Abstract
Canine parvovirus (CPV) is one of the most common infectious agents related to high morbidity rates in dogs. In addition, the virus is associated with severe gastroenteritis, diarrhea, and vomiting, resulting in high death rates, especially in puppies and nonvaccinated dogs. To date, there are 3 variants of the virus (CPV-2a, CPV-2b, and CPV-2c) circulating worldwide. In Mexico, reports describing the viral variants circulating in dog populations are lacking. In response to this deficiency, a total of 41 fecal samples of suspected dogs were collected from October 2013 through April 2014 in the Veterinary Hospital of the University of Guadalajara in western Mexico. From these, 24 samples resulted positive by polymerase chain reaction, and the viral variant was determined by restriction fragment length polymorphism. Five positive diagnosed samples were selected for partial sequencing of the
Canine parvovirus (CPV) is one of the most common infectious agents causing increased morbidity and mortality in domestic dogs worldwide. Clinical signs include severe gastroenteritis, vomiting, diarrhea, and dyspnea causing high mortality in puppies.
12
Canine parvovirus belongs to the
It is hypothesized that CPV genotype 2 (CPV-2) emerged from feline panleukopenia or from a parvovirus in wild carnivores. 2 Soon after the appearance of CPV-2 in the mid-1980s, new variants emerged and replaced the original CPV-2 virus; these new antigenic variants were labeled CPV-2a and CPV-2b, where CPV-2b differs from CPV-2a by a single amino acid substitution (Asn to Asp) at position 426 in the VP2 protein. 15 In the year 2000, a new antigenic variant labeled CPV-2c (Glu at position 426) was identified in Italy. 3 Interestingly, within a few years, this new variant partially displaced CPV-2a and CPV-2b in Italy. 13
When last evaluated, the variants CPV-2a, CPV-2b, and CPV-2c circulate worldwide, and their distributions and genetic diversities fluctuate among countries. 6 For example, surveillance studies have shown that the 3 CPV-2 variants are codistributed in Italy, 13 Spain, 8 and Germany. 23 In India, 14 Korea, 11 and mostly in Asian countries, there is an individual or codistribution of CPV-2a and CPV-2b. In North America, specifically in the United States, 10 the 3 CPV-2 variants are co-distributed in contrast with South America where the most prevalent variant is CPV-2c, as it has been shown in surveillance studies in Uruguay, 18 Brazil, 19 and Ecuador. 1 In this regard, surveillance studies of CPV-2 are necessary in order to understand viral evolution and those factors that influence dispersion and adaptation of CPV-2 variants in dog populations. The aim of the current study was to identify and describe the CPV-2 variants circulating in a dog population from western Mexico. The results demonstrated a dominant frequency of CPV-2c in samples collected for the study.
All clinical samples were collected in the Veterinary Hospital of the University of Guadalajara (Guadalajara, Jalisco, Mexico), which offers veterinary services to the city of Guadalajara and its metropolitan areas comprised of Zapopan, Tlaquepaque, Tonala, and Tlajomulco. Samples of all parvovirus suspect cases were collected from October 2013 through April 2014 by rectal swab from vaccinated and unvaccinated diarrheic dogs. In addition, information such as date of sampling, age, sex, breed, housing location, and clinical history were recorded. Samples were collected in duplicate and frozen at −20°C.
DNA preparation was performed with some modifications, as previously described.18,22 In brief, samples were thawed at room temperature and homogenized in 1 ml of saline solution and clarified by centrifugation at 10,000 RCF for 5 min. Next, samples were diluted 1:10 in distilled water and heated for 10 min at 98°C followed by 5 min at 4°C in a thermal cycler.
a
Polymerase chain reaction (PCR) amplification of the
The RFLP analysis was performed as previously described.3,18 In brief, 5 μl of the 1,042-bp amplicon was digested with 2 U of the restriction enzyme
A total of 41 samples were collected from suspected dogs, and after PCR screening, 24 (58%) were positive for the 1,024-bp amplicon. In a similar way, the live attenuated vaccine showed a same sized band. Nonspecific bands were observed in the 5 samples obtained from healthy dogs. All 24 samples showed a RFLP pattern associated with the viral variant CPV-2c, which consisted of 3 bands of 634, 352, and 56 bp. Live attenuated vaccine was used as an internal control and showed a pattern associated with CPV-2a and CPV-2b that comprised 2 bands of 634 and 408 bp. This last result is coherent because the commercial vaccine used in the current study was the Cornell strain–based vaccine. As shown in Table 1, from the 24 diagnosed dogs with CPV, 16 (67%) were 6 months or younger, 3 (12%) ranged from 7 to 12 months, and 1 (4%) ranged from 13 to 15 months. Fourteen dogs (58%) were referred to as “never vaccinated,” and 7 (29%) dogs received an incomplete vaccine regimen against CPV; none of the dogs treated in the hospital showed a complete vaccination schedule.
Description of breed, age, vaccination status, and outcome for each Canine parvovirus isolate in the current study.*
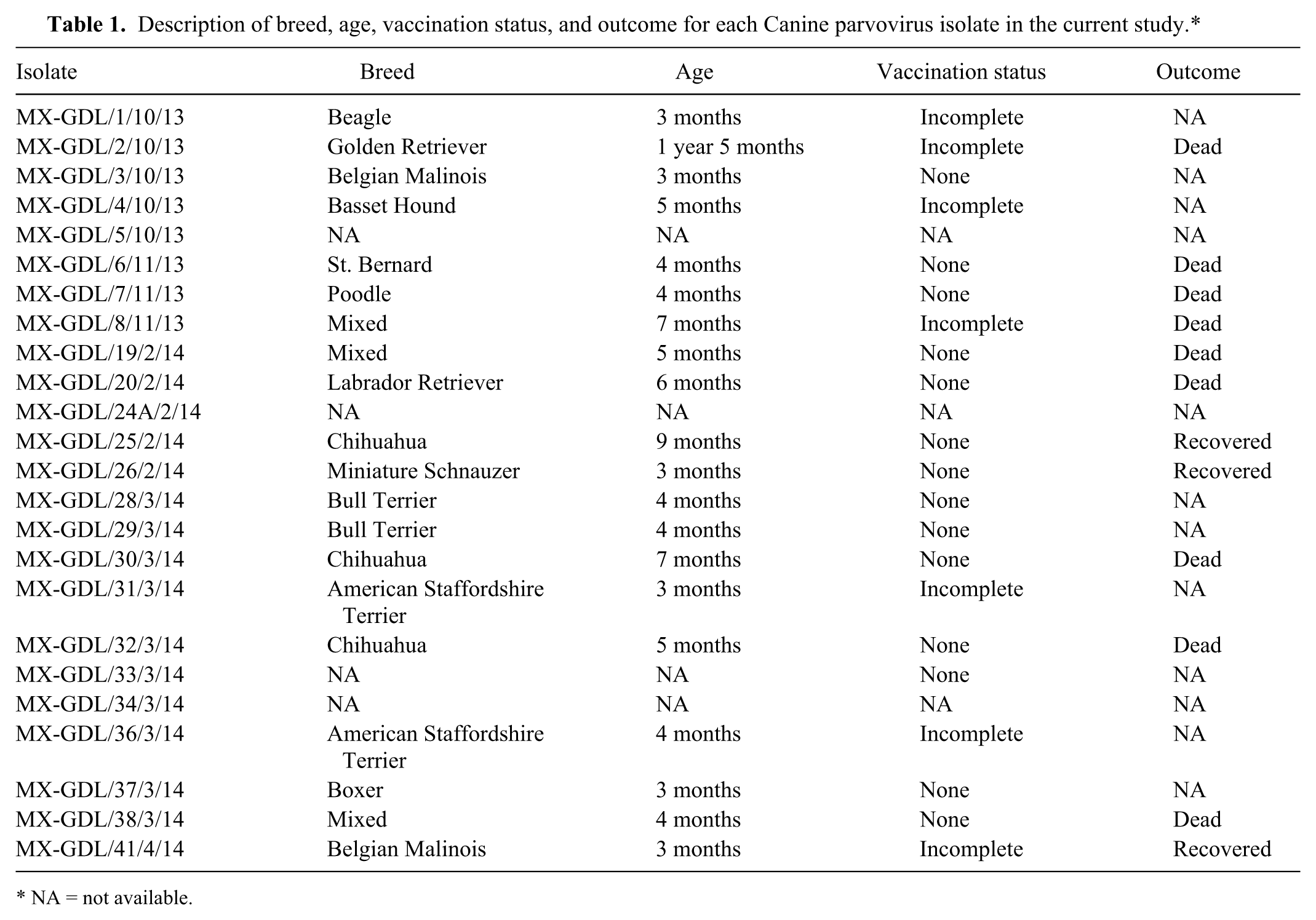
NA = not available.
Five random samples were sequenced, and analysis confirmed the sole presence of CPV-2c viral variant with the characteristic presence of glutamic acid at position 426. DNA BLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi) comparison analysis showed a 98–99% similarity with most sequences reported in GenBank; however, the sequences showed a 99–100% similarity at the amino acid level with most sequences reported worldwide. At the nucleotide level, CPV-2c sequence comparison against isolates from Ecuador, Uruguay, and Italy showed several single nucleotide polymorphisms (SNPs). First of all, a nonsynonymous mutation was identified at position 3442 of the isolate MX-GDL/6/11/13, which showed a transition (AT
Sequence differences of Mexican samples and Canine parvovirus genotype 2c reference strains reported previously.*
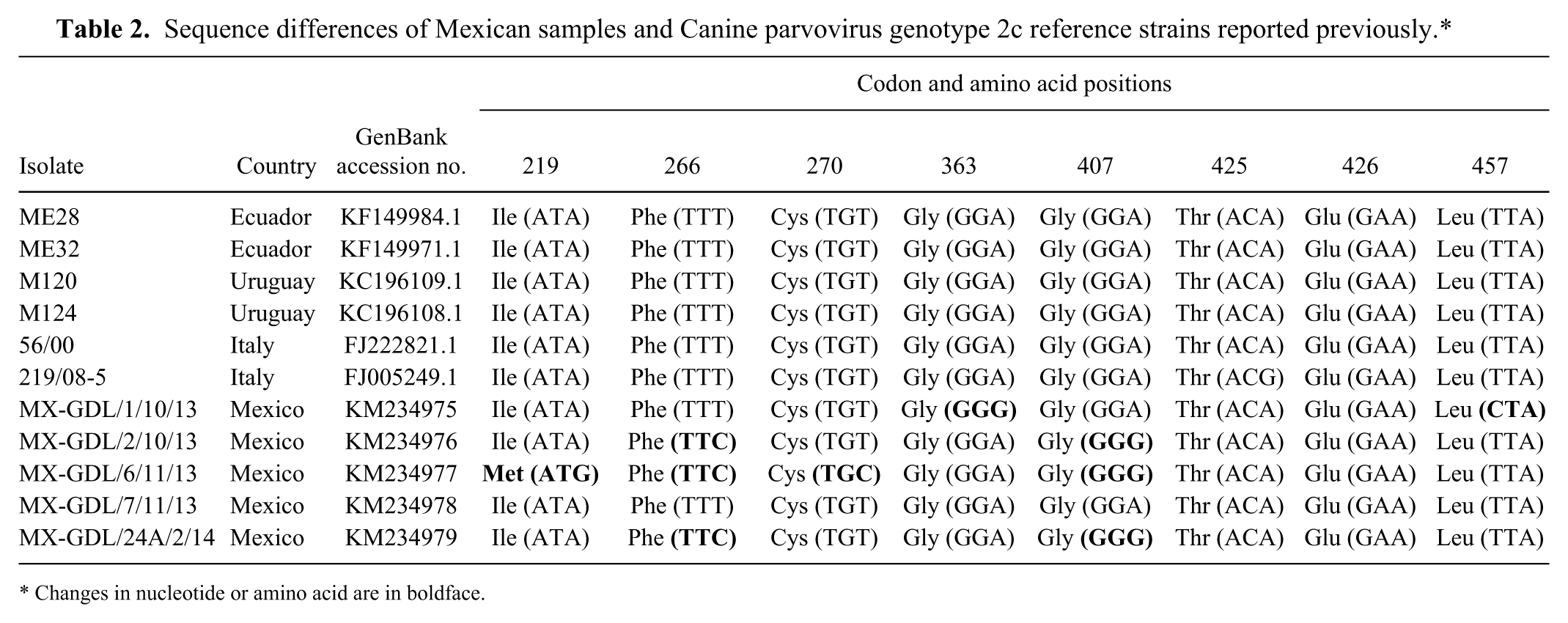
Changes in nucleotide or amino acid are in boldface.
Since its description in 1970, CPV-2 has had a worldwide distribution causing high levels of morbidity and mortality in dog populations. Although dogs from all ages are susceptible to acquire the infection, puppies or dogs less than 6 months of age have a higher predisposition for developing the disease. 12 As shown in Table 1, dogs diagnosed with the virus (67%) were not older than 6 months; this is a relatively low number of infected dogs when compared with reports where an incidence of 95% was determined. 19 However, in the current study, 16% of dogs diagnosed with the virus ranged from 7 to 15 months. This most likely occurred because the majority of dogs tested did not have a vaccination program or were not vaccinated against the virus, therefore increasing the risk of infection.
Canine parvovirus 2c has been distributed worldwide and is cocirculating with the antigenic variants CVP-2a and CPV-2b. In western Europe, for example, samples analyzed from Germany, France, and Bulgaria have shown a cocirculation of CPV-2a and 2c, CPV-2b and 2c, and CPV-2a and 2b, respectively.7,9 In contrast, in Hungary and Albania, the main circulating variant is CPV-2a.4,5 These findings contrast sharply with reports from South American countries where the main CPV variant type is 2c, as it has been reported in Ecuador 1 (54%), Brazil 19 (78%), and Uruguay17,18 (96%). In the current study, all samples collected in western Mexico showed a dominant frequency of the CPV-2c variant as shown in Table 1. This frequency is only comparable with studies reported previously in Uruguay, where 24 of 25 samples analyzed were type 2c. 18 In Mexico, it might be possible that CPV-2c has displaced CPV-2a and CPV-2b in the region and possibly throughout the country. These findings may not be surprising because viral displacement has been reported previously in Italy 13 and Uruguay. 18 However, it is difficult to asseverate this suggestion because there is a lack of previous related studies in Mexico with which to compare the viral dynamics.
Although several nucleotide mutations or SNPs where identified in samples analyzed in the present study, most of them were silent with no impact in the codified amino acid. Interestingly, the isolate MX-GDL/6/11/13 presented a point mutation (AT
Footnotes
Acknowledgements
The authors wish to thank veterinarians Rebeca Granado, Gabriela Ramirez, Pedro Hinojosa, Ramon Carlos, Carmina Varela, and students for collecting feces samples used in this study. Also, the authors thank Michele Brennan for her review of the English language editing.
a.
Thermal cycler, Thermo Fisher Scientific Inc., Waltham, MA.
b.
Primers, Instituto de Biotecnologia de la UNAM, Cuernavaca, Mexico.
c.
PCR reagents, Vivantis Technologies Sdn. Bhd., Subang Jaya, Malaysia.
d.
Parvomune, Holland Animal Health, Cuernavaca, Mexico.
e.
f.
ABI PRISM 3100, Applied Biosystems, Foster City, CA.
g.
FinchTV version 1.5, Geospiza Inc., Seattle, WA.
h.
Serial cloner 2.6.1, SerialBasic Software, France.
i.
BioEdit Biological Sequence Alignment Editor, Ibis Bio-sciences, Carlsbad, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Funding
This work was supported in part by the Department of Veterinary Medicine under the project P3e-2014 with the reference number 220731 approved to CPR.
