Abstract
In recent years, serologic markers for diagnosis and classification of inflammatory bowel disease (IBD) have been used in human medicine. Perinuclear, antineutrophil, cytoplasmic antibodies (p-ANCA) are the most important of these markers. Because of their similar pattern of fluorescence, antinuclear antibodies (ANA) could cause misleading interpretations. The aim of the present study was to evaluate the use of an indirect fluorescent antibody test to detect p-ANCA in dogs with IBD, to compare the presence of p-ANCA in dogs with IBD with the presence of the same antibodies in other dogs, and to analyze the presence of ANAs in the p-ANCA-positive samples. Using a 1:10 dilution as a cutoff point, a sensitivity of 0.34 and a specificity of 0.86 was obtained when dogs with IBD were compared with the other groups as a whole, and specificity increased to 0.94 when dogs with IBD were compared with animals with other chronic gastrointestinal disorders. The lowest specificity value, 0.76, was obtained when the group of dogs with IBD was compared with that of dogs with different inflammatory and infectious disorders. Globally, 78 dogs were positive for p-ANCA when the cutoff was 1:10. Only 1 dog from these 78 animals was also seropositive to ANA. The results suggest that 1) detection of p-ANCA might be included in the IBD diagnostic protocol as another test to differentiate between this disease and other digestive diseases with similar clinical signs, and 2) most p-ANCA- positive dogs are not ANA positive.
Keywords
Introduction
Canine inflammatory bowel disease (IBD) includes a set of diseases characterized by the presence of persistent or recurrent gastrointestinal clinical signs and histologic evidence of intestinal inflammation. The etiology of this group of diseases is unknown, although one of the proposed causes is an alteration in immunologic toleration of luminal antigens (bacteria and dietary components), possibly because of a rupture in the integrity of the mucosal barrier, immune system dysregulation, or alterations of the intestinal flora. 11,12
Clinical signs of this group of diseases include vomiting, diarrhea, and chronic weight loss. Diagnosis can only be made by excluding other possible causes of enteritis. Nowadays, the diagnostic protocol for canine IBD includes a complete blood cell count (CBC), serum biochemical analyses, parasitologic and bacteriologic fecal analysis, serum trypsin-like immunoreactivity (TLI), radiology, and ultrasound. Finally, biopsy samples of intestinal mucosa, usually taken by means of endoscopy, are necessary to reach a definitive diagnosis. 11,12
The diagnostic procedure for IBD in human medicine is very similar to that used in veterinary medicine, but in recent years, serologic markers are also being used to diagnose and classify this group of diseases in humans. Perinuclear, antineutrophil, cytoplasmic antibodies (p-ANCA) are among the most important of these markers. The ANCAs are circulating antibodies mainly directed against neutro-phil granule constituents. Two separate entities within IBD, ulcerative colitis and Crohn disease, have been traditionally differentiated in humans. These antibodies have been detected in both diseases but are more frequently detected in patients with ulcerative colitis (70-80%) than in those with Crohn disease (10-20%). 1,20,22 Because of that finding, ANCAs are also helpful in the differential diagnosis between ulcerative colitis and Crohn disease, 1,3 although ANCA detection is even more useful when combined with detection of other serologic markers, especially with anti—Saccharomyces cerevisiae antibodies (AS-CAs). 19,21,23 The ANCAs have also been used in human medicine, especially as markers to differentiate IBD from other gastrointestinal diseases. 8
The antigens against which these antibodies are directed are well known in the case of primary vasculitis, but they are not well characterized in IBD. Varieties of antigens have been proposed in this disease, although no main specificity has been described. Furthermore, serum from 1 human with IBD may contain ANCAs with different specificities. 19,20
The technique most often employed to detect ANCAs is the indirect fluorescent antibody test (IFAT) on ethanol-fixed neutrophils. Different staining patterns are distinguished depending on the electric charge and cytoplasmic distribution of the antigens recognized by the ANCA. The 2 main patterns are cytoplasmic and perinuclear staining. The most frequent staining pattern in human IBD is perinuclear (p-ANCA), 1 but the target antigens of these antibodies are not well characterized. An “atypical” fluorescence pattern has also been described in this disease. 19
The typical perinuclear fluorescence pattern described in IBD could be due to the detection of either p-ANCA or antinuclear antibodies (ANA). The method of formalin fixation of neutrophils is controversial because it is considered to cause inconsistency, nonspecific effects, and false-positivity owing to the enhanced fluorescence. 6 Formalin fixation does not effectively differentiate true p-ANCA from ANA, which has lead to some authors' interest in determining ANA in p-ANCA-positive samples. 6
There are few publications in veterinary medicine that report the detection of p-ANCA in dogs with IBD. In a previous study, IFAT was used as an ANCA detection method, and it was determined that the principal fluorescence pattern in canine IBD was, as in human medicine, the perinuclear pattern. 2 Another study evaluated the p-ANCA of 26 dogs with IBD before and after immune-suppressive treatment and determined that p-ANCA status was not associated with the activity of the disease in dogs. 17 To date, evaluation of ANA in dogs positive to p-ANCA has not been performed. The objectives of the present study were to evaluate the use of IFAT to detect p-ANCA in dogs with IBD, to compare the presence of p-ANCA in dogs with IBD to other canine populations, and to analyze the presence of ANA in the p-ANCA-positive samples.
Materials and methods
Animals
Three hundred eighty-three dogs, classified into 6 groups, were used in the current study. The groups included dogs with IBD (n = 107); dogs with chronic gastrointestinal disease not associated with IBD (n = 35); dogs with acute gastrointestinal disease (n = 27); dogs with inflammatory and infectious disorders (n = 34); healthy dogs of different breeds and ages (n = 54); and healthy working dogs, specifically police dogs of the Spanish National Police Corps (n = 126).
Dogs with IBD were diagnosed using a diagnostic exclusion protocol that included hematology and blood chemistry tests, parasitologic fecal analysis for 3 consecutive days, TLI to test pancreatic functioning, and digestive endoscopy with intestinal biopsies. Inflammatory bowel disease was diagnosed through histopathologic study of these biopsies. The IBD group consisted of 44 females and 63 males between 12 and 132 months old. The average age was 55.48 months.
Among the 35 dogs with chronic gastrointestinal disease not associated with IBD, 15 were diagnosed with gastrointestinal neoplasms, 8 had pancreatic exocrine insufficiency, 7 displayed parasitism, and 2 were diagnosed with thyroid hormone alterations, 2 with anal sac disorders, and 1 with chronic gastroenteritis that responded to dietary management. The 16 females and 19 males in this group ranged between 8 and 168 months old; the average age was 82.05 months.
Nineteen of the 27 dogs with acute gastrointestinal disorders had a foreign body lodged in the gastrointestinal tract whereas 8 suffered acute gastroenteritis of unknown etiology. In this group of dogs with acute gastrointestinal illness, there were 13 females and 14 males between 5 and 144 months old. The average age was 41.63 months.
The healthy dogs in the present study did not display any signs of disease, and their blood tests did not present any altered parameters. Of the 54 healthy dogs, 31 were females and 23 males. These dogs ranged in age between 5 and 180 months, and the average age was 55.81 months.
The group of dogs with inflammatory and infectious diseases included 12 animals with pyometra, 3 with endometritis, 6 with periodontal disease, 2 with mastitis, 2 with osteomyelitis, 2 with chronic dermatitis, 1 with chronic rhinitis, and 6 that had abscesses in various locations. This group was composed of 21 females and 13 males between 12 and 180 months old. The average age was 95.3 months.
Blood samples
Approximately 3 ml of blood were obtained from each animal in the study. Blood was centrifuged at 430 × g for 10 min to obtain serum. All sera were frozen and stored at −20°C until used.
Detection of p-ANCA by IFAT
Antigen. The antigen was obtained as previously described. 2 Briefly, the first step involved isolating the polymorphonuclear neutrophil leukocytes using a solution a of sodium diatrizoate (13.8% w/v) and dextran 500 (8% w/v). The neutrophils were isolated from 5 ml of blood from a healthy dog. This blood was centrifuged along with the same amount of solution a at 450 × g for 30 min at 22°C. The polymorphonuclear neutrophil leukocyte phase was collected and washed with phosphate buffered saline (PBS) by centrifuging at 200 × g for 10 min at 22°C 3 times. The cells obtained were resuspended in PBS, and the neutrophil content of the solution was verified using May-Grünwald and Giemsa stain. The neutrophil solution was divided among slides that were left to air dry and were later fixed in 96% ethanol at 4°C for 5 min. Slides were frozen and stored at −20°C until used.
Specificity and positive and negative predictive values, comparing the group of dogs with inflammatory bowel disease (IBD) with the other 5 groups studied, individually and together, for a 1:5 dilution of an indirect fluorescent antibody test*
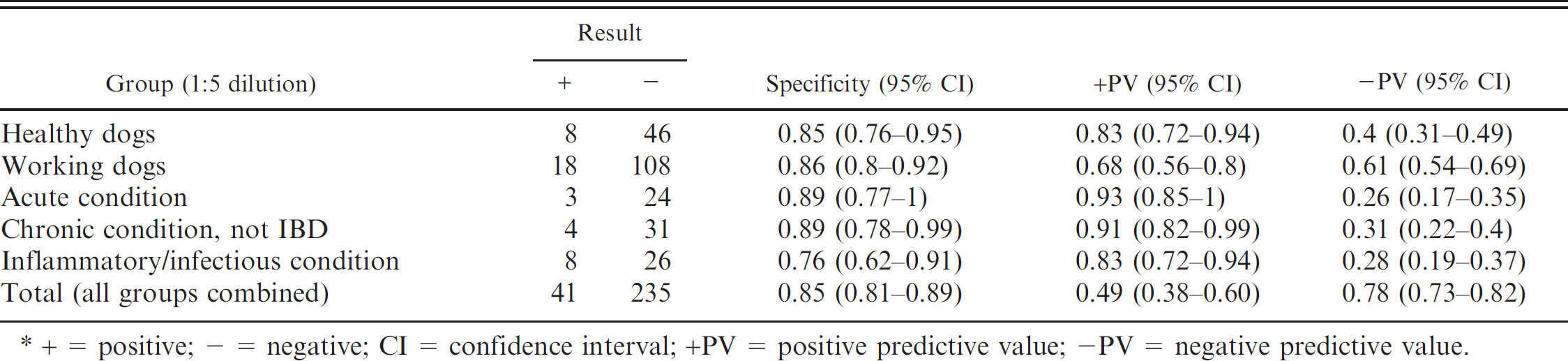
+ = positive; - = negative; CI = confidence interval; +PV = positive predictive value; -PV = negative predictive value.
Indirect fluorescent antibody test. Keeping in mind that the cutoff point used in previous studies in veterinary medicine was 1:10, the working dilutions used by the authors were 1:5, 1:10, and 1:20. The IFAT consists of 2, 1-hr-long incubations in a moist chamber at room temperature, as previously described. 2 The first incubation was done with canine study sera and the second one with an anti-dog immunoglobulin G labeled with fluorescein isothiocyanate b diluted 1:100 in PBS (pH 7.2). After each incubation, the slides were washed 3 times for 5 min with PBS. Finally, the coverslips were mounted with buffered glycerin, and the slides were examined using a fluorescence microscope. The only sera considered positive were those that demonstrated a clear perinuclear fluorescence pattern on the neutrophils (p-ANCA positive). Sera that displayed any other fluorescence pattern were considered p-ANCA negative.
Detection of antinuclear antibodies by IFAT detection in all p-ANCA positive samples
Antigen. The detection of ANA was performed using HEp-2 cells. These cells were obtained from the cell line HEp-2 American Type Cell Culture, c maintained in minimum essential medium, supplemented with 15% bovine fetal serum. The cells were removed after scraping and gently washed × times using PBS (200 × g for 10 min at 22°C). The cells obtained were resuspended in PBS. This solution was placed on immunofluorescence slides and allowed to air dry overnight. Slides were frozen and stored at -20°C until further use.
Indirect fluorescent antibody test. Taking into account that the cutoff for p-ANCA was 1:10, all the samples were analyzed at 1:10 dilution for the detection of ANA. The IFAT for ANA detection was performed following the method previously described for p-ANCA detection.
Statistical analysis
Once all the data were collected, the sensitivity and specificity of the technique (detection of p-ANCA), in addition to the positive and negative predictive values, were determined for the group of dogs with IBD compared with the other groups as a whole and with each group separately. Sensitivity, specificity, positive predictive value, and negative predictive value were calculated applying a confidence interval (CI) of 95%.
Results
Thirty-nine of the 107 dogs with IBD were p-ANCA positive at a 1:5 dilution, indicating that the sensitivity of the technique was 0.36 (0.27-0.46). Thirty-six p-ANCA-positive dogs were identified at a 1:10 dilution. Sensitivity was 0.34 (0.25-0.43) when this dilution was the cutoff point. The number of p-ANCA-positive dogs with IBD at a 1:20 dilution was 19. Sensitivity at that dilution was 0.18 (0.11-0.25).
Comparing dogs with IBD with all the other groups together, the specificity of the IFAT to detect p-ANCA in canine IBD was 0.85 (0.81-0.89), when the cutoff point was a dilution of 1:5; 0.86 (0.82-0.90), when a 1:10 cutoff was used; and 0.91 (0.88-0.94), when the cutoff point was 1:20.
Tables 1–3 present specificity values, together with positive and negative predictive values, comparing the group of dogs with IBD with the other 5 groups studied, considered individually and together, for dilutions 1:5 (Table. 1), 1:10 (Table. 2), and 1:20 (Table. 3). Globally, 78 dogs were p-ANCA positive when the cutoff was 1:10. Only 1 dog from these 78 animals was also seropositive to ANA and belonged to the healthy working dog group.
Specificity and positive and negative predictive values, comparing the group of dogs with inflammatory bowel disease (IBD) with the other 5 groups studied, individually and together, for a 1:10 dilution of an indirect fluorescent antibody test.*
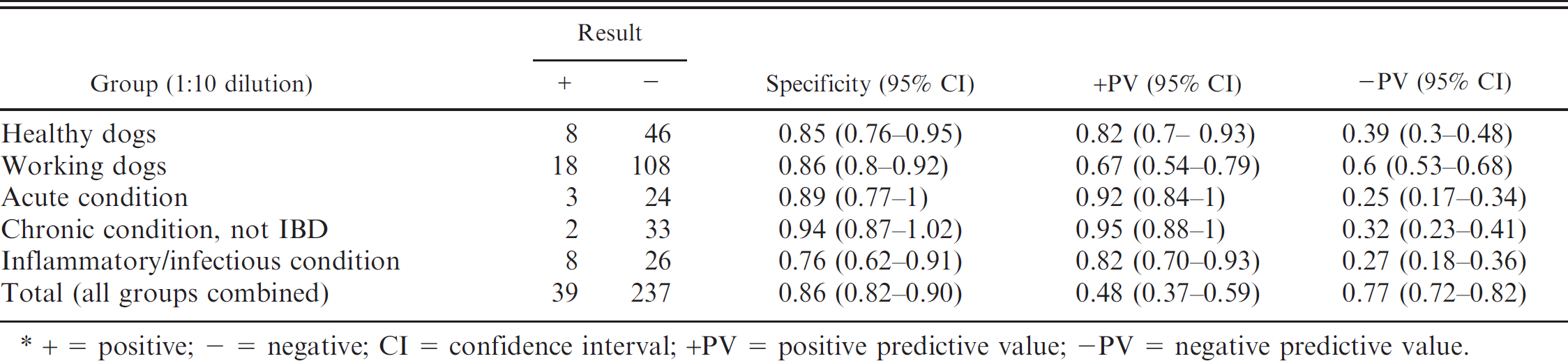
+ = positive; - = negative; CI = confidence interval; +PV = positive predictive value; -PV = negative predictive value.
Discussion
A correct diagnosis of canine IBD is a long and complex process involving techniques that require anesthesia of the animal and collecting intestinal biopsy samples by means of endoscopy or surgery. For this reason, the development of noninvasive diagnostic methods, such as the use of serologic markers, would represent an important step in the diagnosis of this group of diseases.
In the first veterinary study carried out to evaluate the presence of p-ANCA in dogs with IBD using a 1:10 dilution as the cutoff point, specificity ranged between 0.82 and 0.95 in healthy dogs, healthy working dogs, dogs with acute gastrointestinal disorders, and dogs with chronic gastrointestinal conditions not associated with IBD. 2 Sensitivity of the technique in that study was 0.51. If those data are compared with those obtained in the present study using the same dilution, sensitivity in the current study is lower (0.34, 0.25-0.43), although the specificity is very similar, ranging from 0.85 to 0.94 in these same groups. This slight difference could be explained by the analysis of different population samples or by the existence of interlaboratory differences.
In the present study, 3 dilutions of sera to detect p-ANCA were used. Analysis of the results shows that the sensitivity of the technique with the 1:20 dilution (0.18) was much lower than that obtained with the other 2 dilutions. Comparing the results using the 1:5 and 1:10 dilutions, the sensitivity values were very similar to each other (0.36 and 0.34, respectively), although when the cutoff point is established at 1:10, the specificity of the technique improves, especially after comparing it with that of the group of dogs with chronic gastrointestinal conditions not associated with IBD.
The highest specificity and positive predictive values were observed at both the high and low dilutions when dogs with IBD were compared with the 2 groups of animals with gastrointestinal conditions not associated with IBD. These data may indicate the usefulness of p-ANCA in differentiating IBD from other gastrointestinal diseases not associated with this condition, as has been previously described in human and veterinary medicine. 2,8 At every dilution, the lowest specificity value was observed when the group of dogs with IBD was compared with those with inflammatory/infectious conditions. In human medicine, the presence of these antibodies is reported in patients with diverse acute and chronic infectious disorders, which could mean that the prevalence of ANCA in different diseases may be associated with an intense activation of neutrophil leukocytes. 13 It would be interesting to investigate the presence of these antibodies in other well-characterized canine inflammatory diseases.
Specificity and positive and negative predictive values, comparing the group of dogs with inflammatory bowel disease (IBD) with the other 5 groups studied, individually and together, for a 1:20 dilution of an indirect fluorescent antibody test.*
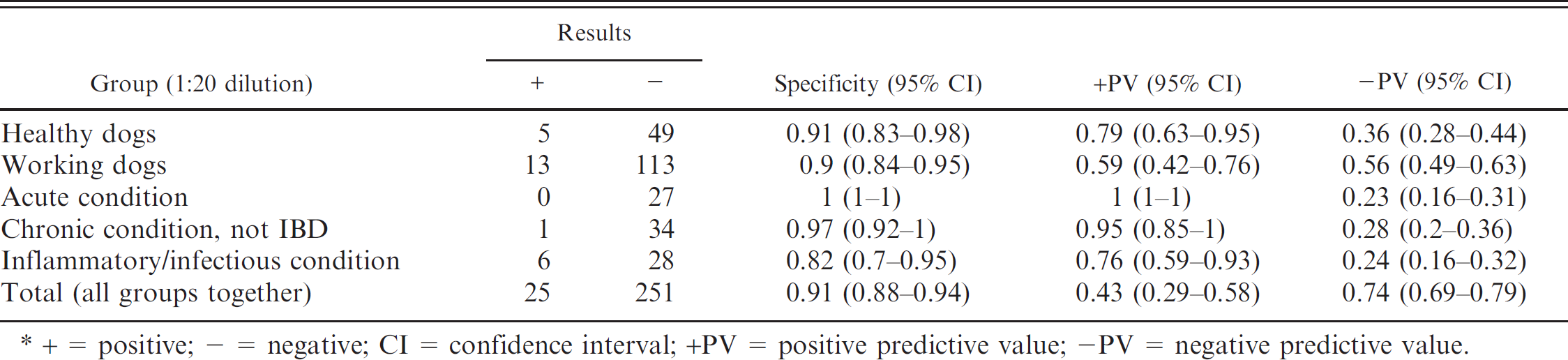
+ = positive; − = negative; CI = confidence interval; +PV = positive predictive value; -PV = negative predictive value.
The ANCAs are circulating autoantibodies directed mainly against neutrophil granule components. Initially, they were described in some immunologic diseases, such as primary systemic vasculitis and Wegener granulomatosis. 9 Later, these antibodies were also found in a large number of autoimmune diseases, including rheumatoid arthritis, autoimmune hepatitis, and systemic lupus erythematosus, as well as in chronic inflammatory processes. Various immune abnormalities have been reported in both human and canine IBD, although IBD is not recognized as an autoimmune disease. 11 Autoimmune intestinal disease may be a result of different mechanisms. A T-suppressor cell failure may result in the escape of an autoantibody-producing auto-reactive lymphocyte clone. Another possibility is an immune response to a foreign antigen in the intestine that shares antigenic determinants with components of their own cells, possibly resulting in an inappropriate attack against one's own tissues. Molecular similarities are likely to be found in the intestine, which constitutes a reservoir of bacterial antigens. A study in humans has demonstrated that ANCAs, and especially p-ANCAs that appear in ulcerative colitis, display a cross-reaction with bacterial antigens, particularly Escherichia coli and Bacteroides caccae. 7 To establish that the disease has an autoimmune origin, it would first be necessary to prove that IBD is associated with specific autoantibodies or autoreactive lymphocytes and that these antibodies or lymphocytes are able to cause tissue damage. It is necessary to clarify whether these autoreactive immune components have a role in the lesions or are simply an epiphenomenon. 11
The perinuclear immunofluorescence pattern of ANCAs detected in IBD could closely resemble the perinuclear staining pattern of ANAs. 15 Several protocols for neutrophil fixation have been described to differentiate these 2 kinds of antibodies (such as formalin, formaldehyde vapor, or formalin ace-tone). 15,16,25 Nevertheless, some authors believe that these assays could add diagnostic confusion and suggest that p-ANCA assay by immunofluorescence using ethanol-fixed neutrophils should be cross-checked with the detection of ANA using Hep2 cells as substrate. 6 In the present study, the detection of ANA using HEp-2 cells was performed to differentiate between p-ANCA and ANA. All the IBD p-ANCA-positive dogs were ANA negative, and only 1 of the p-ANCA-positive dogs belonging to the remaining groups was ANA positive. These results suggest that the p-ANCAs detected in dogs with IBD or in dogs from the other groups included in the present study are different from ANAs.
The pathogenic role of ANCA is well established in systemic vasculitis and Wegener granulomatosis. These antibodies are known to play an important role in the pathogenesis of these diseases, and their titer often parallels the activity of the illness. Furthermore, measurement of the ANCA titer in these patients may be useful in predicting relapses of the disease. 14 However, the pathogenic role of p-ANCA in human IBD has not yet been demonstrated, 10 and most studies do not support the existence of any relationship between the p-ANCA titer and IBD activity. 4,5,18,24 In veterinary medicine, there is only 1 previous study, to the authors' knowledge, that evaluates the relationship between p-ANCA and the activity index of inflammatory bowel disease (in 26 dogs). 17 In that study, the conclusion, as in human studies, was that there is no significant correlation between the ANCA titer and the activity index of IBD.
Although the etiopathogenesis of canine IBD has not been studied in depth, and an autoimmune component is not proved to date, physiopathology of this group of diseases is believed to involve an immune-mediated component. The presence of p-ANCA in canine patients with IBD could be one further indication of the immune-mediated etiology of this group of diseases. More studies on large populations of IBD patients, healthy control patients, and dogs with other diseases are needed to understand the diagnostic value of serologic markers in IBD.
Use of the IFAT to detect p-ANCA in dogs with IBD seems to be a helpful diagnostic tool because of its high specificity when dogs with IBD are compared with those of other groups, as has been previously suggested. 2 It is particularly useful in enabling diagnosticians to differentiate between dogs with IBD and those with other gastrointestinal conditions because of the great specificity of the technique when dogs with IBD are compared with the 2 groups displaying other gastrointestinal disorders. At the present time, this method may be a useful diagnostic support tool, but it cannot substitute for any procedure included in the established diagnostic exclusion protocol that must be carried out in patients with chronic gastrointestinal clinical signs.
Acknowledgements
The authors wish to thank the veterinarians of the National Police Corps, Gloria Gimenez and Silvestre Galera, for their contribution to this work.
Footnotes
a.
Polymorphprep™, Axis-Shield PoC AS, Oslo, Norway.
b.
Jackson Immunoresearch Laboratories Inc., West Grove, PA.
c.
CCL-23, American Type Culture Collection, Manassas, VA.
