Abstract
Reports of neoplasia in captive reptiles are becoming more frequent; however, there is still scarce knowledge of the occurrence of neoplasia in wild reptiles. A wild black rat snake (Pantherophis alleghaniensis) was presented to the Zoological Medicine service of the University of Georgia’s Veterinary Teaching Hospital with a 3 cm in diameter solid mandibular mass that was partially ulcerated. Radiographically, the mass was radiopaque with small bone spicules and partial osteolysis of the adjacent mandible. Histologic examination of the mass revealed a neoplasm composed of cuboidal to polygonal cells arranged in islands, anastomosing cords, and trabeculae of pseudostratified epithelium with a palisading peripheral layer of densely packed columnar cells with cytoplasmic clearing. The neoplastic tissue was separated from the mesenchyme by a prominent band of fine collagen. Neoplastic cells were positive for cytokeratin and negative for smooth muscle actin. Electron microscopy highlighted the presence of tonofilaments and microvilli. These findings led to the diagnosis of ameloblastoma, an odontogenic epithelial tumor known to occur in humans and most veterinary species.
Bacterial stomatitis, parasitic infection (renifers), fungal dermatitis, trauma, and neoplasia are some common differential diagnoses for oral lesions in snakes. 9 Retrospective studies of zoological collections across the United States document rates of neoplasia in captive snakes from 2.9% to 23.1%.5,18 Both epithelial and mesenchymal neoplasms, specifically squamous cell carcinoma and fibroma, have been documented in retrospective studies of snakes at the National Zoological Park and the Sacramento Zoo.5,14 Additionally, squamous cell carcinoma was reported in a wild-caught eastern diamondback rattlesnake (Crotalus adamanteus) on public display in North Carolina. 1 The mass presented herein is most consistent with an ameloblastoma, an odontogenic tumor that, to our knowledge, has not been previously reported in reptiles. Of the odontogenic tumors, ameloblastoma is one of the most commonly reported in humans and veterinary species. In humans, ameloblastomas comprise 1% of head and neck tumors in the United States and Europe, and make up 11.7% of odontogenic tumors, with 80% of ameloblastomas occurring as mandibular tumors.3,12 In veterinary species, odontogenic tumors have been documented extensively in dogs and less commonly in cats.6,7 In nonmammalian species, ameloblastomas have been characterized in captive-reared Chinook salmon (Oncorhynchus tshawytscha). 8
A 1.22 kg, wild, adult black rat snake (Pantherophis alleghaniensis), with a marked oral swelling, was brought to the University of Georgia Veterinary Teaching Hospital, Zoological Medicine Service (Athens, Georgia) subsequent to being found by a good samaritan. On physical examination, a firm, 3 cm in diameter, irregularly spherical mass was noted on the right mandible. Oral examination also revealed stomatitis localized to the right lower arcade. The patient was induced with an intramuscular injection of 10 mg/kg of ketamine a and 0.1 mg/kg of dexmedetomidine hydrochloride, b and anesthesia was maintained with isoflurane. c Lactated Ringer solution (LRS) d was administered at 5 mL/kg intracoelomically for maintenance. Radiographs of the head (4 views) were taken to further characterize the mass. The radiographs revealed an ill-defined, lobular mass with amorphous intralesional mineral arising from the soft tissues ventrolateral to the right mandible. A mild amount of smooth periosteal reaction was present at the ventral margin of the right mandible, adjacent to the mass. The ventral margins of the mass were lobular and irregular. The remaining osseous and soft tissue structures appeared to be normal (Fig. 1A).
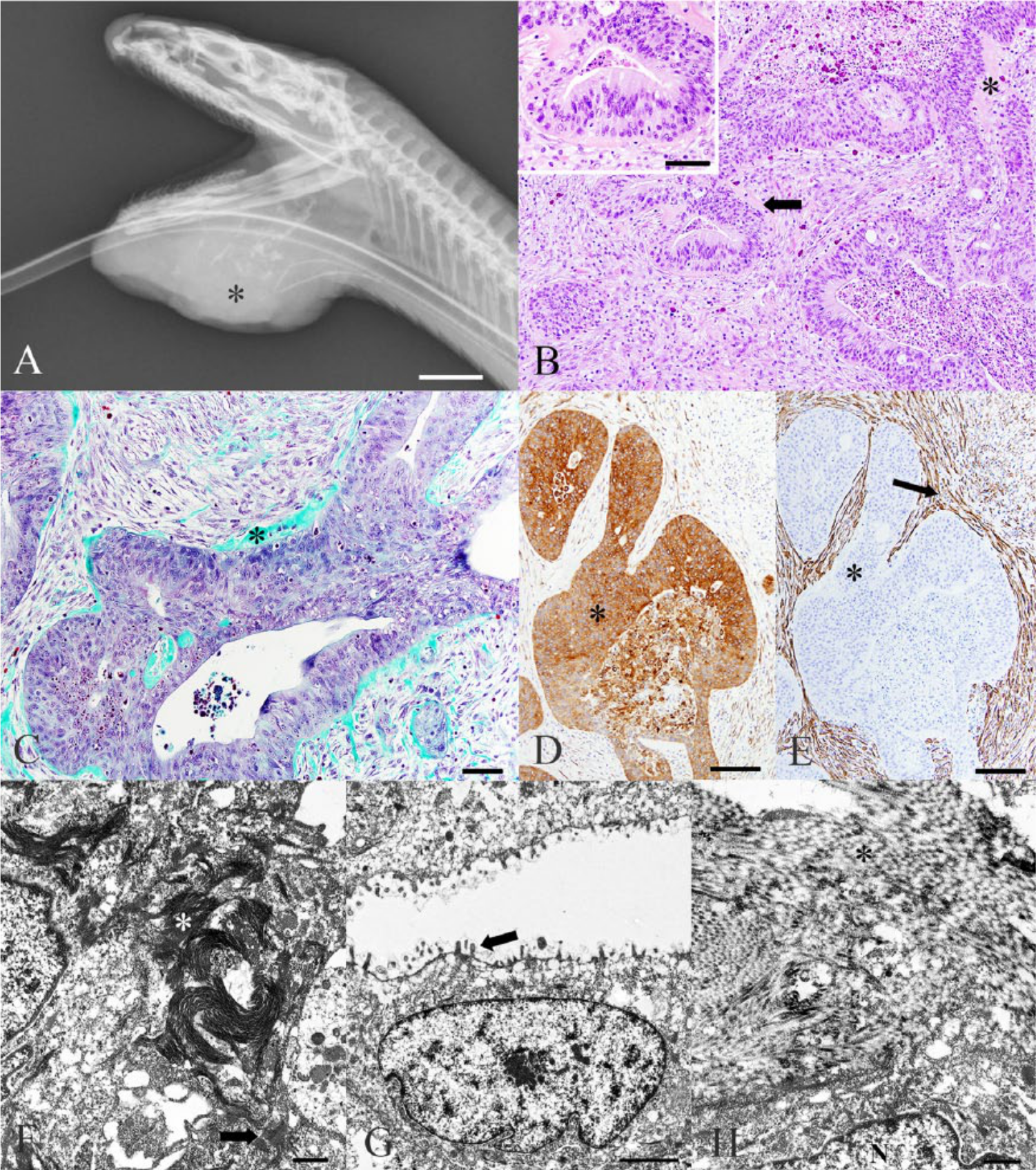
Radiographic, histologic, immunohistochemical, and ultrastructural features of an oral neoplasm from a wild black rat snake (Pantherophis alleghaniensis).
Two, 1-cm long biopsies were taken using a 6-mm biopsy punch. e Purulent exudate was noted on lancing the mass. The wound was cleaned and flushed with chlorhexidine gluconate f and 0.9% sodium chloride solution, g and left open to allow purulent material to drain. The biopsy samples were preserved in 10% neutral buffered formalin h and routinely processed and stained with hematoxylin and eosin.
Treatments received during the patient’s 3 days of hospitalization included the following: tramadol i (10 mg/kg every 48 hr) orally, meloxicam j (0.2 mg/kg every 48 hr) intramuscularly, and ceftazidime k (20 mg/kg every 72 hr) intramuscularly. Additionally, the wound was cleaned and flushed with chlorhexidine–LRSf,d solution twice a day.
Punch biopsy results indicated a highly infiltrative neoplasm with secondary bacterial infection. Because of the poor prognosis, the animal was euthanized. A full postmortem examination was not performed; however, a postmortem tissue sample of the mandibular mass (1.5 cm × 1.0 cm × 0.5 cm) was collected and preserved in 10% neutral buffered formalin. h The sample was quartered, and the 4 sections were routinely processed for histology. Selected sections were also stained with Masson trichrome, Gomori trichrome, and periodic acid–Schiff (PAS) reaction. Immunohistochemistry was performed using polyclonal antibodies against vimentin, l pan-cytokeratin, m smooth muscle actin, n and Ki-67 o protein. A selected paraffin-embedded section of the mass was processed for ultrastructural examination. Ultrathin sections were mounted on copper grids, stained with uranyl acetate and lead citrate, and examined and photographed using transmission electron microscopy (TEM).
Light microscopy revealed a moderately cellular, well-demarcated, unencapsulated, infiltrative neoplasm composed of cuboidal to polygonal cells arranged in islands, anastomosing cords, and trabeculae of pseudostratified epithelium with a palisading peripheral layer of densely packed columnar cells (Fig. 1B). This neoplastic population was embedded in a matrix of spindloid mesenchymal cells separated by moderate amounts of myxomatous material (Fig. 1B). Neoplastic epithelial cells contained moderate amounts of eosinophilic, occasionally vacuolated cytoplasm and exhibited basilar, cytoplasmic clearing, with an apically located, oval nucleus containing stippled chromatin and a single magenta nucleolus. The mitotic rate averaged 5 per one 400× field, and anisocytosis and anisokaryosis were mild. Over the apical surface of the neoplastic epithelium, and sometimes separating epithelial and mesenchymal type cells, there was a moderate amount of eosinophilic homogeneous material that stained magenta by PAS reaction, and light-green (positive for collagen) with Gomori trichrome and blue (positive for collagen) with Masson trichrome (Fig. 1C). The neoplastic cells occasionally formed cystic structures variably filled with necrotic debris. Along one side of the mass, there was a large amount of necrotic debris admixed with numerous degenerated heterophils, occasional colonies of bacilli, and hemorrhage.
The neoplastic population had marked, diffuse, positive cytoplasmic staining with pancytokeratin, m and the mesenchymal component of the mass showed marked, diffuse, positive cytoplasmic staining with smooth muscle actin n (Fig. 1D, 1E), suggesting that the majority of mesenchymal cells were composed of myofibroblasts. Ki-67 o antibodies variably stained the cytoplasm and nuclei of the neoplastic population as well as the stroma, blood vessels, and oral mass mesenchymal cells, providing no meaningful information to further characterize the neoplastic population. Sections of the mandibular mass were diffusely negative for vimentin l staining, while positive control mammalian tissues exhibited the expected cytoplasmic staining, indicating a lack of species cross-reactivity. TEM of the neoplastic epithelial cells revealed numerous apical microvilli on cells lining cystic spaces and occasional desmosomes and tonofilaments (Fig. 1F, 1G). The extracellular matrix was composed by numerous parallel bundles of electron dense, banded, protein fibrils (collagen, Fig. 1H).
The homogenous eosinophilic material separating epithelial and mesenchymal populations, as well as along the apical border of the neoplastic cells, and within cystic spaces, stained positively for collagen (blue with Masson trichrome, and green with Gomori trichrome). 4 PAS reaction with this homogenous eosinophilic material (magenta) also supported that it was not consistent with tooth material such as osteoid, dentin, or enamel. Magenta staining by PAS reaction is characteristic of tissues containing a high proportion of carbohydrates indicating that this material was more likely thickened basement membrane or periodontal ligament. The characterization of this eosinophilic material as basement membrane or periodontal ligament led to consideration of ameloblastoma, an odontogenic tumor that exhibits many of the cellular characteristics seen in this mass. The diagnosis of ameloblastoma is supported by the demonstration that this tumor was not producing enamel matrix or tooth material. 12 Immunohistochemistry for smooth muscle actin n did not stain the neoplastic population; however, it did stain the surrounding vessels and myofibroblasts. In humans, this stain has been used as a marker for ameloblastic carcinoma versus ameloblastoma. The lack of stain uptake in the neoplastic population may indicate that this neoplasm is not malignant; however, it must also be taken into consideration that ameloblastic carcinomas in reptiles may have different staining characteristics than those in humans.2,19 Use of immunohistochemistry and special stains was highly successful as it not only demonstrated several important characteristics of the neoplasm and surrounding population, but it also demonstrated cross-reactivity of many of these stains in reptiles. Vimentin, l however, did not appear to cross-react in this sample, supporting previous claims that this antibody does not cross-react in snakes. 13 Ki-67 o immunohistochemistry was used to determine if the neoplastic population had a greater proliferation rate when compared to the adjacent normal tissues; however, it stained the neoplastic and surrounding cell populations nonspecifically, thus failing to provide information that could support the diagnosis of malignancy of the epithelial component.
Additional characterization of the neoplasm by electron microscopy (EM) demonstrated similarities to previous reports of ameloblastomas in other species.15,17 Examination by EM of the previously discussed homogenous eosinophilic material was confirmed to be collagenous and therefore not enamel matrix. The presence of few but distinct desmosomes on the contact surfaces of the palisading neoplastic cells confirmed the epithelial origination of the neoplastic population and is described in EM evaluations of ameloblastomas.7,17 Visualization of tonofilaments within the cytoplasm of the neoplastic population is consistent with previous reports of both ameloblastoma as well as squamous cell carcinoma. The microvilli that were seen on the apical borders of the neoplastic cells are also a frequently described characteristic of ameloblastomas. 17
The histologic characteristics of this mass showed similarities to both ameloblastoma and acanthomatous ameloblastoma (or acanthomatous epulis). Determination of ameloblastoma versus acanthomatous ameloblastoma, a more specific type of ameloblastoma characterized by increased invasiveness, is not possible in this case. The literature on acanthomatous ameloblastoma is limited to mammalian species, including humans and dogs, and less commonly cats and rabbits.6,10 The invasiveness of this tumor, however, along with the osteolysis and possible presence of periodontal ligament, led to consideration of further classifying the tumor as acanthomatous ameloblastoma.6,11,12,16 Although ameloblastoma is one of the most commonly reported odontogenic tumors in humans, their overall prevalence in domestic animals is low, and to our knowledge, has not been previously reported in a reptile.
Footnotes
Acknowledgements
We would like to acknowledge the staff of the Histology Laboratory of the University of Georgia, College of Veterinary Medicine (Athens, Georgia) for their technical support with histology, immunohistochemistry, and electron microscopy, as well as Faustine Blanville for her help with the treatment and management of this case. We would also like to thank Drs. Elizabeth Howerth, Rita McManamon, Nicole Gottendenker, and Mike Garner for their guidance and opinions on this case.
Authors’ contributions
JR Comolli, HMH Olsen, M Seguel, RW Schnellbacher, SJ Divers, and K Sakamoto contributed to conception and design of the study. M Seguel, SJ Divers, and K Sakamoto contributed to acquisition, analysis, and interpretation of data. JR Comolli, HMH Olsen, RW Schnellbacher, and AJ Fox contributed to acquisition of data. JR Comolli drafted the manuscript. HMH Olsen, M Seguel, RW Schnellbacher, AJ Fox, SJ Divers, and K Sakamoto critically revised the manuscript. K Sakamoto gave final approval and agrees to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Ketaset, Boehringer Ingleheim Vetmedica Inc., St. Joseph, MO.
b.
Dexdomitor, Orion Corp., Espoo, Finland.
c.
IsoFlo, Abbott Laboratories, North Chicago, IL.
d.
Abbott Laboratories, North Chicago, IL.
e.
Miltex Inc., York, PA.
f.
Hibiclens, Mölnlycke Health Care US LLC, Norcross, GA.
g.
Abbott Laboratories, North Chicago, IL.
h.
Cancer Diagnostics Inc., Morrisville, NC.
i.
Amneal Pharmaceuticals of NY, Hauppauge, NY.
j.
Loxicom (5 mg/mL), Norbrook Laboratories Limited, Newry, Co. Down, Northern Ireland.
k.
TAZICEF, Sandoz GmbH for Hospira Worldwide Inc., Lake Forest, IL.
l.
BioGenex, Fremont, CA.
m.
Pancytokeratin (AE1/AE3), Cell Marque, Rocklin, CA.
n.
Cell Marque, Rocklin, CA.
o.
Cell Marque, Rocklin, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The publication of this article was financially supported by the Department of Pathology, College of Veterinary Medicine, University of Georgia.
