Abstract
Hypertrophic cardiomyopathy (HCM) was diagnosed during postmortem examination of 2 captive adult Bennett's wallabies (Macropus rufogriseus rufogriseus). The wallabies were members of a mob (herd) of 3 wallabies, and 2 died spontaneously without clinical signs of heart failure being detected. Gross lesions in both cases included marked concentric hypertrophy of the left ventricle, pulmonary edema, and multifocal hemorrhage and subcutaneous edema of the hind limbs. Histologic lesions of the heart were limited to mild cardiac myofiber disarray and marked cardiac myofiber hypertrophy. A specific etiology for the HCM was not determined in either animal. The cardiac changes are similar to the left ventricular hypertrophy previously described in kangaroos.
The myocardium can be affected by a variety of disease processes, including primary myocardial disorders, degenerative and inflammatory diseases, neoplasia, infarctions, toxicities, and nutritional deficiencies. Idiopathic or primary hypertrophic cardiomyopathy (HCM) refers to a primary myocardial disease characterized by a nondilated and hypertrophied left ventricle in the absence of a known precipitating cause of disease. Secondary HCM is a term used when the cause of the cardiomyopathy is known. Primary myocardial disease is commonly seen in humans and domestic animals, particularly in cats and, to a lesser extent, in dogs, 9 and has been reported 7,10 in kangaroos. Left ventricular hypertrophy (LVH) has been reported 7 in the rock kangaroo (Macropus robustus) and is associated with impaired diastolic function, ventricular arrhythmias, and sudden death. 7,10 The etiology remains unknown. Strenuous jumping was a proposed mechanism; however, similar findings of LVH were identified in the slow-moving wombat. 10
In cases of secondary HCM in Ragdoll 6 and Maine Coon 3 cats, disease-causing mutations have been identified in the cardiac myosin binding protein C gene. Numerous investigations have been undertaken to identify nongenetic causes of HCM, such as hyperthyroidism and nutritional deficiencies. Nutritional deficiencies are associated with dilated cardiomyopathy in some animals. 1,4,9 In the current report, the diagnosis of primary HCM, a condition not previously described in this or any other wallaby species, is described in 2 adult Bennett's wallabies (Macropus rufogriseus rufogriseus).
In a 1-month period during the summer of 2008, 2 of 3 Bennett's wallabies on an exotic animal farm in north-central Oklahoma died suddenly. The animals were fed a diet consisting of 2 different marsupial feeds: Mazuri Kangaroo/Wallaby diet a and Booster Hopper Choice feed. b In addition, the animals received Bermuda grass, apples, sweet potatoes, carrots, and wheat bread. No nutritional supplements were given to the wallabies. No clinical signs of heart disease and/or failure were observed by the owner prior to the death of the 2 animals. The 3 wallabies were housed within the same enclosure and observed daily. Both dead wallabies were female; 1 animal was 3 years old (case 1) and the second animal was 4 years old (case 2). The remaining living animal was an adult male. Both female wallabies were submitted to the Oklahoma Animal Disease Diagnostic Laboratory (Stillwater, OK) for diagnostic necropsy.
Examination of both animals revealed similar gross findings, with significant findings limited to the cardiovascular, pulmonary, hepatobiliary, and musculoskeletal systems. The wallaby in case 1 was moderately obese. The heart was mildly enlarged and on cut surface, marked thickening of the left ventricular free wall and moderate thickening of the interventricular septum were seen. The left ventricular free wall measured 2.0 cm, the interventricular septum measured 1.5 cm, and the right ventricular free wall measured 0.7 cm in thickness. Mild, multifocal hemorrhage and dry musculature were evident in the right hind limb. There was a single liver fracture and approximately 20 ml of blood within the abdomen. The thyroid glands were within normal limits.
In case 2, the wallaby was in an adequate state of nutritional condition. The heart was mildly enlarged, with slight rounding of the cardiac apex, and on cut surface, marked concentric left ventricular hypertrophy of the left ventricular free wall and interventricular septum was seen (Fig. 1). The left ventricular free wall measured 2.0 cm, the interventricular septum measured 2.1 cm, and the right ventricular free wall measured 0.5 cm in thickness. The lumen of the left ventricle was markedly decreased, and the left atrium was mildly dilated. Bilaterally, the hind limbs had a moderate to marked amount of subcutaneous and intramuscular hemorrhage of the quadriceps muscles and long digital extensor muscles. The thyroid glands were within normal limits.
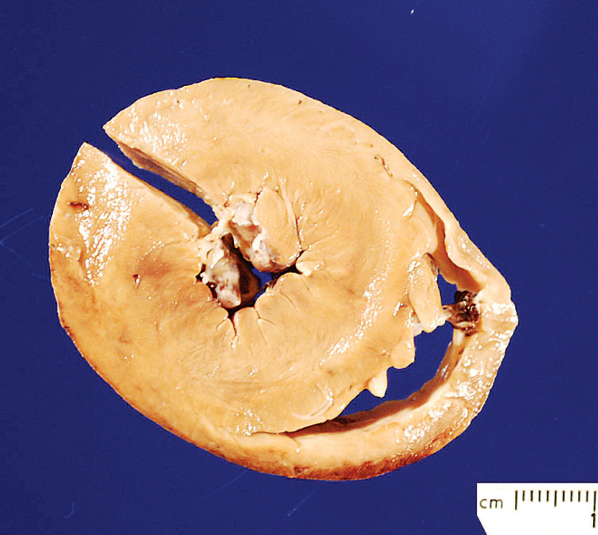
Heart; Bennett's wallaby (Macropus rufogriseus rufogriseus), case 2. Markedly thickened left ventricular free wall and interventricular septum with decreased size of the left ventricular lumen. Section removed from left ventricular free wall for histopathologic studies. The pale discoloration is due to formalin fixation. Bar = 1.0 cm.
To date, there is no published data regarding normal gross or histologic cardiac parameters for any wallaby species. When compared to available cardiac measurements from healthy red kangaroos (Macropus rufa) obtained utilizing echocardiograms, 11 the wallabies in the current report had enlargement of the interventricular septum and left ventricular free wall (Table 1). Additionally, there was asymmetric hypertrophy of the left ventricular free wall in wallaby 1 and symmetric hypertrophy of the left ventricular free wall and interventricular septum in wallaby 2.
Based primarily upon the gross findings of the present case, the diagnosis and cause of death were HCM. Histopathologic examination was performed, and histologic changes were observed in the heart, lungs, liver, and skeletal muscles. In case 1, sections of the left ventricular free wall were examined. Histologic lesions of the heart included mild myofiber disarray, mild myofiber diameter asynchrony, and marked myocardiocyte hypertrophy with no evidence of myocardial necrosis, mineralization, and/or fibrosis. Microscopic findings of the lungs and liver consisted of marked pulmonary and centrilobular congestion, respectively. Skeletal musculature exhibited mild multifocal myonecrosis and multifocal hemorrhage. All other tissues examined were within normal limits. In case 2, sections of the left ventricular free wall, interventricular septum, and right ventricular free wall were examined. Lesions consisted of marked myocardiocyte hypertrophy, with no evidence of myocardiocyte necrosis, mineralization, and/or fibrosis. There was marked pulmonary and centrilobular hepatic congestion. Skeletal musculature exhibited mild multifocal hemorrhage. Heart failure cells were not observed within the lungs in either case. All other tissues examined (haired skin, uterus, ovary, spleen, brain, and kidney) were within normal limits.
Comparison of heart measurements and ventricular ratio [(left ventricular free wall [LV] + interventricular septum [S])/(right ventricular free wall [RV])] compared to healthy mature animals.*
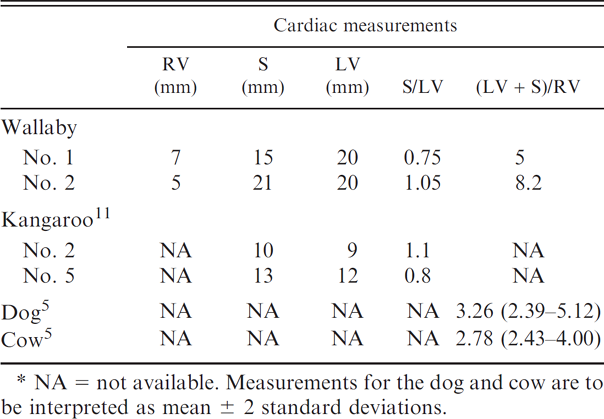
NA = not available. Measurements for the dog and cow are to be interpreted as mean ± 2 standard deviations.
Left ventricular free wall and/or interventricular wall thickening are consistent gross findings in HCM of domestic animals. Pulmonary congestion and edema are commonly associated with left-sided heart failure. Additional, but inconsistent, findings of HCM include left atrial dilation, atrial thrombosis, and aortic thrombosis. The histopathologic hallmarks of HCM are myocardiocyte disarray, cardiac myofiber hypertrophy, and/or cardiac fibrosis. 6 In kangaroos with LVH, myocardial fibrosis has not been observed. 8
As a result of the grossly observed skeletal muscle hemorrhage and dry texture of the hind limb musculature in case 1, fluorescent antibody testing for Clostridium chauvoei, the causative agent of blackleg disease, was performed and was negative. Additional diagnostic testing was undertaken for this case to elucidate a cause of HCM. Although there was no gross or histologic evidence (multiphasic myocyte necrosis, mineralization, and/or fibrosis) to support a nutritional myopathy, vitamin E and selenium analyses were performed on the liver of case 1. Results were 24.45 μg/g dry weight and 1.8 μg/g dry weight for vitamin E and selenium, respectively. No published reference values using dry weight are available for any wallaby species; however, these results were interpreted as within normal limits, as most species have reference ranges of 20–40 μg/g dry weight and 1.0–2.5 μg/g dry weight for hepatic vitamin E concentrations and selenium concentrations, respectively. 8 Pelleted feed was analyzed for ionophores (monensin, lasalocid, and salinomycin) and sodium fluoroacetate (1080); none were detected. Analyses of plasma levels of taurine and carnitine were not performed because appropriate samples for testing were not available.
The association between the sudden death as a result of HCM and the hind limb subcutaneous and intramuscular hemorrhage (cases 1 and 2), myonecrosis (case 1), and liver fracture (case 1) is purely speculative. However, the authors theorize that these changes likely represent acute traumatic injury that occurred prior to death. This may be due to a predator-prey interaction either within the enclosure or surrounding the enclosure, given that coyotes (Canis latrans), bobcats (Lynx rufus), and wild dogs are all present within this region of Oklahoma. The decreased cardiac output secondary to the HCM may have led to exacerbation of the animal's heart disease during forced exertion, resulting in acute left-sided congestive heart failure. The presence of myonecrosis is supportive of capture myopathy as a cause of sudden death in wallaby 1. Another factor that may have exacerbated the existing heart disease in these animals is the weather at the time of death. The average temperature in central Oklahoma during this time was 26.6°C, with temperatures as high as 44.3°C, accompanied by high humidity (http://climate.mesonet.org/monthly_summary.html).
The diagnosis of HCM in these 2 wallabies rests predominantly upon the gross cardiac findings accompanied by a comprehensive effort to rule out other causes of sudden death and to rule out etiologies of some lesions seen. Cardiac histologic findings are considered supportive of the diagnosis, and the interpretation of cardiac histology of these 2 cases should not be considered definitive. Like many other animal species, peer-reviewed literature establishing normal ranges of gross and histologic cardiac parameters of any macropod species is not available, and such data would assist in the diagnosis of similar cases in the future.
Unfortunately, the inciting cause of the HCM for these 2 cases is not known. According to the owner, all wallabies on this farm are from different source farms and are not genetically related. However, because written pedigree information confirming this fact was not available and because any wallabies outside of Australia are part of a limited and unvarying gene pool, a genetic basis for this disease process cannot be excluded. What seems more likely, however, is that the cause of HCM in these animals is environmental. This assumption is based on the occurrence of 2 cases at the same facility, the short time frame between the 2 deaths, the lack of chronic changes, and the degree of the cardiac changes observed. With genetically based (long-term) HCM, more chronic changes, such as cardiac fibrosis, would be expected. One potential source of an environmental cause for development of HCM could be ingestion of a toxic plant. Plants that are found on this farm and potentially ingested by the wallabies include oak (Quercus species), buffalo bur nightshade (Solanum rostratum), mulberry (Morus species), and pokeweed (Phytolacca americana). These plants do have toxic principles but are not known to be associated with cardiac disease. If these cases truly have an environmental source, it is quite possible that the remaining wallaby that is alive at the time of this report has HCM as well. The remaining third wallaby on this farm was lost to follow-up.
With appropriate gross lesions, HCM should be suspected as a cause of mortality in wallabies without clinical signs, and a diagnosis can be made at necropsy. The gross lesions of HCM in the wallabies on this farm are similar to the lesions reported in other domestic animal species; however, histologically, the lack of cardiac fibrosis in kangaroos with unexplained LVH is strikingly similar to that of the wallabies reported in the current case. Additional gross lesions, such as aortic and atrial thrombi, were not observed in the wallabies in the present study. There are no reports of thrombus formation in kangaroos with LVH.
In these 2 cases, no specific etiology for the HCM was identified. Whether there is an underlying infectious agent, nutritional or toxic component, or genetic cause for the HCM is currently not known. As stated previously, the limited export of Australian native species does contribute to a lack of genetic diversity within existing populations of these animals outside of Australia and lends support to a potential genetic cause. Genetically related HCM in domestic cats often occurs in middle-aged cats, and this process could be similar in wallabies. Assuming an average life span of 10–12 years, these wallabies (at 3 and 4 years of age) were approaching middle age. There may also be environmental factors of which the authors are unaware that contributed to the development of HCM in these 2 animals. Alternatively, the changes found in these wallabies may represent LVH similar to that seen in the kangaroo. Although not suspected in the cases reported in the present study, other myocardial diseases in wallabies that may result in acute death include toxoplasmosis and nutritional myodegeneration, 2 both of which exhibit typical histologic findings of inflammation and necrosis (toxoplasmosis) and polyphasic myodegeneration and necrosis (nutritional myodegeneration). None of these findings were present in these 2 cases.
To the authors' knowledge, HCM has not been previously reported in Bennett's wallabies. Based on the information reported herein, further studies should be performed to document normal gross and histologic cardiac parameters in macropods, to elucidate the cause(s) of cardiac changes in wallabies, and to determine whether the wallaby may serve as a potential model for HCM in humans (similar to the use of the kangaroo for studying fatal rhythm disturbances). Such studies should employ electrocardiography, echocardiography, and histopathology to fully evaluate wallabies with cardiac disease.
Acknowledgements. The authors would like to thank Dr. Sandra Morgan and Kelly McCracken for their help in working up this case.
Footnotes
a.
Mazuri®, PMI Nutrition International LLC, St. Louis, MO.
b.
Booster Feed Mill LLC, Tulsa, OK.
