Abstract
Tyzzer disease (TD) is caused by
Tyzzer disease (TD) is caused by
The pathogenesis of TD in horses is not fully understood. The current belief is that ingestion of spores from the environment, most likely from feces of carrier animals, is followed by colonization of the intestine, absorption into the blood circulation, and infection of the liver and other organs.8,21 This mechanism is supported by a study in which TD was reproduced in foals by dosing them orally with feces from infected horses. 29
Although the literature describes a classic triad of necro-inflammatory lesions for TD affecting liver, heart, and colon, the organ distribution of lesions varies greatly among animal species.15,32,36 Most cases of TD in horses seem to affect the liver, and rarely the heart and colon.3,11,21,30,35 However, information about the distribution of lesions in horses with TD is scant and, to our knowledge, there are no detailed descriptions of the distribution of
Here we present a retrospective study of cases of TD in horses, in which we describe in detail the main gross and microscopic lesions with special emphasis on the distribution of
Materials and methods
We reviewed 25 equine cases with a diagnosis of TD submitted for autopsy between 1991 and 2015 to the San Bernardino laboratory of the California Animal Health and Food Safety (CAHFS) Laboratory System, University of California–Davis (Table 1). The diagnosis had been confirmed by the pathologist in charge of each case based on gross and microscopic lesions.
21
A postmortem examination had been performed on every case. Several ancillary tests were performed following CAHFS standard operating procedures. These included, for most cases, aerobic and anaerobic culture of the liver, spleen, lung, small and large intestinal content, ELISA for
Signalment, time of the year, and clinical history of 25 foals with Tyzzer disease.
F = female; M = male; NR = not reported.
Scrolls from archived (1991–2015) paraffin blocks of liver (
Results
Briefly, 24 of 25 foals where <45 d old; the remaining animal was 90 d old (Table 1). There were 12 males and 12 females; the sex was not recorded for one animal. Most animals were Quarter Horses; there were fewer Thoroughbred, Arabian, Paint, and Hanoverian horses. Nineteen of the cases occurred during spring; the remaining cases occurred in late winter or early summer. Of the 16 foals that were presented with acute disease, clinical signs included 1 or more of the following: diarrhea, fever, distended abdomen, depression, weakness, non-responsiveness, failure to nurse, labored breathing, or recumbency. The other 9 foals were found dead without clinical signs being observed.
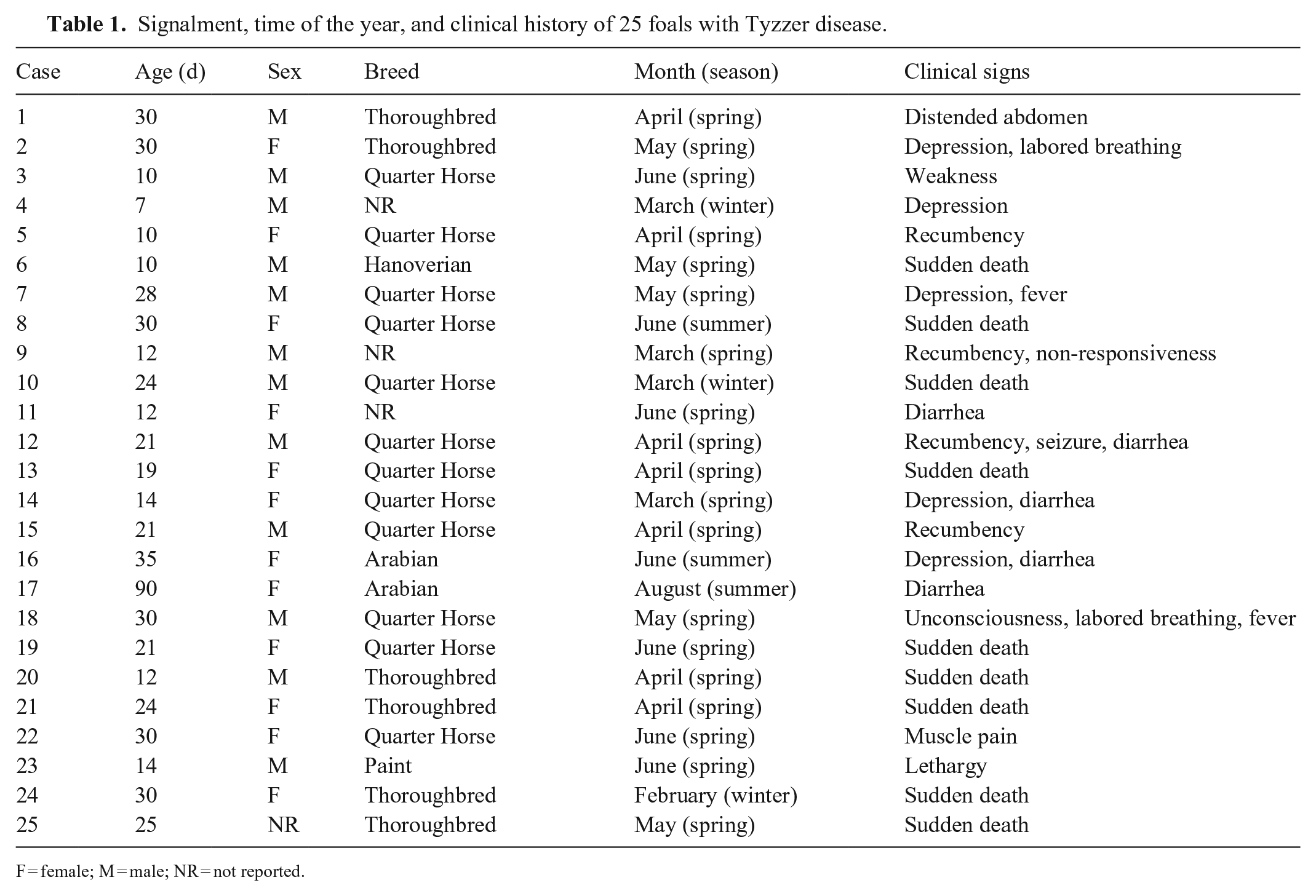
Gross changes were noted primarily in liver, heart, and intestine (Fig. 1; Suppl. Table 1). Briefly, most foals (16 of 25) had enlarged livers with rounded edges (Fig. 2), and a marked acinar pattern (18 of 25) characterized by multiple red-brown or white foci distributed randomly throughout the parenchyma (Fig. 3). Icterus (13 of 25) and serosal petechiation (7 of 25) were common features in various organs. The large intestinal mucosa, primarily colon, was usually diffusely red, and there was liquid or semi-liquid content within the large (Fig. 4) and small intestine. In the heart, subendocardial, and less frequently subepicardial, petechiae and ecchymoses (11 of 25) were observed (Fig. 5). A few animals (5 of 25) had pale foci in the myocardium. Pulmonary edema (9 of 25), splenomegaly (5 of 25), and lymphadenomegaly (5 of 25), mainly of hepatic and mesenteric lymph nodes, were observed occasionally.
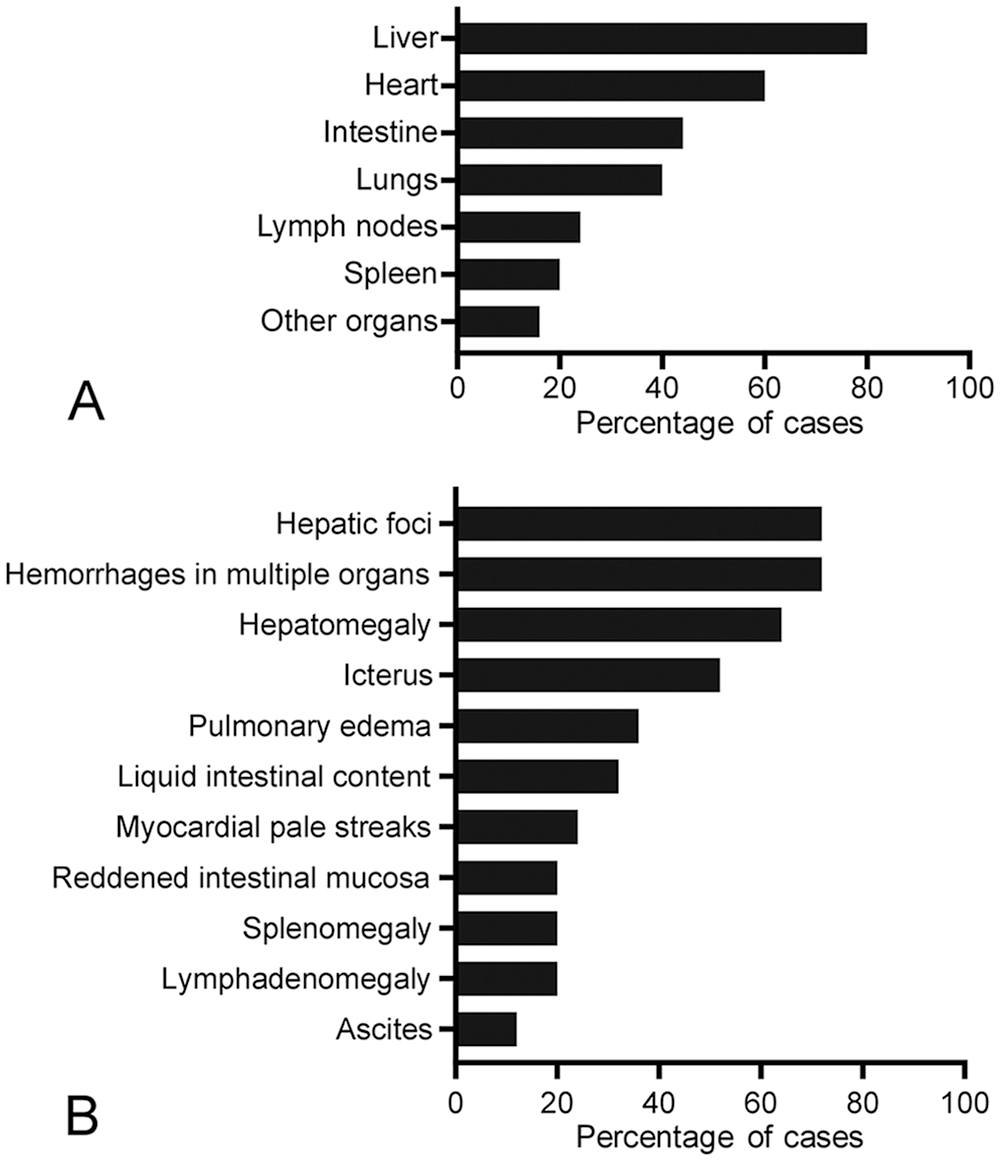
Gross findings in 25 foals with Tyzzer disease.
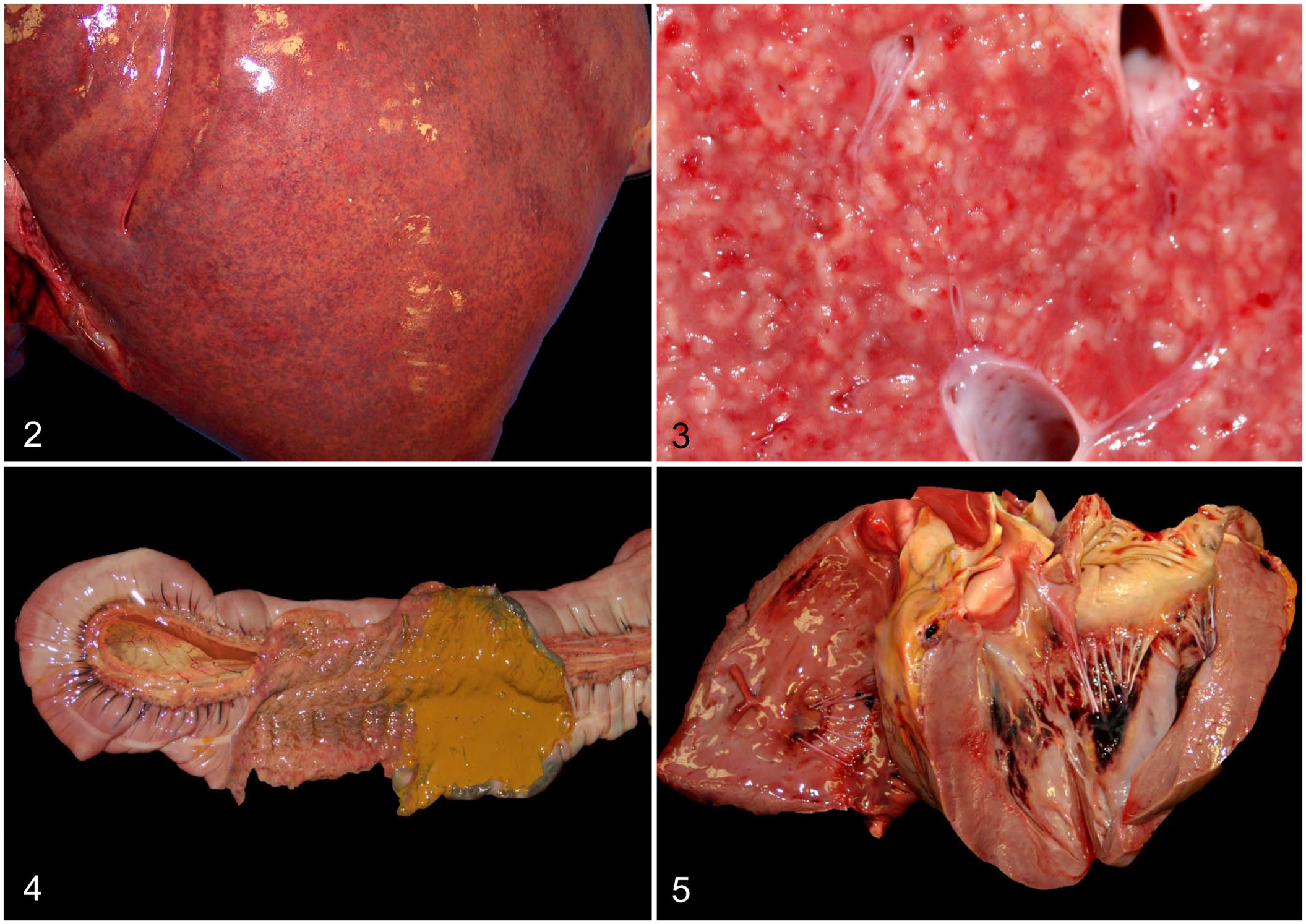
Macroscopic lesions in foals with Tyzzer disease.
Histologically, all foals (Fig. 6) had randomly distributed foci of hepatic necrosis, which often coalesced (Fig. 7; Suppl. Table 2). These foci were characterized by a center of coagulative necrosis admixed with fibrin, viable and degenerate neutrophils (Fig. 7), and occasionally, hemorrhage (7 of 25). The hepatocytes surrounding these foci were often swollen and had approximately round cytoplasmic vacuoles. A variable number of intracytoplasmic, and rarely interstitial, gram-negative filamentous bacilli that tended to form bundles were observed primarily in the hepatocytes at the margin of the necrotic foci (Fig. 7, inset). Steiner silver stain highlighted these filamentous bacteria in the cytoplasm of hepatocytes in all 24 livers stained with this technique (Fig. 8; Table 2).
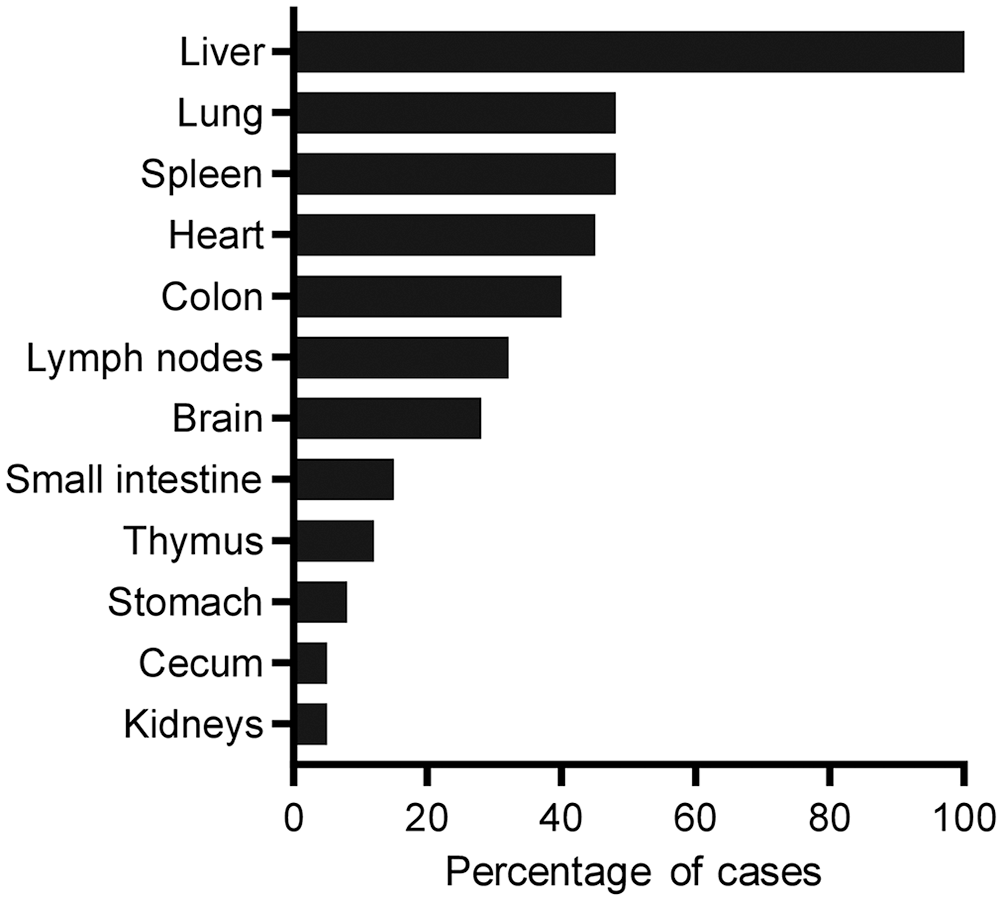
Distribution of microscopic lesions by organ system in 25 foals with Tyzzer disease.
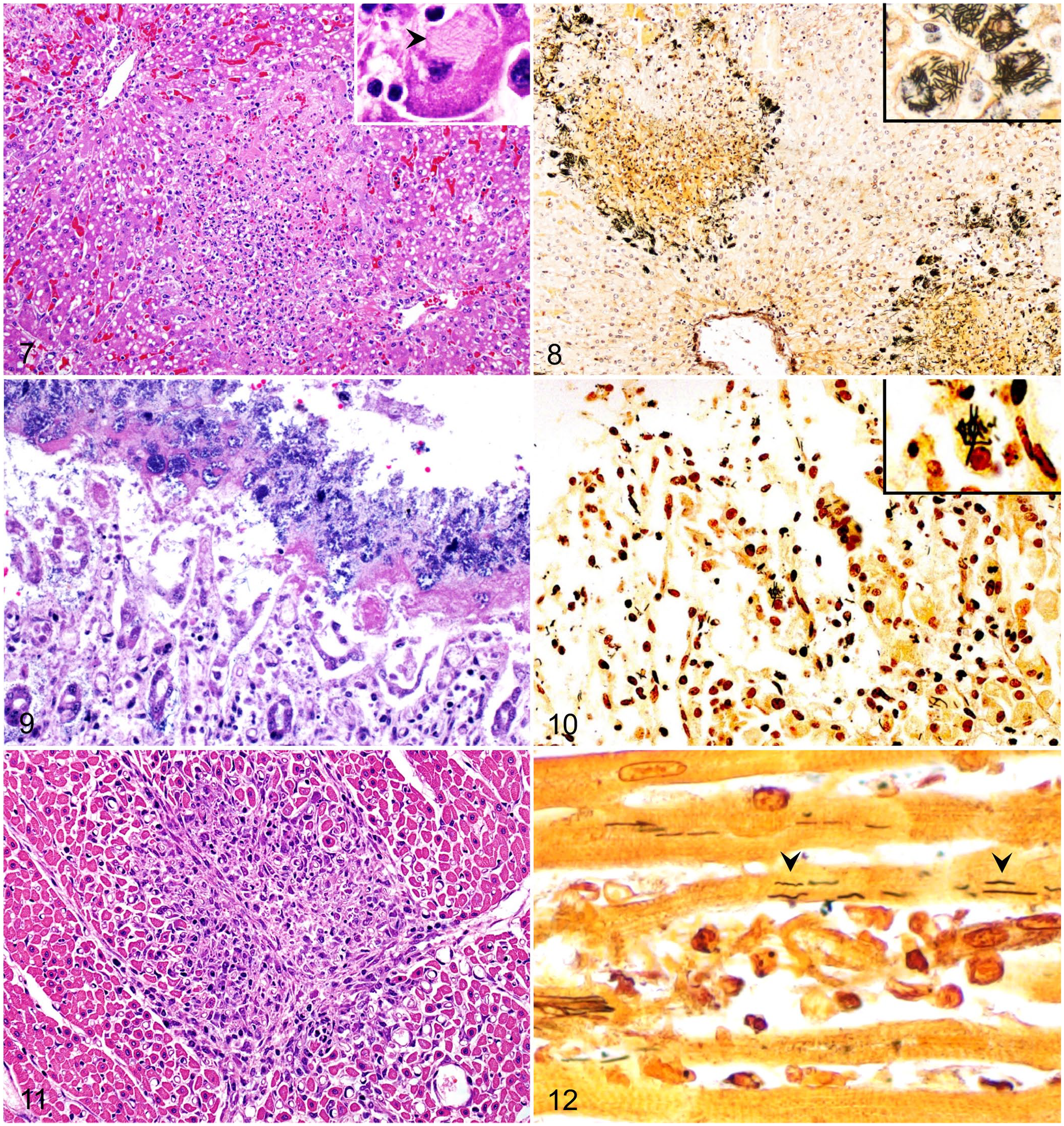
Microscopic lesions in foals with Tyzzer disease.
Steiner staining and molecular results on sections of liver, colon, and heart of 25 foals with Tyzzer disease.
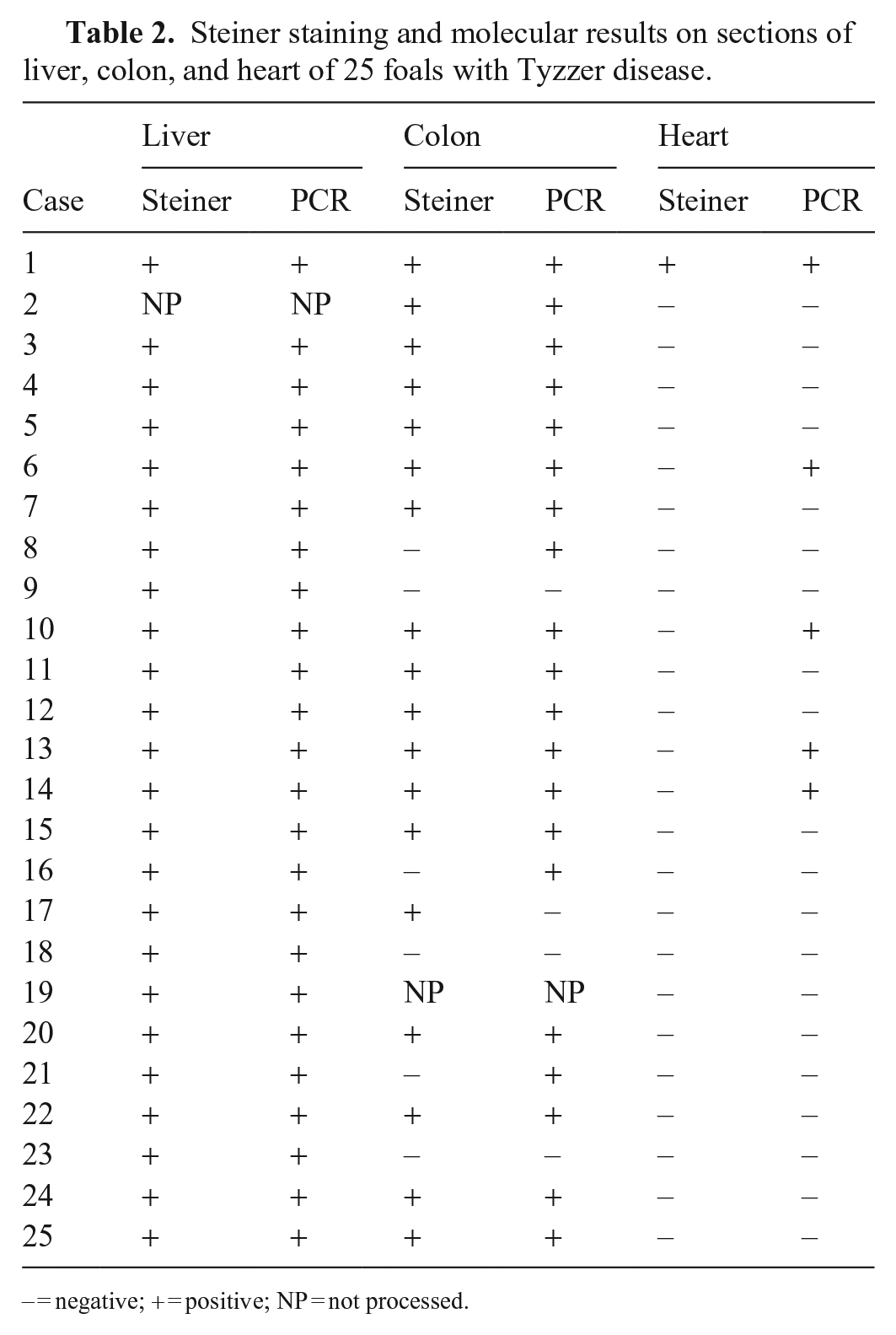
− = negative; + = positive; NP = not processed.
Necrotizing, lymphohistiocytic, multifocal-to-diffuse colitis with crypt necrosis of variable severity was observed frequently (10 of 25). The colonic mucosa was multifocally to diffusely necrotic and sloughed, and rarely covered by a pseudomembrane of cellular debris, fibrin, leukocytes, and mixed bacteria (3 of 25; Fig. 9). The lamina propria was usually infiltrated by moderate-to-abundant lymphocytes and histiocytes, and lesser numbers of plasma cells and neutrophils. In a few cases, the lamina propria was distended by clear edema and was hypocellular (4 of 25). Multifocally, single-to-multiple dilated crypts, with abundant necrotic debris in the lumen and lined with attenuated and/or necrotic epithelium, were observed. The submucosa had scattered, multiple aggregates of lymphocytes, plasma cells, and macrophages. Increased numbers of leukocytes within blood vessels were observed frequently. Fibrin thrombi in the lumen of mucosal and submucosal blood vessels (2 of 25) and lymphocytolysis of gut-associated lymphoid tissue (2 of 25) were seen rarely. Faintly basophilic intracytoplasmic filamentous bacilli were rarely observed in epithelial cells of intestinal crypts or in superficial enterocytes in 4 foals. Similar bacilli were observed in 18 of 24 colon sections stained with Steiner silver stain (Fig. 10; Table 2).
Eight of 25 foals examined had necrotizing myocarditis characterized by foci of myofiber degeneration and necrosis, with vacuolation of fibers, segmental loss of cytoplasm and striations, and infiltration of neutrophils, lymphocytes, and histiocytes (Fig. 11). The myocardium of 2 of these foals had focal fibrosis. Subendocardial and subepicardial, multifocal or focally extensive hemorrhages were observed in 7 foals. Intracytoplasmic filamentous bacteria were observed only in the myocardiocytes of 1 of 24 cases stained with Steiner silver stain (Fig. 12; Table 2). Only 4 foals had lesions in all 3 organs usually considered part of the Tyzzer triad of lesions (i.e., necrosis in liver, heart, and colon).
Other microscopic lesions included: interstitial pneumonia (
Of the 14 animals that were tested for heavy metals, 8 had low marginal selenium concentrations, 3 had high copper concentrations, and 3 had low zinc concentrations. Other heavy metals were within reference intervals. No aerobic or anaerobic bacterial pathogens were isolated from any of the tissues cultured. Fluorescent antibody testing for
Discussion
Hepatic lesions were present in all 25 horses in our study; enteric and myocardial lesions were present in only a few of these animals. Our results indicate that necrotic hepatitis is the hallmark of TD in horses, and that the triad of lesions characteristic of TD in several animal species should not be considered constant in horses. The fact that some horses did show this triad of lesions, whereas others did not, may be related to variations in the virulence of different
Our results are consistent with previous reports in which the liver was the organ most frequently affected by TD in horses. Although the classic triad of lesions was observed in 4 of 25 foals,
Two foals in our study had neurologic clinical signs, and one of them had brain lesions compatible with hepatic encephalopathy 12 ; the other foal had choroiditis of undetermined etiology, which may have been responsible for its neurologic signs. Rare cases of encephalitis have been reported in a bird, a monkey, and a gerbil with TD.18,19,33,37 This was, however, not the case in any of the horses in our study.
Although TD affects foals primarily within the first 45 d of life,29,32 cases may occur in foals up to 90 d old. This was the case in our study, in which the majority of foals were <45 d old, and only one 90-d-old animal was reviewed.
Although we could not retrieve the breed distribution of the CAHFS foal caseload, there are no reports suggesting breed predisposition to TD. Most foals autopsied at CAHFS during the study period were received during the spring. However, the higher incidence of TD observed in the spring likely simply reflects the seasonality of foal submissions.
Immunosuppression has been suggested as a predisposing factor; this was seen in kittens, dogs, a panda, and a human patient.6,13,16,21,28 In 13 foals of our study, lymphopenia (
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211031213 – Supplemental material for Clostridium piliforme infection (Tyzzer disease) in horses: retrospective study of 25 cases and literature review
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211031213 for
Footnotes
Acknowledgements
We thank M. Juliann Beingesser and Jose Polanco Calderon for excellent technical assistance with histotechnology.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study was funded by the California Animal Health and Food Safety Laboratory, UCDavis.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
