Abstract
The present case report describes the isolation of the fungus
A 9-year-old spayed female West Highland White Terrier dog was referred to the Iowa State University Lloyd Veterinary Medical Center (Ames, Iowa) for diagnostic evaluation of refractory protein-losing enteropathy (PLE). The dog had been initially presented to the referring veterinarian 6 weeks earlier for decreased appetite and weight loss. Over the next several weeks, progressive gastrointestinal signs including intermittent vomiting and diarrhea were noted. The dog was treated for presumptive immune-mediated chronic enteropathy. Upon presentation, the dog was bright and alert, weighed approximately 6 kg, and had a body condition score of 5 out of 9. Mild hepatomegaly was noted on abdominal palpation. Digital rectal examination revealed soft stool with hematochezia.
Complete blood cell count, serum biochemistry, and urinalysis were performed. The hematologic and biochemical analyses were consistent with enteric plasma protein loss (i.e., PLE), possibly with gastrointestinal bleeding. Urinalysis was interpreted as normal. Direct and indirect fecal examinations for nematode or protozoal parasites were negative. Abdominal ultrasound revealed hyperechoic, perpendicular mucosal striations along the small intestinal mucosa, suggesting intestinal lymphangiectasia as the cause for the PLE. 11 The intestinal bowel loops appeared uniformly normal in thickness with all layers present. Thoracic radiographs were normal.
The dog was anesthetized, and esophagogastroenteroscopy was performed. Multiple endoscopic biopsies of the stomach and duodenum were obtained for histopathologic examination. Gastric, duodenal, and rectal brush cytology was performed using guarded cytology brushes. Gastric cytology revealed mild neutrophilic inflammation with a mixed bacterial population intermixed with occasional large (approximately 50–75 μm) thick-walled yeasts. Duodenal cytology revealed mixed inflammatory infiltrate. Rectal cytology revealed mixed bacteria and frequent large, round-to-oval, deeply basophilic structures, similar to
Histologic examination of biopsy specimens revealed moderate lymphoplasmacytic gastroenteritis and lymphangiectasia. Periodic acid–Schiff and Warthin–Starry staining did not reveal fungal organisms. The histologic changes were consistent with idiopathic inflammatory bowel disease causing PLE.
No
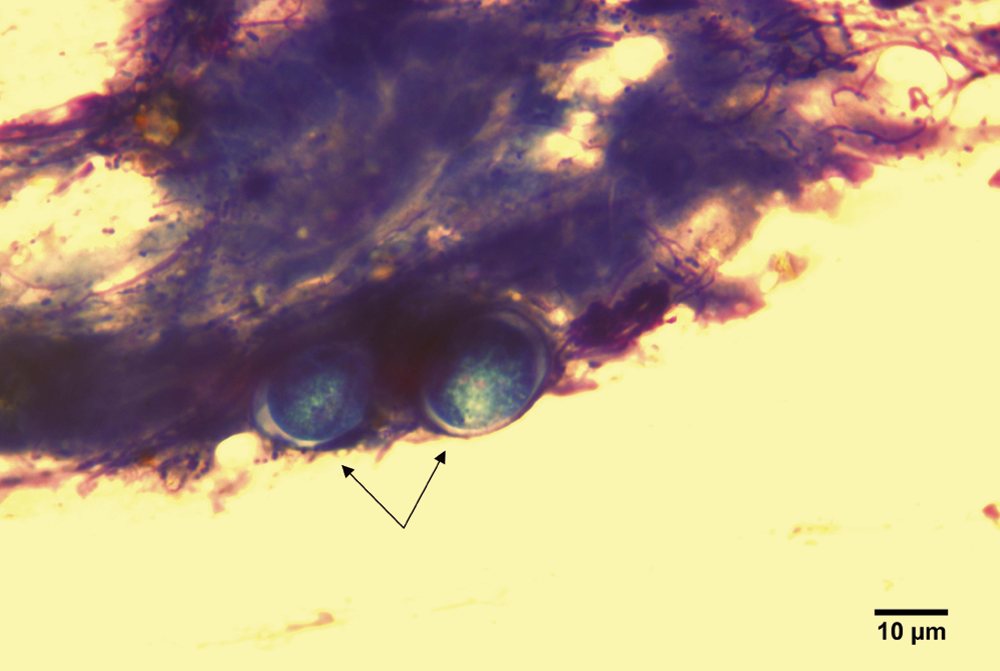
Rectal cytology revealing 2

Earlier case studies have indicated that
Gross changes included a thickened jejunal wall and thick, red–brown luminal contents in the ileum and colon, consistent with intestinal hemorrhage. Gastric and colonic samples were submitted for fungal culture. Histologic examination of the intestines revealed lymphoplasmacytic enteritis. The intestinal crypts were tortuous in areas of more intense lamina proprial inflammation, and moderate numbers of crypt abscesses and dilated villar lymphatic vessels were present. Infectious agents were not observed in periodic acid–Schiff-stained sections. There was no evidence of disseminated fungal disease. Fungal agents were not cultured from either sample.
At room temperature in air,
Differentiation of
A potential pathogenic role of
In human beings, gastrointestinal disease associated with zygomycosis is relatively uncommon. The stomach is the most common area of the gastrointestinal tract affected, followed by the colon. The small intestine and esophagus are rarely affected. 2 Risk factors associated with gastrointestinal zygomycosis include protein-calorie malnutrition, diarrhea, and gastrointestinal ulceration. 8 The dog in the present report exhibited all of these negative risk factors. Other factors contributing to zygomycosis infection include intercurrent disease caused by diabetes mellitus and/or diabetic ketoacidosis, chronic metabolic acidosis, skin or soft tissue damage, and the prolonged use of broad-spectrum antibiotic and antifungal therapies. 2 Three forms of gastric zygomycosis exist: colonization, infiltration, or vascular invasion. Colonization often occurs in preexisting ulcers and is infrequently fatal. The invasive form involves the formation of necrotic gastrointestinal ulcers. Perforation and peritonitis are potential consequences, and ultimately this form of disease is fatal.8,13 Clinical signs of gastric zygomycosis include abdominal pain, nausea, vomiting, diarrhea, hematemesis, melena, and hematochezia. 2
In view of the fact that the dog of the present report had received immunosuppressive doses of corticosteroids in an effort to manage idiopathic inflammatory bowel disease, it is reasonable to hypothesize that the enteric fungal infection occurred secondary to corticosteroid-induced immunosuppression. The diarrhea, intestinal ulceration, and malnutrition were additional risk factors for impaired barrier function that allowed fungal growth. Little is known about the true pathogenicity of
Footnotes
Acknowledgements
The authors acknowledge Dr. Heather A. Flaherty, Dr. Jelena Palić, and Ann E. Greazel for their assistance with figures. Acknowledgements also go to Joann Kinyon and Jessica Schnellbacher for their assistance in culturing the
a.
BD Diagnostic Systems, Sparks, MD.
b.
Remel, Lenexa, KS.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
