Abstract
Jaw infections in macropods are common and will result in mortality if not promptly diagnosed and aggressively treated. They have most often been reported in wallabies; however, in the current case, the gross and histopathologic findings, microbial culture, and management of jaw infections in a population of red kangaroos (Macropus rufus) housed in a zoological park are described. Three red kangaroos, among a group of 23, were submitted for necropsy after death after progressively invasive and nonresolving jaw infections. Extensive bone and soft-tissue inflammation and necrosis were observed in all animals. A mixed population of aerobic and anaerobic bacteria was cultured; however, Pseudomonas spp. was consistently isolated from the sites of infection in all animals. Parental administration of gentamicin and penicillin, along with daily oral flushing of the wounds with Betadine, removal of all rough forage, and hand feeding soft-pelleted feed, was effective in controlling the progression of disease in 1 affected animal. This case documents an important disease in an additional macropod species and identifies predisposing factors, possible etiologies, and treatment and/or management options.
Keywords
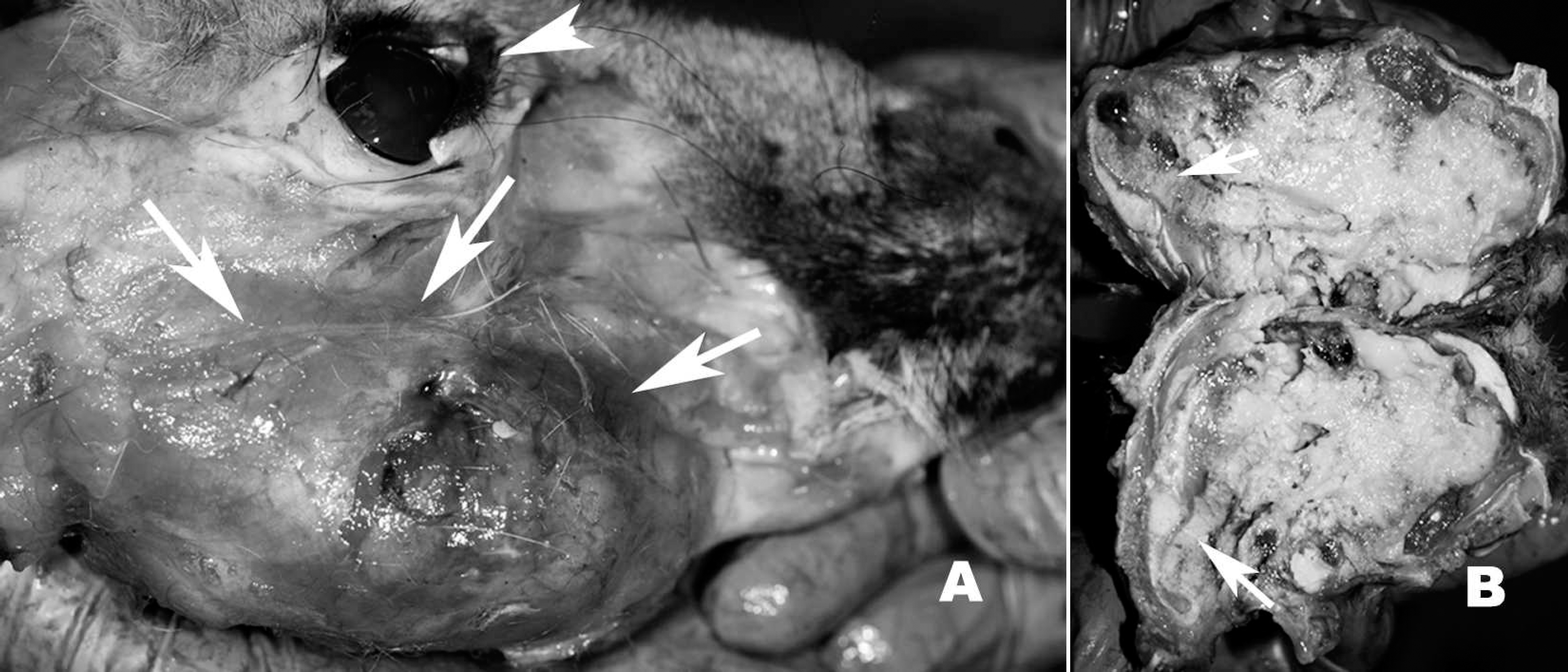
A, gross photograph of a mandibular swelling (arrows), with the skin reflected, in a red kangaroo (Macropus rufus); arrowhead denotes eye for orientation.
Infection of the soft tissues and bones of the face has been reported in many species of wild and domestic ruminants 3 and wild and captive macropods. 2,4,6 This infection has been referred to by many names, including lumpy jaw and jaw disease, but is best described as oral osteomyelitis and myelitis or alveolar osteomyelitis. 1 Coarse forage, dental eruptions, oral abrasions, and poor drainage of oral infections have been implicated as predisposing factors to this type of infection. 2,6 In macropods, Fusobacterium necrophorum (order Fusobacteriales, family Fusobacteriaceae, genus Fusobacterium) is generally considered the primary etiologic agent and often is isolated, along with various other bacteria, such as Streptococcus spp., Actinomycete spp., and Bacteroides spp. 6,7 Red-necked wallabies (Macropus rufogriseus) are the macropods most often reported to be afflicted with this debilitating disease, 4 and past studies unsuccessfully attempted vaccination for F. necrophorum, along with antimicrobial therapy in treatment regimes. Furthermore, consistent isolation of a primary organism has not been successful, and, generally, a mixed genera of bacterial organisms has been cultured. 6 Thus, the etiology and treatment of oral osteomyelitis in macropods remains unclear. Herein, a case of oral osteomyelitis in a captive herd of red kangaroos is described, and a plausible etiology and effective treatment protocol is offered.
Red kangaroos (Macropus rufus; 2–3 years of age; 1 female and 2 males) with a history of unresolved oral osteomyelitis were submitted, within a few hours of death, to the University of Georgia Veterinary Diagnostic and Investigational Laboratory (VDIL; Tifton, GA) for necropsy. The kangaroos were from a captive facility with a previously excellent history of herd health. Herd size at the time of submission was 23 animals, with approximately 20% of the herd affected. The animals were thin, with minimal-to-mild postmortem autolysis. All animals had variably sized firm swellings (2X-4X) of the mandibles (Fig. 1A). These swellings occasionally extended to the temporomandibular joint and the periorbital region. Teeth were generally loose, and the bone was friable and poorly discernible (Fig. 1B), with coalescing areas of caseous and liquefactive necrosis characterized by putrid gray-green exudate. Impacted forage (e.g., course grass and/or hay) was found within the oral lesions. Generally, only minimal body fat was present, but the stomachs were approximately 50% full of forage and feed, and the intestines contained formed feces. Other organs were mildly congested but otherwise unremarkable.
A set of tissues (brain, oral lesion, lung, heart, thymus, lymph nodes, trachea, gastrointestinal tract, liver, kidney, spleen, urinary bladder, and muscle) were collected fresh for bacterial and fungal cultures, as well as for virus isolation. A similar set of tissues were collected into 10% buffered formalin for histologic examination. In addition, swab specimens collected from oral lesions of a live animal were submitted and cultured as fresh tissue specimens for bacterial and fungal organisms. Tissues for aerobic and anaerobic bacterial cultures and fungal cultures were processed by using standard operating protocols at VDIL. Briefly, the samples were inoculated onto a trypticase soy agar plate with 5% sheep blood, Sabouraud dextrose agar, and a mycobiotic agar. The inoculated plates were incubated at 35°C for up to 7 days for bacterial culture and at ambient temperature for 4 weeks for fungal culture. Anaerobic cultures were performed on a CDC (Centers for Disease Control and Prevention) blood agar and were incubated in an anaerobic chamber at 35°C. For virus isolation, a 10% tissue homogenate of the visceral organs was made and filtered directly onto confluent monolayers of multiple cell lines, including Vero, Madin-Darby bovine kidney, and Crandell feline kidney cells. Cultures were observed daily for viral cytopathic effects. If, after 2 weeks, no viral cytopathology was observed, then the inoculum was passaged onto confluent monolayers and observed daily for an additional 2 weeks. Cultures were considered negative if no viral cytopathology was observed at the end of the second passage. Formalin-fixed tissues were routinely processed, embedded in paraffin, sectioned at 4 μm, placed on glass slides, stained with hematoxylin and eosin, and examined under light microscopy.
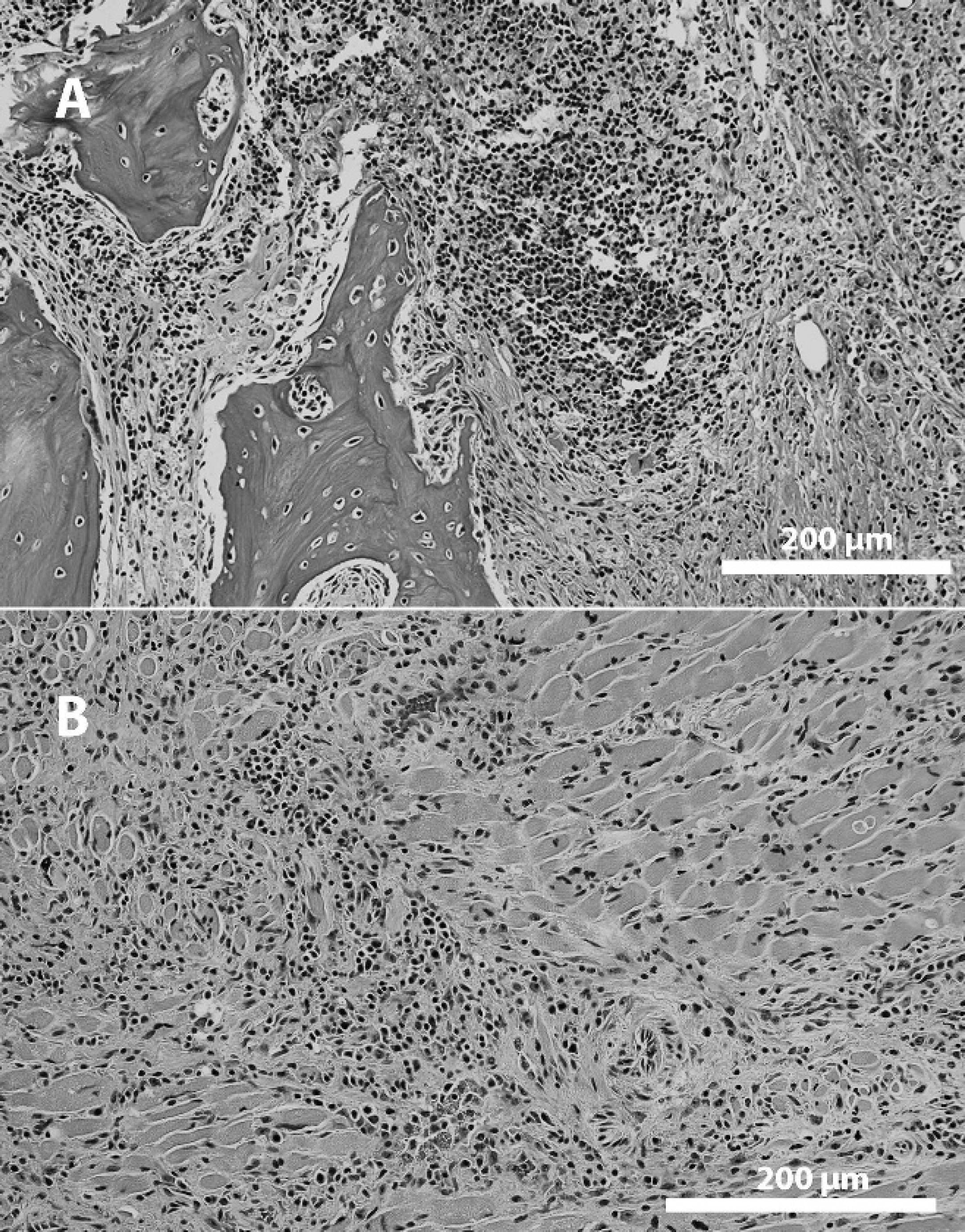
Photomicrograph of the mandibular lesion from a red kangaroo (Macropus rufus). Necrosis and inflammatory cell infiltrates resulted in extensive bone destruction
On histologic examination, extensive necrosis was found within the lesions noted grossly and was accompanied by exuberant mixed (neutrophils, multinucleated cells, macrophages, lymphocytes, and plasma cells) inflammatory cells. This necrotizing inflammatory infiltrate encompassed the bone and surrounding muscles, salivary glands, lymphoid tissue, and skin (Fig. 2). Mixed colonies of bacteria were present within the inflammatory infiltrate. Congestion was noted in other tissues but was generally mild. Bacterial cultures yielded a wide mixture of organisms (Table 1) but Pseudomonas spp. predominated in all cases. Significant bacterial growth was not isolated from other organs. Fungal cultures were negative in all cases. No viruses were isolated.
Results were consistent with oral osteomyelitis, commonly known as lumpy jaw. Although systemic infection was initially suspected, supportive gross, histologic, and bacteriologic evidence was not found. In the current case, morbidity and mortality occurred in the winter months and is similar to what was reported in other macropods. 4 In adults, this is likely because of a tendency to forage on courser vegetation when grasses are limited. This may be similar in young macropods but is also likely compounded by tooth eruption, and thus it is possible that young are equally susceptible year round. Based on the results of the antibiotic sensitivity, a treatment regime of daily subcutaneous injections of 2.2 mg/kg gentamicin and 30,000 IU per 13.6 kg penicillin G was instituted, along with oral flushing of the wounds with Betadine® solution a to a remaining live kangaroo with oral lesions. In addition, all coarse forage (including pampas grass that was growing in the enclosure) was removed, and hand feeding of softened pelleted food was offered daily to ensure adequate dietary intake. Because this species is subject to stress from handling, treatment regimes were modified as necessary. Resolution of clinical disease was achieved in approximately 8 weeks. One year later, no additional cases have been reported. From all these cases, it is conceivable that a sustained injury of the mucosa of the oral cavity, possibly because of abrasions from grazing on rough forage, followed by invasion of organisms present in the environment or oral cavity, may have caused this syndrome. Because the injury at this site is exposed to all oral flora (both aerobic and anaerobic), and also environmental organisms, it could be a colonization and replication of a mixed bacterial population.
Microbial organisms cultured from 4 (1 live and 3 at necropsy) red kangaroos (Macropus rufus) with osteomyelitis.*

Numbers in parentheses are the numbers of kangaroos from which the organism was isolated.
A previous study 6 suggested that F. necrophorum may be the predominant etiology for jaw disease in macropods. However, in the current study, Fusobacterium was isolated from only 1 animal. It is possible that the presence of other aerobic and anaerobic bacteria in large numbers may have precluded the isolation of Fusobacterium from 1 sample. All anaerobes isolated in the current study are indigenous to the oral cavity of humans and possibly animals, 8 because this location provides a suitable habitat for these organisms. Microbiologic analysis for understanding the presence and proportion of these bacterial agents will be useful in deciding the appropriate antimicrobial therapy for these infections. It is also important to know the antimicrobial susceptibility of the organisms to select an appropriate combination of antibiotics. A 2003 report 2 described successful resolution of chronic mandibular osteomyelitis in a Bennett's wallaby (Macropus rufogriseus rufogriseus) by using polymethyl-methacrylate beads impregnated with gentamicin and clindamycin. This treatment required anesthetization and included a 5-day postoperative period of hospitalization with daily subcutaneous administration of carprofen (50 mg), buprenorphine (0.12 mg), and enrofloxacin (150 mg).
In the current case, daily subcuticular injection of a combination of penicillin and gentamicin for therapy based on the results of bacterial antimicrobial sensitivity testing was used. It was speculated that, in addition to the individual effect of each antibiotic, the synergistic effect 5 of this combination might be useful for resolving the infection. In addition, the local treatment with an antibacterial agent (Betadine) likely was helpful in this case, and the removal of course forage items aided in eliminating the predisposition to oral abrasions and in preventing the colonization and invasion of these bacterial agents and recurrence or new infections in the herd. The present case documents an important disease in an additional macropod species and documents an effective treatment and/or management regime.
Acknowledgements. The authors thank the caretakers of Wild Adventures Theme Park for their diligence with patient care, and the staff of the VDIL, Tifton, GA, for assistance in tissue processing, especially, Cindy Watson, Gail Clifton, Diane Rousey, Kim Bridges, and Melissa Parks.
Footnotes
a.
The Purdue Frederick Company, Stamford, CT.
