diarrhea”21,41), or the condition may be idiopathic. As classically understood, canine idiopathic inflammatory bowel disease (IBD) is characterized by persistent or recurrent clinical signs of GI disease of unidentified cause, associated with histologic evidence of inflammatory infiltration of the intestinal mucosa.10,22-24,35 The final diagnosis of an individual patient as food-responsive, antibiotic-responsive, or idiopathic IBD depends on rigorous completion of therapeutic trials to rule out the food- and antibiotic-responsive diseases. In many cases, particularly in retrospective studies, a definitive diagnosis cannot be made from the record available, and thus many authors prefer the less restrictive term “chronic enteropathy.”1,2,6 This term is used in our report because, in many of these cases, a true diagnosis of idiopathic inflammatory disease cannot be made with a high degree of certainty.
Chronic enteropathy is a common cause for presentation of canine patients to veterinarians, with long-term management being challenging in many cases. Given its effects on the intestine, 5-HT may be a therapeutic target to help control the clinical signs of dogs with chronic enteropathies.
The aim of our study was to compare the expression of 5-HT, tryptophan hydroxylase-1 (TPH-1), 5-HT receptor2B (5-HT2B), serotonin reuptake transport protein (SERT), and chromogranin-A (CgA) in the intestinal mucosa between dogs diagnosed with chronic enteropathy (
Materials and methods
Study populations
Immunohistochemistry study
The medical database of our hospital (School of Veterinary Medicine, Oregon State University, Corvallis, Oregon) was searched for dogs diagnosed with chronic enteropathy between 2002 and 2011. Criteria for selection included histologic evidence of intestinal inflammation, the presence of a complete medical record, and adequate availability of paraffin-embedded tissue samples. Unstained control sections
a
(
All hematoxylin and eosin (HE)-stained sections of intestinal biopsy samples from both the chronic enteropathy and control groups were assessed and characterized by use of histologic guidelines for the evaluation of GI inflammation set by the World Small Animal Veterinary Association (WSAVA) International GI Standardization Group. 40 Briefly, these guidelines provide a rubric for scoring of duodenal mucosal pathology through the assessment of morphological features (villous architecture, crypt and lacteal dilation, fibrosis) and the presence and number of intraepithelial and lamina propria lymphocytes, neutrophils, and eosinophils. The scores for these parameters are added to derive a diagnosis and designation of the mucosal pathology as normal, mild, moderate, or markedly inflamed. All HE-stained intestinal mucosal biopsy specimen sections were evaluated by a board-certified veterinary pathologist (BA Valentine) and a small animal internal medicine resident (C Bailey).
The number of cells staining positive for 5-HT and CgA were assessed separately for each animal. For each sample, the number of cells staining positive for the compound of interest was manually counted in 20 random high power fields (HPFs) per slide, and the average of the 20 HPFs were recorded for subsequent analysis. Sections were analyzed entirely via light microscopy and manual counting; no digital image analysis was carried out.
Quantitative RT-PCR study
For this arm of the study, biopsy specimens were obtained from the proximal to middle segments of the duodenum of dogs (
Immunohistochemistry methods
Serotonin
Tissues were sectioned at 4–5 µm, and the sections were collected on positively charged slides and baked at 60°C for 1 h. Slides were deparaffinized and rehydrated through 2 changes of xylene, 2 changes of 100% ethanol, and 1 change of 80% ethanol and water. Slides were washed in Tris-buffered saline–Tween 20 (TBST c ) followed by 3% H2O2 d in TBST for 10 min. Proteinase K e (0.6 U/mL) was applied for 5 min and slides were then washed in TBST; serum-free protein block f was applied for 10 min, and finally the slides were air-dried. The primary antibody to 5-HT (mouse monoclonal antibody) g was diluted 1:40 in a commercial antibody diluent i and applied for 30 min at room temperature. A mouse universal negative control (N1698 f ) was used as the negative control. After washing in TBST, anti-mouse immunoglobulin (Ig)G horseradish peroxidase (HRP) conjugate j was applied for 10 min at room temperature, and slides were again washed in TBST. The chromogen Nova Red k was applied for 5 min; slides were then washed in dH2O followed by hematoxylin l diluted 1:3 in dH2O for 5 min then rinsed in distilled H2O, rinsed in TBST to blue, run down to xylene, and cover slipped.
Chromogranin-A
Tissues were processed as described for 5-HT, including the use of mouse universal negative control, with the following exceptions. High temperature antigen retrieval was performed in a microwave pressure cooker using a commercial antigen retrieval solution m for 10 min after pressure was reached. The pressure cooker was slowly vented, and the container holding the slides was allowed to sit for 20 min at room temperature. The primary antibody to CgA was of rabbit origin (polyclonal). n This antibody is routinely used for detection of canine neuroendocrine cells by our diagnostic pathology service. The antibody was diluted 1:1,000 in antibody diluent i and applied for 30 min at room temperature. Anti-rabbit IgG conjugate o was applied for 10 min at room temperature, and the slides were again washed in TBST.
RNA extraction and complementary DNA synthesis
The GI mucosa was separated from the muscularis and serosal layers by gentle scraping with a sterile razor blade before RNA extraction. Total RNA was extracted from 1 intestinal mucosal biopsy sample from each dog (chronic enteropathy group and control group 2) with a commercial RNA extraction kit. p The concentration of RNA yielded was determined by measuring the absorbance at 260 nm. One microliter of RNA-containing solution was used to synthesize first-strand complementary (c)DNA with a mix of oligo-dT and random primers, q in accordance with the manufacturer’s instructions.
Quantitative RT-PCR assays
Serotonin receptor2B, TPH-1, and SERT were assayed with asymmetric cyanine dye–based 2-step real-time qRT-PCR assays. For these assays, cDNA prepared from each total RNA sample was used. Gene-specific primer sets previously published for canine 5-HT2B and TPH-1 were used. 20 A commercially available qRT-PCR assay r was used to analyze SERT expression.
A commercially available housekeeper gene array s was used to select a combination of housekeeper genes with stable expression in the sample set used. The final analysis used the geometric mean of the expression of the housekeeper genes β-glucuronidase, hypoxanthine phosphoribosyl transferase 1, and TATA box–binding protein. 22
Each cDNA sample was diluted to a concentration of 1.0 ng/μL prior to qRT-PCR assays. Addition of 2.5 μL of cDNA to each 10-μL assay mixture resulted in a final cDNA concentration of 0.25 ng/μL. The reaction mixture also contained forward and reverse primers (5 µM each), Power SYBR mix, t and nuclease-free water. The cDNA samples and negative control samples without template sequences were placed in triplicate in wells of 96-well qRT-PCR assay plates. Serotonin receptor2B and TPH-1 reaction mixtures were exposed to the following thermal protocol: 10 min at 95°C, 40 cycles of 15 sec at 95°C, 30 sec at 60°C, and 30 sec at 72°C, then melt curve analysis at 60°C. Based on the assay manufacturer’s recommendations, SERT reaction mixtures were exposed to the following thermal protocol: 15 min at 95°C, 40 cycles of 15 sec at 94°C, 30 sec at 55°C, and 30 sec at 72°C, then melt curve analysis at 60°C. Quantification cycle values were determined for each target using the thermocycler software, u and relative quantitative analysis was performed utilizing an open source qRT-PCR package v using the ∆∆CT method. Target sequence expression values were normalized to the geometric mean of the 3 housekeeper genes, using a randomly selected, apparently normal dog sample as a reference sample.
Statistical analyses
Normality of the data was evaluated using the D’Agostino–Pearson test. Where data was not consistent with a Gaussian distribution, nonparametric methods were used for data analysis. The difference between study groups in the mean number of 5-HT immunopositive ECCs was analyzed using Student
Results
Study populations
Immunohistochemistry study
Two hundred thirty-seven dogs diagnosed with chronic enteropathy were identified from the hospital database review. Two hundred twenty-four dogs were excluded because of incomplete records, unavailability of records, and/or inadequate sample size of biopsy specimens available for review. Two dogs had only large intestinal biopsy specimens available for review, and were not included in this study. Duodenal biopsy specimens from the remaining 11 dogs were included in the retrospective IHC arm of the study. All of the dogs had evidence of inflammation within the small intestinal mucosa; however, most sections were graded as insignificant to mild levels of disease under the WSAVA criteria for GI inflammation. Histological diagnoses in this group included: predominantly lymphocytic/plasmacytic disease (4/11), predominantly eosinophilic disease (4/11), mixed lymphocytic/plasmacytic and eosinophilic disease (2/11), and plasmacytic disease (1/11). There were 4 spayed females and 7 neutered males. The mean age was 6.2 years (range: 1–14 years). Breeds included 1 of each of the following: Australian Shepherd, Basset Hound, Bernese Mountain Dog, Bichon Frise, Border Collie, Boxer, cockapoo, Labrador Retriever, and Shih Tzu, and 2 with breed not specified.
The control group was derived from banked, paraffin-embedded tissues from 10 apparently healthy control dogs, euthanized for an unrelated project. The control group included mixed-breed, adult dogs of both sexes. Results of physical exams were unremarkable, and results of the routine histologic examination of intestinal biopsy specimens obtained from these control dogs were unremarkable.
Quantitative RT-PCR study population
Gastrointestinal biopsies of a further 10 dogs undergoing assessment for chronic enteropathic disease were included for the prospective arm of the study. Within the 10 dogs included in the prospective study, there were 6 spayed females and 4 neutered males. The mean age was 9.3 years (range: 4–14 years). Breeds included 1 of each of the following: American Staffordshire Terrier, Border Collie, German Shepherd Dog, Golden Retriever, Lhasa Apso, Schnauzer, Shetland Sheepdog, and 3 mixed-breed dogs. Severity of disease in this group, as assessed by the canine IBD activity index (CIBDAI) score, was 4.4 (considered mild disease), with a range of 2–7 (clinically insignificant to moderate disease). Histological diagnoses in this group included: predominantly lymphocytic/plasmacytic disease (4/10), predominantly eosinophilic disease (3/10), mixed lymphocytic/plasmacytic and eosinophilic disease (2/10), and plasmacytic disease (1/10). There was no apparent relationship between histological diagnosis and severity as measured by CIBDAI.
Immunohistochemistry assays
Both the chronic enteropathy group and healthy controls showed individual cells with strong IHC staining for 5-HT (Figs. 1, 2). The number of cells showing positive staining for 5-HT was significantly increased (
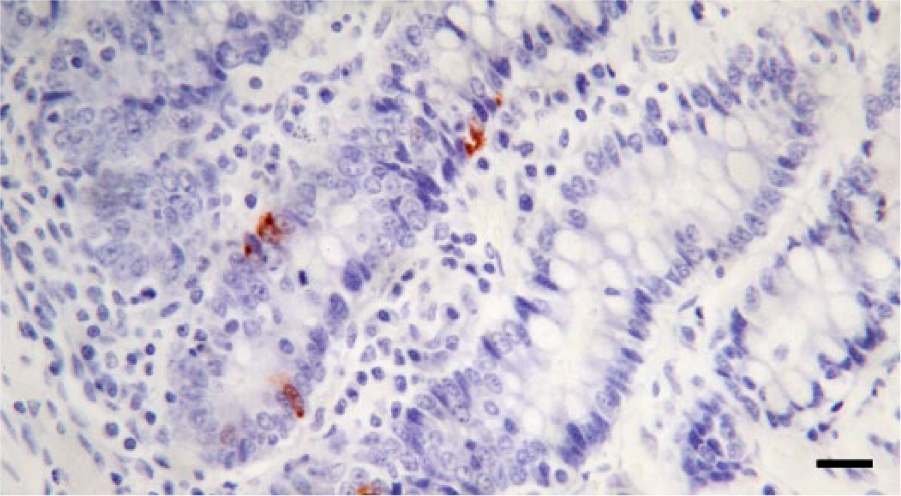
Representative results for detection of serotonin (5-HT) in the small intestinal mucosa of a control dog by immunochemistry. There are scattered intraglandular cells positive for 5-HT. Cells positive for 5-HT are stained with Nova Red chromogen, with Mayer hematoxylin counterstain. Bar = 100 µm.
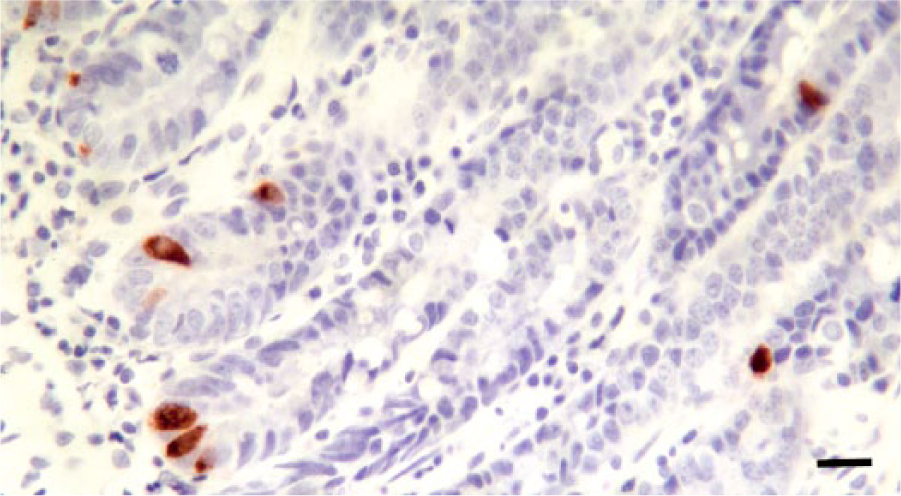
Representative results for detection of serotonin (5-HT) in the small intestinal mucosa of a canine patient with a chronic inflammatory enteropathy. There are an increased number of intraglandular cells positive for 5-HT; note also that cytoplasmic staining intensity of these cells is increased in comparison to those shown in Figure 1. Cells positive for 5-HT are stained with Nova Red chromogen, with Mayer hematoxylin counterstain. Bar = 100 µm.
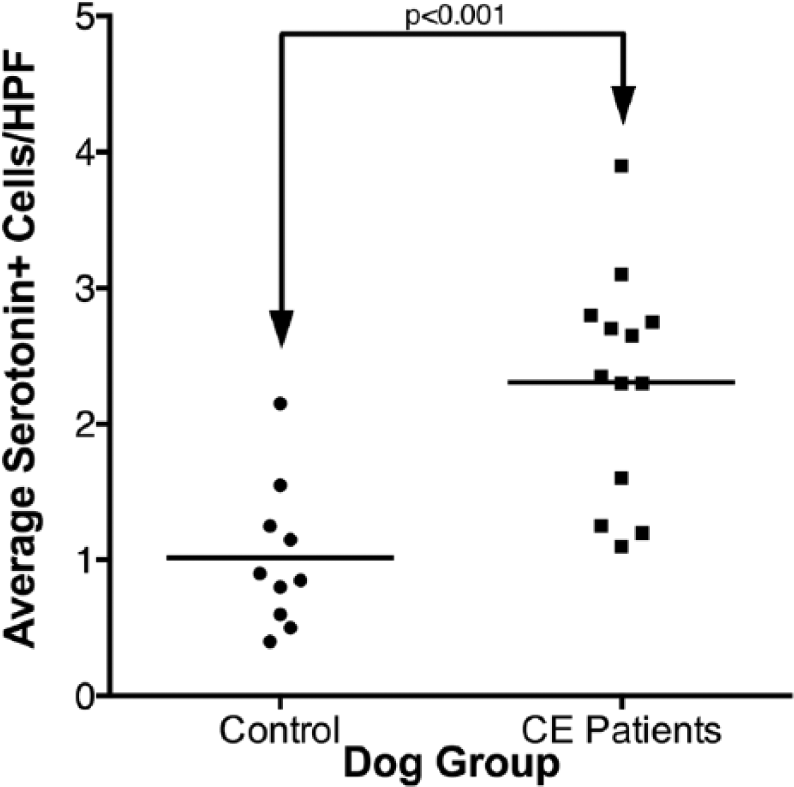
Scattered column plot (bar indicates mean) of serotonin immunopositive cells in the small intestinal mucosa of dogs with chronic enteropathy (CE) and apparently healthy controls. The groups are significantly different (
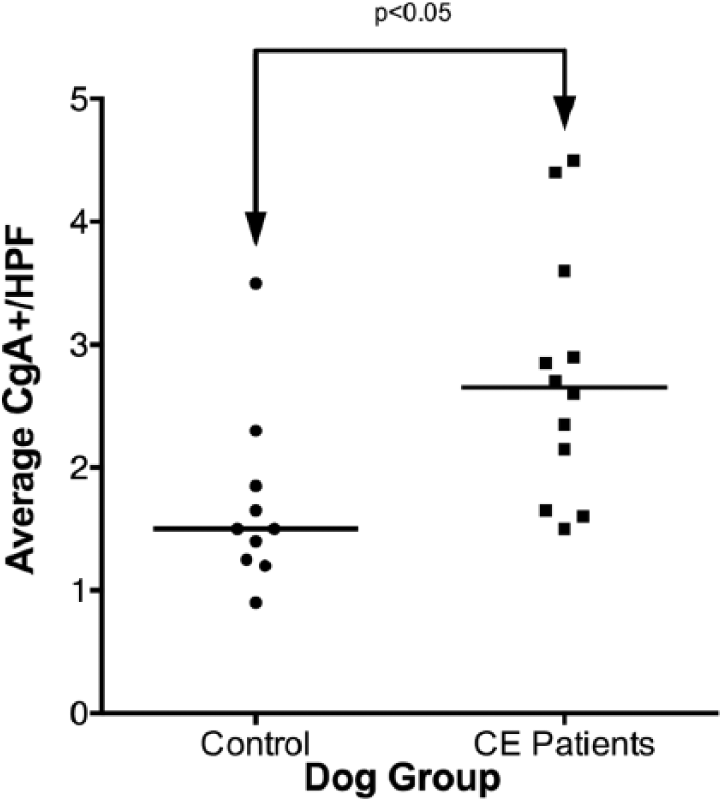
Scattered column plot (bar indicates median) of chromogranin-A (CgA) immunopositive cells in the small intestinal mucosa of dogs with chronic enteropathy (CE) and apparently healthy controls. The groups are significantly different (
Quantitative RT-PCR results
No significant difference in relative expression of SERT, 5-HT2B, or TPH-1 was observed between dogs with chronic enteropathy and the apparently healthy control dogs (Figs. 5–7). The relative expression of both 5-HT2B and TPH-1 were noted to be somewhat more variable in the dogs with chronic enteropathy than in the control dogs (coefficients of variation for normalized relative quantification values were 30.79% vs. 87.55% and 93.24% vs. 136.75% for control vs. chronic enteropathy dogs, 5-HT2B, and TPH-1, respectively).
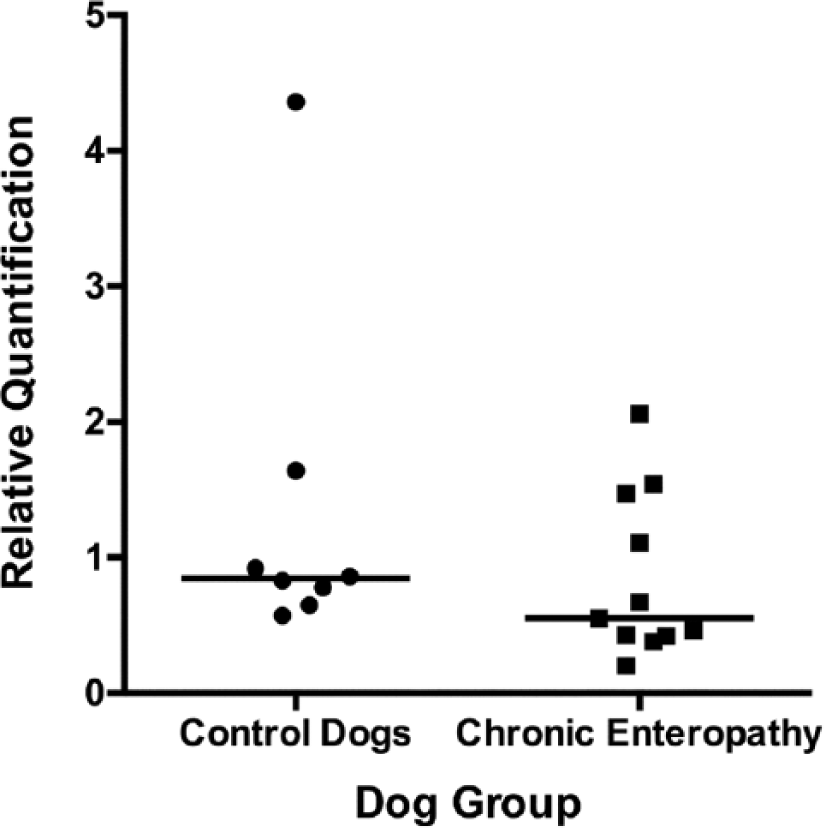
Scattered column plot (bar indicates median) of relative expression of serotonin reuptake transporter protein in the small intestinal mucosa of dogs with chronic enteropathy and apparently healthy controls.
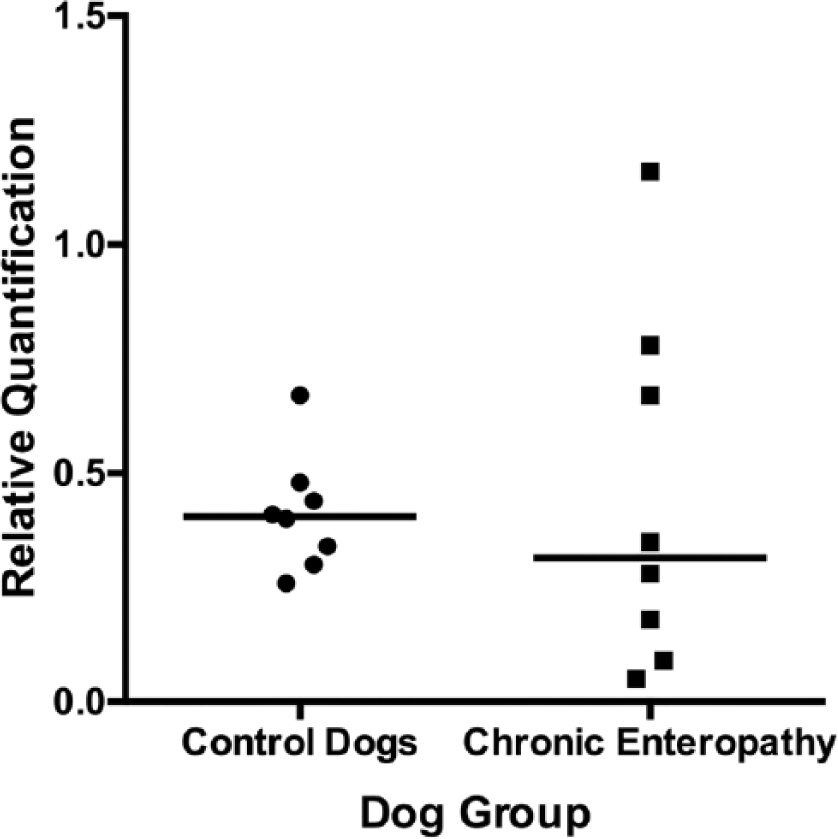
Scattered column plot (bar indicates median) of relative expression of the serotonin receptor2B in the small intestinal mucosa of dogs with chronic enteropathy and apparently healthy controls.
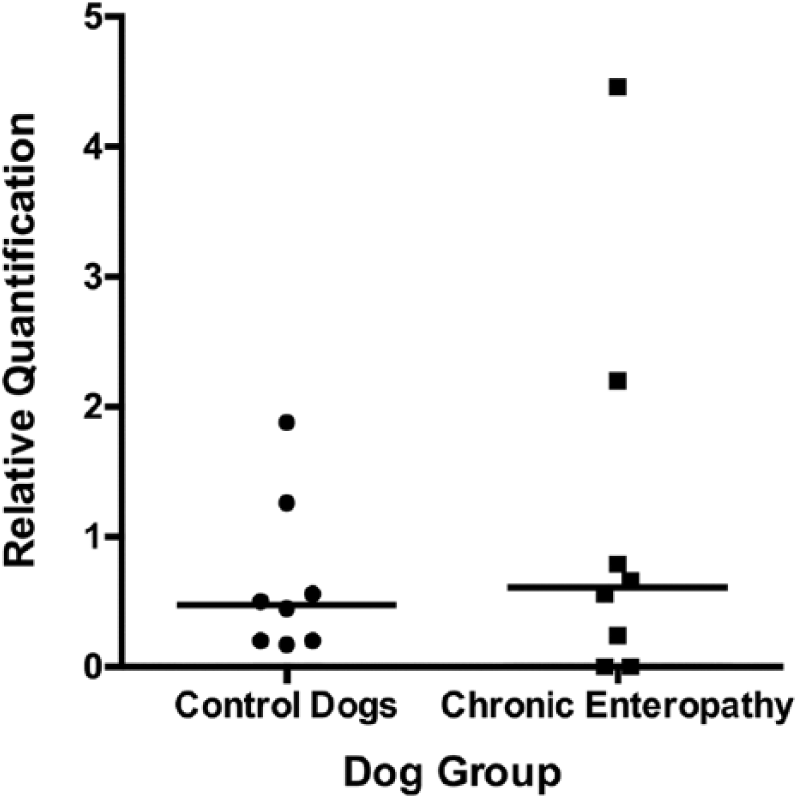
Scattered column plot (bar indicates median) of relative expression of tryptophan hydroxylase-1, the rate-limiting enzyme for serotonin synthesis, in the small intestinal mucosa of dogs with chronic enteropathy and apparently healthy controls.
Discussion
The data reported herein indicates that the duodenal mucosa of dogs with chronic enteropathy contains increased numbers of ECCs expressing 5-HT and CgA, when compared with an apparently healthy control group. No significant differences were detected in the expression of the rate-limiting enzyme for 5-HT synthesis (TPH-1 expression), SERT, or 5-HT2B in the groups of dogs studied.
Serotonin is thought to activate the submucosal sensory branch of the enteric nervous system and increase GI motility via interneurons and motor neurons.15,27 CgA is a protein that is costored and coreleased with monoamines and peptide hormones of various endocrine cells, including those of the GI tract. CgA thus serves as a marker for intestinal endocrine cells. 11 Serotonin-containing ECCs comprise the single largest endocrine cell population in the small intestine. 37
Inflammation of the intestinal mucosa has been shown to affect 5-HT signaling in animal models and in human beings.14,17,29,42 Changes in ECC numbers and in 5-HT content have been reported in association with Crohn’s disease, ulcerative colitis, and lymphocytic colitis in humans.3,4,12,36 Approximately 50% of human patients with IBD in long-standing remission have IBS-like symptoms (diarrhea, constipation, discomfort), which may be related to these inflammation-induced alterations in ECC density and 5-HT signaling. 12 It has also been shown that administration of selective 5-HT reuptake inhibitors is associated with microscopic colitis. 31 Diarrhea-predominant IBS is associated with elevated 5-HT in colonic mucosal biopsies, whereas constipation-predominant IBS is associated with decreased 5-HT in the colonic mucosa.8,38
We hypothesized that the expression of TPH-1 in the chronic enteropathy group would be altered when compared with an apparently healthy control group. However, in this study, there was no significant difference in the mean relative target sequence expression of TPH-1 between the groups. This finding is in contrast to results from a previously published human study, in which levels of messenger (m)RNA–encoding TPH-1 was observed to be decreased in both ulcerative colitis and IBS specimens. 13 IBS is a condition in humans usually diagnosed based on symptoms, including diarrhea, constipation, bloating, and discomfort. Diarrhea-predominant IBS has been associated with elevated 5-HT in colonic mucosal biopsies, whereas constipation-predominant IBS has been associated with decreased levels of 5-HT in the colon mucosa.9,30 Clinical trials using a small molecule inhibitor of THP-1, the rate limiting enzyme for 5-HT synthesis, have shown promise in alleviating the symptoms associated with IBS, particularly in diarrhea-predominant IBS. 3 Treatment with the THP-1 inhibitor decreased blood 5-HT concentrations, relieved discomfort, and improved stool consistency.5,34 Modulation of tryptophan metabolism, especially 5-HT synthesis, may be a novel target for developing therapies for GI disorders. 30 The relatively complex modes of 5-HT synthesis, release, varying receptor types, and specific reuptake pump activity provide a number of potential therapeutic targets. THP-1 expression is a major determinant of 5-HT activity in non-neuronal tissue. Serotonin is produced from tryptophan in a 2-step process, where hydroxylation mediated by THP-1 is the rate-limiting step. 26 To facilitate the actions of 5-HT, and prevent receptor desensitization, intestinal 5-HT transporter (SERT) causes rapid reuptake of 5-HT. Some prior publications have suggested that SERT is down-regulated in some inflammatory or diarrheal motility disorders.9,18
We hypothesized that SERT expression would be altered in the group of dogs with chronic enteropathy. It has previously been reported that mRNA-encoding SERT and SERT protein are diminished in an animal model of colitis. 36 Although there was a trend toward lower expression of SERT in our group of dogs with chronic enteropathy, no statistically significant difference was detected. As with our findings regarding TPH-1, both the low sample size and the relatively low severity of disease in our chronic enteropathy group may have contributed to the failure of this study to detect significance, and further studies in a larger group, including a group with higher disease severity, is warranted.
Inherent limitations of the study reported herein must be acknowledged. A remarkable lack of availability of complete records and satisfactorily banked tissue samples dramatically reduced the sample size of the IHC study. As this study used accessions to a veterinary pathology facility as well as patients at our teaching hospital, records from a large number of samples submitted from outside sources were not available for review.
The major reason for exclusion of samples from our own hospital was availability of only very small tissue samples obtained endoscopically. During the study period, this issue was identified, and we transitioned to larger-jawed biopsy forceps x in an attempt to obtain larger biopsy samples and reduce crush artifact. 19 If these forceps had been used throughout the entire study period, the number of samples that could potentially have been included would have been greater.
Our study demonstrated increased expression of 5-HT and CgA in the small intestine of dogs with chronic enteropathy versus that of a control group, a finding that agrees with previously published studies in humans. This study failed to detect changes in TPH-1, SERT, and 5-HT2B expression, as have previously been reported in rodent models and human beings with GI disease; however, sample numbers were low, and many cases were of only mild severity. Further investigation with a larger more severely affected population is warranted.
Footnotes
Acknowledgements
We thank Kay Fischer, Veterinary Diagnostic Laboratory at Oregon State University, for IHC and HE preparation of sampled tissues.
Authors’ note
A portion of the data presented here was presented previously in abstract form at the American College of Veterinary Internal Medicine Forum, 2013, Seattle, Washington.
Authors’ contributions
C Bailey and C Ruaux contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. BV Stang contributed to design of the study; contributed to acquisition and analysis of data; and drafted the manuscript. BA Valentine contributed to conception of the study and contributed to acquisition and interpretation of data. C Ruaux and BA Valentine critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Kindly donated by Dr. R. Heilmann, Gastrointestinal Laboratory, Texas A&M University, College Station, TX.
b.
RNAlater, Thermo Fischer Scientific, Waltham, MA.
c.
TBS liquid concentrate, Biocare Medical, Concord, CA.
d.
Sigma-Aldrich, St. Louis, MO.
e.
Proteinase K, Dako North America Inc., Carpinteria, CA.
f.
Mouse universal negative control (N1698), Dako North America Inc., Carpinteria, CA.
g.
Serum-free protein block solution, Dako North America Inc., Carpinteria, CA.
h.
Monoclonal anti-serotonin antibody (ab16007, manufacturer data indicates cross-reactivity with 5-HT in the canine), Abcam PLC, Cambridge, MA.
i.
Antibody diluent (background-reducing), Dako North America Inc., Carpinteria, CA.
j.
MaxPoly-One anti-mouse IgG horseradish peroxidase conjugate, MaxVision Biosciences Inc., Bothell, WA.
k.
Nova Red, Vector Laboratories Inc., Burlingame, CA.
l.
Hematoxylin, Dako North America Inc., Carpinteria, CA.
m.
Target retrieval solution, Dako North America Inc., Carpinteria, CA.
n.
Polyclonal rabbit anti-human chromogranin A, Dako North America Inc., Carpinteria, CA.
o.
MaxPoly-One anti-rabbit IgG conjugate, MaxVision Biosciences Inc., Bothell, WA.
p.
RNeasy total RNA mini kit, Qiagen Inc., Valencia, CA.
q.
QuantiTect reverse transcription kit, Qiagen Inc., Valencia, CA.
r.
QuantiTect primer assay (Cf_SLC6A4_3_SG), Qiagen Inc., Valencia, CA.
s.
RT2 Profiler PCR array dog housekeeping genes, Qiagen Inc., Valencia, CA.
t.
Power SYBR Green PCR master mix, Applied Biosystems, Foster City, CA.
u.
Step One Plus real-time PCR system, Applied Biosystems, Foster City, CA.
w.
GraphPad Prism 5.0, GraphPad Software Inc., San Diego, CA.
x.
Radial Jaw 4 jumbo biopsy forceps, Boston Scientific Corp., Marlborough, MA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
