Abstract
Soft tissue sarcomas of the equine distal limb associated with joints, sheaths, or bursae have rarely been reported. Accurate diagnosis of these tumors is challenging in both human beings and veterinary species. Immunohistochemical staining and transmission electron microscopy have been used in human beings to reduce misdiagnosis. The current report describes 2 mature horses presenting with lameness and swelling associated with the dorsal aspect of the metacarpo(tarso)phalangeal joint. In both cases, surgical excision was performed with subsequent histological analysis of the masses to determine the tissue of origin. In both cases, immunohistochemical staining and transmission electron microscopy aided the definitive diagnosis of fibrosarcoma associated with the fetlock joints of 2 horses.
Soft tissue sarcoma (STS) is a broad term encompassing neoplasms such as synovial cell sarcoma, fibrosarcoma, rhabdomyosarcoma, hemangiosarcoma, and leiomyosarcoma. In human beings, synovial sarcomas account for 7–8% of soft tissue sarcomas. 5 Synovial cell sarcomas arise from mesenchymal cells associated with joint capsules, tendon sheaths, and bursae but rarely invade joints; however, such sarcomas may also arise at nonarticular sites in human beings.2,6 Synovial sarcomas can consist of epithelioid or fibroblastic (spindle) cells leading to the classification of 4 morphological types. Synovial sarcomas are uncommon in veterinary species but when diagnosed, are more widely reported in canines than in equines. 9 In addition, there is some controversy as to the existence of true biphasic synovial cell sarcomas in veterinary species. 13 Fibrosarcomas are rare in human beings and extremely rare in equines; cases have been reported in association with subepithelial connective tissues in the head and trunk, or joints, tendon sheaths, and bursae in equines. 15 Hemangiosarcomas, rhabdomyosarcomas, and leiomyosarcomas are other malignant mesenchymal tumors that could be considered differential diagnoses. Morphological and immunohistochemical characteristics of these malignant tumors distinguish them more clearly from fibrosarcomas and synovial sarcomas.
The histological distinction between synovial sarcoma and fibrosarcoma can be extremely difficult in all species. Immunohistochemical staining (IHC) and examination of ultrastructural cell morphology via electron microscopy have been used to differentiate synovial sarcoma from other soft tissue sarcomas in human beings.7,8 Differentiation of STS in equines has been made on the basis of anatomical location and histological examination with light microscopy. A single case report documents the use of IHC in the diagnosis of an equine fibrosarcoma. 15 The current case report documents 2 cases in which IHC and electron microscopy aided the diagnosis of neoplasia associated with the metacarpo(tarso)phalangeal joints of 2 horses.
An 8-year-old Thoroughbred cross mare (case 1) and a 15-year-old cob mare (case 2) were presented to the Philip Leverhulme Equine Hospital (Neston, UK) for investigation of lameness localized to the distal limb and associated with the fetlock joints. A firm soft tissue swelling was identified on the dorsal aspect of the left metacarpophalangeal (MCP) joint in the first case and on the dorsal aspect of the metatarsophalangeal (MTP) joint in the second case. In the first case, diagnostic imaging, including radiography, contrast arthrography, and ultrasonography were used to confirm that the 40 mm × 40 mm mass was located between the common digital extensor tendon (CDET) and the dorsal condyles of the 3rd metacarpal bone (Fig. 1). The mass was located within the thickened joint capsule. In the second case, ultrasonography, radiography, and magnetic resonance imaging were used to characterize the mass over the dorsal MTP joint. Imaging revealed an effusion of the CDET bursa and marked thickening and fibrosis of the subcutaneous tissue and joint capsule. In both cases, surgical excision of the mass was performed under general anesthesia. In case 1, the mass was well delineated and attached to the proximal sagittal ridge of the metacarpal III. In case 2, the mass involved the CDET and thickened dorsal aspect of the MTP joint capsule. At the proximal extent of the mass, an area of caseous necrosis was present within the CDET. Care was taken to avoid penetration of the joint capsule.
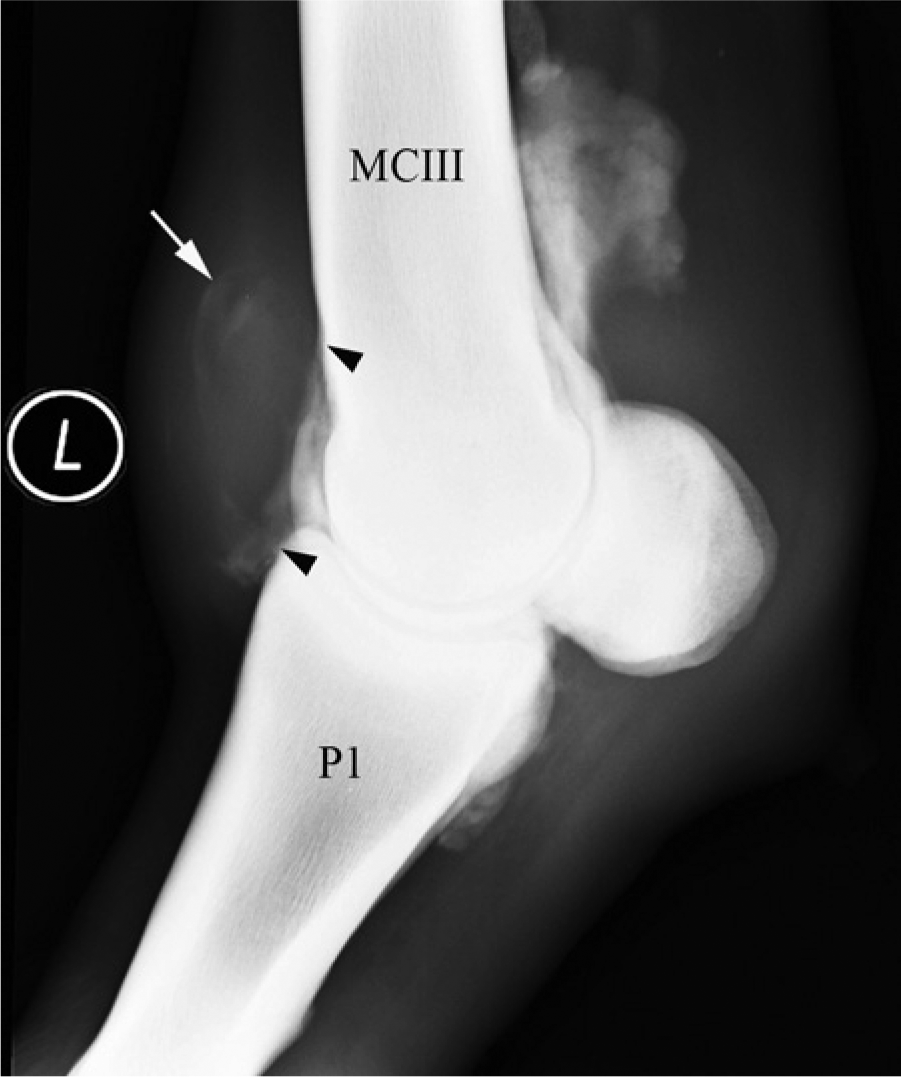
Laterolateral contrast arthrogram of the left metacarpophalangeal (MCP) joint from case 1. A smooth soft tissue density mass (approximately 40 mm × 40 mm) can be seen outlined at the dorsal aspect of the MCP joint (white arrow); this was located within the joint capsule. Periosteal bone reaction is evident (black arrowheads) on the dorsal aspect of the distal metacarpal III bone and on the proximodorsal aspect of the first phalanx (P1).
Excised tissue specimens were fixed for >24 hr in 10% neutral buffered formalin, processed routinely, sectioned at 4 μm, and stained with hematoxylin and eosin for histological evaluation. Immunohistochemical staining was performed using the following primary antibodies (diluted as shown) and standard peroxidase anti-peroxidase methods: pancytokeratin (AE1/AE3; 1:10), vimentin (1:100), desmin (1:2,000), alpha smooth muscle actin (SMA; 1:100), factor VIII (1:1,000), major histocompatibility complex class II (MHC class II; 1:25), and S100 (1:800). Briefly, slides were deparaffinized, and antigen retrieval was performed with 0.05% protease (37°C for 5 min) for pancytokeratin and factor VIII or 10 mM citrate buffer (pH 6, at 96°C for 30 min) for MHC class II; other antibodies did not require antigen retrieval. This was followed by incubation with the primary antibody (16 hr, 4°C), secondary antibody (1:100, 30 min, room temperature), and chromogen (3,3′-diaminobenzidine) visualization. Negative controls, where the primary antibody was replaced with pre-immune rabbit serum (for primary polyclonal antibodies) or murine subtype matched immunoglobulin G unrelated primary monoclonal antibody (for primary monoclonal antibodies), were performed. Positive control stains were performed on appropriate tissues, including equine lymph node for MHC class II (Supplementary Figure available at jvdi.sagepub.com/supplemental). All IHC preparations were counterstained with hematoxylin, then dehydrated, and mounted. Tissue for transmission electron microscopy (TEM) was taken from paraffin-embedded tissue, and paraffin was removed using xylene. Sections were rehydrated in graded alcohols and then fixed in 2.5% glutaraldehyde in 0.1 M sodium cacodylate (pH 7.4) and secondarily fixed in 1% osmium tetroxide(aq) for 90 min. Tissue sections were stained en bloc with 2% uranyl acetate in 0.69% maleic acid for 90 min, then dehydrated in ascending concentrations of ethanol, followed by acetone, before infiltration in 30%, 70%, and 100% (weight/volume) resin in acetone for 1 hr each. Sections were embedded in fresh resin overnight at 60°C to allow polymerization. Ultrathin sections (60–90 nm) were cut and stained with Reynolds lead citrate prior to viewing using a transmission electron microscope at 80 kV. a
Histologically, case 1 demonstrated a solid mass consisting of densely packed interlacing bundles of spindle cells with indistinct cell borders. The cells exhibited pale eosinophilic cytoplasm and large round to oval anisokaryotic nuclei, and an average of 1.5 mitoses per high-power field (Fig. 2). In some areas, the surface of the mass was smooth and convex, whereas in other areas the surface bore a thick fibrinous exudate. There was multifocal necrosis at the margins of the sample. Immunohistochemical staining for pancytokeratin, factor VIII, and S100 was negative; desmin and SMA showed weak staining in approximately 10% of neoplastic cells at the periphery of the mass; low numbers of round cells within the neoplasm were positive for MHC class II, and vimentin was strongly positive within the cytoplasm of neoplastic cells (Fig. 3). The mass had a histological appearance consistent with a STS, with the IHC indicative of a fibrosarcoma. Case 2 was represented by a partially encapsulated, partly infiltrative, densely cellular mass within the tendon. Spindle cells were arranged in streams and bundles, and mitotic figures averaged 1 per high power field. The samples were positive for vimentin on IHC and negative for all other markers (pancytokeratin, desmin, SMA, factor VIII, MHC class II, and S100). As with the first case, positive staining for vimentin was indicative of a tumor of mesenchymal origin.
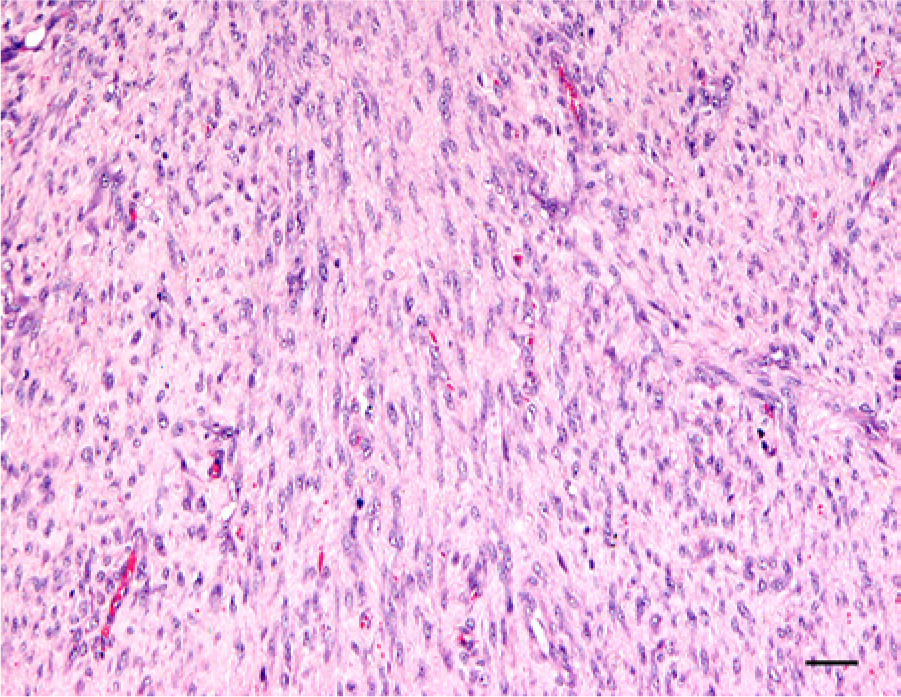
Representative histological section of the tumor from case 1, which was located within the joint capsule on the dorsal aspect of the metacarpophalangeal joint. There are dense bundles and streams of relatively monomorphic spindle-shaped fibroblast-like cells without clefts or myxoid deposition. Hematoxylin and eosin stain. Bar = 50 µm.
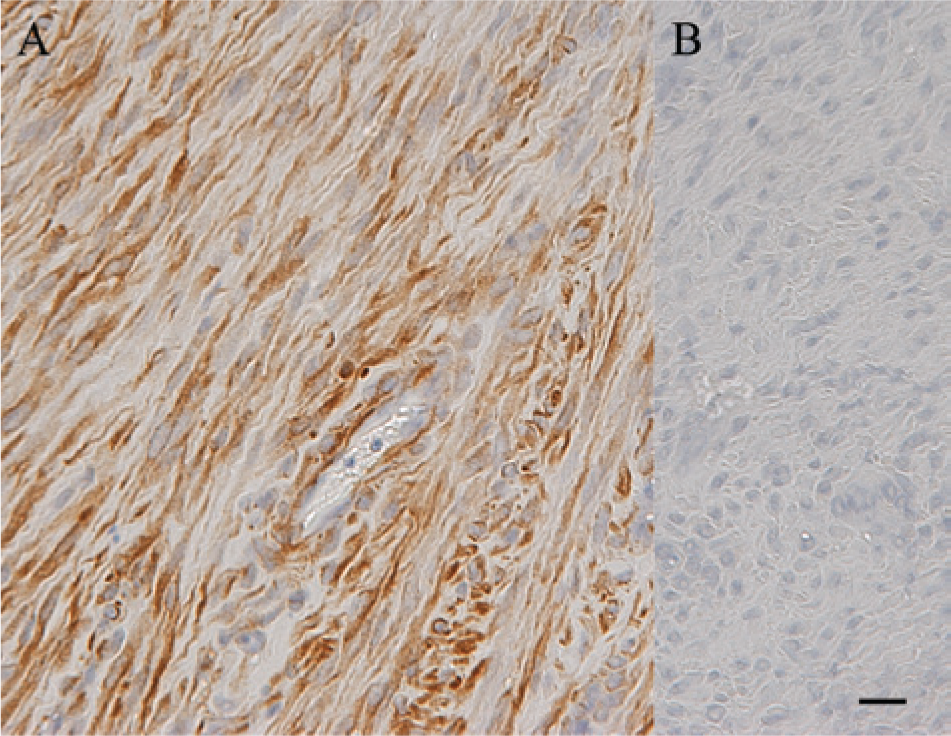
Immunohistochemical staining was utilized to further characterize the tumors. Tumors from both cases were positive for vimentin, confirming the neoplastic cells were of mesenchymal origin. Images shown represent the tumor from case 1; positive vimentin staining (
Multiple ultrathin sections from representative samples of neoplastic tissue from both cases were examined, which displayed similar features; a representative micrograph of tissue from case 1 (in which ultrastructural features were better preserved) is shown in Figure 4. Electron microscopy revealed large amounts of extracellular collagen and spindle cells with abundant rough endoplasmic reticulum, which lacked the short cellular processes in intercellular spaces bordered by cell–cell junctions, which are the typical ultrastructural features of both biphasic and monophasic synovial sarcomas 8 (Fig. 4). Furthermore, the neoplastic cells did not exhibit myofilaments characteristic of leiomyosarcoma, nor organized filaments or Z-bands as seen in rhabdomyosarcomas. 8 Similar to the immunohistochemical characteristics, these ultrastructural features were consistent with the diagnosis of a fibrosarcoma. 7
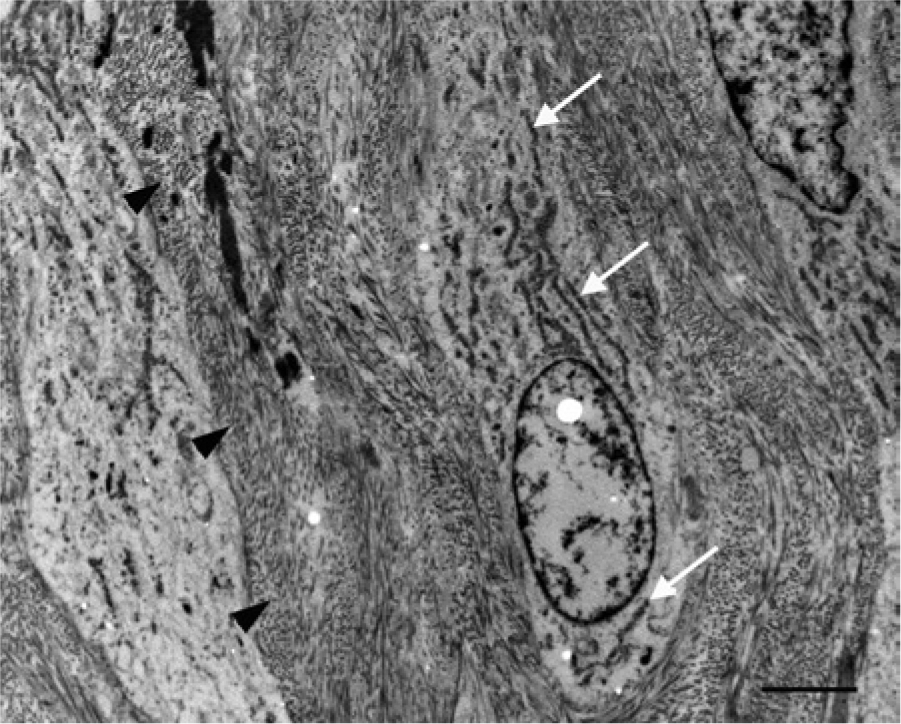
Transmission electron micrograph: photomicrograph shows portions of at least 3 spindle cells. The cell in the center of the micrograph has an oval nucleus with granular chromatin and a distinct nuclear membrane. The cytoplasm is loosely arranged with abundant rough endoplasmic reticulum (white arrows), loosely scattered ribosomes, and a few randomly scattered swollen mitochondria. The other 2 spindle cells have similar cytoplasmic features. Between the cells there are large amounts of densely packed extracellular collagen (black arrowheads). Based on the characteristics described, the spindle cells are most consistent with fibroblasts. Bar = 2 µm.
Soft tissue sarcomas are uncommon in equines and can be hard to differentiate in all species. Combining electron microscopy with IHC resulted in accurate diagnosis of tumor type in 96.5% of cases in 1 human case series. 8 In a 1990 published series, 8 a number of “fibrosarcoma-like” tumors, which did not demonstrate the typical immunohistochemical markers for synovial sarcoma, were identified as synovial sarcomas based on their appearance on electron microscopy. Therefore, the authors felt that it was important to combine IHC and TEM to determine which STS type was present in these cases, as monophasic synovial sarcomas are commonly mistaken for fibrosarcomas. 8 As discussed previously, the monophasic form of synovial sarcoma is reported more frequently in veterinary species, although several authors have contested the existence of biphasic synovial sarcomas in companion animals.3,13
A previous study 4 described 2 equine cases in which histology revealed a mixed cell population containing both fibroblastic and epithelioid cells consistent with a biphasic synovial cell sarcoma. However, no further investigation was pursued. 4 A 2005 study 15 reported moderate to abundant mature collagen in the extracellular space on Masson trichrome stain in a fibrosarcoma, and neoplastic cells were vimentin positive. 15 The cases described in the current report were histologically composed of neoplastic spindle cells and were positive for vimentin, which is consistent with previous reports of fibrosarcomas.14,15
Pancytokeratin and epithelial membrane antigen (EMA) have been previously detected with equal frequency in synovial sarcomas. 8 Conversely, other authors have reported that EMA is more sensitive than pancytokeratin, although the cytokeratin antibody used in the current study (AE1/AE3) was reported as the most sensitive 12 ; no neoplastic cells in the current cases were pancytokeratin positive. Interestingly, case 1 exhibited a low number of round cells positive for MHC class II, a marker of antigen-presenting cells, and some epithelial cells. Synovial cells are either of type A (macrophage-like) and bone marrow–derived with phagocytic and antigen-processing functions, or type B (fibroblast-like). Type A and type B synoviocytes are known to express MHC class II antigens in vitro in response to granulocyte macrophage–colony stimulating factor and gamma interferon, respectively. 1 This may suggest that the tumor in the first case was of synovial origin. However, the MHC class II staining was weak in spindle cells (scattered infiltrating round cells within the section, interpreted as macrophages, exhibited strong staining), and the ultrastructural features of the neoplasm did not support this diagnosis. The tumor from case 1 also exhibited a low number (<10%) of desmin and SMA-positive neoplastic cells in the periphery of the tumor. Fibroblastic tumors are variably positive with muscle markers; myofibroblasts are present within most fibroblastic neoplasms, but a nonuniform staining pattern excludes a tumor of smooth muscle origin.8,10 Other STS such as rhabdomyosarcoma, hemangiosarcoma, and peripheral nerve sheath tumors were excluded by histological appearance and IHC results.
The 1990 published series 8 concluded that IHC had the greatest sensitivity for recognition of synovial sarcomas and rhabdomyosarcomas. However, TEM had greater sensitivity for diagnosis of fibrosarcomas and IHC marker–negative synovial cell sarcomas. On TEM, the ultrastructural components of both monophasic and biphasic synovial cell tumors have been well characterized. 7 The TEM results for the 2 cases described in the current report showed mesenchymal cells with prominent rough endoplasmic reticulum and also abundant extracellular collagen, which, in the absence of intercellular spaces containing short cellular processes characterized previously as present in both monophasic and biphasic synovial sarcomas, 7 are therefore consistent with a fibrosarcoma. Overall, the histological, immunohistochemical, and ultrastructural features led to a diagnosis of fibrosarcoma in both cases.
Interestingly, when a combination of light microscopy, IHC, and TEM were used, fibrosarcomas were the least frequently diagnosed neoplasm in human biengs. 8 Because of the low numbers of synovial sarcomas described in horses, and the current controversy over the morphological appearance of synovial sarcoma in veterinary species, this is an area that requires further research. Future investigation into molecular techniques, such as identification of characteristic genetic translocations as used in human beings 11 may aid diagnosis of equine synovial sarcomas.
Although rare, STS represent a differential diagnosis for soft tissue swelling over the dorsal aspect of the equine metacarpo(tarso)phalangeal joint. It is difficult to differentiate soft tissue sarcomas in all species including equines; therefore, using a combination of diagnostic techniques offers the greatest likelihood of obtaining a positive diagnosis, which can then provide owners with an accurate prognosis and treatment plan.
Footnotes
Acknowledgements
The authors wish to thank the staff of the Histology and Electron Microscopy Laboratories, Veterinary Laboratory Services, School of Veterinary Science, University of Liverpool, for excellent technical assistance, and Dr. Lorenzo Ressel for helpful discussions regarding the article.
a.
Phillip EM2088 transmission electron microscope, Philips, London, United Kingdom.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
