Abstract
Clostridia can cause hepatic damage in domestic livestock, and wild and laboratory animals.
Introduction
Clostridial hepatitis is a general term used to describe diseases produced by several clostridial species in which the liver is the most severely affected organ. In animals, there are 3 well-defined diseases that fulfill this criterion: infectious necrotic hepatitis (INH), caused by
Infectious necrotic hepatitis
INH, sometimes referred to as “black disease,” is an acute toxemic disease of sheep produced by
Etiology
Typing of

− = no toxin produced; + to +++ = increasing amount of toxin produced.
Production of TcnA is related to phage infection. A phage-cured
Pathogenesis
The spores of
Although the literature on the pathogenesis of INH is scant, it is generally believed that migration of immature forms of flukes (mainly
Cytoskeletal disturbance results in cell death, loss of intercellular junctions, and increase in cell-barrier permeability.
56
These effects occur in hepatocytes and endothelial cells, the damage in the latter being responsible for extensive leakage of blood-derived fluid into the connective tissues.
16
The necrotizing and hemolytic beta toxin also contributes to hepatic necrosis.16,44 Because only small amounts of beta toxin are produced by
Epidemiology and clinical signs
INH affects animals of all breeds, sexes, and ages, with the highest incidence in individuals > 4 mo of age and usually in good nutritional condition.4,44
In some areas, INH varies seasonally given its association with fascioliasis and the fluctuation in the concentration of encysted
Despite its strong association with fascioliasis, INH can also occur in areas free of
Clinical signs are rarely seen in sheep or cattle affected by INH given the peracute nature of the disease. In sheep, it is common to find several animals dead without premonitory signs.44,67 When clinical signs are observed, they are usually present for only a few hours, and are nonspecific. Affected animals lag behind the flock or are sternally recumbent for a short time before death. Other clinical manifestations may include drowsiness, anorexia, hyperthermia, tachycardia, and tachypnea.44,67 Cattle with INH have clinical signs similar to those described in sheep.
INH is rare in horses. The few reports available on the disease in this species suggest that the clinical course is of 12–72 h duration,
48
and it is characterized by head tilt, ataxia, reluctance to move, abdominal pain, and, eventually, recumbency.73,82 Interestingly, and contrary to that which occurs in ruminants with INH, horses may sometimes exhibit jaundice, which can be readily observed in the mucous membranes and sclera.73,82 The reason for the occurrence of icterus in horses, but not ruminants, is unknown, but it may be attributed, at least in part, to an apparently increased susceptibility of horses to the action of the hemolytic beta toxin produced by
In ruminants and horses with INH, changes in clinical pathology profiles include neutrophilia with a left shift, increase of several liver enzymes, and nonspecific changes associated with toxemia.5,14,44,48,68
Gross changes
Rapid autolysis is commonly observed in carcasses of animals dying from INH, and unless an autopsy is performed soon after death, postmortem decomposition may mask the gross changes. 67 In sheep, there is usually marked congestion of subcutaneous blood vessels, and hemorrhages, resulting in dark discoloration of the subcutis, from which the name “black disease” is derived.16,44,67,68 Subcutaneous edema may also be seen over the sternum, and in ventral abdominal muscles and the inguinal region. 67 The abdominal cavity and pericardial sac contain variable amounts of straw-colored to serosanguineous fluid. Hemorrhages may be present on the pleura, endocardium covering papillary muscles, and mesentery. 48 The most striking and pathognomonic lesion is found in the liver, which in sheep and cattle consists of multiple, or rarely single, gray-yellow, randomly distributed areas of necrosis, surrounded by a rim of intense hyperemia (Fig. 1). A thin coagulum of fibrin on the hepatic capsule may overlay necrotic areas. Occasionally, the classical necrotic foci may not be seen from the surface of the liver, and slicing of the organ is needed to detect them.16,67 Evidence of immature fluke infestation with necrotic and hemorrhagic tracts in the capsule and hepatic parenchyma is usually present. 16 However, fluke-associated changes are not always seen at postmortem examination.
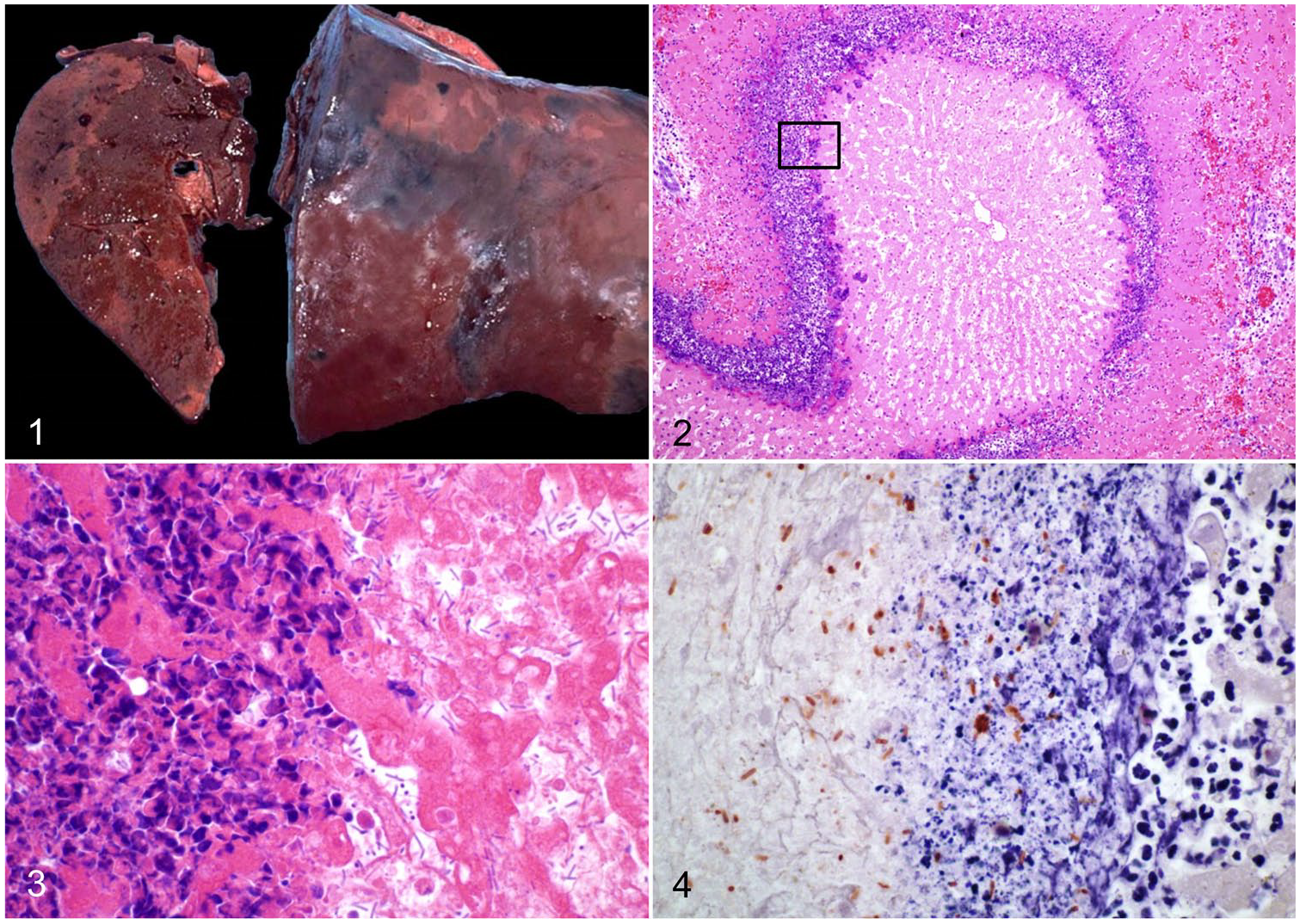
Infectious necrotic hepatitis.
In horses, a single focus of necrosis affecting the left hepatic lobe has been reported. 48 In addition, icteric discoloration of the abdominal serosa as well as adipose and subcutaneous tissue may sometimes be observed in this species.48,82
Microscopic lesions
The microscopic appearance of the liver lesion is considered to be pathognomonic for both INH and BH (see below). It consists of focal or multifocal coagulative necrosis of hepatocytes, surrounded by a leukocytic rim composed mainly of degenerate and viable neutrophils, and a few lymphocytes, plasma cells, and macrophages (Fig. 2). Within the inner margin of this inflammatory rim, there are usually large numbers of large gram-positive rods, many of them with subterminal spores (Fig. 3).16,44 Other histologic changes may include necrotic tracts associated with immature liver fluke migration. 16 Portal fibrosis, bile duct hyperplasia, and portal lymphocytic infiltrates, common features of chronic fascioliasis, may occur concurrently.44,54,55
Cases of equine INH may also exhibit segmental-to-diffuse fibrinoid necrosis of arteries and veins associated or not with fibrin thrombi and hemorrhage in various organs, including liver, lungs, and adrenal glands. 48 In the brain, randomly distributed, perivascular extravasations of eosinophilic, high-protein edema may occur, presumably as a result of the endothelial tropism of TcnA. 48
In horses, there may occasionally be cytoplasmic vacuolation of the renal tubular epithelium, with luminal protein casts, which are Okajima stain positive, consistent with hemoglobin. 48
Diagnosis
Epidemiologic data, clinical signs, and gross and microscopic changes are generally sufficient to establish a presumptive diagnosis of INH. Other causes of sudden death, including, but not limited to, anthrax, blackleg, and
Prevention
In ruminants, given that most cases of INH are associated with liver fluke infestation, control of this parasitosis is vital to help prevent cases of INH. Preventive measures include drainage of soils, limiting access of animals to streams, canals, and ponds, and strategic treatment of animals with flukicides.67,68 In addition, all carcasses of animals dying from INH should be burned, buried, or removed from the premises in order to avoid the dispersal of
Bacillary hemoglobinuria
BH is produced by
Etiology
The main virulence factor responsible for the pathogenicity of
Pathogenesis
The mechanism by which
Epidemiology and clinical signs
BH rarely affects young calves; most cases occur in animals > 1-y-old in good nutritional condition.
72
The disease has low morbidity, but lethality is commonly very high.
45
Cases of BH tend to be more frequent in animals recently introduced into infected pastures because native animals are believed to have some degree of natural immunity.
52
Environmental conditions leading to BH usually involve poorly drained soils and semi-permanent anaerobic, alkaline (pH ≥ 8.0) wetlands, either naturally or artificially created, into which
Animals affected with BH have a rapid onset of clinical signs, which may last from 12 h to 4 d. There is sudden loss of appetite, and cessation of lactation, rumination, and defecation. Affected animals also have anorexia, depression, tachypnea, tachycardia, and fever, as well as icteric mucous membranes, blood-stained feces, and hemoglobinuria.21,66,75,81 Death occurs as a result of severe toxemia and hemolysis-induced hypoxia. 43
Impaired hepatic function in BH leads to significant increases in plasma aspartate aminotransferase (AST) and gamma-glutamyl transferase (GGT) activities, and total bilirubin, and a reduction in the albumin-to-globulin ratio. 32 Hemolysis results in severe anemia.32,43,66 Leukocyte counts vary from initial leukocytosis with a left shift to, in advanced stages, exhaustion leukopenia.32,52
Gross lesions
Carcasses of animals dying of BH are commonly in good nutritional condition but may exhibit signs of dehydration. Edema, petechiae, and ecchymoses are scattered throughout the subcutis, and adjacent fascia and muscle, being most evident on the ventral body wall.21,43,52 Mucous membranes are pale or exhibit mild-to-severe jaundice, the latter which may be prominent in the subcutaneous tissue. The perineal area and tail are stained by dark red, liquid feces.45,52 Variable degrees of jaundice, ecchymoses, and petechiae may be seen on the parietal and visceral peritoneum and the mesentery. 52 The lungs are edematous, and the trachea and bronchi contain blood-tinged, frothy transudate. The pericardial sac and pleural cavity contain large amounts of hemoglobin-stained fluid.21,52
The characteristic, and pathognomonic, lesion is found in the liver. It generally consists of a single, wedge-shaped focus of necrosis with its base on the parietal surface of any of the hepatic lobes (Fig. 5). 45 Multiple foci of hepatic necrosis are seen less frequently. These lesions affect up to ~30% of the hepatic parenchyma, are pale, soft-to-firm, and demarcated by a thin red-to-purple halo. Thrombosis is a common feature in small and medium-sized veins. Affected blood vessels are located within, or adjacent to, the necrotic lesions, and both portal and hepatic branches are involved. 45 The rest of the liver is usually orange-tan, and may have a diffusely distributed, acinar pattern, suggestive of hypoxic degeneration (Fig. 5).21,43 Variable amounts of bright red blood clots may be found in the small and large intestine. There is splenomegaly, and the parenchyma has a gelatinous, purple appearance. The kidneys are dark brown to black (Fig. 6), friable, and may contain numerous petechiae. The urinary bladder is filled with dark red urine.43,45,52
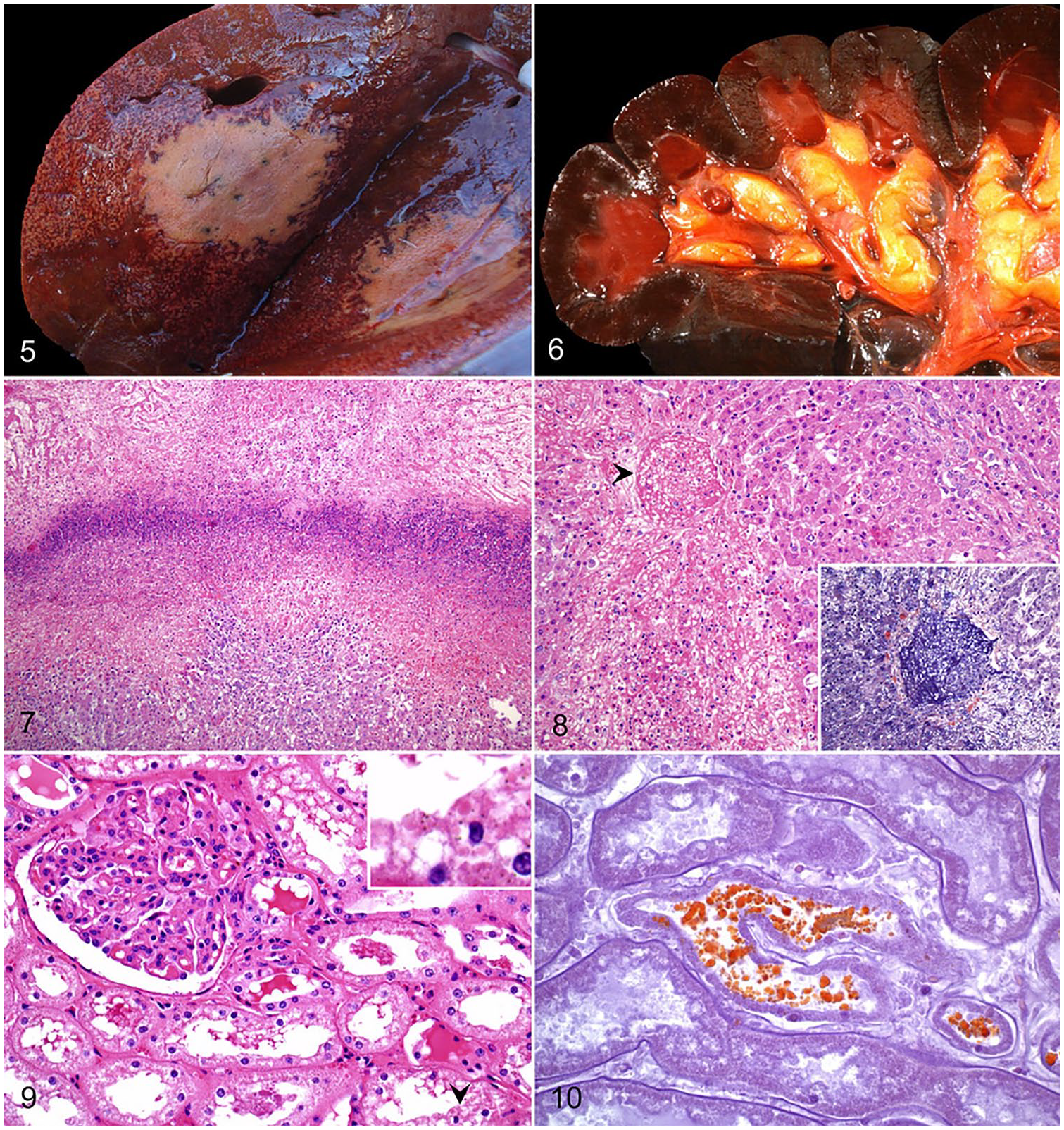
Bovine bacillary hemoglobinuria.
Microscopic lesions
The liver lesions in animals affected with BH are almost identical to those observed in INH (see above; Fig. 7). Thrombosis of hepatic vessels is a consistent feature in affected areas of the liver (Fig. 8), and it was traditionally thought to be the origin of the necrotic damage. However, hepatic infarction is unusual, probably the result of the dual hepatic blood supply, and thrombosis may be a consequence of the expanding necrotic damage, rather than its cause.16,45 Beyond the necrotic foci, areas of moderate-to-severe, probably hypoxia-induced, hepatic centrilobular necrosis are frequently seen. Caspase-3 activation seen in these areas suggests involvement of apoptosis as a mechanism of cell death. 45 As previously described for INH, microscopic changes associated with liver fluke infestation may also be observed.43,54,55
Acute necrosis of proximal and distal convoluted tubules is a common feature in BH and is probably associated with the hypoxia of intravascular hemolysis and subsequent anemia seen in these cases. There is also Okajima-positive hemoglobin pigment in tubular epithelial cells and luminal hemoglobin casts (Figs. 9, 10). 45 In the spleen, sinusoidal congestion and multifocal necrosis, infiltrated with degenerate and viable neutrophils, can be seen throughout the white and red pulp. In addition, signs of vascular damage, in the form of congestion and hemorrhages, are frequently seen in other tissues.45,52,78
Diagnosis
Given the many similarities between BH and INH, diagnostic criteria for these 2 diseases are concordant in many species. Epidemiologic information such as a history of non-vaccination, alkaline and poorly drained soils, history of fascioliasis, and clinical signs including sudden death, hemoglobinuria, and icterus, help facilitate a presumptive diagnosis, supported by gross and microscopic changes. The presence of a large focus of hepatic necrosis, typically single, together with Gram-stained impression smears containing numerous, large bacilli, is usually suggestive of BH or INH. However, the latter finding should be interpreted with caution given that these microorganisms are always present, and proliferate rapidly, as postmortem invaders. 71
As mentioned previously, although IHC for
Prevention
Preventive measures against BH are similar to those described above for INH, including the control and treatment of fascioliasis and other parasites, and the removal of dead animals from the premises. Vaccination with commercial bacterins or toxoids, in mono- and polyvalent preparations, is effective in reducing the incidence of the disease. In areas where the disease is more prevalent, the first 2 doses of vaccine, given 4–6 wk prior to the expected peak of occurrence, are recommended, in addition to the vaccination plan described for INH.43,72
Tyzzer disease
TD is an enterohepatic syndrome cause by
Etiology
Originally termed
Pathogenesis
The posited mode of infection in animals is by ingestion of
Epidemiology and clinical signs
Young animals are the most susceptible to TD, particularly when immunocompromised. Foals are at higher risk when born to mares < 6 y old versus foals born to older mares, suggesting that different colostral quality may be an additional risk factor.
23
In laboratory animals, the disease is mainly predisposed by immunosuppression, stress, high environmental temperatures, overcrowding, poor sanitation, changes in diet, and sulfonamide and corticosteroid administration.7,33 Fecal contamination of bedding with
The clinical presentation in cases of TD may vary across species but reflects hepatic and intestinal disease. Clinical signs include icterus, anorexia, weakness, depression, abdominal pain, watery diarrhea, dehydration, hypothermia, tachycardia, and recumbency.7,12 Neurologic manifestations, including ataxia, tremors, and head tilt, have been reported in a weaver bird and in free-ranging passerine birds.39,40 Sudden death, with no prior clinical signs, may occur in severe cases in rabbits and mice.7,8
Hematologic and biochemical findings reveal non-regenerative anemia, leukocytosis or leukopenia, metabolic acidosis, hypokalemia, hypoglycemia, hypoproteinemia, hyperbilirubinemia, and elevated alanine aminotransferase, AST, GGT, and sorbitol dehydrogenase activities in most domestic and laboratory species.12,24
Gross lesions
In all affected species, the disease is mainly characterized by hepatomegaly with numerous pale foci, 1–5 mm diameter, dispersed throughout the liver parenchyma (Fig. 11). Widely distributed icterus is present in most cases. 24 There is marked mural edematous thickening of the ileum, cecum, and colon, with ecchymoses and petechiae found on the serosal surfaces. The content of the cecum and colon is watery to gelatinous.3,33 Single or multiple, pale, round-to-linear foci may be observed in the myocardium. These lesions, however, are most frequently observed only microscopically. 24 The presence of lesions in liver, intestine, and heart constitute a diagnostic triad of lesions for TD, although their distribution is variable among different species.
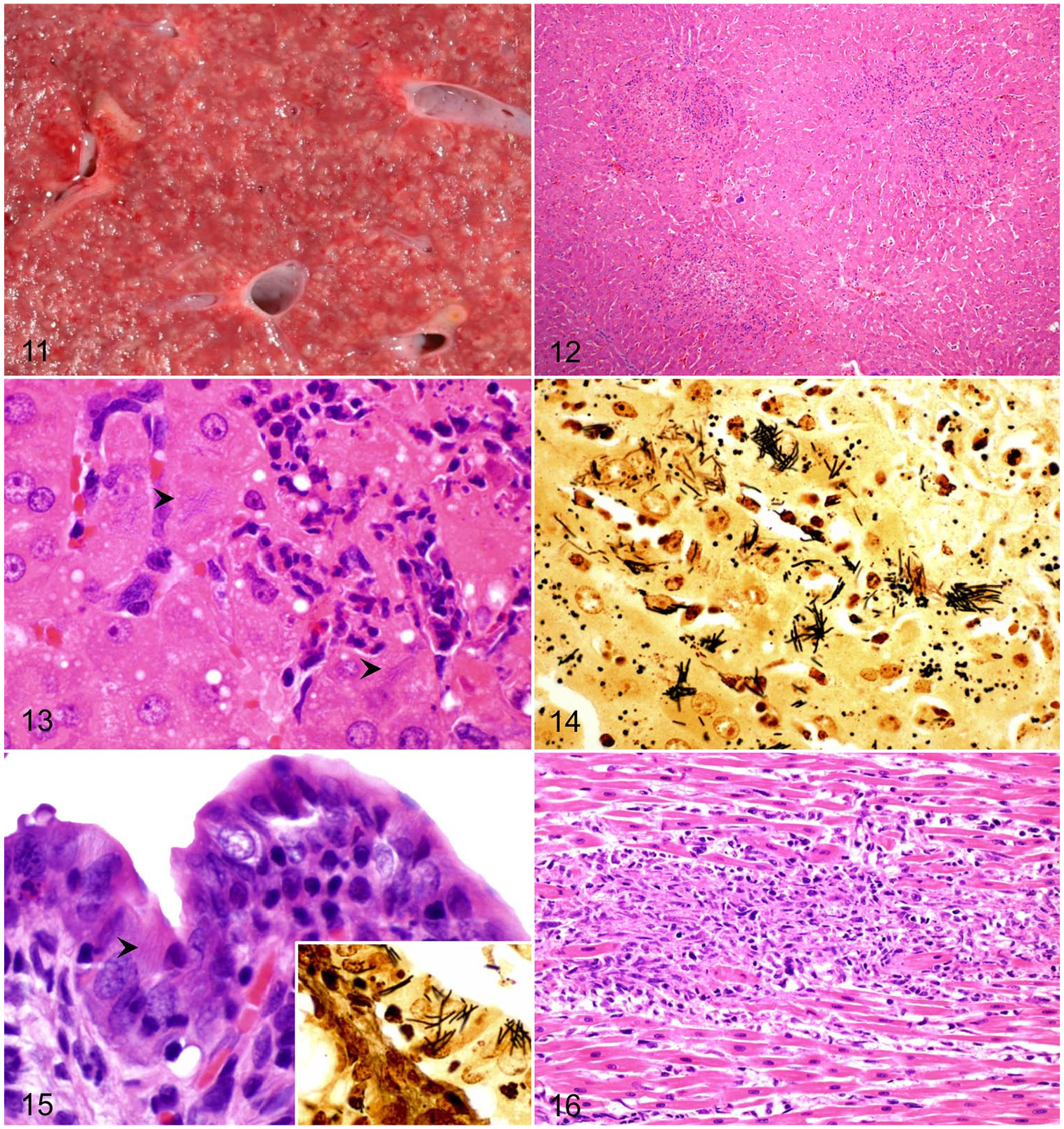
Tyzzer disease.
Microscopic lesions
The characteristic microscopic hepatic lesion in TD is randomly distributed, well-demarcated, focal coagulative necrosis, infiltrated with large numbers of degenerate and viable neutrophils (Fig. 12). Formation of granulomas, composed of aggregates of macrophages, with occasional fibrosis and mineralization, may occur in chronic cases. 24 Numerous filamentous bacilli are visible as bundles or crisscross patterns in the cytoplasm of viable hepatocytes at the margin of the necrotic areas (Figs. 13, 14).3,24,33
In young foals, hepatic lesions are most common, with minor involvement of the intestinal tract and myocardium; the latter 2 tissues are affected more frequently in rodents and lagomorphs. 74 The affected segments of intestine are transmurally necrotic, with submucosal edema and hemorrhage. The mucosa and lamina propria are infiltrated by variable numbers of lymphocytes, plasma cells, and neutrophils, and the crypts are dilated with necrotic debris.3,35 Intracytoplasmic filamentous bacteria can be seen in enterocytes (Fig. 15).24,35
When present, lesions in the heart are characterized by multifocal myocardial necrosis and infiltration by variable numbers of neutrophils, macrophages, and lymphocytes (Fig. 16), with intra-sarcoplasmic filamentous bacteria.13,24,63 On occasion, these lesions may progress to granulomatous myocarditis. 24
Microscopic changes in the central nervous system (CNS) have been reported in a few species including gerbils, 80 a weaver bird, 39 passerine birds, 40 and a marmoset. 84 CNS changes included regionally extensive areas of gliosis, neuropil rarefaction, and microabscess formation, mainly involving the cerebral cortex. At the periphery of these lesions, the characteristic filamentous bacteria may be found in neuronal perikarya.
Diagnosis
Diagnosis of TD relies on gross findings and histologic demonstration of hepatic, intestinal, and/or myocardial lesions. This is further supported by the presence of bacteria in the cytoplasm of infected cells. The bacteria are faintly stained in routine H&E sections; detection of
Prevention
Given that fecal–oral transmission of
Concluding remarks
INH, BH, and TD are the most prevalent clostridial hepatic diseases of animals and, although a presumptive diagnosis can be achieved by a combination of clinical, gross, and microscopic findings, confirmation relies on demonstration of the causative
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
