Abstract
A 1-month-old Domestic Shorthair kitten was submitted for autopsy, with a history of upper respiratory tract infection and diarrhea. This was the third kitten from the same litter that had died with similar clinical findings within a period of 1 month. Severe conjunctivitis, rhinitis, tracheitis, and bronchointerstitial pneumonia were present, together with lymphohistiocytic colitis. Respiratory lesions were caused by infection with Felid herpesvirus 1. Colonic lesions were associated with the presence of long filamentous bacteria, identified as Clostridium piliforme, in the cytoplasm of epithelial cells. Our report describes a case of concurrent C. piliforme infection (Tyzzer’s disease) and feline rhinotracheitis in a kitten.
Tyzzer’s disease is caused by Clostridium piliforme, a long, filamentous, flagellated, spore-forming, Gram-negative, and obligate intracellular rod-shaped bacterium.3,14 Tyzzer’s disease has been described in mice, rats, rabbits, dogs, cats, cattle, pigs, monkeys, rarely in birds, and sporadically in wild species.9,14 C. piliforme has been detected in a Human immunodeficiency virus 1–positive human patient. 13 The pathogenesis of Tyzzer’s disease includes ingestion of spores and/or vegetative forms of C. piliforme, followed by replication in the ileo-ceco-colic area.9,18 In animals with a permissive immune system, the organisms are absorbed and may then travel to the liver and occasionally the systemic circulation, reaching the heart. 11 The typical triad of lesions includes hepatitis, enterocolitis, and myocarditis, although hepatic lesions are seen much more frequently than those in the gastrointestinal tract or the heart. Frequently, the disease results in sudden death, although nonspecific clinical signs such as depression, ruffled hair coat, and diarrhea can be observed before death; the severity of these clinical signs can be highly variable among individuals. 4 Although the incubation period in spontaneous infections is unknown, the disease has an extremely acute clinical course in all animal species.
Species Felid herpesvirus 1 (FHV-1; order Herpesvirales, family Herpesviridae, subfamily Alphaherpesvirinae, genus Varicellovirus) is a DNA virus responsible for feline rhinotracheitis, a very prevalent disease in kittens from shelters.1,15 FHV-1 replicates in the upper respiratory tract epithelium, resulting in necrosis and inflammation because of its cytolytic activity. The acute form of feline rhinotracheitis has an incubation period of 2–6 days. Major clinical signs include sneezing, coughing, oculo-nasal discharge, ulcers on corneal and conjunctival epithelium, weight loss, and fever. In extreme and sporadic cases in kittens up to 4 weeks of age, fatal bronchopneumonia may occur.6,7 Viruses of the Alphaherpesvirinae subfamily remain latent in nervous and lymphoid tissues. FHV-1 is commonly latent in the trigeminal ganglion, and its reactivation in adult cats results mainly from stress, with consequent virus shedding within 4–11 days. 7 Tyzzer’s disease and feline rhinotracheitis have been described separately in cats. However, coinfection by these 2 diseases has not been reported to our knowledge.
A 1-month-old, female, nursing Domestic Shorthair kitten was submitted for autopsy to the California Animal Health and Food Safety (CAHFS) Laboratory, University of California–Davis, San Bernardino branch. The kitten belonged to a shelter and had exhibited dyspnea and marked purulent nasal discharge, diarrhea, and dehydration for ~2 weeks before death, despite treatment with parenteral cefovecin sodium and penicillin, gentamicin eye drops, intravenous Lactated Ringer solution, and kitten milk replacement supplementation via tube feeding. This was the third kitten from the same litter of 3 animals that had died with similar clinical findings within a period of 1 month. The shelter housed 75 cats. Thirty of the kittens became ill (morbidity 40%) over the same period. Twelve of the affected kittens died (mortality 16%). The adult cats present in the shelter, including the dam of the kitten described herein, were clinically healthy. Serological tests for Feline leukemia virus (FeLV) and Feline immunodeficiency virus (FIV) were negative for all cats in the shelter.
A routine autopsy was performed, and representative samples of the conjunctivas, trachea, lungs, heart, liver, spleen, kidneys, pancreas, small and large intestine, and brain were collected and fixed in 10% neutral buffered formalin; 4-µm thick, hematoxylin and eosin (H&E) sections were prepared. Selected sections were also stained with Gram, Giemsa, and Steiner stains.
Immunohistochemistry (IHC) for feline panleukopenia virus (FPV) on jejunum, FeLV on jejunum and colon, and FHV-1 on trachea was performed following standard operating procedures of the Veterinary Teaching Hospital of the School of Veterinary Medicine, University of California–Davis. Briefly, antigen retrieval was performed with target retrieval solution a for FeLV in a steamer for 20 min at 95°C, and proteinase K b for 7 min for FPV and FHV-1 samples. The following primary mouse antibodies were applied to tissue sections: anti–FHV-1 (dilution 1:400), c anti-FeLV (dilution 1:300), d and anti-FPV (dilution 1:500). e Goat anti-mouse immunoglobulins conjugated to peroxidase-labeled dextran polymer f were used as a secondary reagent. All reactions were visualized with NovaRed for peroxidase. g Sections were counterstained with Mayer hematoxylin and coverslipped. Negative mouse serum was used instead of the primary antibodies mentioned above in negative control sections. Positive controls included bone marrow, lymph node, and liver from a FeLV-positive cat, small intestine from a kitten with FPV infection, and nasal epithelium of a cat with FHV-1 infection.
Fresh tissue samples of lung and trachea were submitted to polymerase chain reaction (PCR) analysis for FHV-1, Feline calicivirus, Bordetella bronchiseptica, Chlamydophila felis, and Mycoplasma felis. For extraction of total nucleic acid, an automated system h was used according to the manufacturer’s instructions. For each target gene, 2 primers and an internal, fluorescent-labeled probe were designed using commercial software, i or locked nucleic acid probes were used. Real-time PCR assays targeted the following genes: 1) conserved region of the glycoprotein B (gB) gene of FHV-1 (GenBank accession S66371.1), forward primer 5′-AGAGGCTAACGGACCATCGA, reverse primer 5′-GCCCGTGGTGGCTCTA AAC, and probe 5′-6FAM-TATATGTGTCCACCACCTTCAGGATCTACTGTCGT; 2) ORF1 gene (genome polyprotein) of Feline calicivirus (GenBank accession AY560117); 3) Bordetella bronchiseptica adhesin (fhaB) gene (GenBank accession BX640446); 4) major outer membrane protein A (ompA) of Chlamydophila felis (GenBank accession AB468956); and 5) 16S rRNA gene of Mycoplasma felis (GenBank accession AY274253).
For electron microscopy, a small section of the colon was fixed in 10% neutral buffered formalin from which smaller subsamples were obtained and transferred into modified Karnovsky fixative (1% paraformaldehyde j and 2% glutaraldehyde k ) overnight and processed for transmission electron microscopy per the CAHFS Laboratory standard operating procedure. A transmission electron microscope l was used at 80 kV accelerating voltage for observation. Images were captured with a side-mounted digital camera m using imaging software. n
At autopsy, the carcass was in poor nutritional condition, with no fat reserves and generalized muscle atrophy. The conjunctivas were reddened and covered by large amounts of yellow, fibrinopurulent exudate. The nasal cavity and trachea contained fibrinopurulent exudate, most of adhered loosely to the mucosa, and the lungs were multifocally reddened and firm. The colon was slightly dilated with green, soft contents.
Histologic examination revealed fibrinonecrotizing conjunctivitis, rhinitis, tracheitis, bronchitis, and bronchointerstitial pneumonia, with occasional intranuclear eosinophilic inclusion bodies. In particular, pulmonary lesions were characterized by alveolar wall necrosis, with intraluminal necrotic and desquamated cells, a moderate exudate composed of neutrophils and macrophages, and epithelial syncytial cell formation, with numerous intranuclear eosinophilic inclusion bodies within bronchiolar epithelium and syncytial cells (Fig. 1). Inclusion bodies occupied most of the nuclei and displaced the chromatin to the periphery. Histopathological changes outside the respiratory tract included severe colonic lesions, characterized by expansion of the lamina propria and separation of the crypts by a moderate inflammatory infiltrate, composed of macrophages, lymphocytes, and fewer plasma cells. Numerous crypts were dilated and lined with a low cuboidal to markedly attenuated epithelium, and contained necrotic cell debris and viable and degenerate neutrophils (Fig. 2). Many surface and crypt enterocytes, and goblet cells, contained intracytoplasmic bundles of slender, long rods ~15–20 µm long and 1 µm wide, arranged in a crisscross pattern (Fig. 2, inset). These rods were Gram-negative but stained strongly with Steiner (Fig. 3) and slightly with Giemsa stains. In the submucosa, there was a mild diffuse inflammatory infiltrate composed of lymphocytes, plasma cells, and macrophages. No lesions were identified in the other organs examined.
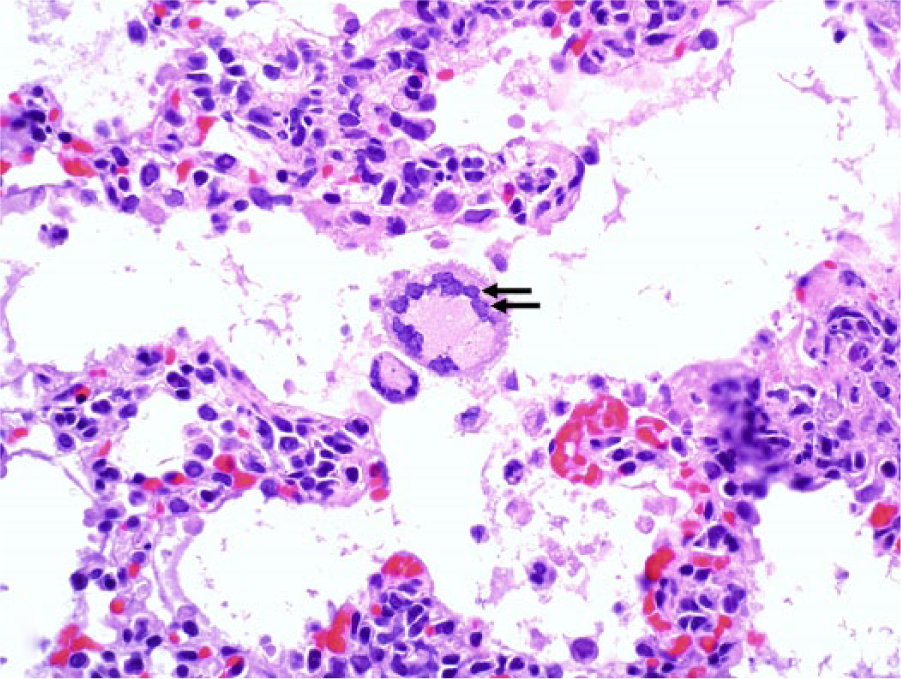
Lung. Alveolar septa are thickened with numerous mononuclear cells. Syncytial cells were present occasionally in alveoli, and contained eosinophilic intranuclear inclusion bodies (arrows). Hematoxylin and eosin. 600×.
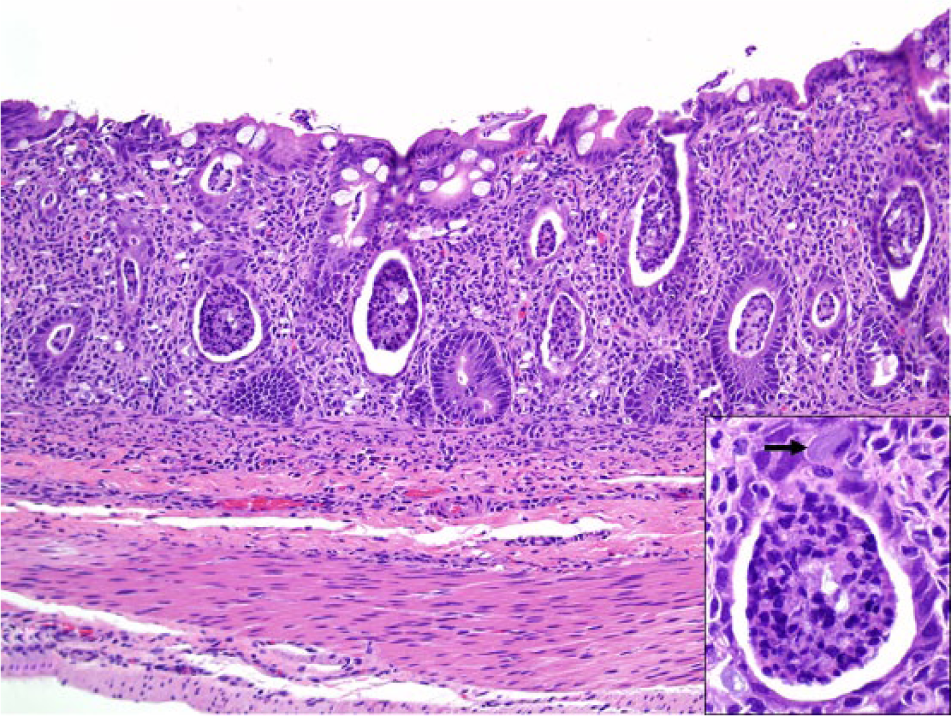
Colon. The lamina propria is markedly expanded by macrophages, lymphocytes, and fewer plasma cells. Abundant necrotic debris is present in crypts. Hematoxylin and eosin (HE). 200×. Inset: in some epithelial cells, faintly staining elongated bacteria are identified (arrow). HE. 600×.
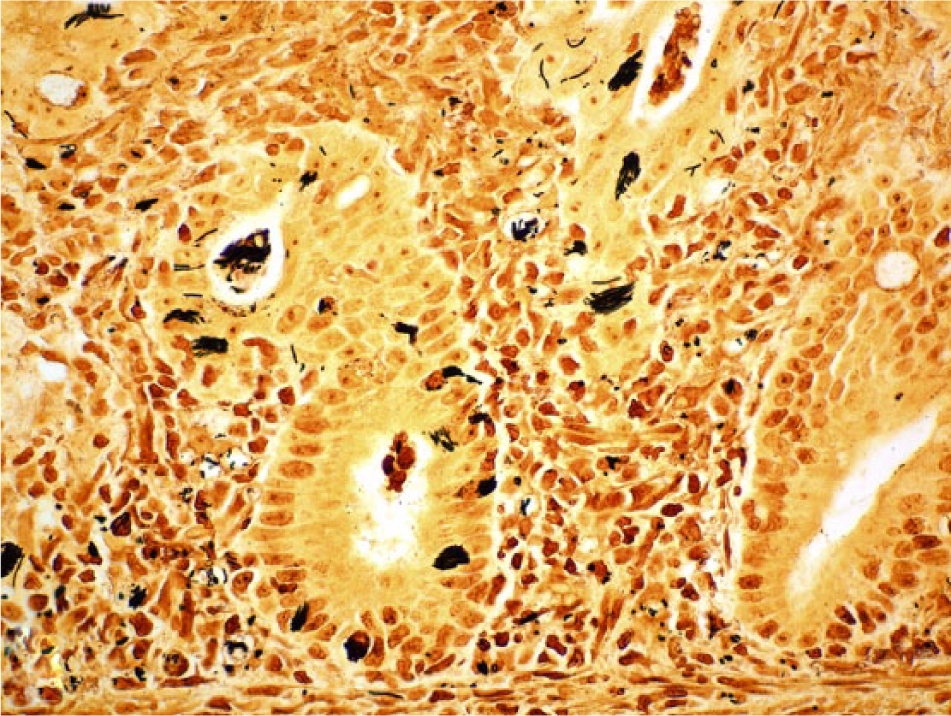
Colon. Numerous argyrophilic, elongated bacteria are present in the cytoplasm of epithelial cells and in the lumen of crypts. Steiner stain. 600×.
Immunohistochemistry for FHV-1 was strongly positive within the cytoplasm of several epithelial cells of bronchi, bronchioles, conjunctiva, and trachea (Fig. 4), and of lung syncytial cells. All other IHC tests were negative. Samples of trachea and lung were positive for FHV-1 by PCR, and negative for all of the other agents included in the PCR panel.
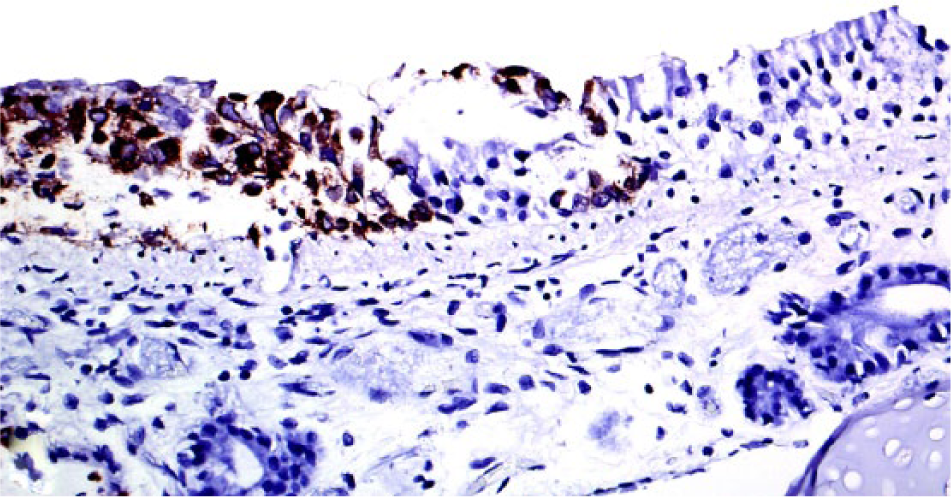
Trachea. Necrotic tracheal epithelial cells have strong cytoplasmic immunopositivity against Felid herpesvirus 1 (FHV-1). Mouse anti–FHV-1 (1:400) immunohistochemistry, Mayer hematoxylin counterstain. 400×.
Transmission electron microscopy of the colon revealed numerous transverse, longitudinal, and oblique sections of both vegetative bacterial forms and spores in the cytoplasm of enterocytes (Fig. 5). Cross-sections of vegetative forms measured 0.3–0.6 µm in diameter, with cell walls ~25 nm thick. Spore forms ranged in diameter from 0.6 to 1.4 µm and contained spore coats that were 90–250 nm thick. The vegetative forms had numerous peritrichous flagella that measured 10–15 nm in diameter (Fig. 6).
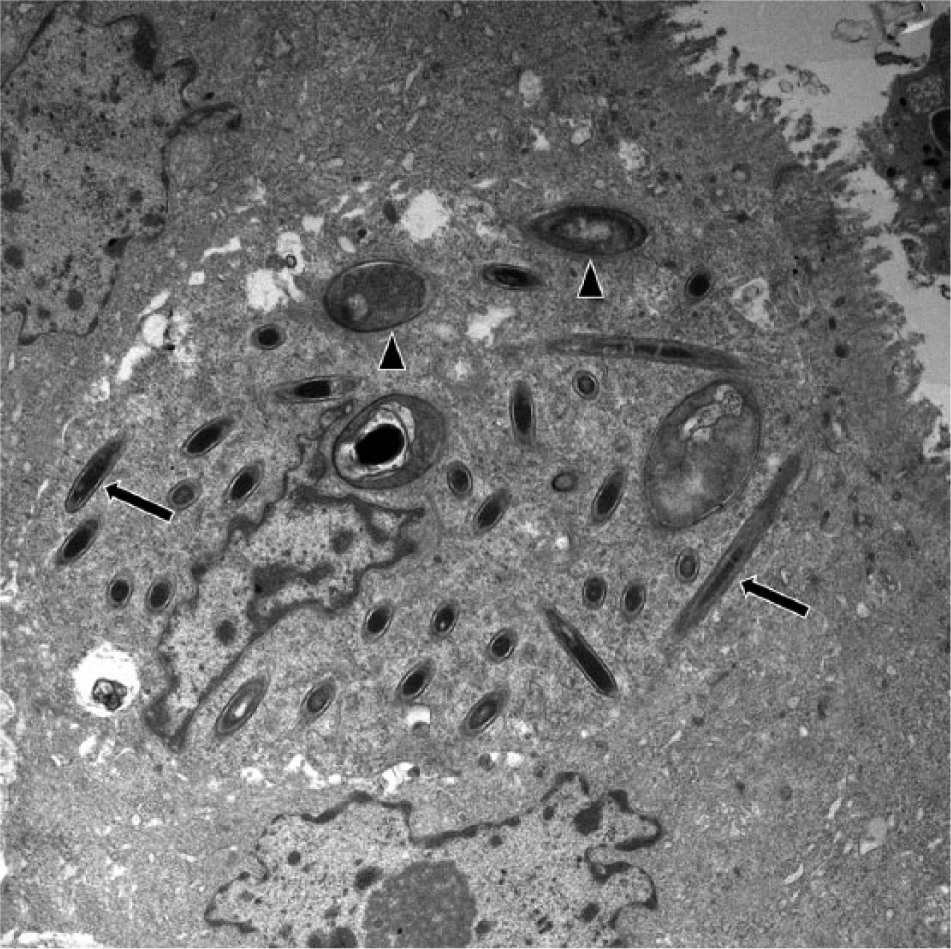
Colon. A crypt enterocyte contains numerous transverse, longitudinal, and oblique sections of both vegetative (arrows) and spore (arrowheads) bacterial forms. Uranyl acetate and lead citrate staining. 7,750×.
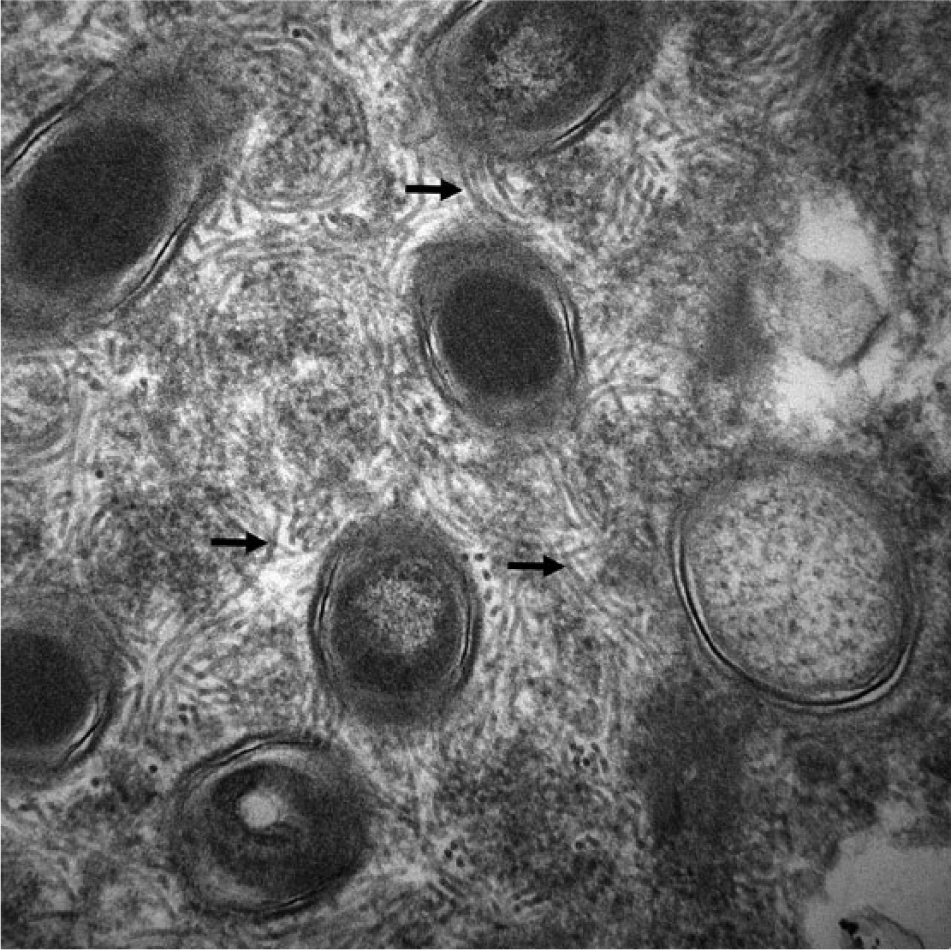
Colon. Cytoplasm of a crypt enterocyte. Bacterial vegetative forms measure 400–680 nm and have numerous peritrichous flagella ~10–15 nm in diameter (arrows), characteristic of Clostridium piliforme. Uranyl acetate and lead citrate staining. 60,000×.
A diagnosis of concurrent Tyzzer’s disease and feline viral rhinotracheitis was established in our case based on gross and microscopic changes, IHC, molecular test results, and ultrastructural features. Additional support for this diagnosis came from ruling out other common respiratory and digestive feline pathogens.
Clostridium piliforme is well known as a cause of severe, fatal enteric, hepatic, and/or cardiac disease in several animal species. Young animals are the most susceptible, and predisposing factors include stressful situations, such as overcrowding, transportation, and poor sanitation. 4 Viral infections can also trigger enough immunosuppression to alter host susceptibility. 12 Despite extensive studies in animal models, little is known about the pathogenesis and immune response in Tyzzer’s disease. The virulence factors of C. piliforme have not been fully characterized although virulence is thought to be related to toxins. Different C. piliforme isolates seem to vary in their degree of cytotoxicity, suggesting that different C. piliforme strains may cause different clinical forms of the disease. The host immune system plays a role in an individual’s susceptibility and disease severity. Interleukin-12 is expressed after C. piliforme inoculation in mice, but its level is substantially higher in resistant murine strain C57BL/6 compared to the susceptible strain DBA/2. 16 This suggests an important role of natural killer (NK) cells and Th1 response during infection and mediation of an immune response.
The ingestion of sporulated forms of C. piliforme is crucial for development of the disease, the source of which is usually feces of wild rodents or rabbits. No lesions caused by C. piliforme were seen outside the alimentary tract in the kitten presented in our report, which may be related to low bacterial strain virulence. Our case is similar to 2 previously described cases of Tyzzer’s disease in kittens, one of which was concurrent with feline panleukopenia, in which no hepatic or cardiac lesions were observed.5,9
The stress factors intrinsic to a shelter environment may be sufficient to trigger physiological stress and consequent immunosuppression. 1 Immunity in neonatal or very young animals is very dependent on passive transfer of maternal antibodies. Exposure to pathogens during this vulnerable period may result in infections with life-threatening outcomes. We hypothesize that environmental factors led to stress-induced immunosuppression in this kitten and most likely also in its littermates. The immunosuppression must have been serious enough to allow concurrent viral and bacterial infection and severe clinical manifestations that led to death.12,15 The histopathological presentation of FHV-1 infection in our case supports this hypothesis, given that bronchointerstitial pneumonia and syncytial cell formation are not commonly seen in classic feline rhinotracheitis. The abundant intraepithelial, intranuclear inclusion bodies also indicate an acute disease that developed concurrently with C. piliforme colonization and intestinal damage. It is also possible that FHV-1 infection acted as a stressor for these animals, making them more susceptible to other pathogens, including C. piliforme.
The diagnosis of C. piliforme can be challenging and depends on recognition of clusters of intracellular bacilli in tissue sections using appropriate staining methods, such as Giemsa and Steiner.4,10 Serology using whole bacterial lysates as antigens, and isolation by inoculation of embryonated hen egg or cell cultures, have been reported. 10 Additional testing used to diagnose this disease are IHC and PCR.3,8,10 PCR can be done from organs and feces and is mainly based on the 16S rRNA gene sequence of C. piliforme.2,8 Electron microscopy is another useful ancillary technique that allows identification of vegetative forms, spores, and flagella of specific shape and size, all of which were recognized in the tissues examined in our study.5,9 Such ultrastructural features are highly compatible with a diagnosis of C. piliforme infection.4,9 Tyzzer’s disease should be included in the differential diagnosis of feline enterocolitis, 17 especially when present in young domestic cats from shelters and/or subject to stressful factors, such as overcrowding, transportation, or virus-induced stress or immunosuppression.
Footnotes
Acknowledgements
We acknowledge Mrs. ME Uzal for the English review of this manuscript.
Authors’ contributions
RT Neto, FA Uzal, and FR Carvallo contributed to conception and design of the study. E Hodzic and M Persiani contributed to conception of the study. S Jolissaint and A Alcaraz contributed to design of the study. FR Carvallo contributed to acquisition of data. All authors contributed to analysis and interpretation of data; drafted the manuscript; critically revised the manuscript; gave final approval; and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Target retrieval solution (pH 9), Dako Denmark A/S, Glostrup, Denmark.
b.
Proteinase K, Dako North America Inc., Carpinteria, CA.
c.
Clone 7-7 (FHV 7-7), Custom Monoclonal International, West Sacramento, CA.
d.
Clone p27 (PF12J-10A), Custom Monoclonal International, West Sacramento, CA.
e.
CPV1-2A1, Custom Monoclonal International, West Sacramento, CA.
f.
Envision+ system-HRP mouse K4001, Dako North America Inc., Carpinteria, CA.
g.
SK-4800, Vector Laboratories Inc., Burlingame, CA.
h.
CAS-1820 X-tractor Gene, Qiagen GmbH, Hilden, Germany.
i.
Applied Biosystems Primer Express software, Life Tech-nologies, Grand Island, NY.
j.
Product no. 18501, Ted Pella Inc., Redding, CA.
k.
Product no. 18427, Ted Pella Inc., Redding, CA.
l.
Zeiss LEO 906E, Carl Zeiss MicroImaging GmbH, Göttingen, Germany.
m.
DVC-2000, Thorlabs Inc., Newton, NJ.
n.
AMT Imaging software, Advanced Microscopy Techniques Corp., Woburn, MA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
