Abstract
The fetal goat tongue cell line ZZ-R 127 and the fetal porcine kidney cell line LFBK-αvβ6 have been reported to have high sensitivity to various Foot-and-mouth disease virus (FMDV) strains. The suitability of ZZ-R 127 cells for FMDV isolation not only from epithelial suspensions but also from other clinical samples has already been confirmed in a previous study. However, to our knowledge, the suitability of LFBK-αvβ6 cells has not been evaluated using clinical samples other than epithelial materials. In addition, both cell lines have never been compared, in terms of use for FMDV isolation, under the same conditions. Therefore, in the current study, the virus isolation rates of both cell lines were compared using clinical samples collected from animals infected experimentally with FMDV. Viruses were successfully isolated from clinical samples other than epithelial suspensions for both cell lines. The virus isolation rates for the 2 cell lines were not significantly different. The Cohen kappa coefficients between the virus isolation results for both cell lines were significantly high. Taken together, these results confirmed the suitability of LFBK-αvβ6 cells for FMDV isolation from clinical samples other than epithelial suspensions. The levels of susceptibility of both cell lines to FMDV isolation were also confirmed to be almost the same.
Keywords
Foot-and-mouth disease (FMD) is a highly contagious disease of cloven-hoofed domestic and wild animals that occurs sporadically or endemically in numerous countries around the world. The FMD virus (FMDV; order Picornavirales, family Picornaviridae, genus Aphthovirus) causes the disease. 7 There are 7 serotypes, A, O, C, SAT1, SAT2, SAT3, and Asia1, with each serotype also having several topotypes; the topotypes are classified according to a comparison of their viral protein 1–coding sequences. 7
Virus isolation is described as one of the diagnostic methods for FMD according to the World Organization for Animal Health (OIE; http://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/2.01.05_FMD.pdf). Several primary cells and established cell lines can be used for virus isolation according to the OIE manual, and primary bovine thyroid (BTY) cells have generally higher sensitivity for virus isolation than other primary cells and cell lines (http://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/2.01.05_FMD.pdf). However, because BTY cells are derived from a thyroid collected from a calf <7 days old, 3 it is difficult to routinely prepare them. The sensitivity of the BTY cells for virus isolation also varies between batches. 3 Furthermore, BTY cells cannot be cryopreserved without a decrease in their sensitivity for virus isolation. 3 Therefore, BTY cells are less convenient for diagnostic work on FMD in FMD-free countries because, in general, such diagnostic work is not performed frequently in such countries.
Two established cell lines have been reported by different research groups to have high sensitivity to various FMDV strains, namely the fetal goat tongue cell line ZZ-R 1272 and the fetal porcine kidney cell line LFBK-αvβ6.8,9 The ZZ-R 127 cell line has naturally high susceptibility to FMDV 2 ; however, the LFBK-αvβ6 cell acquired greater susceptibility than the original LFBK cell as it has been modified to artificially express a bovine αvβ6 integrin, 8 which is one of the receptors of FMDV. 11
The recommended materials for diagnostic work on FMDV are epithelium or vesicular fluid, and ideally at least 1 g of epithelial tissue is collected from an unruptured vesicle or a vesicle immediately after rupture on the tongue, buccal mucosa, or foot because viral titers of epithelial and vesicular materials are generally higher than those of other clinical samples (http://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/2.01.05_FMD.pdf). However, it is generally difficult to collect fresh epithelial and vesicular materials in the field. In general, apparent lesions tend not to develop or to disappear rapidly in small ruminants. 1 The OIE also recommends that animals are sedated before any samples are collected in order to avoid injury to personnel collecting the samples as well as for animal welfare reasons (http://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/2.01.05_FMD.pdf). However, sedating animals in order to collect samples in the field is not convenient in an FMD epidemic area because various control measures must be performed as soon as possible.
For the above-mentioned reasons, it is important to evaluate any primary cells and cell lines for virus isolation using clinical samples such as sera, saliva, nasal swabs, and oropharyngeal fluids, which can be collected more easily than epithelial materials. The susceptibility of ZZ-R 127 cells to virus isolation has already been evaluated using clinical samples collected from animals infected experimentally with a FMDV strain. 5 However, the susceptibility of the LFBK-αvβ6 cells to virus isolation has not been evaluated using clinical samples other than epithelial materials, to our knowledge. Therefore, an evaluation of the susceptibility of LFBK-αvβ6 cells to virus isolation with sample types other than epithelium was the first objective of this study. Both the ZZ-R 127 and LFBK-αvβ6 cell lines have levels of susceptibility to FMDV isolation similar to that of BTY cells.2,8 However, the susceptibility levels of both cell lines have never been directly compared under the same conditions. Therefore, a comparison of the susceptibility of these cell lines to FMDV isolation using clinical samples collected from animals infected experimentally with FMDV and using cell culture supernatants of several FMDV strains was the second objective of this study.
The ZZ-R 127 a and LFBK-αvβ6 b cells were maintained using a commercial medium c supplemented with 10% fetal bovine serum (FBS). An FMDV isolate, which was isolated in the 2010 epidemic in Japan, was used in the experimental infections in our study (isolate O/JPN/2010-1/14C). 4 Cell culture supernatants of O/JPN/2010, 4 O/JPN/2000, 11 O1 Manisa, d O1 BFS 1860, d O/TAW/97, e O/TUR/5/2009, d A15 TAI 1/60, d A22 IRQ 24/64, d A/IRN/1/2011, d A/TAI/10/2011, d C PHI 7/84, d Asia1 Shamir (ISR 3/89), d Asia1/TUR/49/2011, d SAT1/KEN/117/2009, d SAT2/SAU/6/2000, d and SAT3/ZIM/3/83 d strains were also used in order to compare progress of virus replication in the ZZ-R 127 and LFBK-αvβ6 cells over time and a range of virus dilutions.
Clinical samples used in our study were collected following experimental infections. In experiment 1, three 3-month-old Holstein cattle were administered an inactivated FMDV vaccine (serotype O, O Manisa strain). f Then, the cattle were inoculated with 1 mL of 106 50% tissue culture infectious dose (TCID50) of O/JPN/2010-1/14C by an intradermal route. The cattle were observed for ~1 month after inoculation. Full details of animal experiments 2–4 have been previously published.6,10 Brief details of these experiments are as follows. For experiment 2, two 6-month-old Holstein cattle, which were housed in separate cubicles, were inoculated with 1 mL of 106.2 TCID50 of O/JPN/2010-1/14C by an intradermal route. At 1 day postinoculation (dpi), two 6-month-old Holstein cattle were housed with the inoculated cattle. The cattle were housed in the same cubicle for ~1 month. 10 For experiment 3, two 4-month-old Japanese Saanen goats were inoculated with 1 mL of 106.2 TCID50 of O/JPN/2010-1/14C by an intradermal route. At 1 dpi, two 4-month-old Japanese Saanen goats were housed together with the inoculated goats. The goats were housed in the same cubicle for ~1 month. 10 In experiment 4, three 2-month-old pigs or four 2-month-old pigs were each inoculated with 1 mL of 103 or 106 TCID50 of O/JPN/2010-1/14C by an intraoral route. The pigs were housed in the same cubicle for ~2 weeks. 6 All experimental infections were carried out in a high-containment facility at the National Institute of Animal Health (NIAH; Japan). All animal procedures were approved prior to initiation of the experimental infections by the Animal Care and Use Committee of the NIAH (authorization nos. 12-027, 12-056, 13-024, and 13-054).
Sera were collected from cervical veins using a vacuum blood collection tube. g Saliva was collected from oral cavities using a roll-shaped synthetic saliva collector h and forceps. Nasal swabs were collected from nasal cavities using a cotton swab. i Oropharyngeal fluids were collected using a probang cup. Collection of the clinical samples was performed daily until 10 dpi and at 3–4-day intervals after that. All collected samples were stored at −80°C until tests were performed.
Virus isolation was performed according to the OIE manual (http://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/2.01.05_FMD.pdf). Cells, which were seeded using the commercial medium
c
supplemented with 10% FBS in 24-well plates 1 day before the virus isolation was performed, were washed once using a different commercial medium
j
supplemented with 2.4 g/L of tryptose phosphate broth, 1% of 7.5% NaHCO3, and 1% of 2.92%
Table 1 shows the detection rates of viral genes for the RT-PCR assay and the virus isolation rates for both cell lines. A total of 372 samples were collected from the experimentally infected cattle. Viral genes were detected in 116 (31.2%) of 372 samples in the RT-PCR assay. Viruses were isolated from 50 (13.4%) and 59 (15.9%) of 372 samples by the ZZ-R 127 and LFBK-αvβ6 cells, respectively. All of the samples from which the viruses were isolated were among the RT-PCR–positive samples. The virus isolation rates from each sample and the total virus isolation rates for both cell lines were not significantly different. Time course data of detection of viral genes by the RT-PCR assay and isolation of viruses by both of the cell lines are shown in Supplementary Tables 1–4.
Comparison of detection rates of viral genes obtained by the reverse transcription polymerase chain reaction (RT-PCR) assay and virus isolation rates obtained by the ZZ-R 127 and LFBK-αvβ6 cell lines.
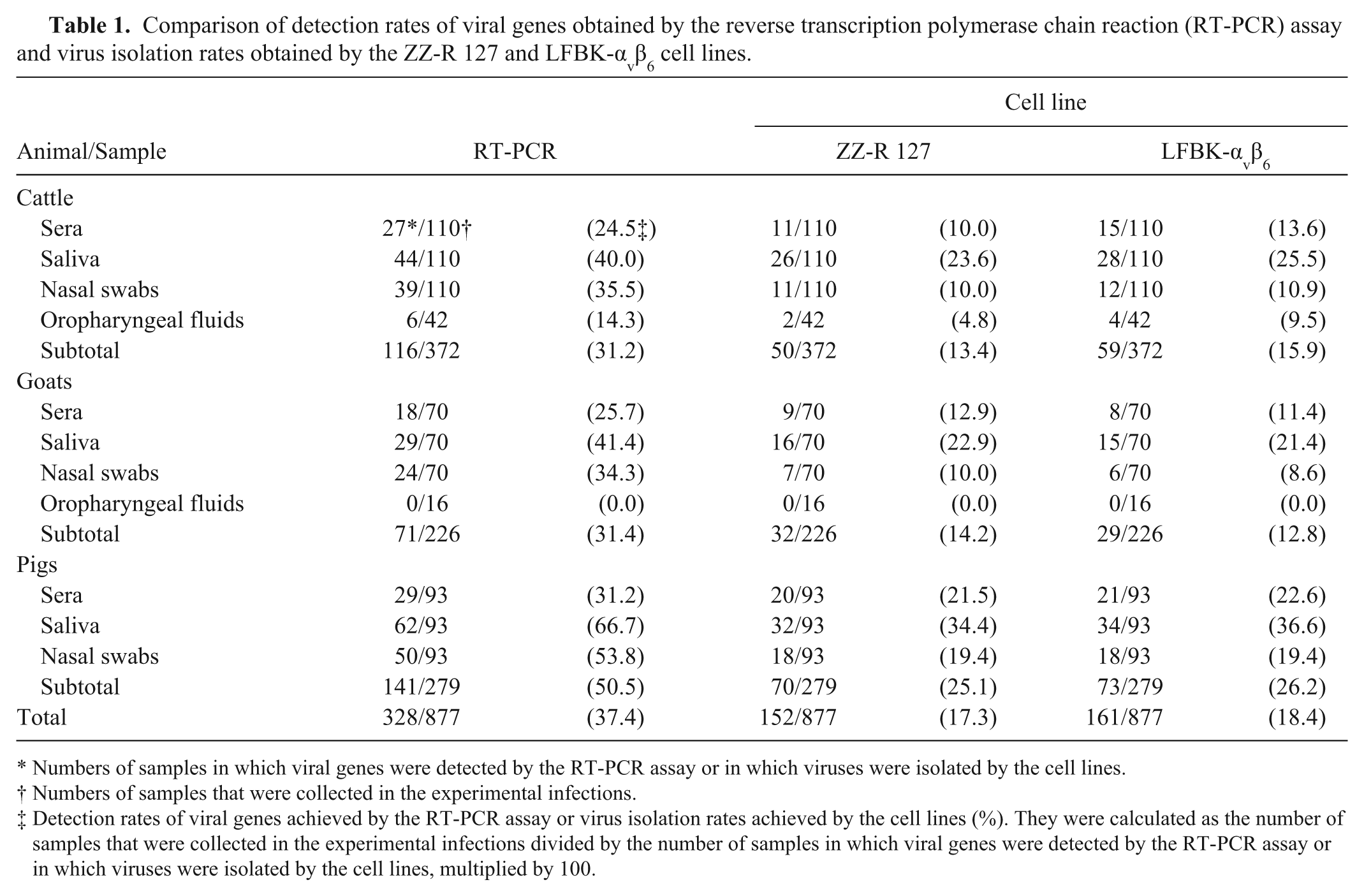
Numbers of samples in which viral genes were detected by the RT-PCR assay or in which viruses were isolated by the cell lines.
Numbers of samples that were collected in the experimental infections.
Detection rates of viral genes achieved by the RT-PCR assay or virus isolation rates achieved by the cell lines (%). They were calculated as the number of samples that were collected in the experimental infections divided by the number of samples in which viral genes were detected by the RT-PCR assay or in which viruses were isolated by the cell lines, multiplied by 100.
In total, 226 and 279 samples were collected from the experimentally infected goats and pigs, respectively. As with the experimental infections using cattle, viruses were isolated from approximately the same number of samples by both cell lines. In addition, all of the samples from which the viruses were isolated were among the RT-PCR-positive samples. Furthermore, the virus isolation rates from each sample and the total virus isolation rates of both cell lines were not different statistically. Supplementary Tables 5–8 show time course data of detection of viral genes by the RT-PCR assay and isolation of viruses by both cell lines in the goats and pigs. Table 2 shows progress of virus replication of 16 FMDV strains over time and a range of virus dilution in both cell lines. Virus replication could be detected in both cell lines after 24 hr by recognition of development of a CPE. The development of the CPE progressed further after 48 hr; however, the development was not different between both cell lines. Table 3 shows a comparison of the virus isolation results obtained by both cell lines. The concordance rates between the virus isolation results obtained by both cell lines in cattle, goats, pigs, and cell culture supernatants of the FMDV strains were 97.0%, 98.7%, 98.9%, and 100%, respectively. In addition, the Cohen kappa coefficients between the virus isolation results obtained by both cell lines in cattle, goats, pigs, and cell culture supernatants were 0.88, 0.94, 0.97, and 1, respectively, and were significantly high.
Development of a cytopathic effect (CPE) arising from the inoculation of cell culture supernatants of 16 Foot-and-mouth disease virus (FMDV) strains in the ZZ-R 127 and LFBK-αvβ6 cell lines.*
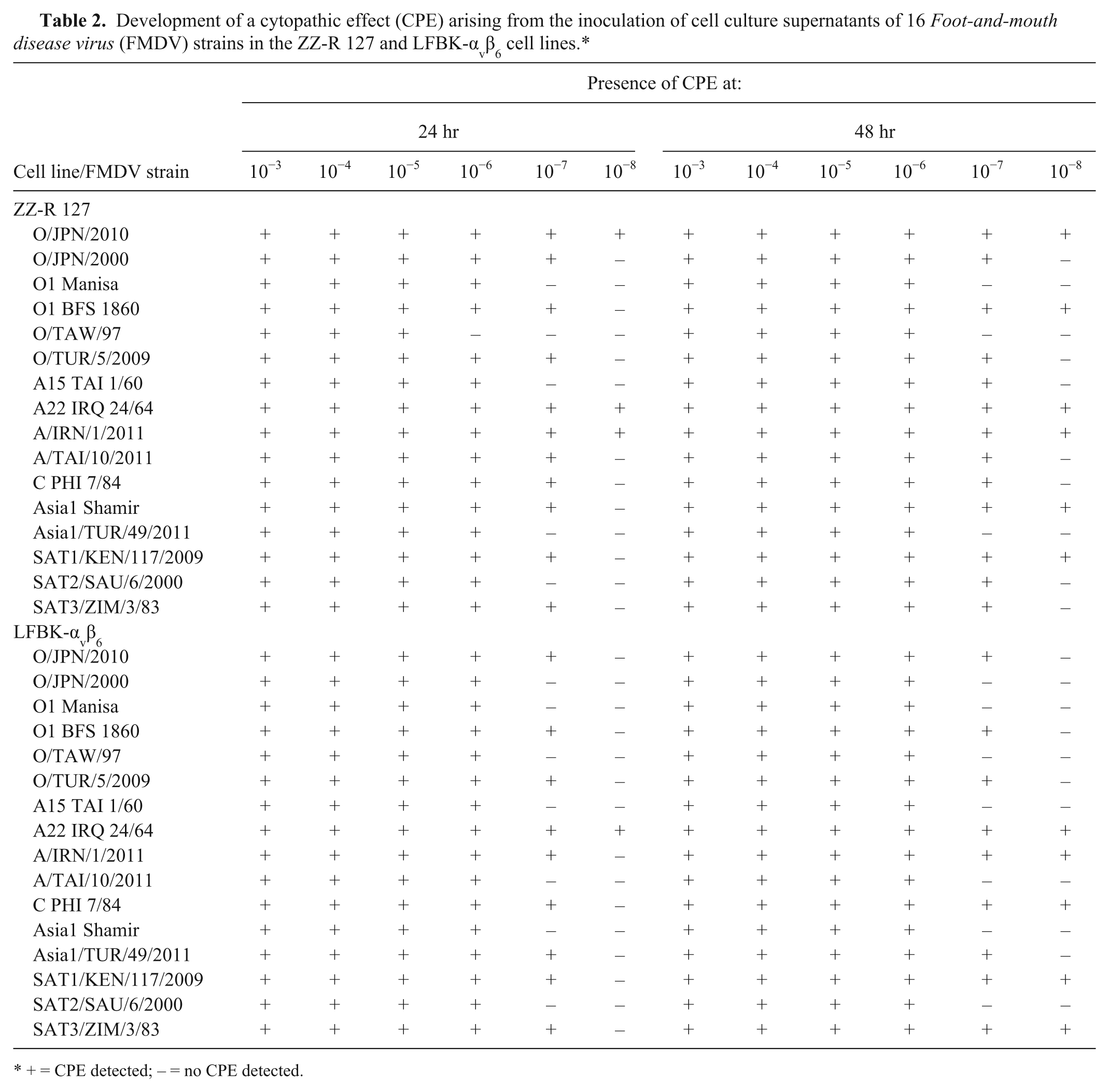
+ = CPE detected; – = no CPE detected.
Comparison of the results of virus isolation by the ZZ-R 127 and LFBK-αvβ6 cell lines.
Concordance rate between the results of virus isolation by both the cell lines in cattle was 97.0% (361/372).
Concordance rate between the results of virus isolation by both the cell lines in goats was 98.7% (223/226).
Concordance rate between the results of virus isolation by both the cell lines in pigs was 98.9% (276/279).
Concordance rate between the results of virus isolation by both the cell lines in cell culture supernatants was 100% (16/16).
The suitability of ZZ-R 127 cells for FMDV isolation not only from epithelial suspensions but also from other clinical samples such as sera, saliva, nasal swabs, and oropharyngeal fluids has already been confirmed. 5 However, the suitability of LFBK-αvβ6 cells for virus isolation from clinical samples other than epithelial suspensions has not been evaluated, to our knowledge. In the current study, although slight difference in the virus isolation rates was observed between the ZZ-R 127 and LFBK-αvβ6 cell lines across the animal species from which the clinical samples used in this study were collected, viruses were isolated at almost the same rate from samples by the ZZ-R 127 and the LFBK-αvβ6 cell lines. The suitability of the LFBK-αvβ6 cells for FMDV isolation from clinical samples other than epithelial suspensions was confirmed in our study.
It has been reported that the ZZ-R 127 and LFBK-αvβ6 cell lines have similar sensitivity for FMDV isolation as that given by BTY cells.2,8 However, the sensitivity levels of both cell lines have never been compared under the same conditions. The virus isolation rates from the clinical samples collected from the cattle and pigs were slightly higher for the LFBK-αvβ6 cells than for the ZZ-R 127 cells. On the other hand, those from the samples collected from goats were slightly higher for the ZZ-R 127 cells than for the LFBK-αvβ6 cells. The ZZ-R 127 cells and LFBK-αvβ6 cells originated from a fetal goat tongue and a fetal porcine kidney, respectively.2,9 In addition, the bovine αvβ6 integrin is expressed artificially on the LFBK-αvβ6 cells. 8 Therefore, the results of the current study were not unexpected. Although the virus isolation rates obtained by both cell lines were not different statistically and the Cohen kappa coefficients between the results of virus isolation by both cell lines in each animal were significantly high, there were several samples from which viruses were isolated only by one or the other of the cell lines. Therefore, diagnostic work for FMD could be improved by using a combination of these cell lines.
The ZZ-R 127 cells have several disadvantages that the LFBK-αvβ6 cells do not have. First, the FMDV O/TAW/97 strain, which is porcinophilic, grows well in LFBK-αvβ6 cells but does not grow well in ZZ-R 127 cells.2,8 Also, LFBK-αvβ6 cells grow very quickly in culture; however, ZZ-R 127 cells grow slowly in culture.2,8 Although the LFBK-αvβ6 cells are able to detect swine vesicular disease virus (SVDV) and vesicular stomatitis virus (VSV), which show clinical signs similar to those of FMD (http://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/2.01.05_FMD.pdf), the ZZ-R 127 cells are able to detect only high titers of VSV and are not able to detect SVDV.2,8 In contrast, the only potential disadvantage of the LFBK-αvβ6 cells is that they are contaminated with Bovine viral diarrhea virus (Rodriguez LL, personal communication, 2014). These disadvantages may not be problems in diagnostic work on FMD in many cases; however, both the cell lines should be utilized for diagnostic work on FMD with a good understanding of their characteristics. In conclusion, diagnosing FMD could be improved by using both ZZ-R 127 and LFBK-αvβ6 cell lines because the suitability of both cell lines for FMDV isolation from clinical samples other than epithelial suspensions and cell culture suspensions of strains representing all 7 serotypes of FMDV was confirmed in this study.
Footnotes
Acknowledgements
We are grateful to Dr. Matthias Lenk (Friedrich-Loeffler-Institute, Collection of Cell Lines in Veterinary Medicine, Germany) for supplying the ZZ-R cells, Dr. Peter W. Krug (Plum Island Animal Disease Center, USA) for supplying the LFBK-αvβ6 cells, and Mr. Kohei Honda (Yamagata Prefectural Central District Animal Hygiene Service Center, Yamagata, Japan) and Mr. Tomohiko Shimoda (Mie Prefectural Central District Animal Hygiene Service Center, Mie, Japan) for technical assistance. We would also like to thank Mr. Hiroki Kimura, Mr. Masayuki Kanda, Mr. Shinya Sato, Mr. Kenichi Ishii, Mr. Tatsuo Nakamura, and Mr. Shigeo Mizumura for their care of the animals.
Authors’ contributions
K Fukai and T Kanno contributed to the conception and design of the study, and critically revised the manuscript. K Fukai contributed to acquisition, analysis, and interpretation of data. K Morioka, M Yamada, T Nishi, and K Yoshida contributed to acquisition of data. T Kanno contributed to acquisition and analysis of data. R Kitano and R Yamazoe contributed to interpretation of data. K Fukai, K Morioka, M Yamada, T Nishi, K Yoshida, and T Kanno drafted the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Friedrich-Loeffler-Institute, Collection of Cell Lines in Veterinary Medicine, Greifswald-Insel Riems, Germany.
b.
Plum Island Animal Disease Center, Greenport, NY.
c.
Dulbecco’s modified Eagle medium: nutrient mixture F-12, Life Technologies, Carlsbad, CA.
d.
Institute for Animal Health, Surrey, UK.
e.
Animal Health Research Institute, New Taipei, Taiwan.
f.
Aftpor, Merial SAS, Lyon, France.
g.
Venoject II, Terumo Corp., Tokyo, Japan.
h.
Salivette, Sarstedt KK, Tokyo, Japan.
i.
Men-tip, JCB Industry Ltd., Tokyo, Japan.
j.
Eagle’s minimum essential medium, Nissui Pharmaceutical, Tokyo, Japan.
k.
Indirect sandwich enzyme-linked immunosorbent assay for detection of antigens of foot-and-mouth disease virus and swine vesicular disease virus, Biological Diagnostic Supplies Ltd., Ayrshire, UK.
l.
High pure viral RNA kit, Roche Diagnostics, Basel, Switzerland.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a research project on improving food and animal health of the Ministry of Agriculture, Forestry and Fisheries of Japan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
