Abstract
A neuropathologic survey was conducted on mink brains from the 5 licensed mink farms in Ireland. The survey was part of a transmissible spongiform encephalopathy surveillance study. Aleutian disease (AD) was present on 4 of the 5 farms (80%). Neuropathologic features of nonsuppurative meningoencephalitis were common in mink from the 4 affected farms but were absent in the mink from the fifth farm, which was free of AD. The meningoencephalitis was characterized by infiltrates of lymphocytes and plasma cells, which were present in meninges, perivascular spaces, and the brain parenchyma. Fibrinoid necrotizing arteritis was seen in 11 mink brains, all of which were obtained from a single farm. Aleutian mink disease virus (AMDV) sequences for the capsid protein VP2 were obtained from brain samples from all affected farms. Although containing previously unreported amino acid residues, similarities with European and North American isolates were observed in the hypervariable regions within VP2, suggesting Irish AMDV is related to those isolates. The predicted amino acid residues, suspected of conferring pathogenicity at certain positions of the VP2 sequence, were present in the viral nucleic acid sequences.
Aleutian disease (AD) is a common chronic disease of captive mink (Mustela vison) caused by Aleutian mink disease virus (AMDV; family Parvoviridae, subfamily Parvovirinae, genus Amdovirus). 5 The disease is characterized by persistent viremia, hypergammaglobulinemia, and infiltration of tissues with lymphocytes and plasma cells. 1,11,14 Cases of AD can, on occasion, present as neurologic disease associated with a nonsuppurative meningoencephalitis. 7,11 Aleutian disease causes severe economic losses for mink farms. In addition, concerns have been raised that mink that have escaped captivity may be a potential source of the virus in wild carnivores. 7,13 Irish mink farms were established in the early 1950s, and subsequent to their escape, mink have been a common sighting on Irish waterways for decades (Ross A: 2009, Invasive alien species in Northern Ireland. Available at http://www.habitas.org.uk/invasive/species.asp?item=5131. Accessed August 5, 2009).
In Ireland, there are currently 5 licensed fur farms containing approximately 75,000 brown mink. During a neuropathologic survey of these farms, undertaken to screen for transmissible mink encephalopathy, a nonsuppurative meningoencephalitis was found to be common. The objectives of the present study were to determine whether the meningoencephalitis was associated with AMDV infection, to establish the spectrum of neuropathologic changes, and to compare the viral sequences obtained from the brains of infected mink with known sequences obtained from AD-infected mink in other countries.
The 5 Irish mink farms were each visited at the time of pelting during a period of 6 weeks. The information on AD status on these farms available to the authors was that serum from farms 1–4 had been consistently positive for AMDV antibody for a number of years, and serum from farm 5 had always tested negative for AMDV. Economic losses attributed to AMDV infection were reported on farm 2, where the average litter size was only 2.5 kits per female, as opposed to 5 kits per breeding female on the other farms. For histologic examination, brains were collected at random from 157 mink, aged between 1 and 3 years (Table 1). Following formalin fixation, routine processing, and paraffin embedding, hematoxylin and eosin–stained sections of each brain (including the cerebrum, basal ganglia, thalamus, midbrain, medulla at the level of the pons, medulla at the level of the obex, and cerebellum) were examined.
For polymerase chain reaction (PCR) detection of AMDV, brains from mink on each of the 5 farms were stored in liquid nitrogen vapor (-196°C). The unfixed brain tissue was available for individual mink from farm 1 (n = 13) and farm 2 (n = 16). Because of a change in procedures during pelting, only pooled samples of 10 unfixed brains were available from each of the other 3 farms. A DNA extract was prepared from 25 mg of each specimen (whether individual or pooled). a The isolate ADV-G b was used as a positive control for all PCR analysis. 16
The occurrence of nonsuppurative meningoencephalitis per number of brains examined by histopathology on 5 Irish mink farms.
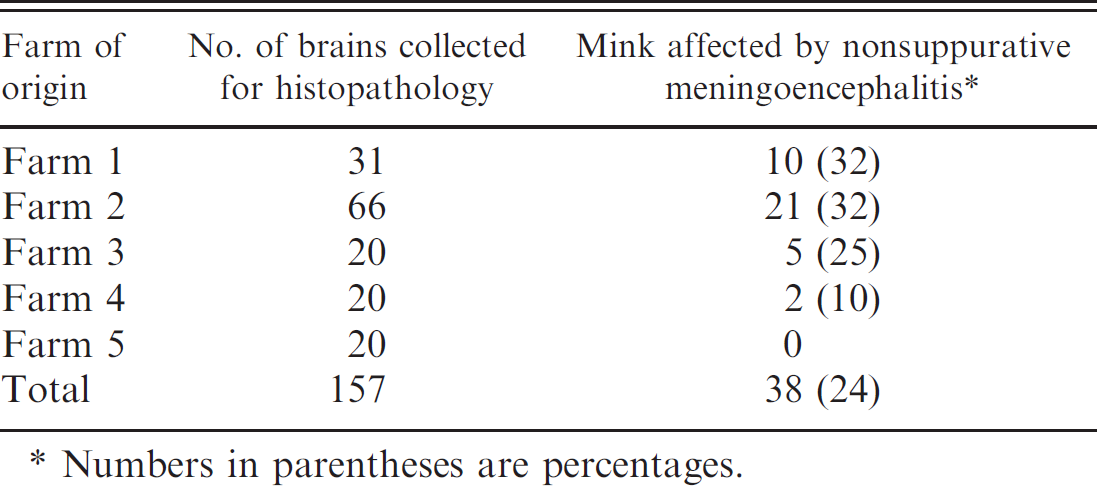
Numbers in parentheses are percentages.
Twenty-eight specimens yielded an AMDV-specific PCR product: 11 out of 13 samples from farm 1 (85%), 16 out of 16 samples from farm 2 (100%), and the pooled samples from farms 3 and 4. Eleven of these specimens were selected for sequencing, representing brain material collected from cases with or without neurologic lesions from the 4 AMDV-positive farms. The PCR products were prepared for sequencing using 5 pairs of forward and reverse primers based on the ADV-G genome 4 (S. Porcella, J. Wolfinbarger, and M. E. Bloom, National Institute of Allergy and Infectious Diseases, Rocky Mountain Laboratories, personal communication): P1 (corresponded to nucleotide [nt] positions 2293–2312 and nt 2774–2755), P2 (nt 2587–2608 and nt 3279–3252), P3 (nt 3060–3084 and nt 3546–3528), P4 (nt 3422–3445 and nt 4063–4041), and P5 (nt 3900–3921 and nt 4473–4455). c For each primer pair, 20 ng per 100 bases of purified PCR product, derived from separate PCR assays for each positive specimen, were vacuum-dried and sent for sequencing. d
The forward and reverse sequences were edited, aligned, and compared with ClustalW. 22 Amino acid sequences corresponding to the 11 partial VP2 nucleotide sequences were predicted by EMBOSS transeq 20 and compared with other published sequences for the VP2 of North American and European viruses (ADV-G NC001662, Utah-1 U39015, ADV-K X77084, Pullman U39014, ADV-TR U39013, ADV-TH5 AF124791, ADV-SL3 ×97629, ADV-LL6 AF205382). The Irish sequence data were then submitted to GenBank under accession numbers DQ630715–DQ630724.
Aleutian mink disease virus was detected in brain tissues from farms 1 to 4, but no virus was detected in the pooled samples from farm 5. This highlights the widespread distribution of AMDV in the Irish farmed-mink population, concurring with the serologic results supplied by the farmer.
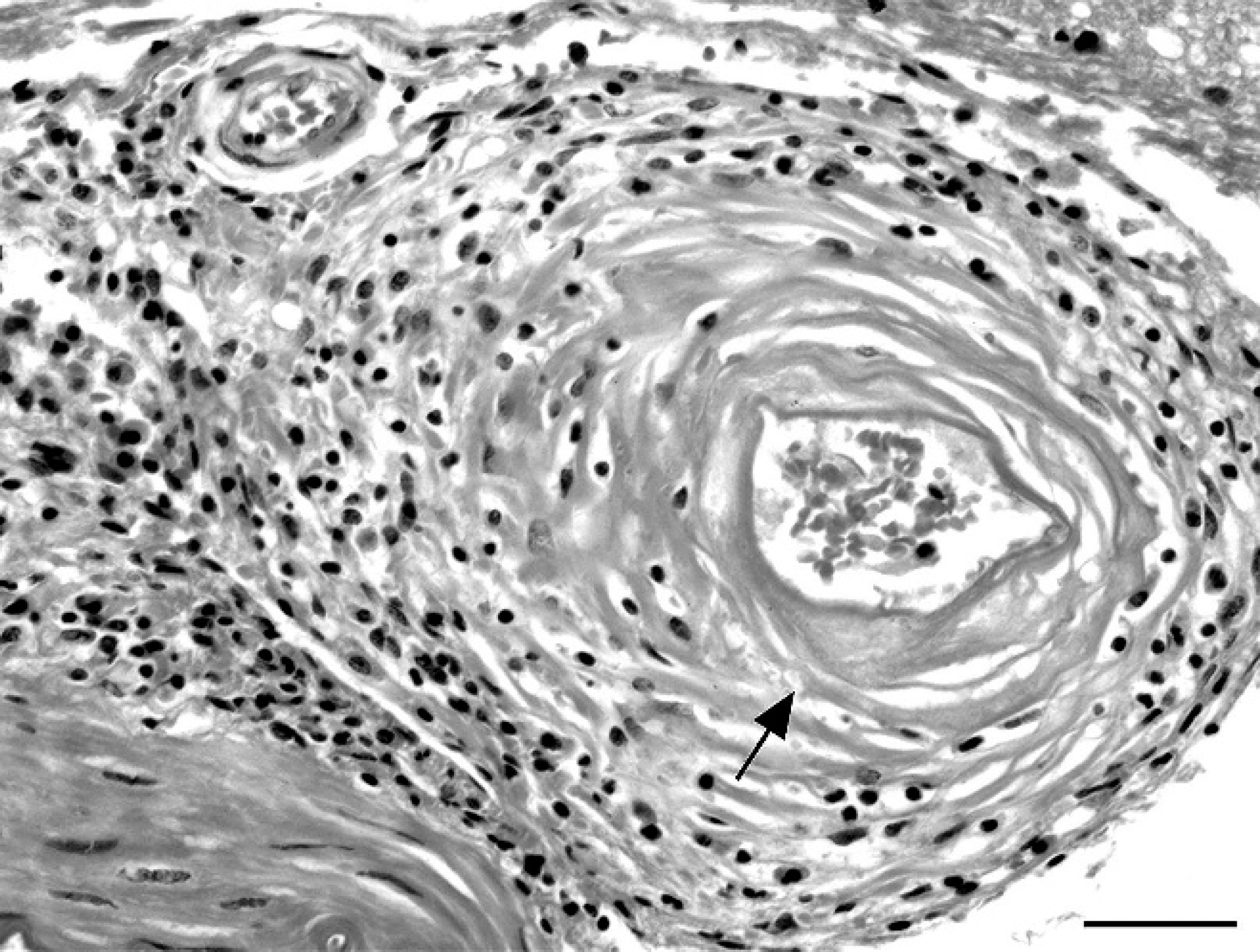
Cerebral meninges; mink (Mustela vison). Fibrinoid necrosis of arteriole wall characterized by eosinophilic, amorphous material expanding the arteriolar media (arrow), with peripheral infiltration of lymphocytes and macrophages. Hematoxylin and eosin. Bar ° 25 μm.
Nonsuppurative meningoencephalitis was observed in farms 1–4 only. The prevalence of meningoencephalitis within the specimen examined varied among the different farms, with up to 32% of individual mink having lesions in 2 farms (Table 1). The histopathologic changes were similar among mink brains from the 4 affected farms. Lymphocytic, plasmacytic, and histiocytic infiltration into the meninges was predominantly seen in the ventral aspect of the brain at the level of the medulla, midbrain, and thalamus and, to a lesser extent, in the meninges covering the frontal cortex. Rarely, there was cerebellar meningeal involvement. Perivascular infiltrates of similar cells were aggregated around large meningeal arterioles. The choroid plexus in the lateral ventricles and the ependymal cells in the fourth ventricle were often infiltrated with plasma cells and lymphocytes and, in the adjacent subependymal tissue, a lymphocytic plasmacytic vasculitis was observed. In 11 of the 21 affected mink from farm 2 (52%), extensive necrotizing arteritis in the meninges was also observed (Fig. 1). This was characterized by fibrinoid necrosis of the media of arterioles, and despite significant damage to vascular walls, thrombi impinging on lumina, infarctions, or hemorrhages were not observed. The changes in the grey and white matter of the brain parenchyma were distributed throughout the brain, favoring the thalamic region, midbrain, and medulla. These were characterized by a lymphocyte and plasma cell infiltration in the perivascular spaces (Fig. 2) and occasionally in the vessel wall of arterioles, rather than capillaries, accompanied by multifocal glial nodules or diffuse gliosis, sometimes including swollen astrocytes. Within these clusters of inflammation, some vessel walls had ruptured, and hemorrhage was seen. Despite the extensive neuropathology in the mink examined in the current study, the owners reported that there was no overt evidence of neurologic disease.
The meningoencephalitis found in the present study is similar to that previously reported in mink with AD with or without neurologic signs. 23 Necrotizing arteritis, apparent in some of the affected mink from farm 2, is a commonly reported finding in the brains of AD-affected mink, where the incidence can vary between 12% and 40%. 15,19,23 Although the pathogenesis of fibrinoid necrosis of arterioles is not clear, the postulation is that when production rates of viral immune complexes exceed clearance rates, complexes get deposited on vascular walls and instigate inflammation and necrosis. 3,12 Interestingly in the present study, these affected mink came from a single farm (farm 2), and as highlighted later, this farm had isolates suggestive of multiple infection sources rather than a single virus type as found on farm 1.
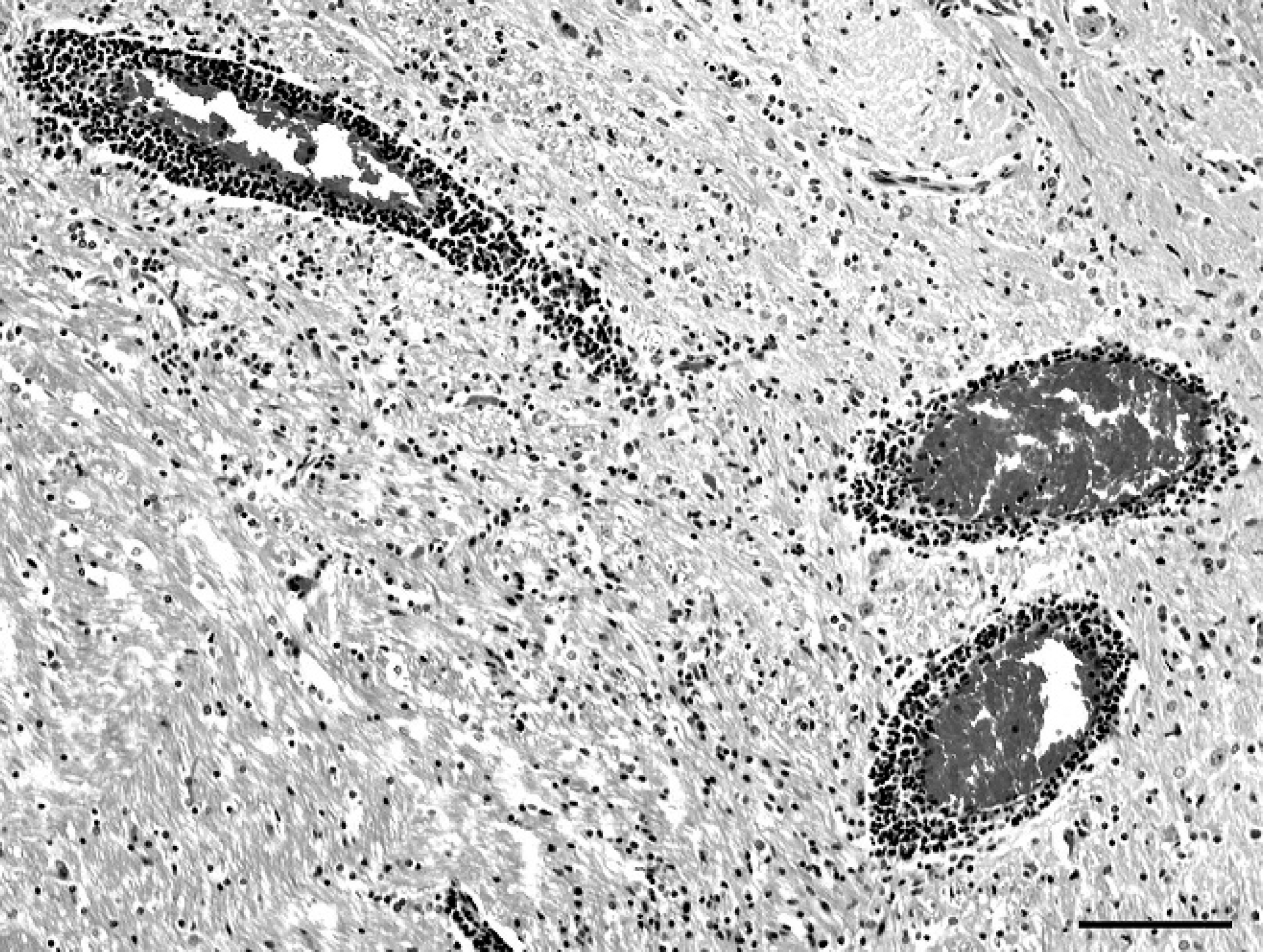
Thalamus; mink (Mustela vison). Perivascular infiltration of lymphocytes and plasma cells. Hematoxylin and eosin. Bar = 100 μm.
Sequence analysis of 11 PCR products obtained in the current study revealed 10 different previously unreported partial sequences for the capsid protein VP2. However, the differences between the Irish and the published sequences are mainly due to changes of up to 10% of single amino acid residues. The significance of this is not clear because the mutation rate of AMDV is not known, but studies on other parvoviruses revealed that apparently minor sequence differences can be correlated with host range and differences in replication. 18
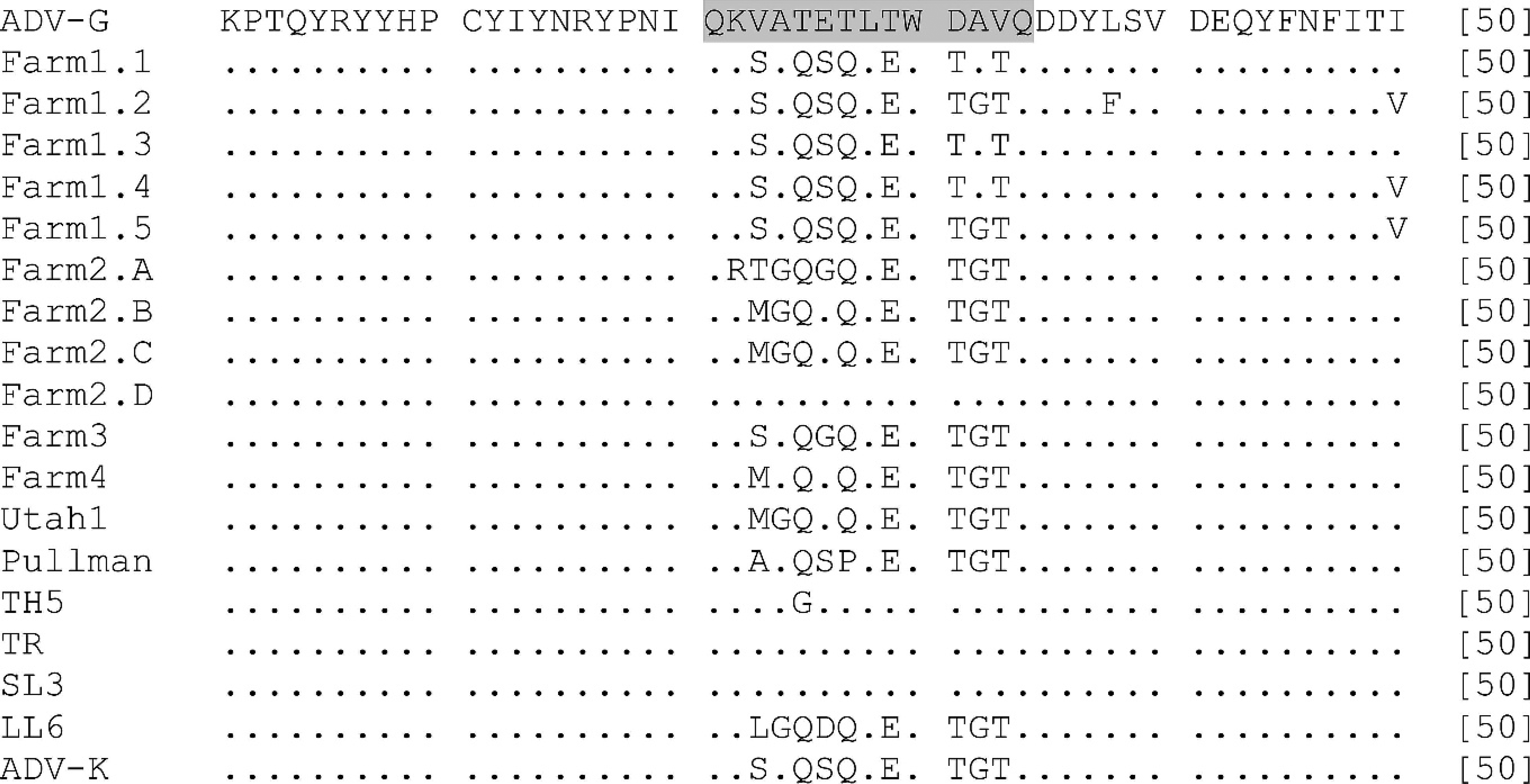
Sequence analysis and comparison of the hypervariable region as part of the VP2 region (231–244 amino acid sequence highlighted in grey) of Aleutian mink disease virus (AMDV) using the sequence analysis program ClustalW 22 (predicted 50 amino acid sequence from 11 Irish sequences [VP2 amino acid residues 211–250]). ADV-G, Utah-1, Pullman, TH5, TR, SL3, LL6, and ADV-K represent AMDV sequences from different countries published in GenBank.
More important, it is known that the VP2 sequence of the virus contains a hypervariable region at positions 230–244 4 of the amino acid sequence, which defines sequence types and is, therefore, potentially useful for identifying different AMDV isolates and their origins. 16 Similarities were observed in the hypervariable region among the viruses detected in Irish farmed mink and viruses detected elsewhere (Fig. 3), suggesting that Irish AMDV isolates are closely related to European and American AMDV isolates. The 5 sequences available from farm 1 were all very similar at the hypervariable region, whereas one of the sequences (A) from farm 2 showed 3 amino acid changes at positions 231, 232, and 235, which has not, to our knowledge, been reported previously. Another sequence (D) from farm 2 had the same amino acid sequence as the German SL3 and, therefore, differed considerably from the other sequences obtained from the same farm, which had the same sequence as the American virus Utah-1. Such features may suggest that in farm 1, all 5 sequences were from a single viral source, whereas those analyzed from farm 2 may represent multiple viral infections. Considering that fibrinoid arteritis was only observed in mink from farm 2, which also reported a low average litter size, this may indicate that the presence of multiple strains induces more severe pathologic changes in mink. Other studies have reported several sequence types within 1 animal and on 1 farm in 1 outbreak, but the connection between the occurrence of multiple viruses on an affected farm and the severity of pathologic changes is unclear. 9,17 The implications of the variability of the Irish isolates are not clear because many previous studies report only single-sequence analyses. 2,4,10 The above findings are the result of sampling at 1 time point and provide a reference as to the status of AMDV and AD in Ireland. It is clear that more long-term and detailed epidemiologic studies, combined with virus and tissue sampling, would shed more light on the dynamics of AMDV and the possible relationship of the presence of multiple viral strains and the severity of pathologic changes. However, as escaped mink could potentially infect wildlife, this Irish AMDV reference will be important when it comes to mapping infectious diseases in wildlife reservoirs.
The VP2 region was also analyzed in the present study because it contains amino acid residues at 5 positions (352, 395, 434, 491, and 534) that have been identified as highly conserved among pathogenic isolates when compared with the cell culture–adapted nonpathogenic strain ADV-G. 4,8,21 In the present study, these specific amino acid residues were predicted for those positions of the VP2 sequence analyzed. One exception was a single isolate from farm 2, which had a unique amino acid substitution predicted for position 395. Results of the current study, therefore, confirm that the Irish isolates are pathogenic viruses containing certain amino acid residues that confer the abilities of the virus to replicate in vivo and cause pathologic changes.
Furthermore, it has been suggested that some AMDV strains might preferentially cause disease in the nervous system. The VP2 sequence was used to characterize the ADV TH5 strain that was isolated from an outbreak of meningoencephalitis on a mink farm. 6 In that study, it was suggested that specific amino acid residues at positions 452 and 623 of the VP2 sequence could potentially confer neurotropism. However, in the present study, none of the sequences had the same unique 2 amino acid residues that characterized the TH5 strain, suggesting that such amino acid substitutions are not a prerequisite for neurotropism or neuropathogenicity.
The present study showed that AD is widespread among Irish farmed mink and that nonsuppurative meningoencephalitis is a common feature in mink with AMDV infection in Ireland. Novel VP2 sequences were obtained by PCR on brain tissues, which showed variability, and the presence of multiple sequence types from a single farm was observed.
Footnotes
a.
QIAamp® DNA Mini Kit, Qiagen Ltd., West Sussex, UK.
b.
Kindly supplied by M. E. Bloom and J. B. Wolfinbarger, NIAID, Rocky Mountain Laboratories, Hamilton, MT.
c.
Sigma-Genosys Ltd., Haverhill, UK.
d.
MWG Biotech AG, Ebersberg, Germany.
