Abstract
Primary hyperaldosteronism is an increasingly recognized syndrome in cats, and diagnosis can be difficult. A potential diagnostic method has been reported, utilizing oral fludrocortisone administered twice daily for 4 days followed by collection of urine. In the current study, we sought to determine if blood sampling and a shorter dosing period would provide a possible means to test for primary hyperaldosteronism. Also, cortisol concentrations were measured to assess the potential of fludrocortisone to act as a glucocorticoid in cats. In phase I, 8 healthy laboratory cats were studied in a placebo-controlled, crossover design. Serum aldosterone and cortisol concentrations were measured before and on the second, third, and fourth day of treatment and compared within groups. In phase II, based on the results obtained in phase I, 8 healthy client-owned cats were administered 3 doses of fludrocortisone or placebo. Serum aldosterone and cortisol concentrations were compared before and after treatment within groups. In both phases, serum aldosterone and cortisol concentrations were significantly suppressed in fludrocortisone-treated cats. Thus, it was determined that oral administration of fludrocortisone causes suppression of serum aldosterone in healthy adult cats after only 3 doses. Further research is needed to determine the effects of oral fludrocortisone in cats with primary hyperaldosteronism and cats with other disorders causing hypertension and/or hypokalemia to determine if this protocol can be used as a tool for the definitive diagnosis of primary hyperaldosteronism.
Introduction
Primary hyperaldosteronism (PHA) in cats has been reported in the veterinary literature since 1983, but with increasing frequency over the past decade.1,2,4,7–10,14,17,19–21 It is an important feline disease because of significant associated morbidity and mortality attributable to resultant hypokalemia and hypertension. Furthermore, it has been implicated as a mediator of progression of renal disease 10 and hypertension 11 in cats.
Diagnosis of PHA in humans involves screening via the aldosterone-to-renin ratio (ARR), followed by confirmation with a functional test.15,23 In contrast, in cats, suspicion of PHA arises with identification of hypokalemia, hypertension, and elevated plasma aldosterone concentration; diagnosis ultimately relies on identification of a discrete tumor within the adrenal glands.1,2,4,7–9,14,17,19–21 However, in the absence of a discrete tumor, such as in cats with adrenocortical nodular hyperplasia, minimal or no adrenal gland changes may be detected via ultrasound. 10 In fact, in humans, following a diagnosis of PHA, further testing such as a computed tomography scan and adrenal vein blood sampling is conducted to differentiate between unilateral and bilateral hyperplasia, adenoma or adenocarcinoma.15,23 Such testing is essential in determining the treatment plan. With increasing awareness of PHA and the identification of a non-tumorous form of PHA in cats, early diagnosis of PHA will likely present a growing challenge without a definitive confirmatory test, as abnormalities such as hypokalemia and azotemia are not specific for PHA, and aldosterone concentrations may not be markedly elevated in early cases or in those with hyperplasia. To our knowledge, a feline plasma renin activity assay is currently not offered commercially in the United States, making ARR measurement infeasible for the diagnosis of feline PHA. A plasma renin activity assay for nonhuman species is offered by a single clinical laboratory, but no reference intervals are provided. a In addition, the ARR can fluctuate in and out of the reference interval in cats with PHA. 10
A test requiring administration of fludrocortisone, a synthetic corticosteroid with mineralocorticoid activity, has been evaluated in cats to determine if it is a definitive, reliable confirmatory test with high specificity for PHA.5,6 Fludrocortisone should indirectly suppress the renin–angiotensin–aldosterone system (RAAS), and thereby serum aldosterone concentrations, in healthy cats either by increasing blood pressure or causing sodium retention; conversely, aldosterone concentrations in cats with PHA would not be expected to decrease as aldosterone secretion is autonomous. Urine was collected as a reflection of average aldosterone secretion over time, potentially eliminating the problem of fluctuations in serum concentrations throughout the day. 6 Oral fludrocortisone administration for 4 days to healthy cats significantly suppressed the urinary aldosterone-to-creatinine ratio (UACR), with a median suppression of 78% compared with baseline. 6 In addition, 1 cat with confirmed metastatic adrenocortical adenocarcinoma causing PHA had an increased UACR following fludrocortisone administration. 6 The test was further evaluated in cats with hypertension with or without hypokalemia, and a diagnosis of PHA could be excluded in cats in which the UACR on day 4 was suppressed by more than 50% compared with baseline. 5
Thus, a fludrocortisone suppression test has merit as a diagnostic tool in feline PHA. However, alternative protocols warrant investigation. Urine is not the route of excretion of major aldosterone metabolites in cats, 22 and, therefore, urinary aldosterone concentrations may not accurately reflect plasma aldosterone concentrations. Indeed, in a previous report, baseline plasma aldosterone concentrations more clearly separated cats with and without PHA than did the UACR. 5 Additionally, urine collection from cats in a hospital setting typically requires cystocentesis, which can be difficult with fractious patients or patients with insufficient bladder size, and collection at home can be cumbersome or impossible, especially in multi-cat households. Finally, the protocol evaluated requires twice daily medication for 4 days.5,6 As cats can be difficult to medicate orally, a test requiring a shorter treatment duration would be advantageous.
Fludrocortisone possesses both mineralocorticoid and glucocorticoid activity. Compared with the indirect suppression of mineralocorticoid secretion caused by fludrocortisone administration, the glucocorticoid activity of fludrocortisone could directly inhibit adrenocorticotrophic hormone (ACTH) secretion either by affecting the pituitary or hippocampus, ultimately causing hypocortisolemia, as occurs in humans.3,13,18 Abrupt cessation of administration after 4 days could, therefore, cause clinical signs of cortisol deficiency. To our knowledge, the potential effect of fludrocortisone administration on serum cortisol concentration in cats has not been previously reported.
We therefore conducted a pilot study in healthy cats to identify a potential alternative protocol for a fludrocortisone suppression test. The objectives were 1) to determine if monitoring of serum aldosterone concentration could be used instead of urine aldosterone concentrations in a fludrocortisone suppression test; 2) to define the number of days of fludrocortisone administration required for suppression of serum aldosterone concentrations; and 3) to determine if fludrocortisone administration suppressed cortisol secretion. Our hypothesis was that a mineralocorticoid suppression test measuring serum in place of urine aldosterone concentrations would allow for a shorter duration of fludrocortisone administration, and thus a more convenient protocol for a suppression test. Additionally, we hypothesized that fludrocortisone administration would suppress endogenous cortisol concentration based on the fact that this steroid has both glucocorticoid activity in addition to mineralocorticoid activity in humans.
Materials and methods
Cats
The study was carried out in 2 phases. Phase I was a blinded, crossover, placebo-controlled study using healthy, adult laboratory cats. Phase II was a blinded, placebo-controlled study using healthy client-owned cats. Animal use was approved by the Auburn University Institutional Animal Use and Care Committee. All client-owned cats were used with client consent.
Phase I
Health was ensured in 8 adult intact female cats via physical exam, serum biochemistry, complete blood count, and urinalysis within 1 week prior to study initiation. Median weight of the cats was 2.9 kg (range: 2.4–3.6 kg), and the median age was 2.7 years (range: 2.2–3.7 years). Aliquots of serum and urine were saved for baseline aldosterone and urinary creatinine assays. Cats were randomly assigned to receive either fludrocortisone acetate (group F) or placebo (group P). Per a previously evaluated protocol for a fludrocortisone suppression test,
6
fludrocortisone acetate
b
(0.05 mg/kg every 12 hr orally) was administered to cats in group F; cats in group P received placebo. Both were given at approximately the same time each day (8:30
Phase II
Thirteen clinically healthy client-owned neutered cats >1 year old, weighing >3 kg, were recruited. Baseline physical exam, serum biochemistry, complete blood count, urinalysis, and blood pressure measurement were performed to ensure health. Aliquots of the samples were saved for baseline aldosterone and urinary creatinine assays. Exclusion criteria included evidence of cardiac, liver, or renal disease; presence of hypertension (defined as systolic blood pressure >180 mm Hg as measured by Doppler sphygmomanometry); or being too fractious to be handled. Five cats were excluded: 2 because of temperament, 1 because of detection of azotemia and proteinuria, and 2 because of detection of liver enzyme activity above the reference interval, one of which also had glucosuria. Of the 8 cats enrolled, 2 were spayed females and 6 were neutered males. The median weight was 5.8 kg (range: 4.4–7.7 kg). Age was known for 7 cats; median age was 3 years (range: 1–10 years). The other cat was a young adult.
Based on the results of phase I, a dosing scheme for fludrocortisone of 0.05 mg/kg every 12 hr orally for 3 doses was chosen. Cats were assigned to 1 of 2 groups in alternating fashion as they were admitted into the study, with 1 group receiving fludrocortisone acetate and the other receiving placebo sugar capsules (every 12 hr orally). Both were given at approximately the same time each day (8:30
Sample collection
Phase I
Cats were sedated with dexmedetomidine c (40 µg/kg intramuscularly) for sample collection, and sedation was reversed with an equal volume of atipamezole d intramuscularly once collection was complete. Serum was collected daily on days 2–4 for measurement of aldosterone, cortisol, sodium, and potassium concentrations, and urine was collected daily for measurement of UACR. Sample collection was performed 4 hr after the morning administration of fludrocortisone or placebo. Blood was collected from a jugular vein, and urine was collected via manual expression of the bladder. Blood was allowed to clot and was centrifuged within 1 hr of collection, and the serum was separated. Urine was centrifuged within 1 hr of collection and the supernatant removed. Prior to storage, an aliquot of urine to be used for aldosterone measurement was mixed with boric acid as a preservative for storage per manufacturer’s recommendations. All samples were stored at −80°C until assayed. Samples for measurement of urinary and serum aldosterone were stored a maximum of 5.5 months and 1 month, respectively. Samples for measurement of cortisol were stored for a maximum of 7 months.
Phase II
A blood sample was drawn 4 hr after administration of the third dose of fludrocortisone or placebo for measurement of aldosterone and cortisol concentrations. Urine was not collected for measurement of UACR based on the results in phase I. Chemical restraint was not used, and blood was collected via jugular venipuncture. After collection, the blood was allowed to clot and was centrifuged within 1 hr. The serum was separated and stored at −80°C until assayed. No sample was stored for longer than 1 month.
Sample analysis
Serum aldosterone concentration was measured using a radioimmunoassay e previously validated for use in cats. 24 Urine aldosterone was measured using the same kit as previously reported. 22 Acid hydrolysis with hydrochloric acid to extract aldosterone metabolites from urine was not performed as per manufacturer instructions, as it does not significantly increase the recovery of aldosterone in feline urine. 22 Use of the kit for measuring urine aldosterone without acid hydrolysis was validated in the present study by determining the intra- and interassay coefficient of variation, which were 4.1% and 7%, respectively. Dilutional parallelism was also demonstrated by measuring the aldosterone concentration of serially diluted samples. The sensitivity of the aldosterone assay was 69 pmol/L. Serum cortisol concentration was measured using a radioimmunoassay f previously validated for use in cats. 12 The sensitivity of the assay was 5 nmol/L. 12 All samples were run in duplicate. Serum samples were run separately from urine samples; however, all serum samples from each phase were assayed in a single batch, as were all urine samples. Serum sodium and potassium and urine creatinine concentrations were measured by an automated analyzer. g The UACR was calculated as the ratio of urinary aldosterone (pmol/L)/urine creatinine (mg/dL).
Statistical analysis
A statistics package
h
was used for statistical analysis. Descriptive statistics were calculated. Continuous data are reported as mean and standard deviation, unless not normally distributed, in which case median and range are provided. For values below the detection limit, the mean between 0 and the detection limit was entered for statistical analysis (i.e., 35 pmol/L and 3 nmol/L for aldosterone and cortisol, respectively). As we had no expectation of what the concentrations below the detection limit would be, we chose the 50% mark; likely some concentrations, if they had been measurable, would have been higher and some lower. Significance was set at the
Phase I
Sample size was calculated prior to the study in order to detect a 50% difference in peak aldosterone concentration using the assumptions of a type I error of 0.05 and a power of 0.8 with an accepted error of 0.1. Data for serum and urine aldosterone as well as serum cortisol were nonparametric. Serum aldosterone and cortisol concentrations and UACR values were compared within groups over time using a repeated measure analysis of variance (ANOVA) on ranks. A post-hoc analysis was performed using the Tukey test when a statistically significant difference was identified. Serum concentrations of aldosterone and cortisol on the first day of administration of fludrocortisone or placebo were compared to those of the first day after the crossover using a signed rank test. Data for serum sodium and potassium concentrations were parametric. Results were compared within and between groups using a repeated-measures ANOVA. Data are given as median (range).
Phase II
Data were parametric. The mean serum aldosterone and cortisol concentrations were compared within groups using a 1-tailed paired
Results
Serum aldosterone concentration
Phase I
Baseline aldosterone concentration was not significantly different between groups (443 pmol/L [277–744] for group F and 376 [61–662] pmol/L for group P). Serum aldosterone concentrations were not significantly different between day 1 and the first day after the crossover (data not shown). In group F, the serum aldosterone concentration changed significantly over time (
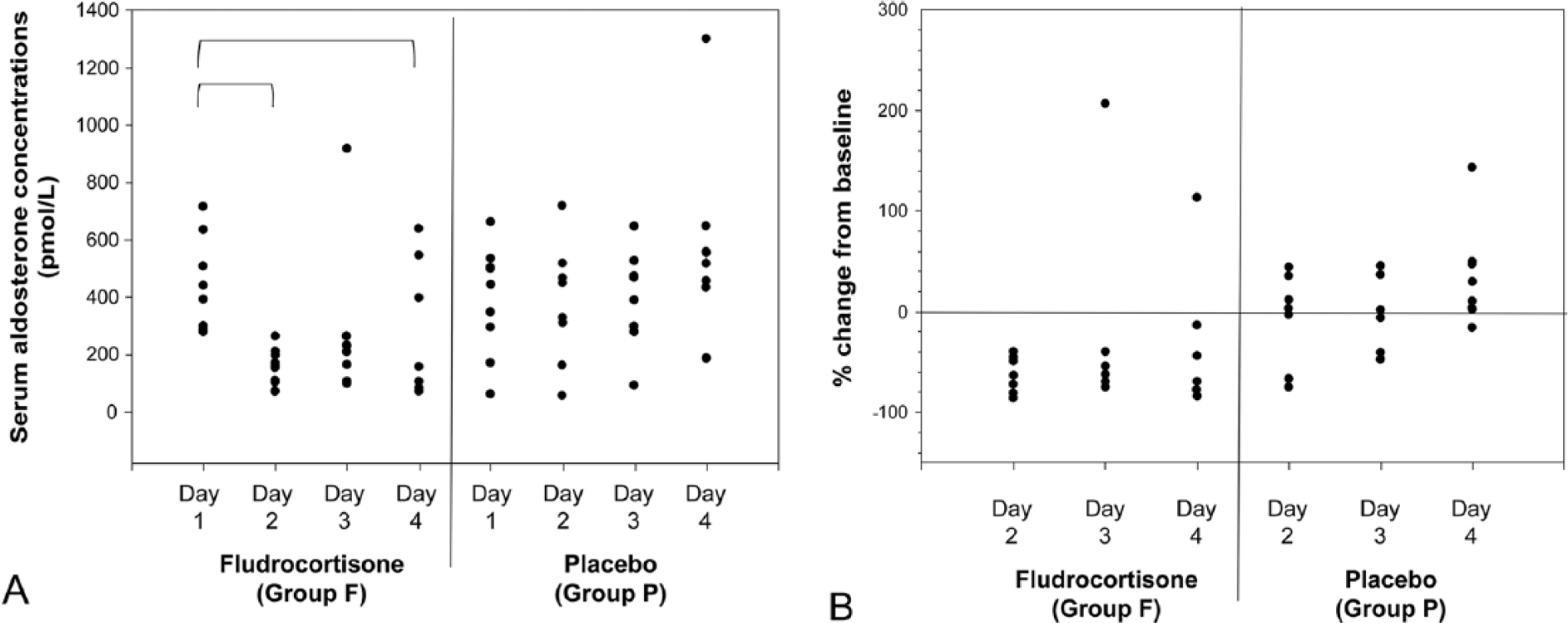
Phase II
One cat in the fludrocortisone-treated group had a baseline serum aldosterone concentration below the sensitivity of the assay. As a diagnosis of PHA would be ruled out in a cat with such a baseline serum aldosterone concentration and the degree of suppression in response to fludrocortisone was impossible to determine, the cat’s data were removed from statistical analysis. Thus, this group included data from 3 cats.
Baseline serum aldosterone concentration was 175 ± 82 pmol/L in cats treated with fludrocortisone and 202 ± 106 pmol/L in cats treated with placebo. The post-treatment serum aldosterone concentration measured 4 hr after the third dose of fludrocortisone was below the sensitivity of the assay in the cats treated with fludrocortisone, and 116 ± 53 pmol/L in cats treated with placebo. The decrease in aldosterone concentration in the fludrocortisone-treated cats was statistically significant (
Urine aldosterone concentration and aldosterone-to-creatinine ratio
Although UACR appeared to decrease over time, no significant change in UACR was detected at any time point for cats treated with fludrocortisone (
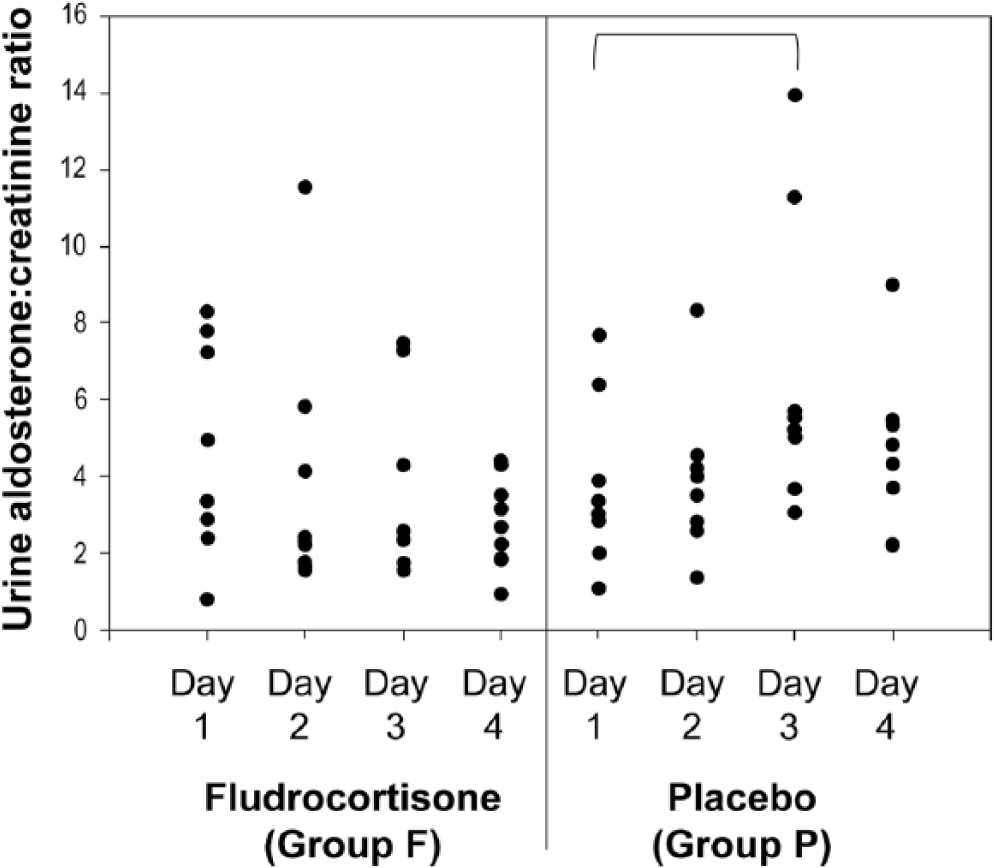
Urine aldosterone-to-creatinine ratio in 8 cats treated with placebo and fludrocortisone for 4 days. Bracket indicates significant difference between the 2 days.
Serum sodium and potassium concentrations
No significant change in serum sodium or potassium was detected in either treatment group at any time point compared to baseline in phase I (for cats treated with fludrocortisone:
Serum cortisol concentration
Phase I
Baseline cortisol concentration was not significantly different between groups. No significant difference was detected between cortisol concentrations after the crossover compared with the start of phase I. Baseline serum cortisol concentrations were 51 (13–96) nmol/L and 58 (30–143) nmol/L for group F and P, respectively. Serum cortisol concentration decreased significantly (
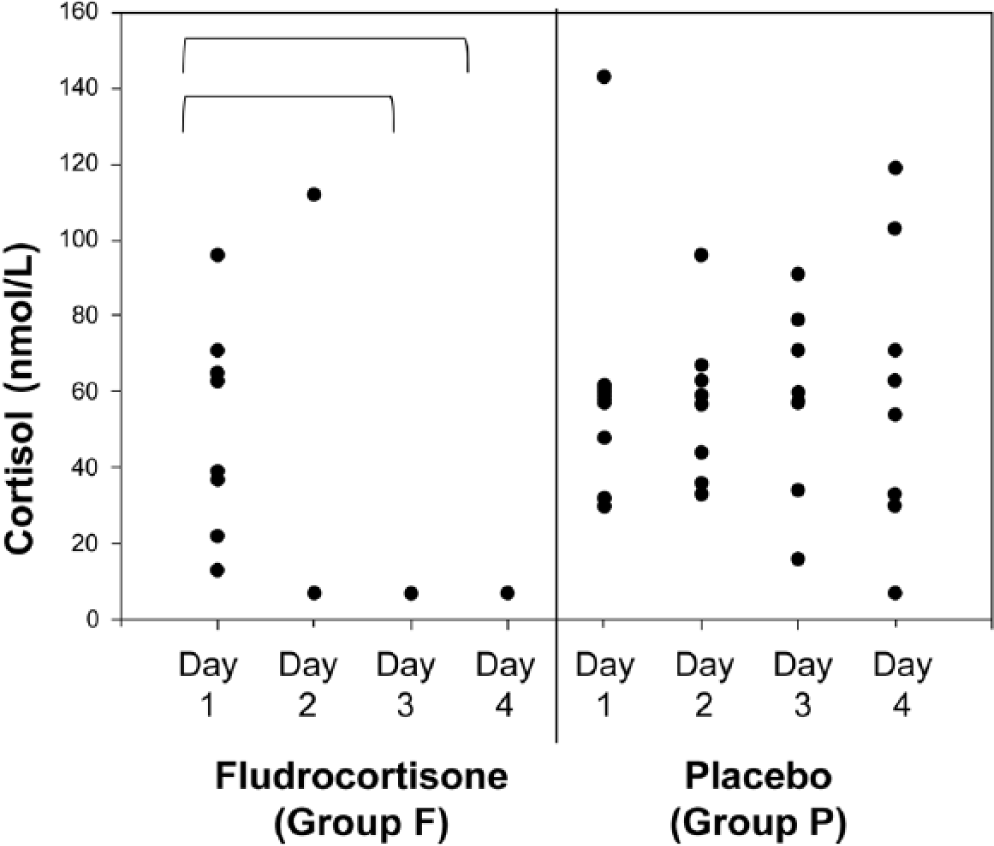
Serum cortisol concentration in 8 cats treated with placebo or fludrocortisone for 4 days. Brackets indicate significant difference between the 2 days.
Phase II
Baseline cortisol concentration was 141 ± 106 nmol/L in cats treated with fludrocortisone and 296 ± 34 nmol/L in placebo-treated cats. Post-treatment cortisol concentration was 10 ± 7 nmol/L and 202 ± 101 nmol/L in cats treated with fludrocortisone and placebo, respectively. The decrease in cortisol concentration was significant in cats treated with fludrocortisone (
Discussion
The present study demonstrates that oral administration of fludrocortisone acetate (0.05 mg/kg every 12 hr) causes significant suppression of serum aldosterone concentration in healthy cats. Furthermore, maximal suppression occurs on day 2 of treatment, after 3 doses have been administered, although suppression was also significant on day 4. On days 2 and 4, 8 out of 8 and 6 out of 8 cats had >40% suppression of serum aldosterone concentration, respectively, compared with baseline. The lack of significance in serum aldosterone suppression on day 3 may be due to a single cat having an increase in aldosterone concentration on day 3. The aldosterone concentration decreased by 51% between baseline (299 pmol/L) and day 2 (152 pmol/L) in this cat; the aldosterone remained above baseline on day 4 (638 pmol/L). As the samples were assayed in duplicate and results were similar between the 2 tubes, the values from days 3 and 4 are unlikely to be laboratory error. The cause of the increase in aldosterone concentration despite continued fludrocortisone administration is unexplained. Diet remained unchanged in the laboratory cats; vomiting or diarrhea (i.e., causes of dehydration that could increase serum aldosterone concentration), were not observed.
A previous study determined that UACR was significantly suppressed after 4 days of treatment with 0.05 mg/kg fludrocortisone every 12 hr. 6 In comparison, in our study, UACR was not significantly suppressed on any day in phase I. The difference between the studies potentially may be explained by disparities between the assays used or omission of the hydrolysis with hydrochloric acid and extraction steps in the current study. Acid hydrolysis allows measurement of aldosterone-18-glucuronide, a major metabolite of aldosterone in human urine. 22 However, as compared to humans, acid hydrolysis of urine did not significantly affect recovery of urine aldosterone in cats. 22 Therefore, omission of the acid hydrolysis and extraction steps in our study is unlikely to account for the difference. Given the trend in decreasing UACR over time in our study, it may have been underpowered to detect a difference; the previous study used 15 cats. 6 As no effect of fludrocortisone on UACR was found in phase I, it was not evaluated in phase II.
Urine has been proposed as the ideal sample to reflect the average aldosterone concentration over time so as to avoid difficulties in interpretation of variation throughout the day. 6 However, urine can be difficult to obtain from cats in the clinical setting if the owner is unable to keep their cat from urinating prior to an appointment, and collection of urine at home can be difficult or cumbersome for some clients. In fact, all of the client-owned cats used in phase II of the current study had insufficient urine for collection on presentation for their initial evaluation, requiring them to stay in the hospital for several hours until a sample could be obtained. Difficulty in urine collection would be particularly troublesome if a urine sample must be collected at a specific time following drug administration. Using serum for the test sample allows clinicians to obtain a sample at any time.
Phase II was performed in order to determine if the findings of phase I, which used laboratory cats, would be similar in client-owned cats and to ensure that the use of dexmedetomidine in phase I did not affect the results. Dexmedetomidine significantly decreases blood pressure in cats, 16 which, in turn, can theoretically increase aldosterone concentration through activation of the RAAS. Significant reduction in blood pressure, however, does not occur until 90 min after dexmedetomidine administration, 16 which was much longer than the time it took to collect blood and urine samples in our laboratory cats. Given that the findings of the 2 phases were similar, the findings in phase I appear valid.
Cats were monitored for adverse effects of fludrocortisone administration including changes in serum potassium and sodium concentrations. In 19 cats with PHA or with hypertension with or without hypokalemia not due to PHA, fludrocortisone administration (0.05 mg/kg every 12 hr for 4 days) minimally affected blood pressure. Hypokalemia, however, was exacerbated or induced in 7 cats, leading to muscle weakness in 1 cat. 5 In the current study, no significant change was noted in serum sodium and potassium concentrations; nor did any cat develop hypernatremia or hypokalemia. Blood pressure measurements were attempted, but the fractious nature of the laboratory cats and need for sedation for blood collection made it impossible to obtain reliable measurements. Although no adverse effects were noted in healthy cats, the safety of fludrocortisone administration to cats with PHA or other conditions such as renal disease cannot be determined from our study. As the cats in the previous study 5 were treated with fludrocortisone for 4 days, a suppression test with fewer doses of fludrocortisone may reduce the potential for such side effects.
In humans, fludrocortisone administration significantly decreases ACTH and cortisol secretion.3,13,18 Thus, we sought to assess the effects of fludrocortisone on cortisol secretion in cats. Suppression of cortisol by fludrocortisone with abrupt withdrawal at the end of the test could lead to signs of cortisol deficiency in the same manner as abrupt cessation of glucocorticoid therapy. Oral fludrocortisone administration significantly suppressed basal serum cortisol concentration on days 3 and 4 in phase I. Furthermore, in phase I, serum cortisol concentration in all but 1 cat on day 2 was below the sensitivity of the assay; however, because of the low number of cats and a reduced statistical power, the concentrations on day 2 were not significantly different from baseline. In phase II, serum cortisol concentration was significantly suppressed after only 3 doses of fludrocortisone. Thus, in the majority of cats, significant cortisol suppression occurs with 3 doses of fludrocortisone. The full extent of suppression cannot be determined, as ACTH stimulation tests were not performed. However, as the cats were in a stressful situation (i.e., in a clinic having blood drawn), cortisol concentrations would be expected to be elevated. Thus, at the least, a relative cortisol deficiency was present. As the baseline cortisol concentrations after the washout period in phase I were not significantly different from those at the start of phase I and all were measurable, adrenal cortisol secretion recovered quickly (i.e., by 4 days after cessation of fludrocortisone administration [0.05 mg/kg every 12 hr orally for 4 days]).
The effect of fludrocortisone on cortisol secretion in cats with PHA may depend on whether there is concurrent hyperadrenocorticism. Two cats were reported to have concurrent hyperprogesteronism in addition to hyperaldosteronism.2,4 To our knowledge, there are no reports of cats having concurrent hyperaldosteronism and hypercortisolism, but this is possible in theory. In such a case, fludrocortisone is unlikely to affect cortisol concentrations because cortisol secretion would presumably be autonomous.
In both phases of the study, up to 25% of the cats in the placebo group had a >40% suppression of serum aldosterone concentration on day 2. This is likely a result of the fluctuation in serum aldosterone concentration that occurs throughout the day. The laboratory cats in phase I were fed a consistent diet, but access to water or food could have been affected via competition, as the cats were housed in a group. Client-owned cats would potentially have access to other pets’ food, treats, or table scraps, which could affect aldosterone concentrations. Random fluctuation may be less important in cats being tested for PHA as compared with the healthy cats used in this pilot study. Cats with PHA are expected to be resistant to feedback, 5 and >40% suppression of aldosterone concentration is unlikely. Cats with PHA and those suspected to have PHA need to be studied for verification. As a pilot study, our study was not intended to prove utility of the fludrocortisone suppression test as a definitive test for diagnosing PHA. Rather, it was intended to determine if a suppression test was feasible, in particular, using serum as a substrate for measuring aldosterone. Further research using clinically affected cats will be needed to determine utility of the test, including the likelihood of obtaining false results.
The results of our pilot study suggest that an oral fludrocortisone suppression test may be a useful diagnostic tool, and if serum is used for measurement, a shorter protocol than previously reported5,6 may be possible. However, further study is needed. First, the findings of our pilot study should be confirmed in cats with PHA as well as in those with a history and biochemical changes suggestive of PHA but that do not have the disease. Second, the basal aldosterone concentrations were higher in phase I than in phase II, potentially because of a difference in diet between laboratory and client-owned cats. Thus, the effect of diet on aldosterone concentrations and suppression by fludrocortisone should be evaluated. It is likely important that the diet remain unchanged during the course of a fludrocortisone suppression test so sodium intake remains consistent. Finally, in order to optimize the utility of a fludrocortisone suppression test, clear indications for performing such test must be determined. In people, indications for screening for PHA include the following: concurrent hypertension and hypokalemia; severe hypertension; treatment-resistant hypertension; hypertension in combination with the finding of an incidental adrenal mass; early onset hypertension; and in patients being evaluated for secondary hypertension such as renovascular disease or pheochromocytoma.15,23 Similarly, potential indications for screening in cats could include: hypokalemia concurrent with hypertension or azotemia, the finding of an incidental adrenal mass, and the presence of refractory hypertension.
In conclusion, we found that oral fludrocortisone acetate administration significantly suppresses basal serum aldosterone concentrations in healthy cats after 3 doses (0.05 mg/kg every 12 hr). Suppression to <40% of baseline occurred on day 2 in 8 out of 8 laboratory cats and in 3 out of 3 healthy client-owned cats. Cortisol concentration was significantly suppressed by fludrocortisone administration, but the concentrations returned to baseline within 4 days (i.e., the end of the washout period). No other adverse effects were noted. Thus, an oral fludrocortisone suppression test may have applicability in the diagnosis of PHA. More specifically, the use of serum as the test substrate may allow for a shorter fludrocortisone treatment period and may be a preferred alternative to urine. Further studies in cats with renal disease, hypertension, and aldosterone-secreting tumors are needed to assess the ideal duration of fludrocortisone administration and to confirm the test’s ability to differentiate between PHA and other conditions associated with RAAS activation.
Footnotes
Acknowledgements
We gratefully acknowledge Drs. Nancy Cox and Doug Martin of the Scott-Ritchey Research Center for providing resources for the project.
a.
Ohio State University Wexner Medical Center University Reference Laboratories, Columbus, OH.
b.
IMPAX Laboratories Inc., Hayward, CA.
c.
Dexdomitor, Zoetis Inc., Florham Park, NJ.
d.
Antisedan, Zoetis Inc., Florham Park, NJ.
e.
Coat-A-Count aldosterone, Siemens Healthcare Diagnostics, Los Angeles, CA.
f.
Coat-A-Count cortisol, Siemens Healthcare Diagnostics, Los Angeles, CA.
g.
Hitachi 911, Boehringer Mannheim Corp., Indianapolis, IN.
h.
SigmaPlot 12, Systat Software Inc., Chicago, IL.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded in part by the Nestle Purina Corp.
