Abstract
A 2.5-year-old, male, neutered cat presented with a 5-month history of progressive right hind limb lameness and an enlarged right popliteal lymph node. Radiographs revealed significant bony lysis of the tarsus and distal tibia, and fine-needle aspirate of the bone lesion and lymph node revealed a neoplastic population of cells with uncertain origin. Amputation was elected, and the mass was submitted for histology and cellular culture for better characterization. Histologic examination revealed a mixture of spindle-shaped cells and larger, round to polygonal cells. All cells were immunoreactive for vimentin, and only the larger polygonal cells were also positive for cytokeratin. All cells were negative for desmin, smooth muscle actin, cluster of differentiation (CD)3, CD18, CD79a, macrophage antibody (MAC)387, and glial fibrillary acidic protein. Cultured neoplastic cells failed to express CD18, and were not able to secrete the pro-inflammatory cytokines tumor necrosis factor-α, interleukin-1 (IL-1)β, and IL-6 when stimulated by lipopolysaccharide, disproving that the cells originated from the macrophage or monocyte line. Ultrastructurally, neoplastic cells were characterized by abundant rough endoplasmic reticulum, interdigitating cellular processes, and membrane condensations. Based on location and cytologic, histologic, ultrastructural, and functional studies, this neoplasm was considered a synovial cell sarcoma.
A neoplastic process involving the joint can arise from any of the several structures that compose the joint or are present in its close proximity. The World Health Organization classification of bone and joint tumors lists synovial sarcoma (SS) as the only primary joint neoplasm. 26 Classification of SSs in animals has mostly been extrapolated from the human medical literature 19 ; however, controversy exists on the origin of SSs in humans. The name “synovial sarcoma” stems from the early literature as a result of the frequent periarticular location and the microscopic resemblance to developing synovium. 9 Despite the name, immunohistochemical and ultrastructural studies have shown enough differences between normal synovium and SSs to disprove the synovial origin of the tumor.17,24 The most evident difference is that both the epithelial and spindle cell elements of SS show reactivity for cytokeratin 9 and vimentin by immunohistochemical staining (IHC), 24 while normal synovial lining cells display strong vimentin positivity but fail to bind antibodies against cytokeratin. 17 In addition, many human SSs occur at sites distant from joints. 9 For this reason, the tissue origin of this tumor remains uncertain. 5
Synovial sarcomas are described as a mixture of 2 morphologically different cell populations: epithelial and spindle cell. Synovial sarcomas are classified in 1 of 3 ways depending on the cellular composition of the tumor. Monophasic SSs are almost entirely composed of 1 cell type, biphasic are composed of a mixture of the 2 cell types, and a classification of “poorly differentiated synovial sarcoma” is used when it is impossible to distinguish between the 2 cell types. 9 While similarities between SSs in animals and humans have been noted, discrepancies in the suspected tissue of origin exist. In animals, SSs are still believed to arise from 1 of the 2 cell types present in the synovium,6,20,29 even though the cytokeratin and vimentin dual positivity remains unexplained. Similar to humans, SSs in animals are often described as a mixture of synovioblastic and/or epithelioid and fibroblastic (spindle-cell) populations. 20 The relative proportion of each type can vary between tumors and even within the same tumor, 29 and some suggest that the different cell morphologies may reflect regional pleomorphism of malignant tumors rather than a biphasic cell population.20,29 The mitotic rate and the presence of bi- and multinucleated cells are highly variable. Individual cells may be surrounded by an eosinophilic matrix resembling osteoid, 29 perhaps explaining the origin of the matrix noted by cytology. Cavities or cleft-like spaces containing proteinaceous fluid or mucoid material and lined by a poorly defined layer of epithelioid cells may form within sheets of tumor cells.20,29 In cats, SSs have been rarely reported.3,7,10,12,13,15,18,25,28 The diagnosis of SS is challenging, given the similarities to other tumors. IHC, especially for cytokeratin, has been used to distinguish this tumor from others, such as histiocytic sarcoma, which can resemble the epithelioid component of SS. 19 Given the existing confusion regarding primary synovial tumors in domestic animals, our study responds to the need of a more in-depth characterization of tumors arising from the joint. 19
A 2.5-year-old, male, neutered cat was presented to the University of Georgia Veterinary Teaching Hospital (Athens, Georgia) with a 5-month history of progressive right hind limb lameness. At presentation, the cat had severe, non–weight-bearing, right hind limb lameness and notable instability of the right tarsus and distal tibia. The limb lesion was painful when palpated. Enlargement of the right popliteal lymph node was also noted. Radiographically, there was severe bony lysis of the tarsus and distal tibia, with fracture and luxation of the tarsocrural joint, as well as focal lytic lesions throughout the tibia (Fig. 1). Fine-needle aspirates of the bone lesion and of the enlarged lymph node were submitted for cytologic review. Aspirates of the lytic bone lesion and of the popliteal lymph node were applied on glass slides, routinely stained with modified Romanowsky stain, and reviewed by a board-certified, clinical pathologist. Cytologically, exfoliated cells were present in moderate quantities as individualized cells and in loose, cellular aggregates. These cells were large, pleomorphic, and had ill-defined, stellate to round to fusiform borders. The nuclei were central, round to ovoid to angular, and had finely stippled chromatin and single to multiple, large, prominent nucleoli. The abundant cytoplasm was moderately basophilic and occasionally contained vacuoles. This aberrant, pleomorphic population demonstrated strong criteria for malignancy including: bi- and multinucleation; prominent nucleoli that approached the same size as an erythrocyte; oddly shaped, multiple nucleoli; variable nucleus-to-cytoplasm ratios; marked anisocytosis and anisokaryosis; and occasional mitotic figures. Rarely, extracellular, eosinophilic matrix was associated with the cells. Aspiration of the popliteal lymph node revealed the presence of similar cells admixed with large numbers of lymphocytes (Fig. 2). A diagnosis of metastatic neoplasia was made, and differential diagnoses included the following: sarcoma of uncertain origin (which could include SS and osteosarcoma), histiocytic sarcoma, anaplastic carcinoma, and amelanotic melanoma.
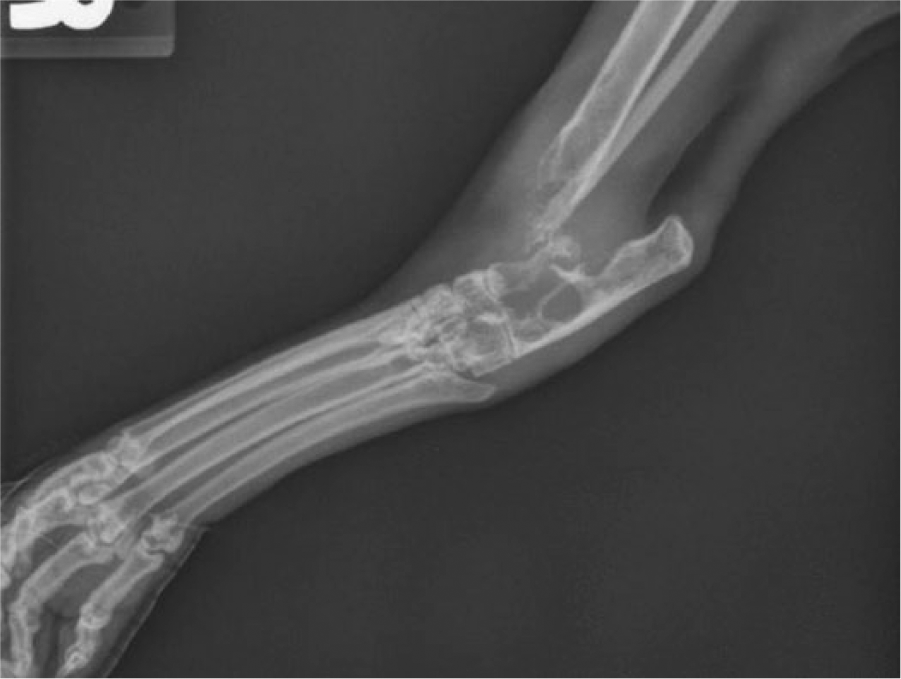
Joint tumor; cat. Radiograph illustrating the aggressive polyosteolytic lesion centered on the tarsocrural joint.
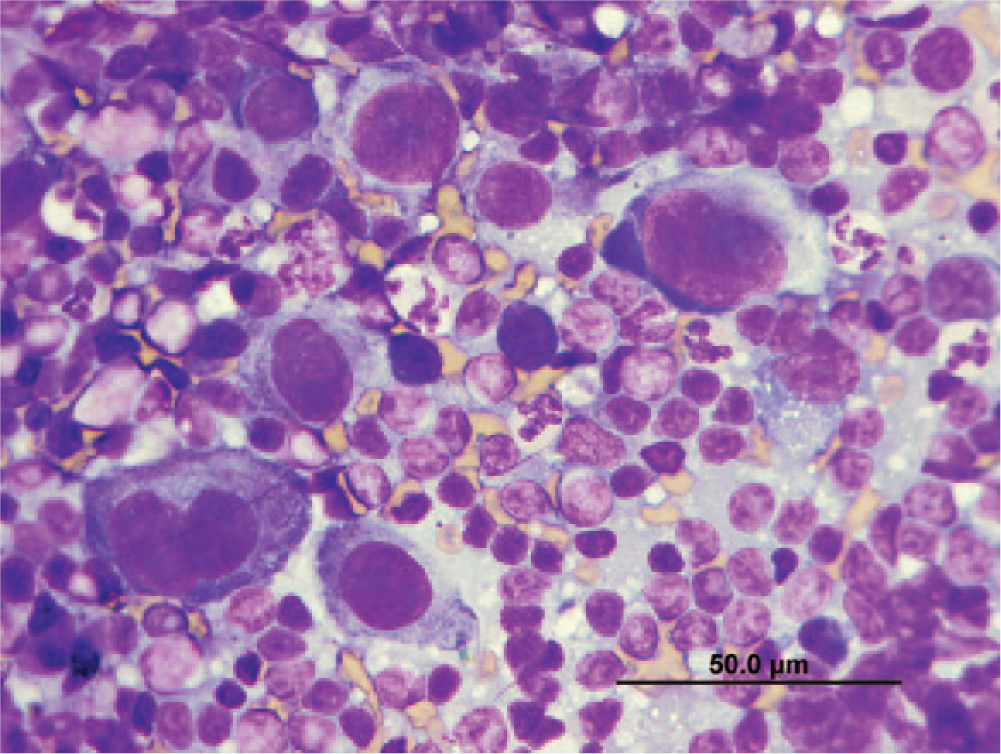
Popliteal lymph node cytology; cat. Among small mature lymphocytes and a few neutrophils are large, pleomorphic cells with ill-defined, round to polygonal borders and abundant cytoplasm occasionally containing small vacuoles. The nucleus is central, round to ovoid, and has finely stippled chromatin and 1 or 2 indistinct nucleoli. Modified Romanowsky stain. Bar = 50 µm.
Limb amputation was elected by the owners, and the leg was submitted for histology and IHC. On gross examination, the tarsocrural joint was mildly swollen because of periarticular proliferation of pale, firm, flexible, fibrous tissue that infiltrated and expanded the distal tibia and fibula and crossed the joint space to infiltrate the tarsal bones. A section of the pale tissue was collected for cell culture. A section of formalin-fixed tissue, and cells from the established cell line, were also submitted for electron microscopy. Formalin-fixed, paraffin-embedded tissue from the lesion was cut at 5 μm thickness and stained with hematoxylin and eosin stains. Histologic examination of the joint revealed that almost all of the preexisting bone had been replaced by cells of 2 different morphologies: proliferating spindle cells (Fig. 3) and foci of large pleomorphic cells (Fig. 4) that resembled those seen on the cytologic preparation. The spindle cells were thin with a small, ovoid nucleus. The cells were arranged in short, interweaving bundles and infiltrated the surrounding tendons. The larger cells were present in small, solid foci or randomly spread throughout the spindle cells, and they were occasionally dispersed by hyaline material. The larger cells were characterized by abundant, eosinophilic cytoplasm that was occasionally vacuolated around the nucleus, and large, round, irregular nuclei with atypia. The nuclei were hyperchromatic with 1 or 2 large nucleoli. Anisocytosis and anisokaryosis were marked, and many bi- and trinucleated cells were noted. Occasional cells with abundant eosinophilic cytoplasm and several, round to oval, relatively uniform nuclei were noted. These cells were interpreted to be osteoclasts. Foci of necrosis surrounded by hemosiderin-laden macrophages and multifocal infiltration by lymphocytes were also present. The popliteal lymph node was multi-focally replaced by 2 populations of cells similar to those observed in the joint and by fine-needle aspiration cytology. At this time, differentials included SS or histiocytic sarcoma.
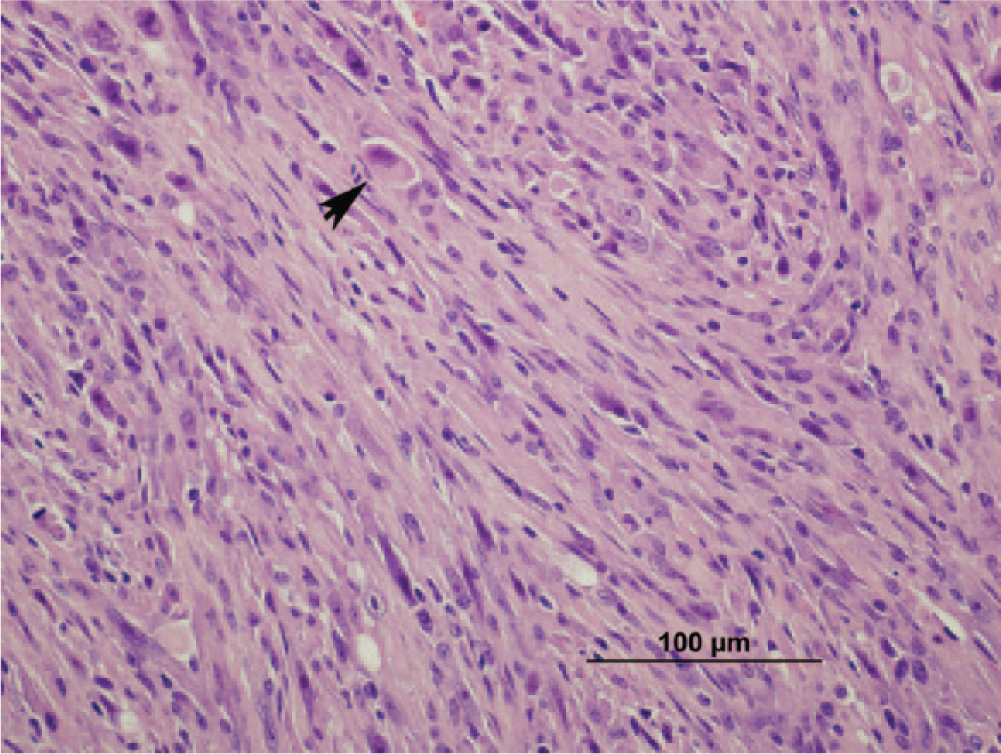
Joint tumor; cat. Interweaving bundles of plump, spindle-shaped cells. Small numbers of large cells are also present (arrow). Hematoxylin and eosin. Bar = 100 µm.
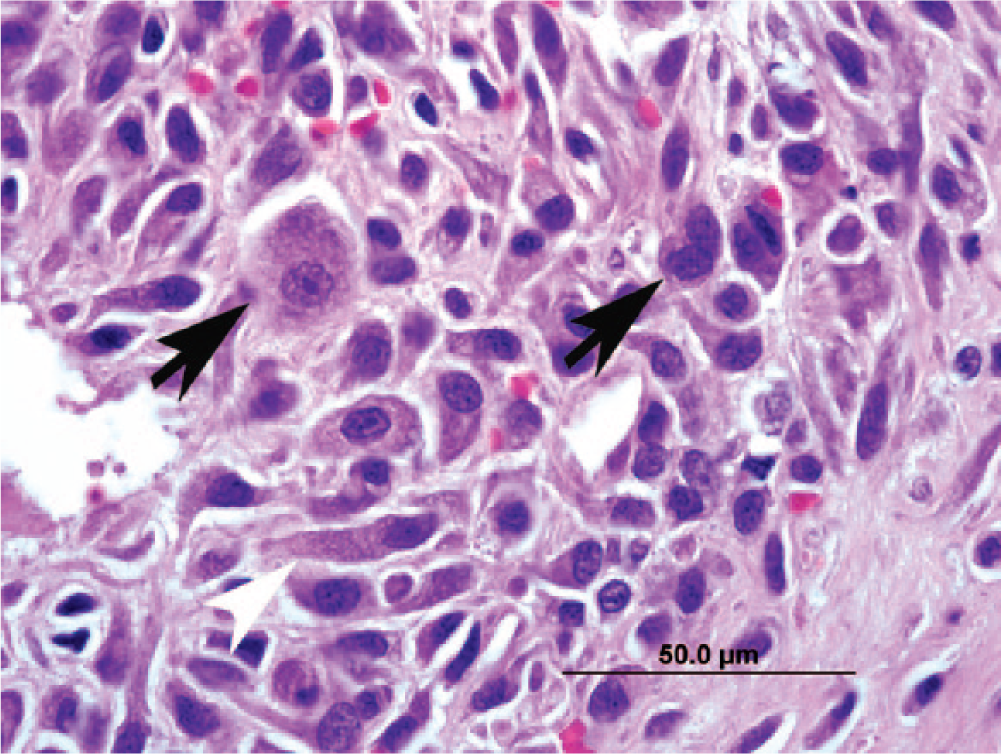
Joint tumor; cat. An aggregate of large pleomorphic cells (arrows) similar to those noted in the cytologic specimen are present next to more spindle-shaped cells (white arrowhead). Hematoxylin and eosin. Bar = 50 µm.
To better identify the origin of the neoplastic cells, IHC was performed (Fig. 5; Table 1). Healthy synovial membrane from another cat with no clinical or gross evidence of joint disease was collected during a routine postmortem examination and used as a control. Serial sections were deparaffinized and rehydrated for routine IHC to detect expression of cluster of differentiation (CD)3 a (1:800), CD79a b (1:50), glial fibrillary acidic protein c (GFAP; 1:800), desmin d (1:10), smooth muscle actin e (SMA; 1:500), macrophage antibody (MAC)387 f (1:500), CD18 g (1:50), vimentin h (1:600), and cytokeratin A1/A3 i (1:200). Antigen retrieval methods for CD3, CD79a, CD18, MAC387, desmin, GFAP, and vimentin consisted of heat-induced epitope retrieval (HIER) using citrate buffer at a pH of 6.0. j Antigen retrieval methods for cytokeratin A1/A3 consisted of proteinase K k treatment. Endogenous peroxidase was blocked using 3% hydrogen peroxide. l Protein blocking was performed using Power Block. m As a negative control, the primary antibody was eliminated and substituted with purified rabbit immunoglobulin n (for CD3) and purified mouse immunoglobulins o (for CD18, CD79a, SMA, MAC387, GFAP, and desmin). The substrate chromogen system used was 3,3′-diaminobenzidine. The tissue sections were counterstained with Gills II hematoxylin and bluing, dehydrated sequentially in 70%, 95%, and 100% ethanol, cleared in xylene, and mounted with xylene-based mounting medium. Immunostaining for CD18, MAC 387, CD3, and CD79 were used to identify leukocyte origin, desmin and SMA for muscle origin, and GFAP for perivascular wall origin. While all of the neoplastic cells had cytoplasmic immunoreactivity for vimentin, only some of the larger cells demonstrated a double positivity for vimentin and cytokeratin, and all other markers were negative (Fig. 5). CD18 IHC, which is not shown in the figure because of high background staining, was also considered negative relative to positive and negative controls. IHC results are summarized in Table 1.
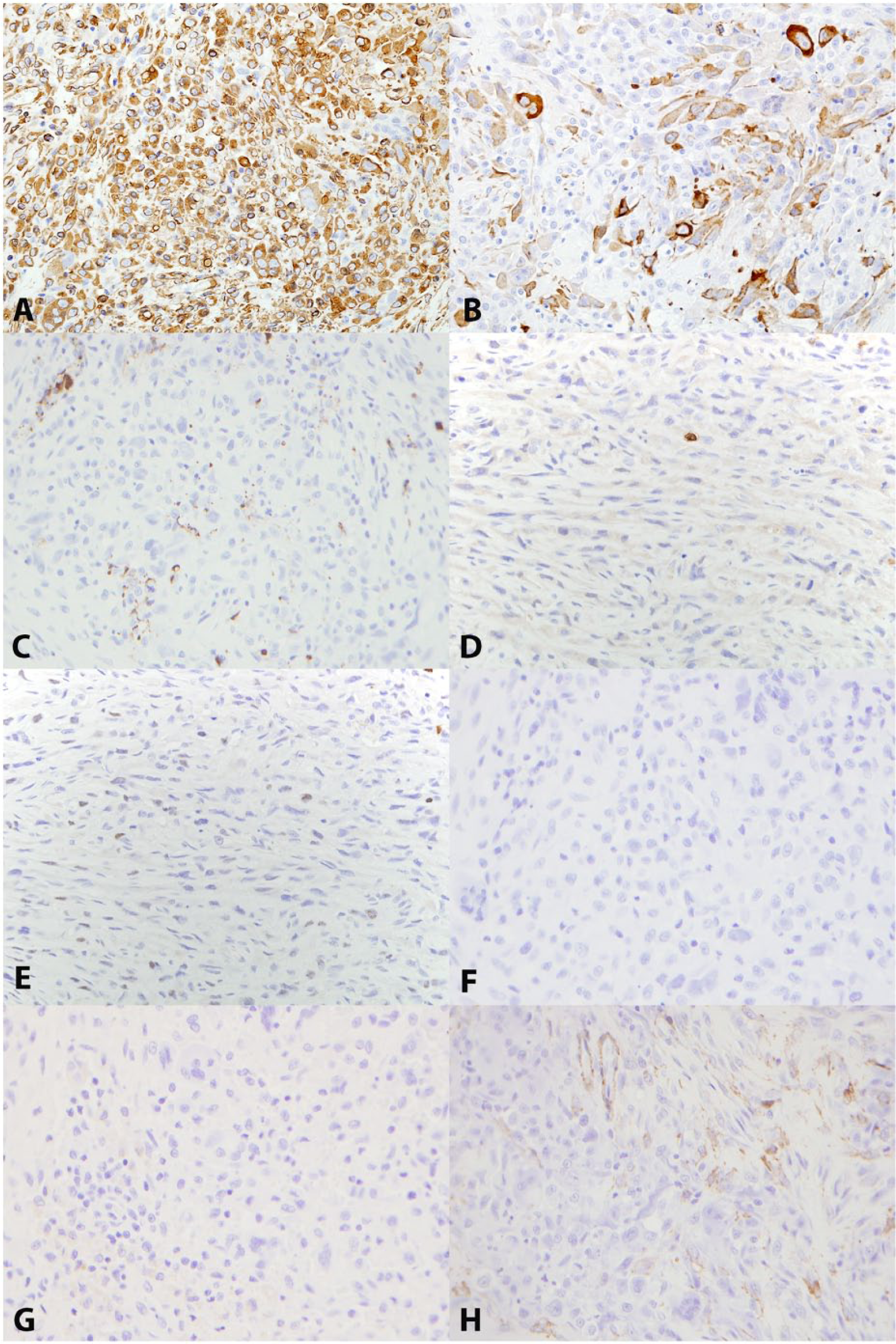
Joint tumor; cat.
Immunohistochemical staining (IHC) performed on the formalin-fixed tissue obtained from the mass, the tumor cell line, and a section of healthy synovium obtained from an unrelated cat. The mass and the tumor cell line were only positive for vimentin and cytokeratin (AE1/AE3 and Lu-5), while the healthy cat synovium lacked cytokeratin expression.*
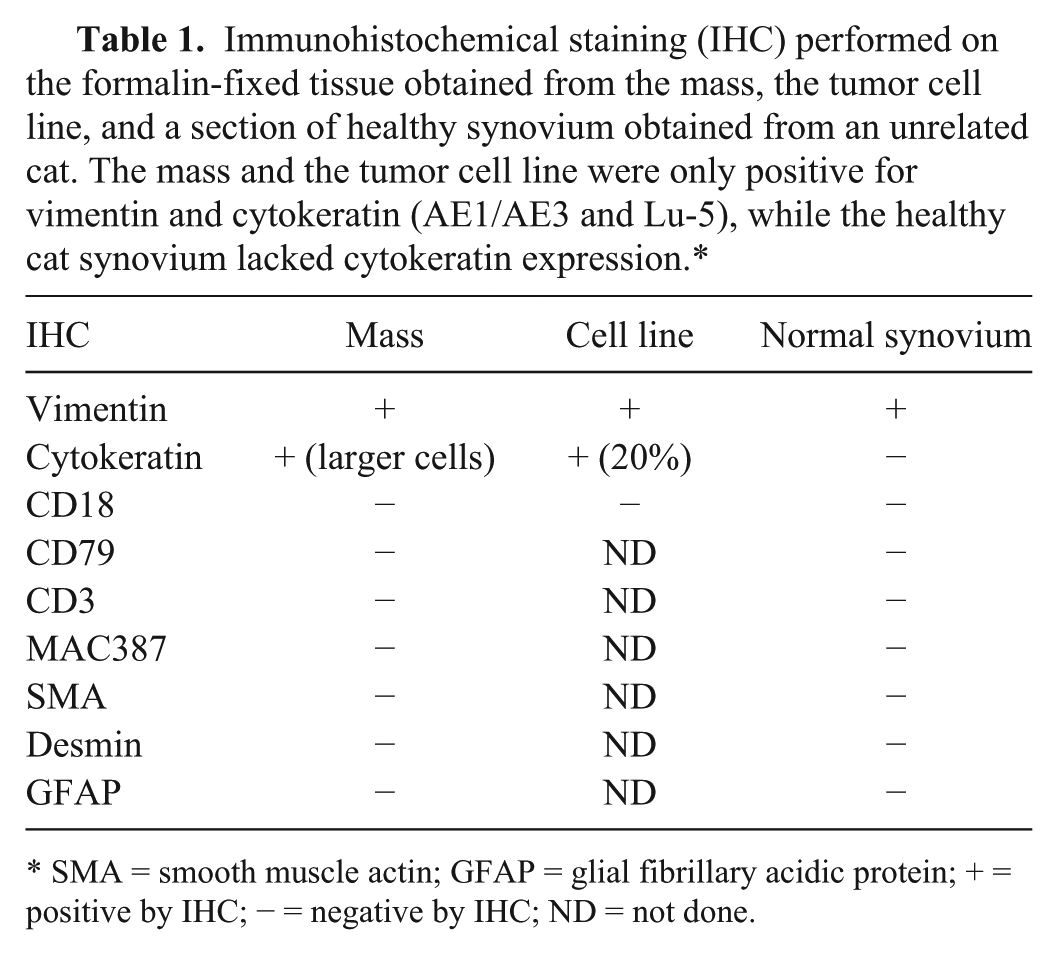
SMA = smooth muscle actin; GFAP = glial fibrillary acidic protein; + = positive by IHC; − = negative by IHC; ND = not done.
Fresh neoplastic tissue from the submitted amputated limb was also prepared for tissue culture, adapting previously published protocols.4,23,27 The aim was to establish a tumor cell line and apply functional tests and IHC with the goal of better characterizing the cell of origin. The cells from the monolayer observed were removed, mixed with cell culture medium (1% nonessential amino acids, 1%
A lipopolysaccharide (LPS) stimulation assay was performed on the culture cell line to test whether the cells were of histiocytic origin and capable of producing proinflammatory cytokines after stimulation. After 10 passages, the cells were split, and ~5 × 105 cells were added to each of 6 wells in a 24-well plate. At the same time, feline peripheral blood mononuclear cells (fPBMCs) were collected as a positive control, and the cells were equally split between 6 wells. Three wells of the tumor cell line and of the fPBMCs received medium only and were used for a negative control, while 3 wells of each were stimulated with LPS (100 ng/mL) for 24 hr. The supernatant was removed and stored at −20°C until used for enzyme-linked immunosorbent assay for detection of feline tumor necrosis factor (TNF)-α, interleukin-1 (IL-1)β, and IL-6. p All assays were conducted in accordance with the protocol provided by the manufacturer. All supernatant samples were assayed in triplicate. Optical density was determined with a plate reader and associated software q ; absorbance was set at 450 nm. The LPS stimulation assay revealed that while concentrations of the major proinflammatory cytokines (TNF-α, IL-1β, and IL-6) were significantly higher for LPS-stimulated fPBMCs used as a positive control, LPS-stimulated tumor cells failed to secrete any detectable concentration of proinflammatory cytokines in response to LPS.
At the same time, the cells were grown over sterile round glass coverslips. Two coverslips from each group were also stained immunocytochemically r with mouse anti-feline CD18 monoclonal antibody s and isotype control antibody. t The round coverslips were fixed by incubation in acetone for 3 min at room temperature (~25°C). Each coverslip was incubated at room temperature for 1 hr with mouse anti-feline CD18 (1:20) or an isotype control antibody (1:20). The coverslips were then counterstained with Gill hematoxylin stain, air-dried, mounted, and examined. None of the cells demonstrated plasmalemmal staining for CD18 (Fig. 6A), while control fPBMCs showed diffuse plasmalemmal and cytoplasmic staining. Results obtained from the LPS stimulation assay and from the negative CD18 IHC led to the conclusion that the neoplastic cells were not histiocytic in origin, and that histiocytic sarcoma was to be excluded as a differential in the present case.
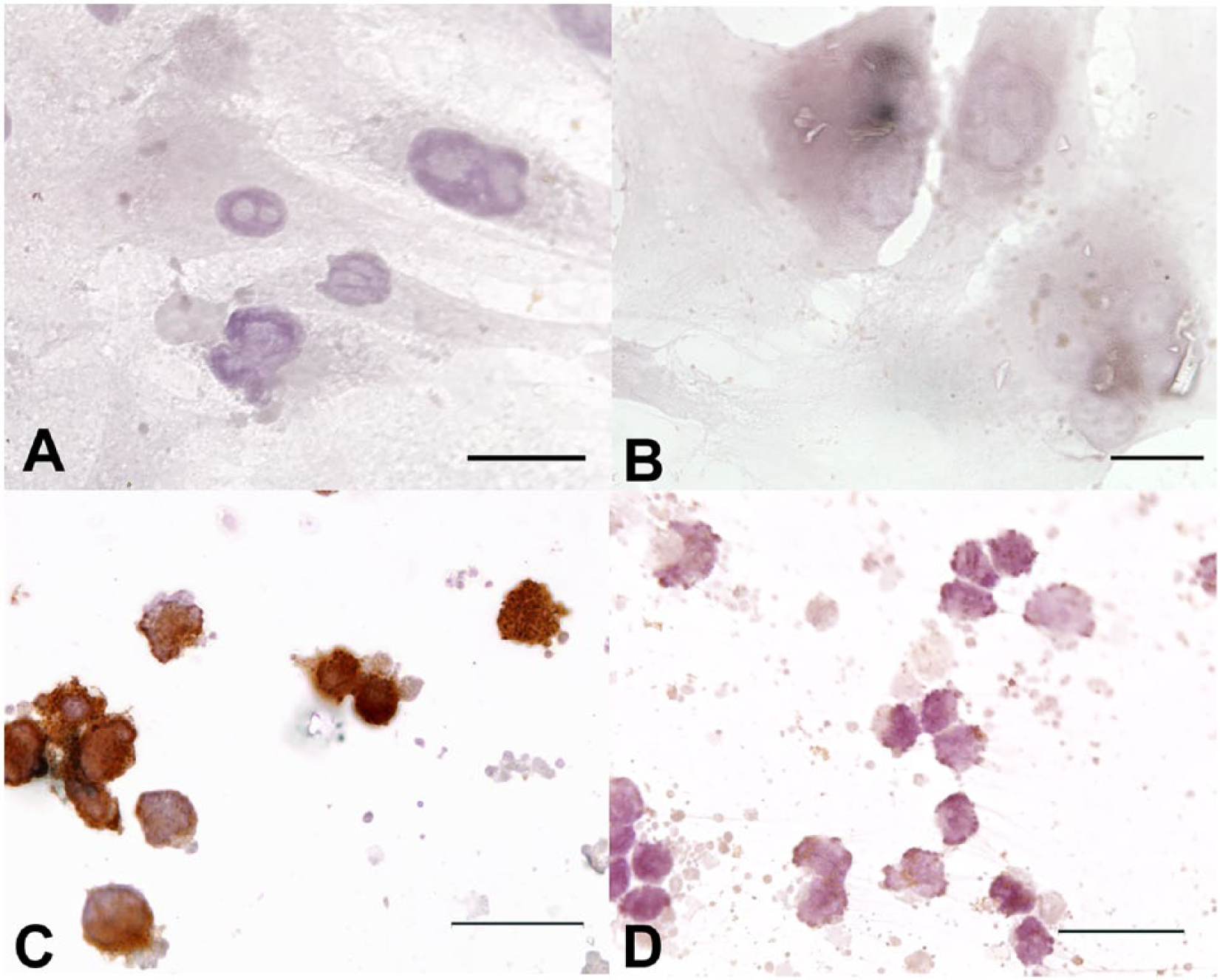
Photomicrographs of CD18 immunocytochemistry of tumor cell line samples (
To perform further IHC on culture cells, a pellet was created using HistoGel u according to the manufacturer’s protocol. The pellet was then embedded in paraffin and cut at 5 μm thickness and stained with hematoxylin and eosin stains. IHC for cytokeratin i and vimentin h was also performed. Similar to the original mass, all cells present stained with vimentin, while only ~20% of the cells were positive for cytokeratin (Fig. 7; Table 1).
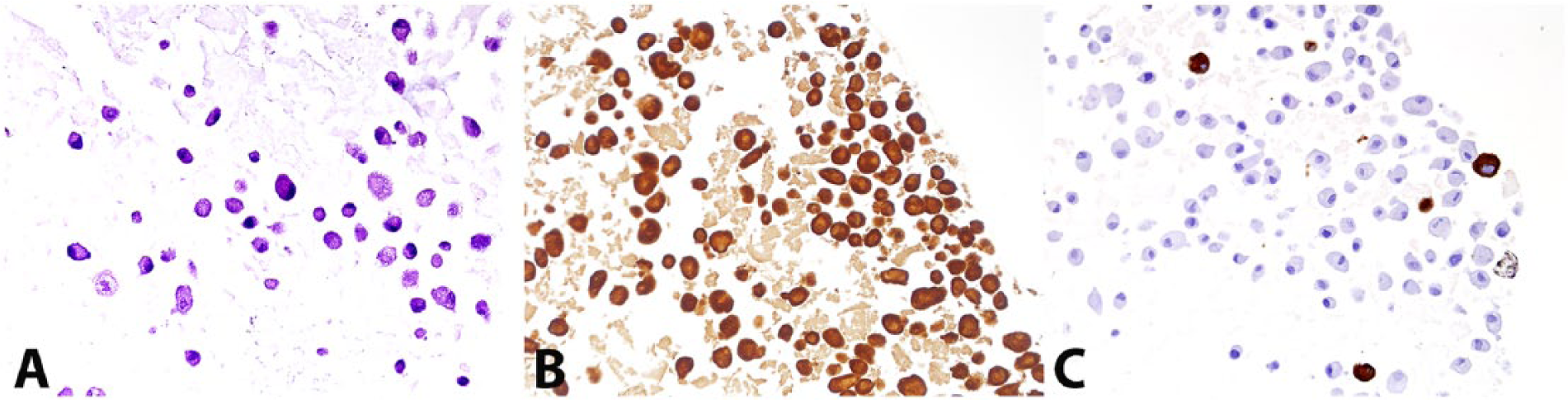
Cultured neoplastic cells; cat. Photomicrographs of hematoxylin and eosin (
An electron microscopy study of the formalin-fixed mass and of the cultured cells was performed in an attempt to compare the neoplastic cells with other reports of synovial cells. The culture cells were pelleted, fixed, and prepared for electron microscopy as previously described. 21 The grids were viewed using a transmission electron microscope. v
Ultrastructurally, the tumor cells from both the mass and the cell culture were morphologically similar. Cells from the mass ranged in size from 10 to 15 µm, had abundant cytoplasm, and were spindloid with several interdigitating cellular processes (Fig. 8, inset asterisks). Membrane condensations were often present in these processes (Fig. 8, inset arrows). Short projections were also present on the cell surface. Numerous mitochondria with flattened cristae were admixed with moderate numbers of elongated profiles of rough endoplasmic reticulum (RER) and intermediate filaments. The nucleus was centrally to eccentrically located, oval to irregularly outlined, with margination of the chromatin along the nuclear membrane (Fig. 8). No basement membrane was present.
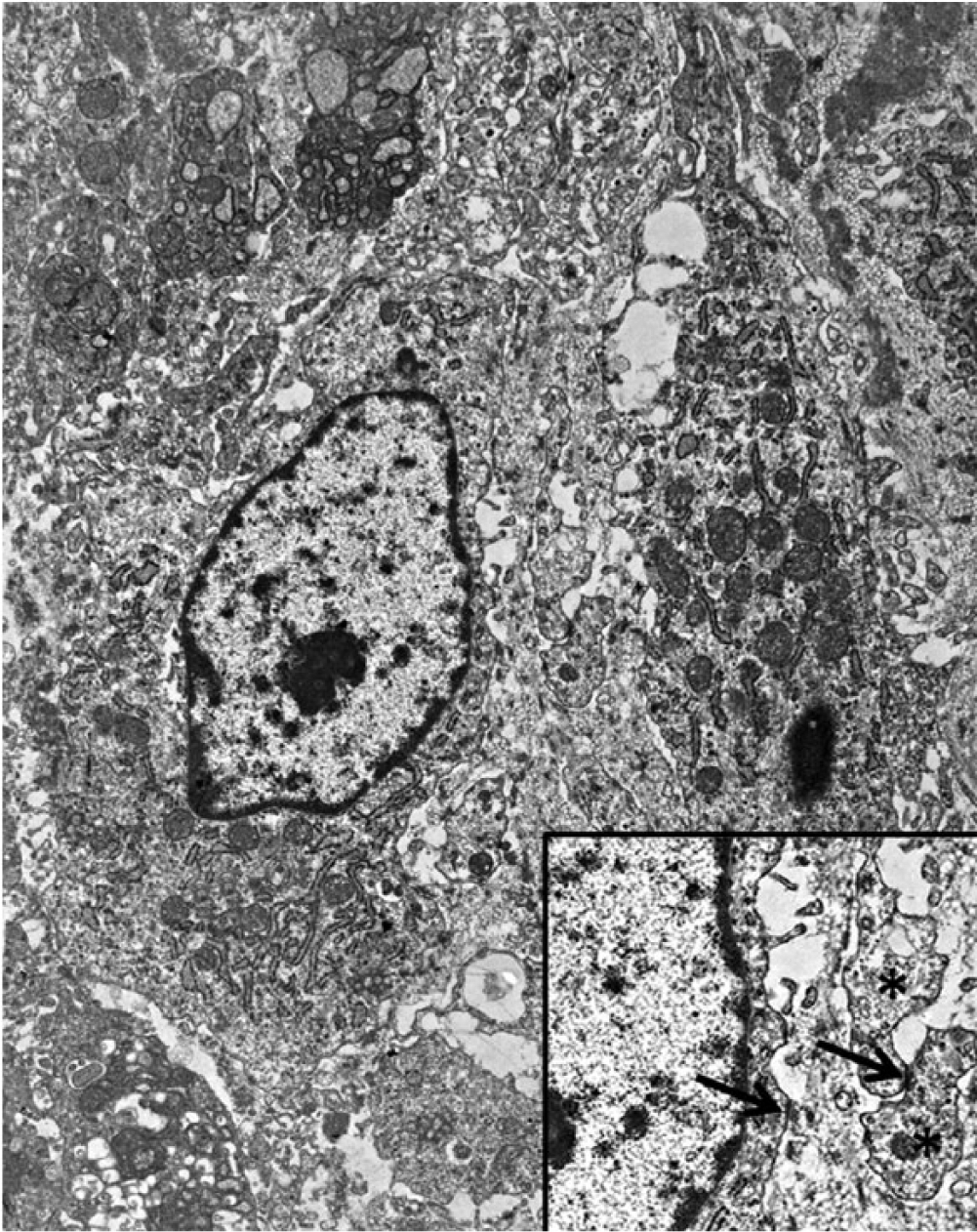
Joint tumor; cat. Ultrastructural appearance of the neoplastic cells shows the presence of interdigitating processes (inset, asterisks) and membrane condensations (arrows).
Ultrastructurally, culture cells appeared plump and round to spindle-shaped, and ranged in size from 10 to 50 µm, with numerous cytoplasmic processes. The cytoplasm contained large numbers of ribosomes, RER, abundant mitochondria, and occasional coated vesicles. Anisocytosis, anisokaryosis, and multinucleation were common in all passages examined. Similar to the biopsy sample, each centrally to eccentrically located, oval to irregular nucleus contained at least 1, commonly multiple, large nucleolus, with margination of the chromatin along the nuclear membrane, frequent mitotic figures, and membrane condensations where cells abutted each other (Fig. 9).
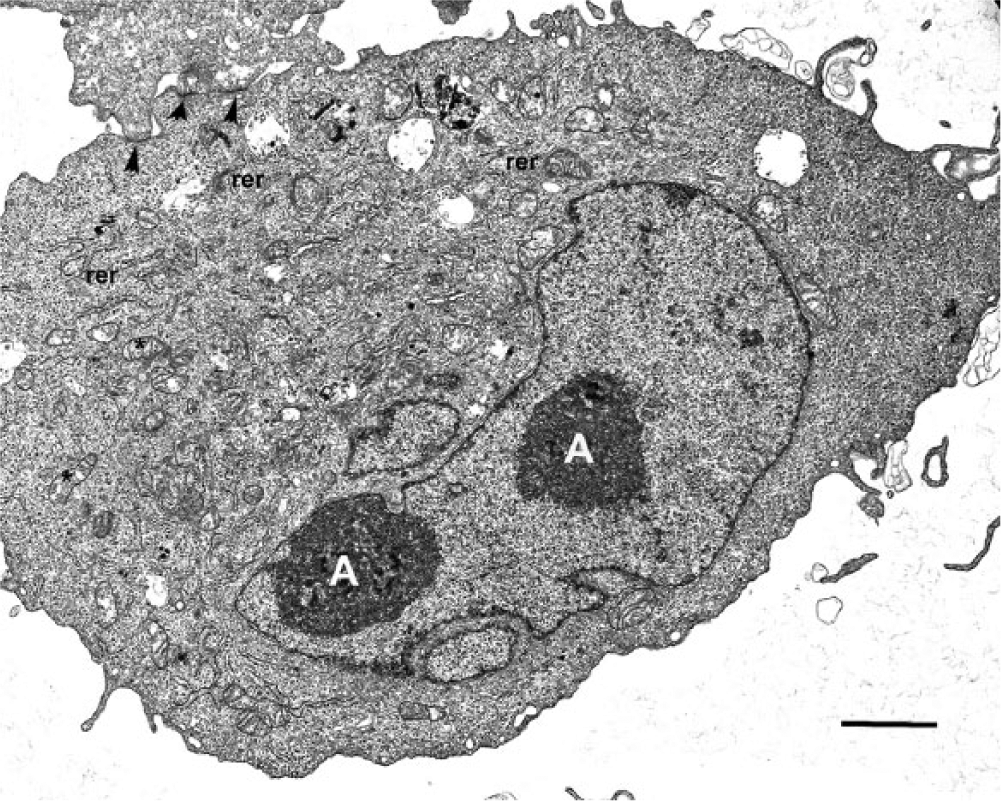
Cultured neoplastic cells; cat. Photomicrograph depicting the ultrastructural morphology of the tumor cell line. Notice the multiple nucleoli (A), the numerous mitochondria (*), the rough endoplasmic reticulum (rer), and membrane condensations (arrowheads). Uracil acetate and lead citrate. Bar = 2 µm.
Although typical of epithelial tumors, cytokeratin expression has been documented in a variety of sarcomas. 11 In some cases, such as with SSs and epithelioid sarcomas, true epithelial differentiation is suspected, while in other types of sarcomas, cytokeratin expression is considered an anomaly. 11 The neoplasm in the present report demonstrated cytokeratin expression without the presence of a basement membrane, which is less consistent with epithelial origin. The cytokeratin expression is in accordance with some reports of synovial cell sarcomas6,9; however, this feature is controversial and should be used with caution when making a diagnosis of SS.
Based on the ultrastructural study, the tumor cells in this case are compatible with published descriptions of synoviocytes and SS cell lines obtained from various species.8,14,22 More specifically, the morphology is similar to the type B synoviocyte, which has been described in rats, horses, and humans, among other species. 14 These cells are characterized ultrastructurally by abundant RER and cytoplasmic processes. Functionally, they are considered to secrete collagens, fibronectin, and other glycosaminoglycans into the joint cavity. Immunohistochemical markers for this specific cell type are not completely known, but it has been suggested that protein gene product (PGP) 9.5 is a reliable marker for this cell type, at least in horses. 14 The membrane condensations seen ultrastructurally in the neoplastic cells resemble primitive intercellular junctions seen in monophasic SSs and are supportive of epithelial differentiation. 2
Based on the location of the tumor, and the morphologic (Figs. 2–4), immunohistochemical (Table 1; Figs. 5–7), and ultrastructural findings (Figs. 8, 9), the neoplasm in the present case is most consistent with a SS. Although histiocytic lineage was initially a differential diagnosis in this case, the tumor cells failed to express CD18, and were not able to secrete proinflammatory cytokines when stimulated by LPS, arguing against origination from the macrophage or monocyte line. Osteosarcoma was also discounted as a diagnosis, as the cytologic features were not consistent with osteoblasts (lacked eccentrically placed nuclei, cytoplasmic eosinophilic dusting [osteoid], and extracellular osteoid). Furthermore, although the location of this neoplasm is potentially consistent with a vaccine-associated sarcoma, the latter tends to contain a myofibroblast component that is smooth muscle actin positive1,16; this was not a feature of this case. To our knowledge, establishment of a feline SS cell line has not been previously described or published. This cell line could be useful in the further characterization of SS and, with comparison studies using normal tissue, may help acquire additional information regarding the synovial function.
Footnotes
Acknowledgements
We would like to acknowledge Abbie Butler for her help and assistance with immunohistochemistry, Mary Ard for her assistance with electron microscopy, and Llewelyn Sellers for technical assistance.
a.
Polyclonal rabbit anti-human CD3 clone A0452, Dako North America Inc., Carpinteria, CA.
b.
Monoclonal mouse anti-human CD79αcy clone HM57 M7051, Dako North America Inc., Carpinteria, CA.
c.
Monoclonal mouse anti-GFAP clone MU020-UC, BioGenex Laboratories Inc., San Ramon, CA.
d.
Monoclonal mouse anti-desmin clone MU072-UC, BioGenex Laboratories Inc., San Ramon, CA.
e.
Monoclonal mouse anti-human SMA clone M0851, Dako North America Inc., Carpinteria, CA.
f.
Monoclonal mouse anti-human MAC387 clone M0747, Dako North America Inc., Carpinteria, CA.
g.
Monoclonal mouse anti-feline CD18 clone FE3.9F2, Leukocyte Antigen Biology Laboratory, Davis, CA.
h.
Monoclonal mouse anti-vimentin clone MU074-UC, BioGenex Laboratories Inc., San Ramon, CA.
i.
Monoclonal mouse anti-cytokeratin cocktail clone MU071-UC, BioGenex Laboratories Inc., San Ramon, CA.
j.
HK086-9K, BioGenex Laboratories Inc., San Ramon, CA.
k.
S3020, Dako North America Inc., Carpinteria, CA.
l.
H312-500, Fisher Scientific, Fair Lawn, NJ.
m.
HK085-5K, BioGenex Laboratories Inc., San Ramon, CA.
n.
NC495H, Biocare Medical LLC, Concord, CA.
o.
NC494H, Biocare Medical LLC, Concord, CA; K3466, Dako North America Inc., Carpinteria, CA.
p.
DuoSet, R&D Systems Inc., Minneapolis, MN.
q.
Gen 5, Biotek Instruments Inc., Winooski, VT.
r.
Envision system, Dako North America Inc., Carpinteria, CA.
s.
Clone Fe3.9F2, provided by Dr. Peter Moore, Department of Pathology, Microbiology, and Immunology, School of Veterinary Medicine, University of California–Davis, Davis, CA.
t.
Mouse IgG1 negative control, AbD Serotec, Raleigh, NC.
u.
HistoGel, HG-4000-012, Thermo Fisher, Kalamazoo, MI.
v.
JEM-1210 transmission electron microscope, JEOL USA Inc., Peabody, MA.
Authors’ note
Paola Cazzini and Karelma Frontera-Acevedo contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
