Abstract
A commercial facility producing hamsters with a history of infection by dwarf tapeworm (Hymenolepis nana) submitted 15 animals for necropsy and postmortem parasitological and microscopic examination. No tapeworms were detected grossly or microscopically. Fecal examination including gastrointestinal mucosal smears demonstrated mixed intestinal bacteria and low numbers of Giardia sp. Histologic examination of small intestine demonstrated filling of the small intestinal crypts by large numbers of 7–9 µm × 3 µm, rod to crescent or teardrop-shaped flagellates consistent with Spironucleus sp. These organisms had two 1-µm, basophilic, oval nuclei and multiple superficial flagella-like structures. Much larger 10–15 µm × 8–10 µm, oval to pear-shaped organisms were also present in lower numbers and usually located with the crypts. These larger flagellates had multiple flagella and a basophilic rod-shaped nucleus. The larger flagellates included Giardia sp., which had an intimate interface with the surface of the mucosal epithelium, bilaterally symmetry, and binucleation. Lower numbers of trichomonads were also present and were distinguished by an undulating surface membrane and a single nucleus. The mucosa was hyperplastic and moderately inflamed. Although the tapeworm infection was resolved, diagnosis of multiple intestinal flagellates by fecal examination is complicated by the varying sensitivity and diagnostic accuracy of different types of fecal analysis for different flagellate types. Key differences in the morphology and location of the different types of flagellates as observed by histology of intestinal tissues provide important additional diagnostic information to distinguish trichomonads, Spironucleus sp., and Giardia sp.
The University of Florida–Veterinary Medical Center (UF-VMC) Surgical Pathology Service received 4 samples of jejunum from Syrian hamsters (or golden hamster; Mesocricetus auratus). The samples were submitted by a referring veterinarian who had performed an unspecified set of fecal analyses and postmortem examinations on 15 animals. The hamsters were owned by a commercial vendor selling to the pet trade that had received reports that their stock contained the dwarf tapeworm, Hymenolepis nana. Following treatment for tapeworms, the referring veterinarian reported that fecal analysis using various, but unspecified, methods were negative. To provide a more intensive parasitological workup, the referring veterinarian euthanized 15 animals of various ages for a postmortem examination; no gross abnormalities were reportedly found on opening and observing content within the entire length of the gastrointestinal tract, and direct fecal smears and mucosal impression smears of unspecified regions revealed unremarkable microflora and Giardia sp. Samples of small intestine from multiple animals described as varying in age, but without specific clinical histories, were submitted in 10% neutral buffered formalin for microscopic examination. The referring veterinarian indicated that histology was requested to rule out the presence of tapeworm cysts within the bowel walls, which was an ongoing concern for the vendor.
Four sections of jejunum ranging in length from 14 cm to 8 cm and having a white to tan serosa and mucosa were received in 10% neutral buffered formalin at UF-VMC. The tissues were routinely processed, paraffin-embedded and examined as hematoxylin and eosin–stained, 4-µm sections by light microscopy.
Microscopic examination yielded a diagnosis of moderate lymphoplasmacytic to eosinophilic enteritis with bacteria and flagellates. The lamina propria had a multifocal patchy, rather than diffuse, inflammatory pattern characterized by mild to marked infiltrates of lymphocytes, plasma cells, fewer histiocytes, and eosinophils within the lamina propria separating the crypts and infiltrating the glands and crypts (Fig. 1A, 1B). Increased amounts of mucus were present at inflamed areas on the surface with extension into crypts. The crypt epithelium within inflamed areas was hyperplastic with cellular crowding and moderately increased numbers of mitotic figures (Fig. 1C). Within crypts in some areas, there were very large numbers of 2 µm × 6 µm elongated flagellates consistent with Spironucleus sp. organisms (Fig. 1D). Higher magnification of the Spironucleus sp. organisms within hyperplastic crypts demonstrates their small size and elongated or crescent shape with no ventral flattening and without a close adherence to the mucosal surface (Fig. 2A), and several locally extensive areas had large numbers of Gram-negative rods attached to the superficial luminal mucosal epithelium (Fig. 2B). Two types of larger flagellates are also present in lower numbers within the crypts and on the bowel surface. The first are trichomonads characterized as 8–14 µm piriform (pear-shaped) organisms with a pointed posterior end continuous with 3–5 flagella, a round profile, and an undulating surface membrane (Fig. 3A, 3B). The second are Giardia sp. observed as 10–20 µm × 5–15 µm piriform, bilaterally symmetric organisms with flagella (Fig. 4A). The Giardia sp. can be located within the lumen or may be present as narrow, ventrally flattened profiles when attached to the mucosal epithelium by a ventral sucking disk (Fig. 4B). The Giardia sp. have 2 nuclei, each of which has an oval intranuclear endosome giving the appearance of “2 eyes” (Fig. 4C). Inflamed areas of hyperplastic mucosa tended to have a large number of 1 type of flagellate rather than 2 or more types; however, combinations most often consisted of large numbers of Spironucleus sp. with lower numbers of one of the larger flagellates.
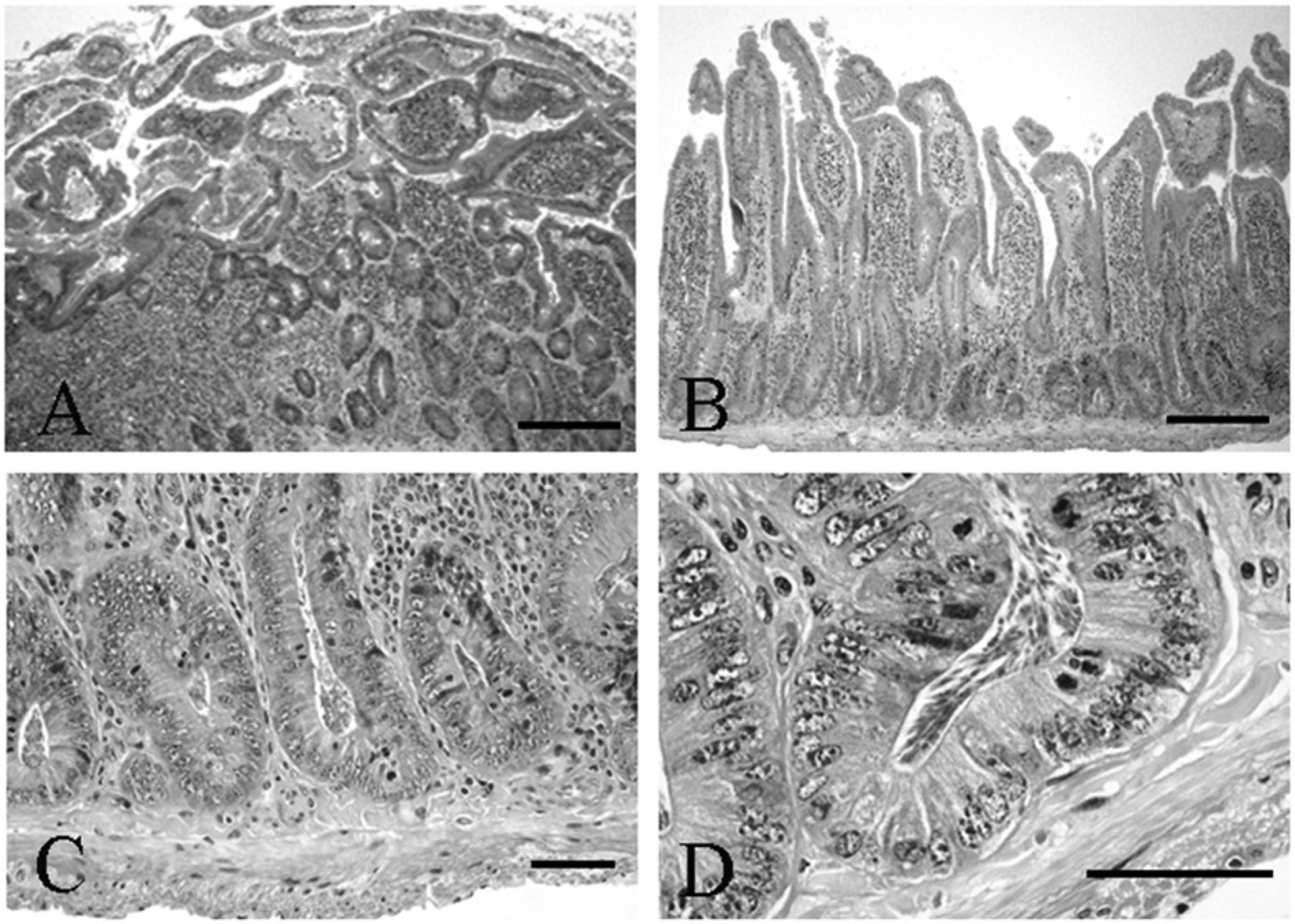
Syrian hamster (Mesocricetus auratus); microscopic overview of small intestinal changes and parasite burden.
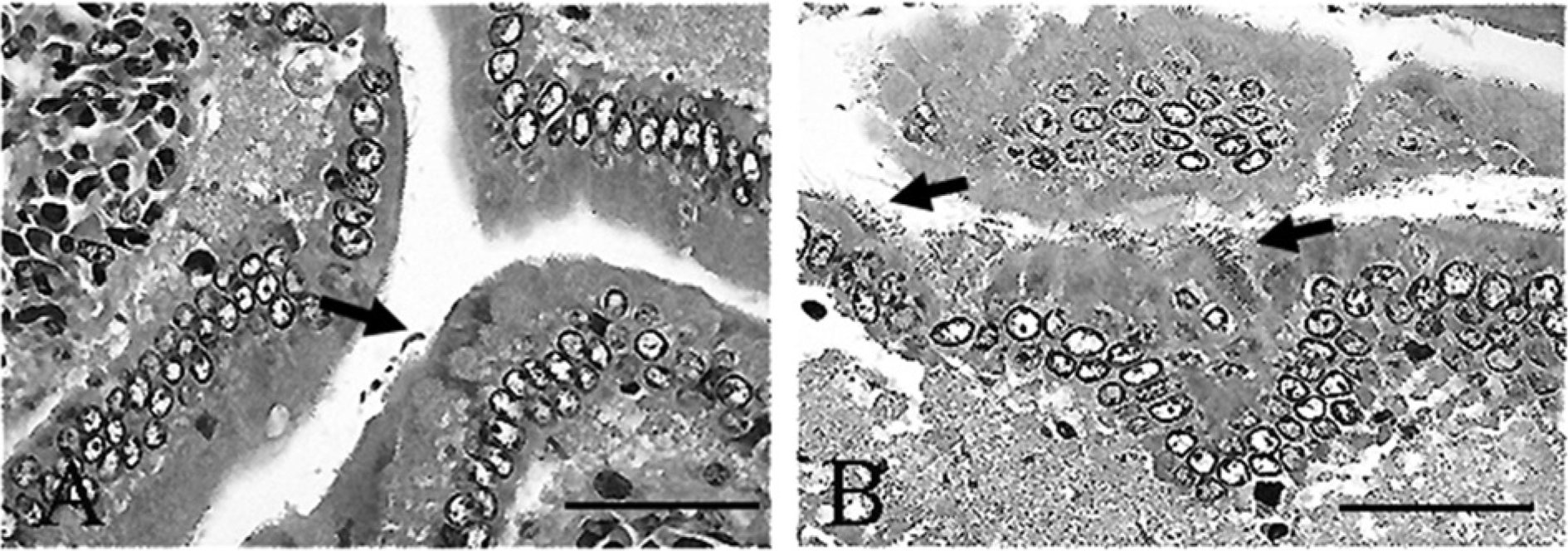
Syrian hamster (Mesocricetus auratus); small intestine.
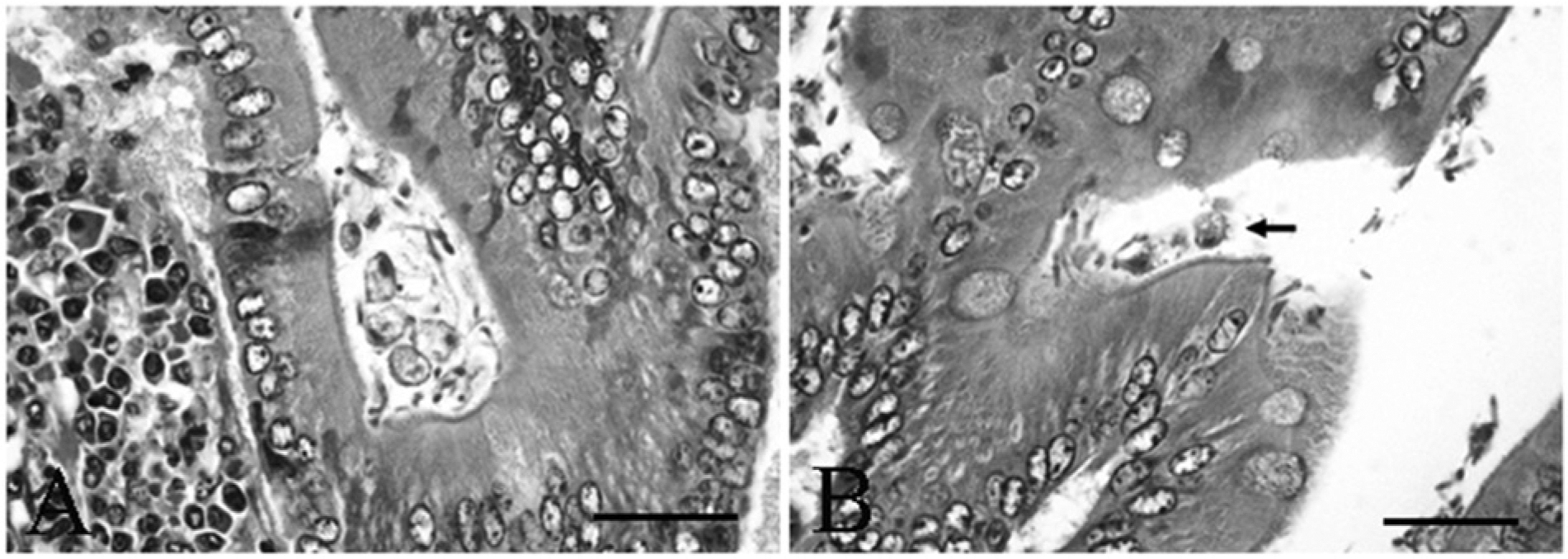
Syrian hamster (Mesocricetus auratus); small intestine with trichomonads.
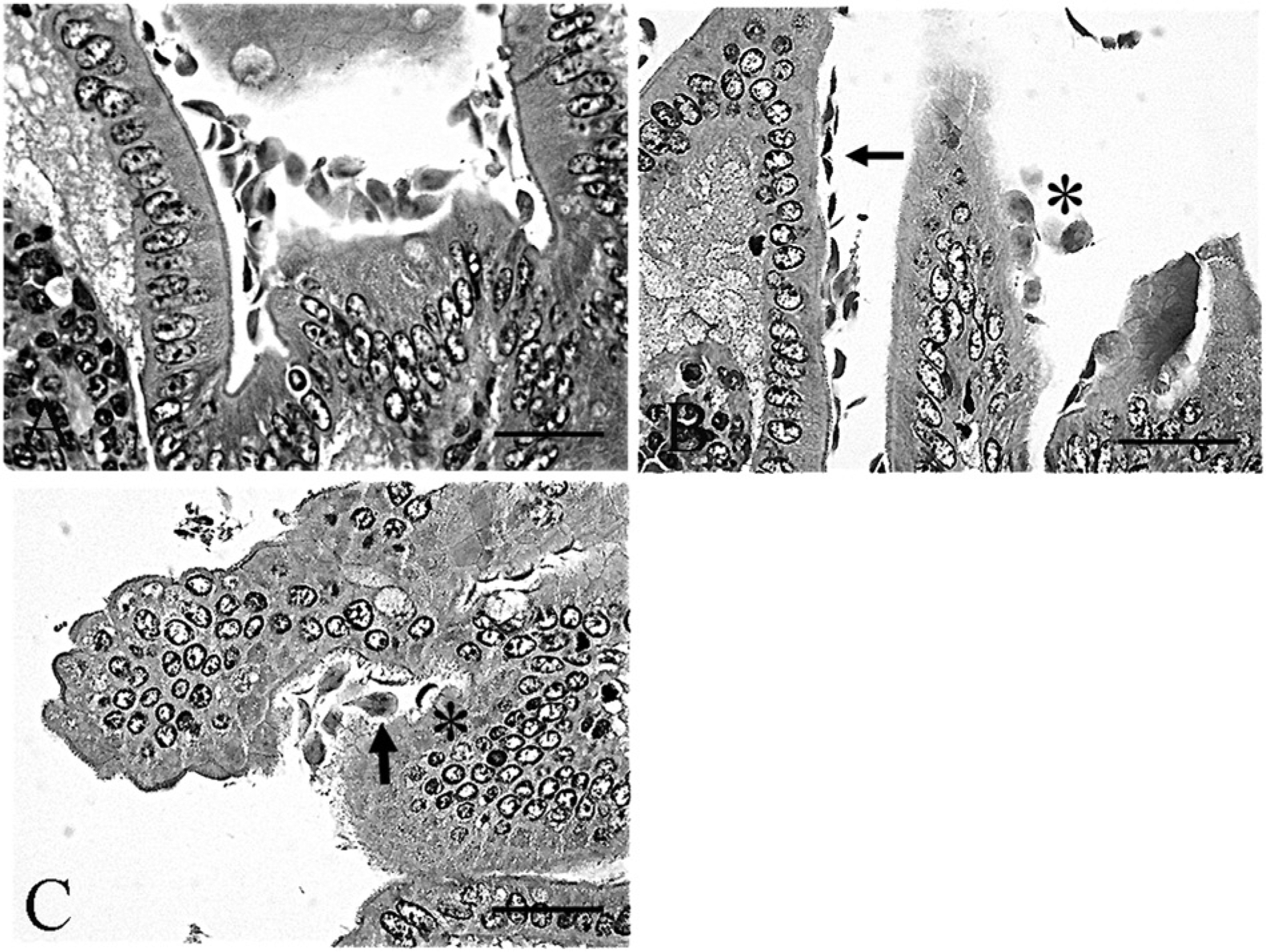
Syrian hamster (Mesocricetus auratus); small intestine with Giardia sp.
Although 3 different types of flagellates were found in abundance within the histology tissue sections, the 3 organisms are not all equally detectable by any 1 specific type of fecal analysis. Flagellates vary in both their morphology observed by histology and their detection in feces as compared in Table 1.
Comparison of the distinguishing characteristics of and diagnostic methods for flagellates observed in the small intestine of the Syrian hamster (Mesocricetus auratus).
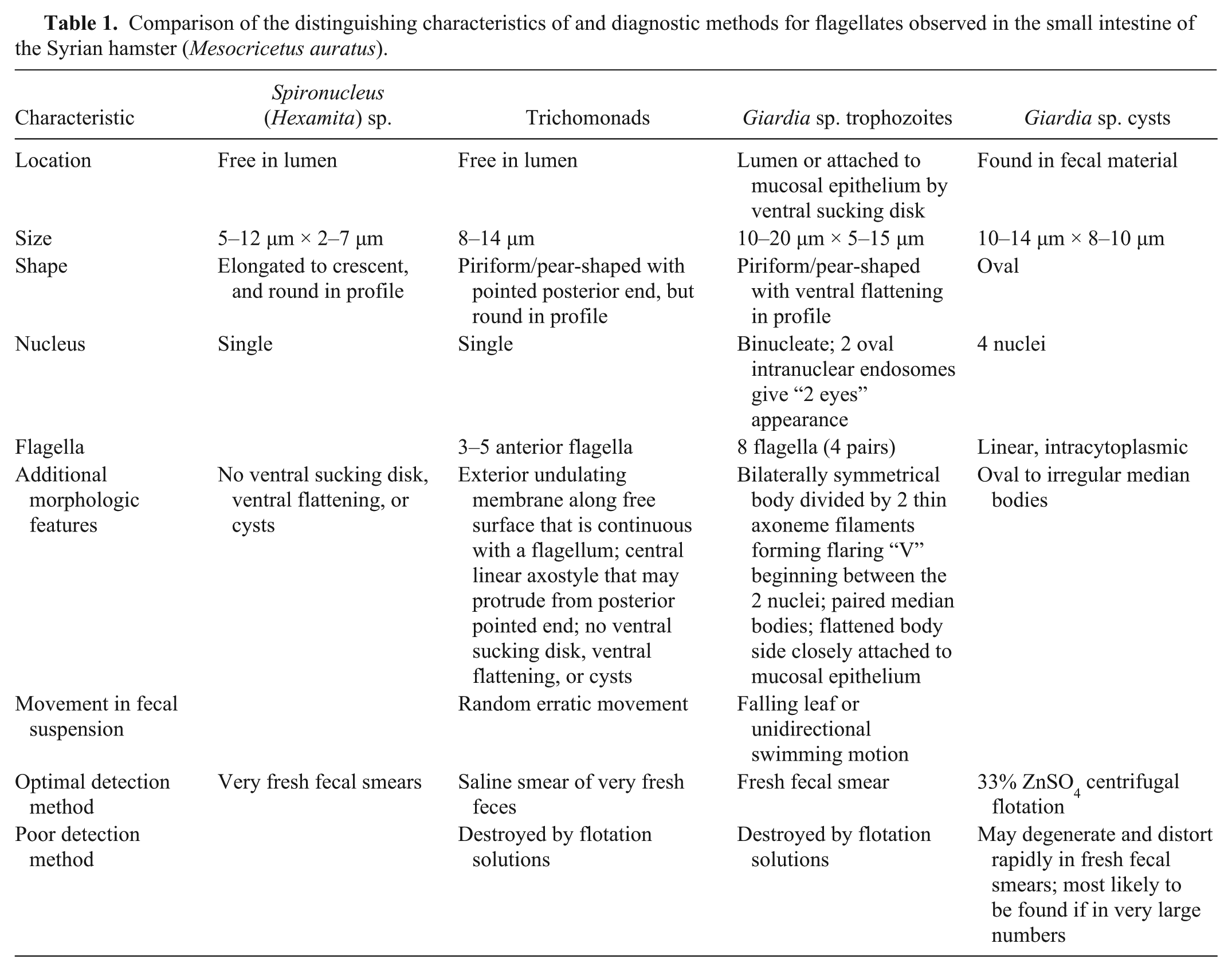
The 3 types of parasites observed in the bowel of the hamsters each have distinguishing characteristics, some of which are apparent in tissue section, while others are better characterized in fecal preparations. Organisms in fecal preparation are superior to tissue sections for demonstration of more detailed features, such as those of Giardia sp., including division of the bilaterally symmetrical body by 2 thin axoneme filaments forming a flaring “V” beginning between the 2 nuclei, and the 8 flagella. Spironucleus (= Hexamita) sp. (5–12 µm × 2–7 µm trophozoites) are the smallest observed organism. Trichomonads (8–14 µm trophozoites) and Giardia sp. (10–20 µm × 5–15 µm trophozoites and 10–14 µm × 8–10 µm cysts) are of similar size, but have specific morphologic differences.3,10
Spironucleus sp. organisms are small piriform flagellates that can be identified in fresh fecal smears and lack a sucking disk.5,10 Trichomonads are larger pear-shaped flagellates with a pointed posterior end. Internal structures include a single nucleus and a linear axostyle that may protrude from the posterior end; external features are an undulating membrane visible along the length of the free surface that is continuous with a posterior flagellum, and multiple anterior flagella numbering from 3 to 5. Identification to species is generally based on the host, location, flagellar number, and, on occasion, more specific morphology or genetic characterization. Trichomonads can be distinguished from Giardia sp. in tissue section because they are rounder in profile without ventral flattening in association with the small intestinal mucosal surface, lack a cyst stage, presence of an undulating membrane on the surface, and a single nucleus.1,2,10 Trichomonad trophozoites in fresh feces can be identified in a saline smear preparation, but are destroyed by flotation solutions. 10 Giardia sp. cysts are oval, with 2 nuclei, linear intracytoplasmic flagella, and oval to irregular median bodies. The Giardia sp. trophozoites have a distinctive teardrop or piriform shape and bilateral symmetry. The 2 nuclei have clear oval endosomes mimicking the appearance of 2 eyes, and the body is divided by 2 thin axonemes, a pair of median bodies, and 8 flagella (4 pairs). In tissue section, the organism is sometimes found closely apposed to the small intestinal mucosal epithelium where it can be visualized in profile having a ventrally flattened shape due to the ventral adhesive disk, which is adapted for close association with the mucosal epithelium.2,7,8,10 Giardia sp. cysts are most likely to be detected viable and without distortion by using 33% ZnSO4 centrifugal flotation, as they degenerate rapidly in feces making fresh fecal smears less helpful. However, Giardia sp. trophozoites can be identified in a fresh fecal smear where they have a characteristic falling leaf or wobbling swimming motion, which assists in distinguishing them from trichomonads. 10
The adult dwarf tapeworm, Hymenolepis nana (previously known as Vampirolepis nana), is found in the small intestine. Rodents become infected after consuming cysticercoid-infected intermediate beetle or flea hosts that ingested embryonated eggs found in feces of rodents, primates, or human beings. 9 Humans and rodents can be infected directly by ingestion of food or other items contaminated by egg-laden feces and can autoinfect within their own digestive tracts perpetuating the infection. The infection in pet rodents can become objectionable to owners or clients if they notice tapeworm proglottids on the perineum or passed in the feces. Praziquantel is used when 30–50-µm, oval, clear-shelled eggs containing an embryo with 6 hooks are detected in a fecal flotation4,10; however, the hamster is immediately susceptible to subsequent reinfection following treatment.
Although the intestinal flagellates are generally no longer found in hamsters purchased for scientific studies, animals provided for the pet trade as “pocket pets” may still contain several types of intestinal parasites. Hymenolepis nana is generally an incidental finding in hamsters, but has been associated with constipation if large numbers are present within the small intestine, and is a public health concern because of transmission to human beings. 6 Spironucleus muris is common in commercial hamster stocks in large numbers without clinical signs, but can be associated with mucosal damage and depression. 6 Hamsters can transmit S. muris to mice in whom it is often asymptomatic, and is more likely to present a problem in weanlings or immunosuppressed animals, where it causes diarrhea and clinical signs. Giardia muris is the most common species of Giardia in hamsters where it is generally asymptomatic, although association with emaciation and diarrhea is possible. 6 Trichomonads have most commonly been associated with reproductive and digestive tract pathology; however, many are asymptomatic and are incorrectly blamed for diarrhea as they tend to multiply in fluid feces. 2
With regard to flagellates in hamsters, the characteristics in tissue, movement in fecal preparations, and knowledge of the optimal techniques for diagnosis are important. The diagnosis of these organisms can be made by light microscopy based on some key aspects of morphology if they are present in adequate numbers and are intact; however, fine detail requires examination at 100× magnification, and diagnosis by fecal analysis can be difficult unless the method of analysis is suitable for the given organism. In the current case, the presence of H. nana was a useful indicator that there were most likely additional types of intestinal parasites. Both Giardia sp. and H. nana have zoonotic potential and would be of concern to clients.
Footnotes
Acknowledgements
The authors thank Mimi Barton for technical assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
