Abstract
Inflammation of the blood vessel wall has been reported infrequently in dogs, and it may occur without apparent cause (primary vasculitis) or as a pathologic reaction to a range of initiating insults (secondary vasculitis). The aims of our study were to report histologic, clinical, and survival data from a large series of cases with primary and secondary vasculitis, and to compare the clinical parameters and outcome data between groups. Clinical data was collected retrospectively from the medical records of 42 client-owned dogs with a histologic diagnosis of primary or secondary vasculitis, and follow-up information was obtained. Cases were grouped according to clinical and histologic descriptors, and biochemical, hematologic, and survival data was compared between groups. Several forms of primary vasculitis were observed, and vascular inflammation was observed in conjunction with numerous other diseases. Female dogs were more likely to develop primary vasculitis, and serum globulin concentration was greater in dogs with primary vasculitis compared to those with underlying disease. All dogs with primary vasculitis of the central nervous system died or were euthanized shortly after presentation, but other forms of primary vasculitis could be managed effectively. In conclusion, presentation of clinical cases in this series was variable, and there did not appear to be well-defined vasculitic syndromes as described in people.
Introduction
Vasculitis refers to inflammation of the blood vessel wall that may develop without apparent cause (primary vasculitis), or in response to a range of initiating insults (secondary vasculitis). 19 Vasculitis has characteristic histologic features, including infiltration of the vessel wall by inflammatory cells and development of fibrinoid change, 6 but little is known in dogs of the initiating events and pathologic mechanisms that result in these changes.
Primary systemic vasculitides are widely reported in human medicine, and a nomenclature system has been formulated to describe the typical features of several distinct forms of the disease. 14 Such vasculitides were traditionally classified according to histological criteria, such as the size of affected vessels and the nature of the inflammatory infiltrate. More recent descriptions of inflammatory vascular disease also take into account epidemiologic factors and serologic markers of autoimmune disease, including antineutrophil cytoplasmic antibodies (ANCA). Clinical manifestations of primary vasculitides in humans depend on the organ(s) affected and the severity and chronicity of vascular changes. 33
There are few defined syndromes in veterinary medicine that are referable to primary vasculitis, but these include the systemic necrotizing polyarteritis syndrome described in colonies of laboratory Beagle dogs, 12 pulmonary arteritis,26,31 and cutaneous vasculitis.18,22 Two previous reports17,27 described cases with clinical or histologic features reminiscent of the human primary vasculitides Wegener’s granulomatosis and angiitis of the central nervous system (CNS), but the relative frequencies of these syndromes have not been clearly established.
Secondary vasculitis has been reported in association with a wide range of infectious, inflammatory, and immune-mediated diseases in animals and humans. 19 Of these, reported causes include autoimmune diseases such as systemic lupus erythematosus (SLE), 21 infection with Leishmania spp., 24 Rickettsia spp., 15 Angiostrongylus vasorum, 9 and Dirofilaria immitis, 30 and exposure to therapeutic products such as carprofen 20 and meloxicam. 23
The aims of the current study were to describe the histologic and clinical findings from a large series of dogs with vasculitis and to compare these with features reported in human vasculitides. A secondary aim was to determine whether primary forms of vasculitis occurred with increased frequency in dogs of any breed, sex, or age group.
Materials and methods
Data collection
The free text of the clinical records of a veterinary tertiary referral hospital was searched for cases that had a histologic description of vasculitis, derived either from a biopsy sample or from autopsy examination. Affected animals were presented over a period of 7 years, between January 2004 and October 2011. All samples were reviewed by a single board-certified pathologist (SL Priestnall) for the purpose of our study. Cases were diagnosed with vasculitis if fibrinoid change was observed, with or without infiltration of leukocytes into the vessel wall. Fibrinoid change was defined by the presence of eosinophilic, homogeneous, nonstructured, hyaline or fibrillar material within the vessel wall, with or without pyknotic nuclei of either endothelial or smooth muscle cells.
For each case, the following data was collected: signalment; types of vessel affected with vasculitis and types of infiltrating leukocytes (if any); results of hematologic and serum biochemical analysis of blood samples and any other clinicopathologic tests undertaken; and final histologic or clinical diagnosis. Referring veterinarians were contacted by telephone to obtain follow-up data.
A control population (n = 120) was assembled by randomly selecting dogs presented to the same institution over the same time period. Age, breed, and sex were also recorded for these patients. The clinical records of these dogs were studied to ensure that they did not have a histological diagnosis of vasculitis, nor clinical signs that could have been suggestive of vasculitis, nor were diagnosed with an infectious, autoimmune, or neoplastic disease that has been associated with vasculitis previously.
Statistical analysis
All statistical analyses were performed using a commercial software program, a with a significance level of ≤0.05. The probability that animals with primary vasculitis belonged to particular breeds, age groups, or sex was assessed using logistic regression. Cases were stratified into 4 balanced age groups (0–2.0 years, 2.1–5.0 years, 5.0–9.7 years, and 9.8–17.0 years) and according to sex (male and female). Breeds were grouped into 6 categories according to a classification system based on published data describing the extent of haplotype sharing between canine breeds. 32 The following groups were used: mixed breeds; sight hounds and herding dogs; retrievers, small terriers, and mastiff-like dogs; working dogs; toy dogs, spaniels, and scent hounds; and ancient breeds (Supplementary Table 1 available at http://vdi.sagepub.com/content/by/supplemental-data). Only 1 dog among the control population belonged to an ancient breed, and this animal was excluded from analysis of breed predisposition. Age, breed, and sex were entered individually as categorical variables with case or control as the binary outcome.
Clinical variables were assessed for normality by visual inspection of histograms and use of the Shapiro–Wilks test. Normally distributed variables were compared using Student t tests, whereas nonnormally distributed continuous variables were compared with Mann–Whitney U tests or Kruskal–Wallis tests. Categorical variables were compared with chi-square or Fisher exact tests.
Literature search
The PubMed, CAB abstract, and ISI Web of Science databases were searched for entries using the following algorithm: (arteritis OR vasculitis OR angiitis OR Takayasu OR Wegener OR Kawasaki) AND (dog OR canine), and the reference lists of relevant articles were also reviewed to identify further records of interest. The search was conducted on April 7, 2014. Abstracts were scanned by one of the authors (JW Swann) to identify those that described cases of primary or secondary vasculitis diagnosed on the basis of histologic examination. Data was abstracted in tabular format so that the histologic and clinical classification schemes described above could be applied. Articles were excluded if they were not published as complete reports in a peer-reviewed journal or if they were published before 1960.
Results
Descriptive statistics
Vasculitis was identified on histologic examination of samples from 42 dogs, of which 9 (21.4%) were intact males, 11 (26.2%) were neutered males, 6 (14.3%) were intact females, and 16 (38.1%) were spayed females. Twenty-six different breeds were represented, of which the 4 most common were Labrador Retriever (n = 5), German Shepherd Dog (3), Weimaraner (3), and Staffordshire Bull Terrier (3). The mean age at first presentation was 6.2 years (standard deviation [SD] = 3.5, standard error [SE] = 0.5).
Among the control dogs, the mean age was 5.4 years (SD = 4.3, SE = 0.4), with similar numbers of intact male (n = 35, 29.2%), neutered male (37, 30.8%), and spayed female dogs (33, 27.5%), and a smaller number of intact females (15, 12.5%). Forty-six different breeds were represented, of which the 4 most common were Labrador Retriever (n = 10), Boxer (9), Cavalier King Charles Spaniel (6), and West Highland White Terrier (6).
Twenty cases (47.6%) were diagnosed antemortem after analysis of biopsy samples, and the remaining 22 cases were diagnosed after postmortem examination. Over the same time period, 1,358 canine autopsies were performed, and 14,192 canine biopsies were analyzed at the same diagnostic laboratory; vasculitis was detected in 1.62% of cadavers and 0.16% of surgical biopsies. Of the cases that underwent autopsy examination, 14 (58%) showed evidence of vasculitis in 2 or more organs, whereas only 1 organ or tissue was affected in the remaining 8 cases.
Description of clinical cases
Eleven cases were diagnosed with primary vasculitis after diagnostic tests excluded initiating factors or concurrent diseases. In 6 cases, vascular changes were solely observed in the CNS, resulting in some combination of acute onset paresis, spinal hyperesthesia, cranial nerve deficits, and/or altered mentation (Supplementary Table 2). Breeds affected included Weimaraner (n = 2), Boxer (1), Miniature Schnauzer (1), lurcher (1), and cross-breed (1). Autopsy examination revealed that blood vessels of the brain were affected in 1 case, the spinal cord in 2 cases, and multiple parts of the CNS in 3 cases. Complete blood cell counts, serum biochemical profiles, and imaging studies did not reveal underlying causes for vasculitis in any of these dogs. Blood samples were also submitted for measurement of serum antibodies directed against Neospora caninum (n = 4), Toxoplasma gondii (3), Ehrlichia spp. (2), Leishmania spp. (1), Borrelia burgdorferi (1), Babesia canis (1), Rickettsia spp. (1), and Canid herpesvirus 1 (1); these tests yielded negative results in all cases. All of these dogs died or were euthanized while hospitalized because of acute deterioration or poor response to treatment.
Two dogs, including 1 Rhodesian Ridgeback and 1 Jack Russell Terrier, were presented with ulcerative lesions of the tongue and/or oral cavity that were found to be caused by vasculitis (Supplementary Table 3). The Rhodesian Ridgeback also had an ulcerative lesion on 1 footpad that was referable to vasculitis. Both cases were discharged from the hospital and treated with glucocorticoids and antibiotics; the Rhodesian Ridgeback also received pentoxifylline. Both cases were lost to subsequent follow-up.
The remaining 3 dogs suffered vasculitis in the cervical musculature (n = 1, Border Collie), in conjunction with a nonhealing cutaneous wound (1, Labrador Retriever), or in multiple organs (1, Bichon Frise; Supplementary Table 3). No underlying cause was identified for the vasculitis of multiple organs, but the dog was presented because of increased hepatocellular enzyme activity and persistent vaginal discharge caused by concurrent cystic endometrial hyperplasia. Signs of arteritis were detected in the uterus, in liver biopsy samples, and in a mammary adenoma that was removed during the same surgical procedure. The dog was treated with prednisolone, ursodeoxycholic acid, and s-adenosyl methionine, and was euthanized after 410 days.
The Labrador Retriever was presented with a nonhealing wound on the right flank, which was noted ~1 month previously. Surgical closure had been attempted at the referring veterinary practice, but the wound subsequently broke down. Surgical biopsy revealed vasculitis, with necrotic dermatitis and thrombosis of dermal vessels. A concurrent infection with methicillin-resistant Staphylococcus aureus was also detected, but this was considered to be an environmental contaminant. The dog was treated with oxytetracycline, niacinamide, and pentoxifylline, resulting in complete resolution of clinical signs. The dog was alive and asymptomatic when follow-up information was obtained 930 days after initial presentation.
The Border Collie had a 3-week history of pyrexia, lethargy, and inappetence when presented to the referral center. Physical examination revealed cervical pain, and changes suggestive of myositis were observed on magnetic resonance imaging of the neck. An incisional biopsy revealed chronic mixed inflammatory panniculitis, with myositis and vasculitis. The dog was treated with prednisolone, resulting in resolution of the clinical signs, and was still living when follow-up data was obtained 946 days after initial presentation.
Of the dogs that developed vasculitis secondary to another disease process, the majority were diagnosed with systemic or localized inflammatory or infectious disease (Table 1). One dog was exposed to xylitol immediately before the development of vasculitis affecting multiple organs, and 1 dog had been receiving regular injections of cytosine arabinoside for management of meningoencephalitis of unknown etiology for a period of 2 months prior to development of vasculitis in the small intestine, liver, lungs, and in the dermis and hypodermis at the site of injections in the dorsal interscapular region.
Description of cases of secondary vasculitis according to associated clinical syndrome.

To the authors’ knowledge, these disease processes have not previously been associated with vasculitis in dogs.
Age, breed, and sex predispositions
Logistic regression models were constructed to determine whether animals with primary vasculitis were more likely to have been of particular age, sex, or breed group. Females were 4.0 times (95% confidence interval = 1.0–15.8, P = 0.048) more likely to have vasculitis than males when compared to a groups of control dogs, but there was no effect of neutering status. There was no association between development of primary vasculitis and breed or age.
Laboratory data
In dogs with either primary or secondary vasculitis, neutrophils represented the most frequent type of inflammatory cell infiltrate in the vessel wall (Table 2, Fig. 1A), although 36% of primary cases and 10% of secondary cases had mixed inflammatory cell infiltrates. The mixed infiltrates included combinations of neutrophils, macrophages, lymphocytes, and plasma cells (Fig. 1B), although 1 case with vasculitis associated with inflammatory rhinitis had a mixed eosinophilic and neutrophilic infiltrate. A smaller number of cases (1 dog with primary vasculitis and 3 [10%] with secondary vasculitis) had predominant lymphocytic or plasmacytic infiltrates or a combination of these 2 cell types. Leukocytoclastic vasculitis (the presence of leukocyte fragments within the vessel wall with concurrent fibrinoid changes) was not definitively observed in any dog in our study.
Classification of vasculitis cases according to histologic criteria.*
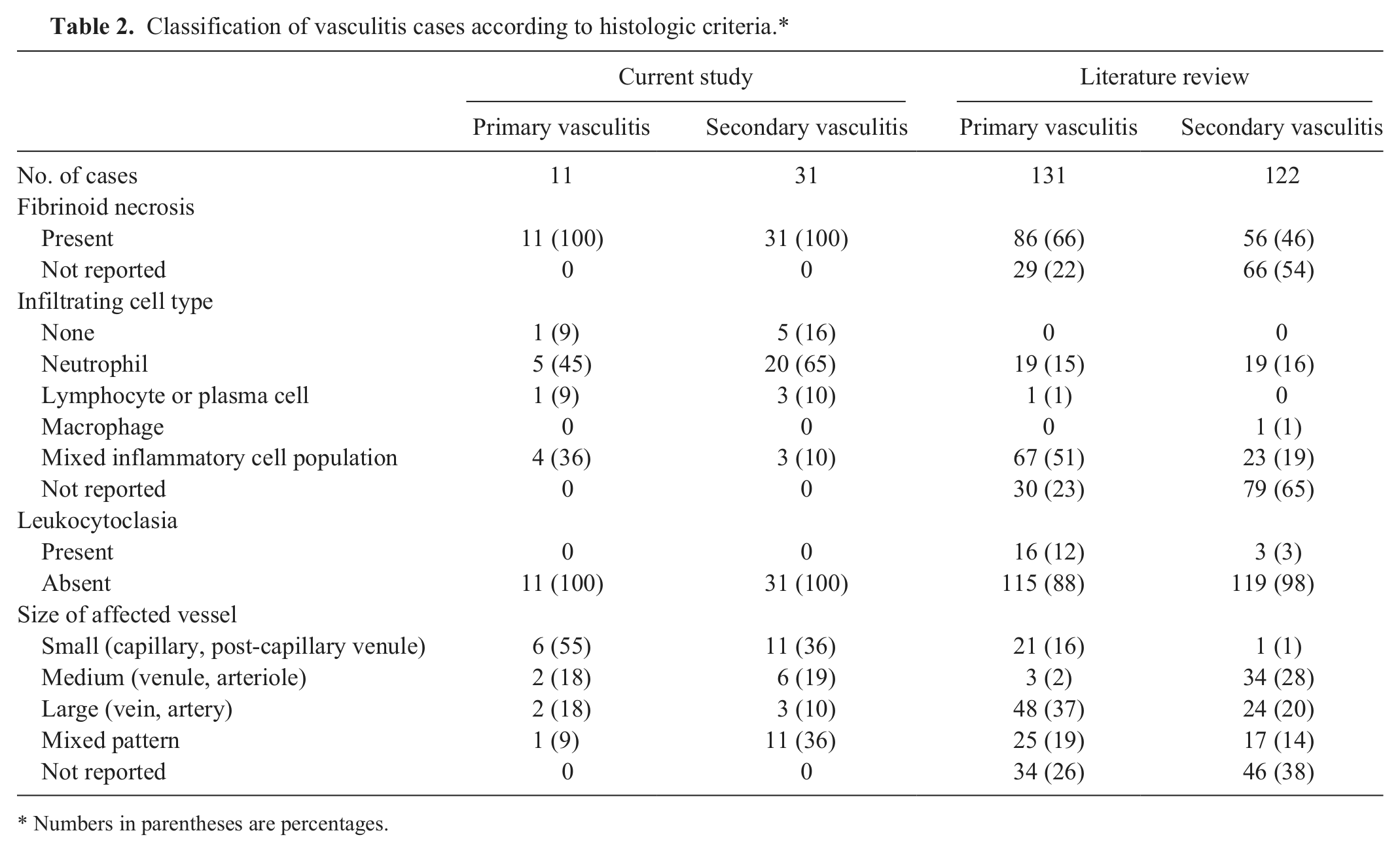
Numbers in parentheses are percentages.
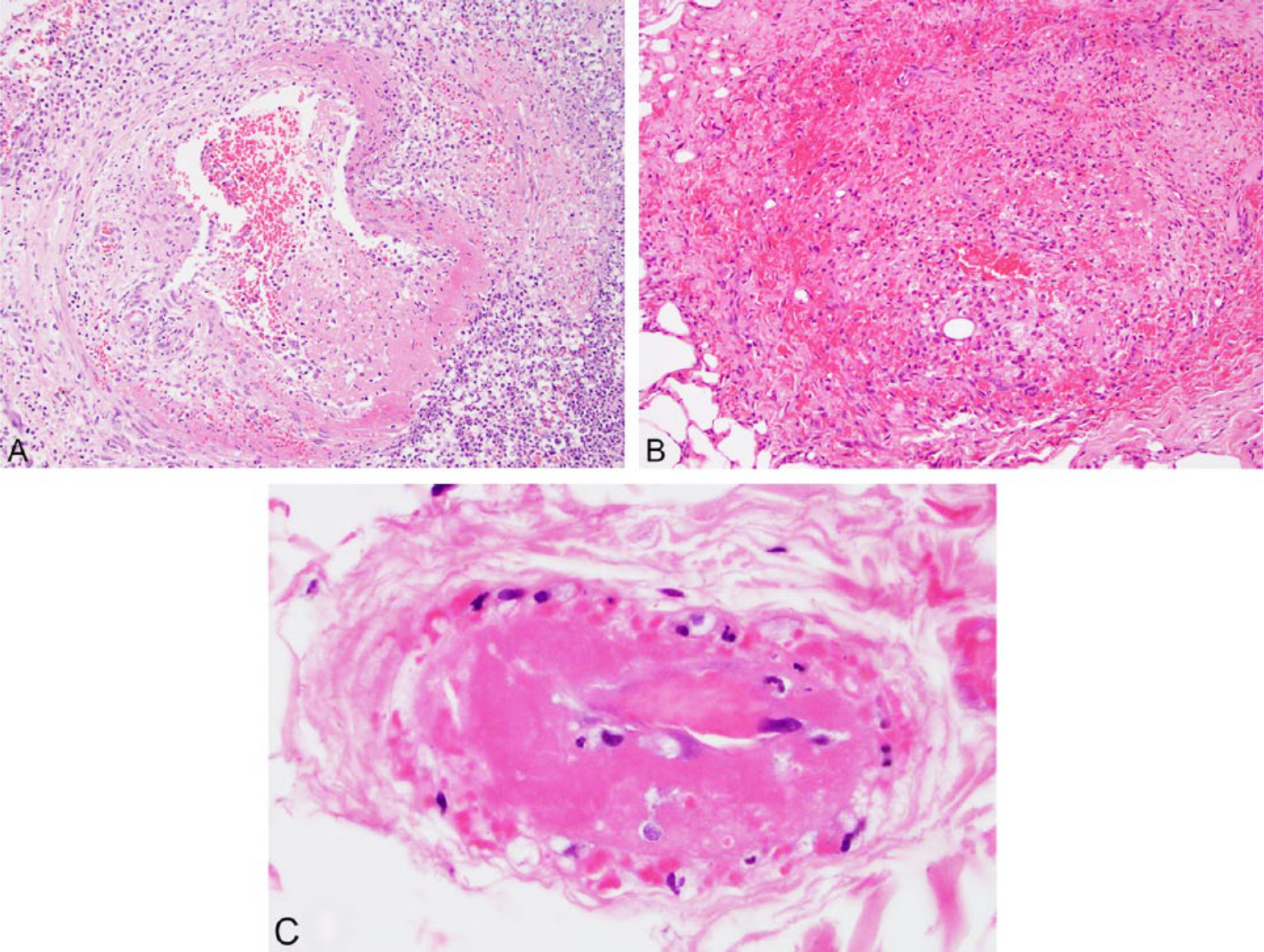
Representative images from cases with a histologic description of vasculitis. Hematoxylin and eosin.
In more than half (55%) of the cases with primary vasculitis, capillaries or postcapillary venules were predominantly affected (Fig. 1C), with medium and large vessels affected in smaller numbers of dogs (18% for each, Fig. 1A). A greater proportion of dogs with secondary vasculitis had changes in more than 1 type of vessel (36%, compared to 9% of primary cases), and the proportion of cases with vasculitis of the small vessels (36%) was correspondingly lower.
The serum concentration of globulin was greater in dogs with primary vasculitis (median = 34.4 g/l, interquartile range = 24.4–41.7) than those with secondary vasculitis (median = 28.2 g/l, interquartile range = 15.0–40.4, P = 0.040), but there were no other significant differences in the distributions of serum biochemical or hematologic values between these groups.
Outcome data
Follow-up data was obtained from referring veterinarians for 31 of 42 (73.8%) cases, and the remaining 11 cases were lost to follow-up after discharge. There was no difference in percentage mortality while hospitalized or at 1 year after discharge when comparing groups defined according to the size of the affected vessel or the predominant inflammatory cell type observed on histologic specimens (data not shown).
Literature search
Seventy-seven articles were identified that fulfilled the criteria for inclusion in the review of previously published data. A summary of the data presented in these studies is shown in Table 2. In contrast to the findings of our study, the majority of cases of primary vasculitis described in the veterinary literature affected medium or large blood vessels and involved mixed inflammatory cell infiltrates, but many studies failed to describe the changes in sufficient detail to enable classification. Fibrinoid changes were reported in approximately two-thirds (66%) of dogs with primary vasculitis but in less than half (46%) of those with secondary vasculitis.
Discussion
Our study describes the clinical and histologic features of a large number of cases of primary and secondary vasculitis in dogs. Primary vasculitis resulted in various clinical signs and pathologic changes depending on the organ system affected, with some cases showing signs of disease localized to just 1 area. Vascular inflammation also occurred in conjunction with a wide range of underlying diseases, some of which had not, to our knowledge, been reported previously. Perusal of the veterinary literature revealed that several forms of primary and secondary vasculitis had been described previously, but only small numbers of animals appeared to be affected, and there was little consistency in the reporting of histologic features of disease.
There were several similarities between the cases with primary vasculitis in our study and those reported previously in the veterinary literature. Several different forms of primary vasculitis have been reported to affect the CNS of dogs, of which the most comprehensively described example is necrotizing arteritis of Beagles. The lesions in Beagles and individuals of other breeds appear to primarily involve meningeal vessels,5,12,28 as was observed in cases 2, 3, 5, and 6 in our study. The white matter changes observed in case 4 were similar to those reported previously in 1 Weimaraner dog that had leukoencephalitis and vasculitis primarily affecting vessels within the white matter tracts of the brain. 3
Oral or lingual ulcerations have been reported previously in cases with primary vasculitis but not as a sole feature of the disease. In a case series of 5 dogs with primary vasculitis, 2 had oral or lingual ulcers in addition to cutaneous lesions. 25 One of the dogs in our study had concurrent vasculitis of a digital pad, but vasculitis of other organs could not be excluded in either of the 2 dogs because diagnosis was based on local biopsy samples.
Several novel associations were observed between vasculitis and the development of inflammatory diseases, including diskospondylitis and immune-mediated hemolytic anemia (IMHA). To our knowledge, development of vasculitis has not been reported previously in dogs that have ingested xylitol, although injections of cytosine arabinoside have been reported to cause vasculitis in people. 2
Almost half of the cases considered herein were diagnosed after examination of biopsy samples, and vasculitis affecting other organs or underlying disease processes could have been missed in those cases where complete autopsy was not performed. The nature of investigative tests performed also differed considerably between cases. Because classification of cases into primary or secondary vasculitis was chiefly based on this clinical information, some cases considered to have developed primary vasculitis in our study may have been grouped incorrectly depending on the investigations that were performed.
Females were 4 times more likely to have primary vasculitis than males, and similar associations have been reported across a wide range of human immune-mediated diseases, 7 including several that are associated with the development of vasculitis, such as SLE and rheumatoid arthritis. Similar associations have been reported for several canine immune-mediated diseases, including IMHA 16 and hypoadrenocorticism. 10 In people and laboratory animals, this female predisposition for development of immune-mediated diseases is related to the effect of estrogens. Presence of these hormones is associated with active Th1 responses, greater numbers of CD4+ T cells, and higher immunoglobulin concentrations than occur in males. 1
No association was detected when comparing breed frequencies between cases and controls, but our approach to grouping breeds according to haplotype sharing may not have been valid if the pathogenesis of primary vasculitis is related to genes that are conserved across groups. Attempts to investigate breed associations with primary vasculitis are also complicated by the heterogeneous nature of this syndrome, and examination of previous reports of primary vasculitis in dogs suggests that certain breeds or kindreds develop distinct forms of disease that may not share common pathologic mechanisms.
There was some variation in the relative frequencies of histologic features of vasculitis in this sample compared with the composite sample assembled from previously published studies. Among the primary vasculitis cases, these differences are most likely attributable to the relative overrepresentation of juvenile polyarteritis syndrome in laboratory Beagles in previously published studies.11,28,29 This disease is consistently associated with mixed inflammatory infiltrates and fibrinoid necrosis affecting small and medium-sized muscular arteries, 29 and this pattern of changes can be observed in the data presented in Table 2 for the group of primary cases identified from the literature review.
Fibrinoid changes in the vessel wall were considered to be the defining characteristic of vasculitis in our study, but these changes have not been described consistently in previous reports, particularly in those cases that were considered to have vasculitis secondary to another disease process. Because leukocytes observed in blood vessel walls may be migrating into the surrounding tissues rather than causing active inflammation, it is possible that some of the cases in this group may not have had vasculitis if the vessel wall was unchanged.
The data suggests that few forms of primary vasculitis occur consistently in dogs, and this situation contrasts with that observed in human medicine where several defined syndromes have been extensively described. According to the nomenclature system promulgated at the second Chapel Hill Conference in 2012, 14 the primary vasculitides of humans are grouped according to the size of the affected blood vessel, though this system is unlikely to reflect the immunopathogenesis of the syndromes described. 13 Given the wide variation in clinical presentation and histologic changes observed in our study, we do not consider that this grouping system will be helpful in further investigation of canine cases, but there is a need for consensus regarding histologic reporting of similar cases in the future.
An important tool in the diagnosis and classification of human vasculitides is measurement of the serum concentration of ANCA. This parameter is increased in several forms of small-vessel (ANCA-associated) vasculitis, and increased concentrations also correlate with disease activity and the risk of relapse in some patients. 4 Serum ANCA was not measured in any of the dogs included in our study, but it was found to be increased in 2 of 3 Beagles suffering from juvenile polyarteritis syndrome, 8 suggesting that some forms of vasculitis in dogs may be associated with the production of these antibodies. Although serum ANCA testing is not available for clinical use in veterinary practice, it could be used in the future as a clinical or research tool to explore the pathogenesis of canine vasculitis.
In conclusion, our study reports data from a large series of dogs with primary or secondary vasculitis treated at a single institution. Secondary vasculitis may be an underdiagnosed feature of several diseases, which could affect the outcome compared with uncomplicated cases. The causes of vasculitis in dogs are poorly understood, and a greater understanding is required of the pathogenesis of several forms of this disease in dogs.
Supplemental Material
DS1_JVDI_10.1177_1040638715587934 – Supplemental material for Histologic and clinical features of primary and secondary vasculitis
Supplemental material, DS1_JVDI_10.1177_1040638715587934 for Histologic and clinical features of primary and secondary vasculitis by James W. Swann, Simon L. Priestnall, Charlotte Dawson, Yu-Mei Chang and Oliver A. Garden in Journal of Veterinary Diagnostic Investigation
Supplemental Material
DS2_JVDI_10.1177_1040638715587934 – Supplemental material for Histologic and clinical features of primary and secondary vasculitis
Supplemental material, DS2_JVDI_10.1177_1040638715587934 for Histologic and clinical features of primary and secondary vasculitis by James W. Swann, Simon L. Priestnall, Charlotte Dawson, Yu-Mei Chang and Oliver A. Garden in Journal of Veterinary Diagnostic Investigation
Supplemental Material
DS3_JVDI_10.1177_1040638715587934 – Supplemental material for Histologic and clinical features of primary and secondary vasculitis
Supplemental material, DS3_JVDI_10.1177_1040638715587934 for Histologic and clinical features of primary and secondary vasculitis by James W. Swann, Simon L. Priestnall, Charlotte Dawson, Yu-Mei Chang and Oliver A. Garden in Journal of Veterinary Diagnostic Investigation
Footnotes
Authors’ contributions
JW Swann drafted the manuscript. JW Swann and OA Garden contributed to conception and design, and to acquisition, analysis, and interpretation of data. C Dawson contributed to design. YM Change contributed to analysis of data. SL Priestnall and C Dawson contributed to acquisition and analysis of data. JW Swann, SL Priestnall, C Dawson, YM Chang, and OA Garden critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
IBM SPSS Statistics for Windows v. 20.0, IBM Corp., Armonk, NY.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
