Abstract
A 9-year-old Golden Retriever dog was presented to the Veterinary Medical Center with a 3-week history of grand mal seizures and was subsequently euthanized. At autopsy, a discrete, firm, expansile mass was found in the right pyriform lobe, which compressed the ipsilateral hippocampus, thalamus, and cerebral cortex. Histologically, the mass was composed of well-differentiated adipose tissue supported by fibrous and mucinous stroma. Adipocytes exhibited strong immunoreactivity for vimentin and were negative for pancytokeratin (AE1/AE3), glial fibrillary acidic protein, neuron-specific enolase, and synaptophysin. These findings are most compatible with an intracranial lipomatous hamartoma, which is an extraparenchymal lesion that has been identified in several species. The current report describes an intracerebral lipomatous hamartoma in a veterinary species.
A 9-year-old, male, neutered Golden Retriever dog was presented to the Western College of Veterinary Medicine Veterinary Medical Center (Saskatoon, Saskatchewan, Canada) because of several grand mal seizures that had occurred over the preceding 3 weeks. There was no history of any previous neurologic abnormalities, but the dog had recently finished a course of cephalexin and prednisone prescribed for recurrent skin disease by the referring veterinarian. Other than evidence of osteoarthritis, no abnormalities were detected on physical examination; neurologic examination was unremarkable. Complete blood cell count was unremarkable; serum biochemistry showed mild hypercholesterolemia (6.62 mmol/L, reference [ref.] range: 2.70–5.94) and a mild increase in alkaline phosphatase (251 U/L, ref. range: 9–90). Urine was hyposthenuric (urine specific gravity 1.007) but otherwise was unremarkable. Changes were attributed to recent prednisone therapy. Thoracic radiographs and abdominal ultrasound were unremarkable. Transverse and sagittal magnetic resonance imaging (MRI) revealed a contrast-enhancing, intramedullary, pyriform lobe mass adjacent to the right caudal hippocampus that was hyperintense with T2-weighted images. These MRI findings were interpreted as consistent with a glial cell tumor. Radiation was recommended to the owner but declined; conservative medical management was chosen. The dog was initially treated with phenobarbital (3 mg/kg every 12 hr) a and prednisone (0.5 mg/kg every 24 hr), b but continued to have seizures ~1–2 times a week with occasional cluster seizures. The phenobarbital and prednisone dose was sequentially increased over several weeks to a final dose of 6.5 mg/kg every 12 hr and 0.5 mg/kg every 12 hr, respectively, to maintain reasonable control. Four months after initial presentation, the owners elected for euthanasia because seizures had become refractory to medical therapy.
Autopsy of the dog revealed severe generalized muscle atrophy despite the presence of abundant subcutaneous and visceral fat stores. The liver was mildly enlarged, yellow, and had several (≤1 cm in diameter) pale foci extending throughout the parenchyma. Gross lesions consistent with osteoarthritis were not identified. Removal of the brain revealed that the right pyriform lobe was mildly enlarged with a corresponding loss in distinction of cerebral gyri and sulci. Transverse sections of the brain revealed a well-circumscribed (1.5 cm × 1.8 cm × 1.7 cm), firm, yellow mass within the right pyriform lobe that extended rostrally to the level of the thalamus and caudally to the level of the rostral colliculi. The mass was attached to the overlying leptomeninges. There was compression of the ipsilateral thalamus (resulting in midline shift) and of the hippocampus and cerebral cortex (Fig. 1A). Mild hydrocephalus was present. No additional masses were found on postmortem examination.
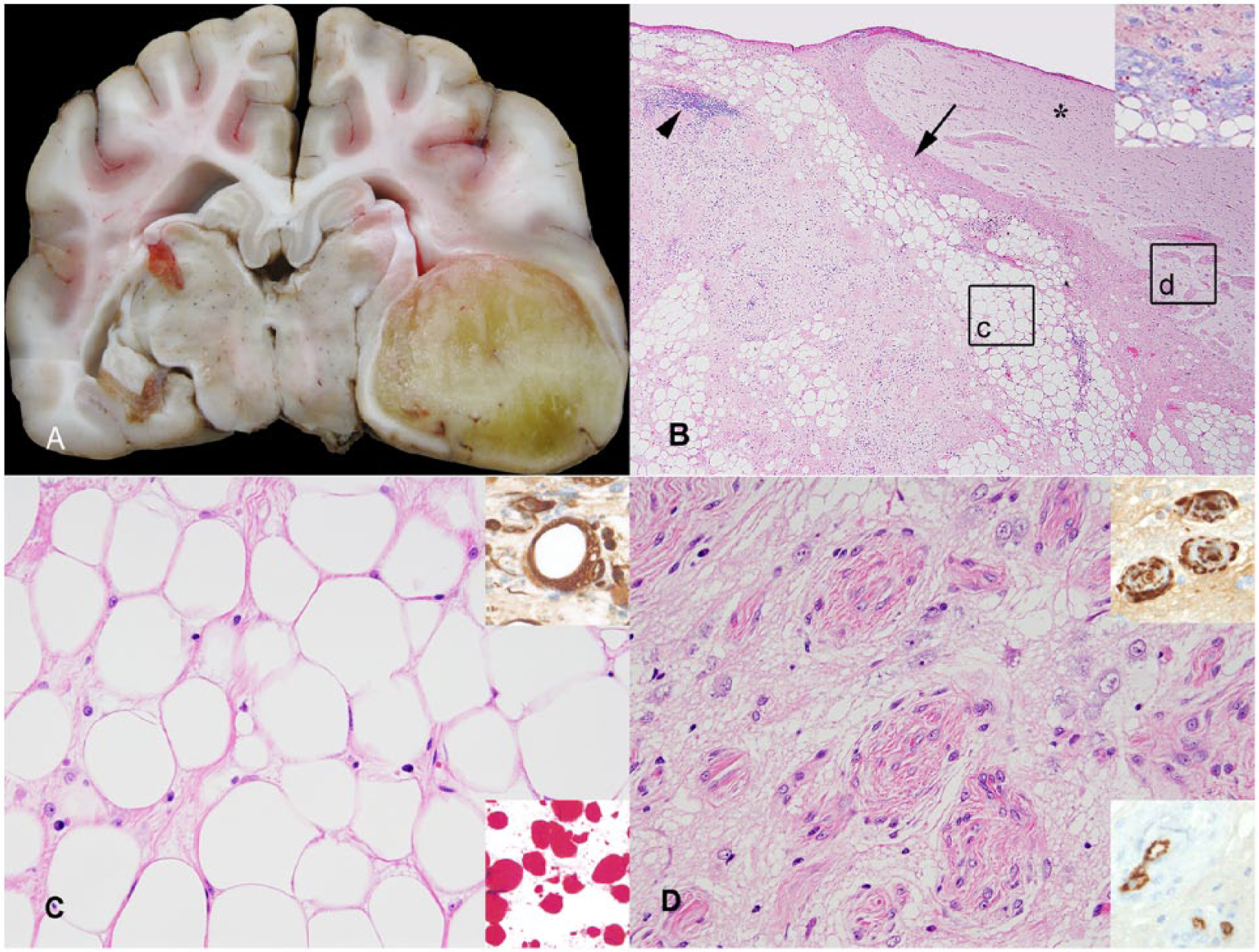
Brain mass in a dog.
Representative sections of the brain and all major tissues were collected and fixed in 10% neutral buffered formalin, embedded in paraffin, cut into 5-μm sections, and stained with hematoxylin and eosin (HE) for routine histological examination. The pyriform lobe mass was further evaluated with oil red O (in frozen sections), Masson trichrome, and alcian blue counterstained with periodic acid–Schiff (PAS). Immunohistochemical staining was performed for detection of vimentin, c pancytokeratin (AE1/AE3), d glial fibrillary acidic protein (GFAP), e neuron-specific enolase (NSE), f synaptophysin, g von Willebrand factor (vWF) antigen, h and smooth muscle–specific actin (SMA) i using a streptavidin–biotin amplification system (Table 1). j Binding of the primary antibody was detected using biotinylated horse anti-mouse immunoglobulins k and an avidin-biotin immunoperoxidase complex reagent, k with 3,3′-diaminobenzidine tetrahydrochloride (DAB) l as the chromogen. Electron microscopy was performed on formalin-fixed specimens of the brain mass that were postfixed in 5% glutaraldehyde followed by 1% osmium tetroxide. Sections were embedded in resin, contrasted with uranyl and lead acetate, and examined with a transmission electron microscope. m
Antibodies and their respective immunohistochemical staining method.*
Superscripts refer to the Sources and Manufacturers list.
Histologically, the parenchyma of the pyriform lobe was expanded and replaced by a well-demarcated, unencapsulated mass that compressed the periventricular white matter. The mass was lipomatous with collagenous to mucinous stroma (Fig. 1B), which was confirmed with Masson trichrome and alcian blue stains. The predominant tissue consisted of lobules and sheets of well-differentiated adipocytes. Most adipocytes contained a single, large, clear vacuole that displaced and compressed the nucleus (Fig. 1C) and was positive for lipid with oil red O stain (Fig. 1C, lower inset). The stroma was present as bands or islands within the fatty tissue and at the interface between the mass and the remaining brain parenchyma. Much of this stroma was collagenous and associated with bland spindle cells that were interpreted as fibroblasts, myofibroblasts, or both. Along the periphery of the mass the collagenous stroma was often arranged as perivascular collars (Fig. 1D), which in 1 area coalesced into a compact band that extended to the meninges. Other areas of stroma interpreted as mucinous were pale blue–gray on HE stain and alcian blue positive. Present throughout the fibrous and mucinous stroma was a mild multifocal mixed inflammatory infiltrate, consisting of moderate numbers of plasma cells, lymphocytes, and neutrophils and small numbers of eosinophils and Mott cells. There were multifocal areas of hemorrhage, which occasionally contained hematoidin crystals and extracellular and intrahistiocytic hemosiderin. The lipomatous mass did not extend beyond the pyriform lobe, but in the adjacent nonaffected cerebral cortex and hippocampus formation, there was mild vacuolation of the neuroparenchyma and small numbers of reactive astrocytes. Additional findings on histologic examination included hepatocellular vacuolar degeneration, resolving renal infarcts, nodular splenic hyperplasia, and atrophy of skeletal muscles.
Adipocytes within the mass and stromal spindle cells exhibited strong cytoplasmic expression of vimentin, consistent with their mesenchymal origin (Fig. 1C, 1D, upper insets). Endothelial cells in areas of collagen deposition exhibited cytoplasmic expression of vWF antigen (Fig. 1D, lower inset), and were variably surrounded by smooth muscle cells exhibiting cytoplasmic expression of SMA. GFAP expression was limited to astrocytic processes that surrounded the mass (astrogliosis) and scarce numbers of reactive astrocytes among adipocytes. Synaptophysin and NSE were expressed by few entrapped neurons at the periphery of the mass. Staining for pancytokeratin was negative.
Adipocytes were lined by a complete basal lamina and contained a large non–membrane-bound unilocular, electron-lucent vacuole and a small rim of peripheralized cytoplasm. Nuclei were eccentric (signet cell appearance), and there were numerous mitochondria within the cytoplasm.
The histologic, immunohistochemical, and ultrastructural features of the pyriform lobe mass described in our report resemble those of an intracranial lipomatous hamartoma—an uncommon lesion reported in humans, dogs, laboratory mice and rats, ducks, and in a single horse, pig, and humpback whale.1,2,4,6,9,10,12,13 Intracranial lipomatous hamartomas are composed predominantly of mature adipose tissue with varying amounts of intervening fibrous tissue or other mesenchymal elements; however, in contrast to the mass presented herein, these lesions are typically extraparenchymal and arise from meninges or are associated with midline structures of the brain such as the corpus callosum and ventricles.1,2,4,6,9,10,12,13 In humans and other species, additional anomalies, including agenesis or hypoplasia of the corpus callosum and septum pellucidum, often occur in association with lipomatous hamartomas, but these were not present in our case.6,12
Although the histogenesis remains unclear, it is generally regarded that intracranial lipomatous hamartomas are congenital malformative lesions that arise from pluripotential cells of the meninx primitive—the embryonic mesenchymal covering of the brain that gives rise to the pia-, arachnoid-, and dura mater. 12 The intraparenchymal location of the lipomatous mass in the dog in the current study may indicate that it originated from the meninx primitiva associated with meningeal vessels extending into the cerebral cortex. On the other hand, if this mass arose from heterotopic mesenchymal cells of the meninx primitiva then a diagnosis of a choristoma may be more appropriate, but we are unaware of how this diagnosis could be confirmed.
In human and veterinary medicine, intracranial lipomatous hamartomas are generally asymptomatic and considered incidental findings on autopsy; however, compression of the adjacent neuroparenchyma can result in debilitating neurologic disease, including seizures, head tremors, and ataxia.1,2,4,6,9,10,12,13 They are most often encountered within the first few years of life, but an intracranial lipomatous hamartoma has been diagnosed in a mature horse and in elderly humans.1,2,4,10,12 The age of the dog described herein and the absence of concurrent neurologic deficits indicate that this mass was likely slow growing. 5 Although areas of necrosis were not observed in the sections examined, the presence of moderate numbers of neutrophils and scattered areas of hemorrhage may indicate occurrence of necrosis, which may have contributed to the recent onset of seizures in this dog.
Based on the anatomic location and histologic features of the mass described in our report, differential diagnoses included an invasive metaplastic meningioma, or a lipidized variant of an astrocytoma. Although meningiomas often exhibit positive immunoreactivity for vimentin, this diagnosis was considered unlikely given the purely intraparenchymal location of the mass, lack of cellular atypia, and absence of desmosomes, a common ultrastructural finding in meningiomas. 8 In dogs, astrocytomas are typically positive for GFAP and neoplastic cells containing bundles of intermediate filaments and glycogen granules, neither of which were features in this case. 11 A diagnosis of a lipoma was considered unlikely because of the highly collagenous and mucinous stroma within the mass. A metastatic (or possibly primary) liposarcoma was not considered given the absence of cellular atypia and because a primary tumor was not identified elsewhere at autopsy.
The extension of leptomeningeal fibrous stromal cells into the neuroparenchyma, along with evidence of concurrent perivascular collagen deposition was considered a reactive change in response to the lipomatous hamartoma. Meningioangiomatosis (MA) was initially considered as a differential for these areas; however, this was considered unlikely as MA is typically diagnosed in dogs within the first year of life; the prominent vascular component of MA was lacking as was necrosis at the interface of invading meningoendothelial cells and the neuroparenchyma. 3 A malignant neoplastic process was considered unlikely given the fatty tissue maturity and absence of cellular atypia; however, the distinction between some hamartomas and benign neoplastic processes is at times arbitrary.
Additional lesions in this dog, specifically hepatocellular vacuolar degeneration and muscle atrophy, were likely secondary to prednisone therapy. Steroid hepatopathy is a well-known sequela of long-term glucocorticoid therapy due to both activation of glycogen synthase and suppression or inhibition of glycogen phosphorylase. Hepatocytes become swollen and are characterized by microvacuolated cytoplasm due to increased glycogen synthesis and storage. 5 In our case, glycogen accumulation was confirmed by demonstrating abundant PAS-positive and diastase-sensitive intracellular material within hepatocytes. Although this dog’s prednisone dose was relatively low, we speculate that it may have been responsible for the generalized muscle atrophy because there was no evidence of inflammatory muscle disease or denervation on histologic examination of masticatory, tricep, and bicep femoris muscles. Glucocorticoid-induced muscle atrophy is commonly encountered in both humans and domestic species, possibly mediated through upregulation of myostatin, a negative regulator of skeletal muscle. 7 Cachexia associated with cancer was not considered because this dog still had ample fat stores and because intracranial tumors are rarely associated with generalized wasting. Renal infarcts and splenic nodular hyperplasia were considered incidental findings.
To our knowledge, an intraparenchymal brain mass resembling an intracranial lipomatous hamartoma or choristoma has not been previously described in any species. By providing the macro- and microscopic, immunohistochemical, and ultrastructural findings of this rare lesion, this case report serves as an important stepping stone in our attempt to better understand uncommon lesions affecting the central nervous system of dogs.
Footnotes
Acknowledgements
We thank Dr. Jeffrey Joseph, neuropathologist at the Clark H. Smith Brain Tumor Centre (Calgary, Alberta, Canada), for providing his invaluable insight regarding this case. We also thank Prairie Diagnostic Services Inc., Saskatoon, Saskatchewan for performing immunohistochemistry.
Authors’ contributions
SJ Scott, K Elliot, H Philibert, and E Simko contributed to conception and design of the study. SJ Scott and K Elliot drafted the manuscript. All authors contributed to acquisition, analysis, and interpretation of data; critically revised the manuscript; gave final approval; and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Pharmascience Inc., Montréal, Quebec, Canada.
b.
Teva Canada Ltd., Toronto, Ontario, Canada.
c.
Sigma-Aldrich Canada Ltd., Oakville, Ontario, Canada.
d.
BioGenex Laboratories Inc., San Ramon, CA.
e.
Immunostar Inc., Hudson, WI.
f.
NeoMarkers Inc., Fremont, CA.
g.
Leica Microsystems Canada Inc., Richmond Hill, Ontario, Canada.
h.
Dako Canada Inc., Mississauga, Ontario, Canada.
i.
Vector Laboratories Inc., Burlingame, CA.
j.
BMK iVIEW DAB paraffin detection kits, Ventana Medical Systems Inc., Tucson, AZ.
k.
Vector Laboratories Inc., Burlingame, CA.
l.
Electron Microscopy Science, Ft. Washington, PA.
m.
Philips 410LS, Philips/FEI Corp., Eindhoven, The Netherlands.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
