Abstract
The current study was directed at developing and validating an indirect fluorescent antibody test (IFAT) capable of detecting antibodies specific for the agent of epizootic bovine abortion (aoEBA). Sensitivity and specificity was determined by comparing antibody titers from 114 fetuses infected with aoEBA with 68 fetuses diagnosed with alternate infectious etiologies. Data established specificity at 100% and sensitivity at 94.7% when cutoff criteria for a positive test were assigned at a titer of ≥1,000. Potential cross-reactivity was noted in samples from 3 fetuses with antibody titers of 10 or100; all were infected with Gram-positive organisms. The remaining 65 fetuses infected with microbes other than aoEBA, and an additional 12 negative reference sera, did not have detectable titers. The IFAT-based serology assay is rapid, reproducible, and unaffected by fluid color or opacity. Total fetal immunoglobulin (Ig)G was also evaluated as an aid for diagnosing EBA. Significantly higher concentrations of IgG were identified in fetuses infected with aoEBA as compared to those with alternate infectious etiologies. The presence of IgG is a sensitive indicator of EBA and increases the specificity of FAT-based serologic diagnosis when titers are 10 or 100. Taken together, serology and IgG analyses suggest that the incidence of EBA may be underestimated.
Introduction
Epizootic bovine abortion (EBA), also known as foothill abortion, is a tick-borne disease prevalent in cattle pastured during the first and second trimesters of pregnancy in the dry foothill and mountainous regions of California, 3 northern Nevada, 7 and southern Oregon.5,16 Grazing practices place beef cattle in the same “foothill” habitat as the vector Ornithodoros coriaceus, 14 whereas dairy cattle are typically confined to environments that do not harbor this unique tick. A prolonged incubation period following infection of the dam typically results in third trimester abortions, premature delivery, or the birth of weak calves that fail to thrive. Aborted fetuses present with gross pathologic changes that may include splenomegaly, hepatopathy, ascites, and dramatic lymphadenopathy. Characteristic lesions include lymphoid hyperplasia with macrophage infiltration, acute vasculitis, and pathognomonic thymic lesions. 10 Diagnosis has been historically based on histology, elevated fluid immunoglobulin (Ig)G, 13 and dam history. The etiologic agent of EBA (aoEBA) is now known to be a novel organism in the class Deltaproteobacteria within the order Myxococcales, 11 and diagnosis is currently being supplemented with immunohistochemical staining (IHC) 1 and polymerase chain reaction (PCR)4,11 assays to detect the microbe.
Serum antibodies binding aoEBA have been identified in aborted bovine fetuses through application of IHC and fluorescent antibody test (FAT) techniques on infected necropsy tissues.1,2 Fluorescent antibody test cross-reactivity, using serum derived from fetuses infected with aoEBA, 2 was minimal when applied to a variety of bacteria, including the closest known relative, Sorangium cellulosum. Weak binding was noted only with Staphylococcus aureus and presumed to be due to antibody binding to protein A on the surface of this organism. Application of IHC, 1 using fluid derived from fetuses infected with aoEBA, onto Lawsonia intracellularis–infected porcine necropsy tissues demonstrated cross-reactivity (unpublished data). Lawsonia intracellularis is the only other animal pathogen identified within the Deltaproteobacteria class. While L. intracellularis has been detected in a small percentage of diarrheic calf feces using PCR, 8 it is not considered a disease of cattle and is not known to infect fetuses of any species.
A diagnostic assay for identifying antibodies specific for aoEBA has not been available due largely to the inability to successfully propagate the bacteria in vitro. Bacteria obtained from bovine fetuses are opsonized with Ig, precluding use of anti-bovine Ig secondary antibodies. 2 Bacteria propagated in severe combined immunodeficient (SCID) mice are free from opsonization by Ig, 2 but bacteria are difficult to purify. Attempts to develop a reproducible enzyme-linked immunosorbent assay–based serology test have been thwarted by unacceptable and highly variable ratios of host to bacterial proteins.
The primary objective of the current study was to validate an indirect FAT (IFAT) for detection of antibodies specific for aoEBA in postmortem fetal and precolostral neonatal fluids. A secondary goal was to better document the diagnostic potential of IgG concentrations in aborted bovine fetuses by comparing fluids derived from fetuses infected with aoEBA with those diagnosed with alternate etiologies.
Materials and methods
Fetal fluid samples
A total of 340 fluid samples, harvested from the blood vasculature or peritoneal cavity, were obtained from fetuses and precolostral neonates. Animals are collectively referred to as “fetuses” and fluids as “fetal fluid” from this point forward. Fluids were stored at −20°C. Fetal fluid quality ranged in opacity from clear to cloudy and in color from straw to black (secondary to hemolysis). This full range of fluid quality was represented in each diagnostic category described (Table 1).
Summary of bovine fetus and neonate diagnoses and corresponding agent of epizootic bovine abortion (aoEBA) indirect fluorescent antibody test (IFAT) serology.
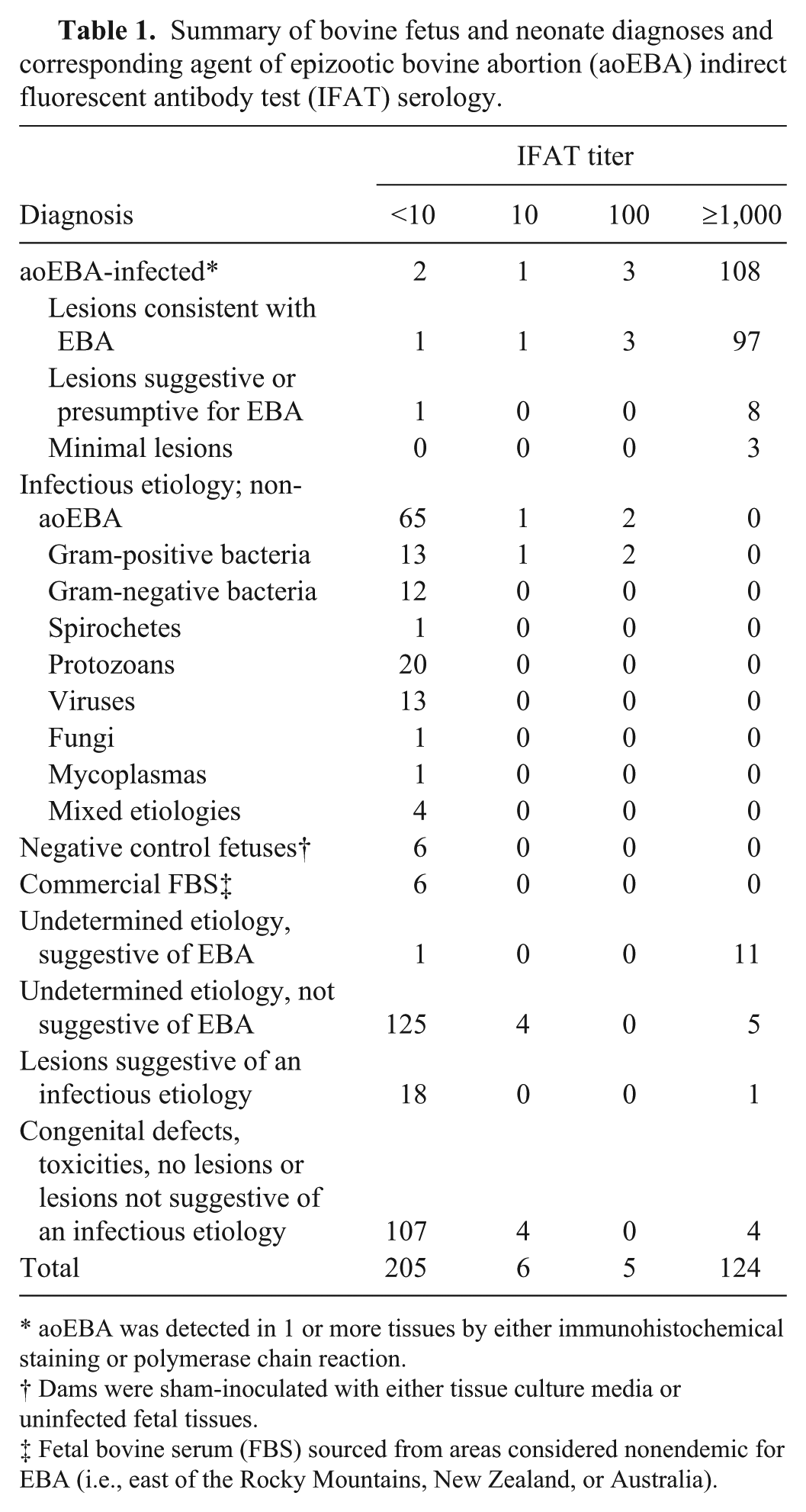
aoEBA was detected in 1 or more tissues by either immunohistochemical staining or polymerase chain reaction.
Dams were sham-inoculated with either tissue culture media or uninfected fetal tissues.
Fetal bovine serum (FBS) sourced from areas considered nonendemic for EBA (i.e., east of the Rocky Mountains, New Zealand, or Australia).
Fetal fluids were obtained from 1 of 4 sources. The majority were collected from submissions to the Davis and Tulare branches of the University of California Animal Health and Food Safety (UC-CAHFS) Laboratory System (n = 253). An additional 75 fetuses were recovered from experimentally infected dams. Heifers were inoculated between 85 and 125 days gestation with either fetal bovine tissues (n = 23) or murine tissues (n = 52) infected with aoEBA as previously described. 2 Two fetuses were taken at the time of dam euthanasia while all others were collected following natural delivery. Experimentally infected cattle were maintained on irrigated pasture at the Main Station Field Laboratory, University of Nevada, Reno (UNR-MSFL), free from natural exposure to the tick vector. All cattle research was conducted in compliance with the U.S. Federal Animal Welfare Act and the Health Research Extension Act and under approval by the UNR Institutional Animal Care Use Committee.
Twelve fluid samples served as negative reference sera. Six fetal bovine sera (FBS) were purchased from 3 commercial vendorsf,g,h and were sourced from New Zealand, Australia, or U.S. states considered nonendemic for EBA. An additional 6 fluids were obtained from fetuses recovered from dams maintained at UNR-MSFL that had been sham-inoculated with either tissue culture media or uninfected control fetal bovine tissue.
Clinical diagnosis
All fetuses, both experimentally infected and field-derived, underwent diagnostic evaluation at UC-CAHFS. Diagnoses were based on gross and histologic examinations, supported by standard assays to detect viral, fungal, or bacterial agents that may cause bovine abortion. Fetal fluids were tested for antibodies directed against common bovine abortigenic pathogens including infectious bovine rhinotracheitis (IBR; Bovine herpesvirus 1), Bovine viral diarrhea virus (BVDV), Neospora caninum, and Leptospira spp. Tissues from fetuses with lesions consistent with or suspected of EBA and fetuses with IFAT antibody titers ≥1,000 but undetermined etiologies were examined using IHC as previously described. 1 A quantitative PCR assay specific for aoEBA 4 was conducted on frozen necropsy tissues in cases where IHC results were equivocal or negative and frozen tissues were available. Thymus, spleen, prescapular lymph node, and mesenteric lymph node were the preferred tissues for PCR analysis.1,4
Fetuses were assigned a definitive diagnosis of EBA if aoEBA bacteria were detected in 1 or more tissues (aoEBA-infected). The remainder was categorized as either those diagnosed with infectious etiologies from microorganisms other than aoEBA or those with undetermined etiologies (Table 1). Fetuses with undetermined etiologies were subdivided into those that presented with lesions consistent with or suggestive of EBA but without detection of the bacterium, those suspected of an infectious etiology but lesions were not suggestive of EBA, and those lacking evidence of an infectious etiology. The latter group included those with congenital defects, toxicities, minimal lesions, or no noted lesions.
Bacterial antigen and slide preparation
Four- to 5-week-old female SCID mice (BALB/c-SCID a ) were infected with the aoEBA. 2 The mice were inoculated by intraperitoneal injection (IP) with aoEBA-infected murine splenocytes. Spleens were harvested following a prolonged incubation period (8–10 weeks) in which the most obvious sign of infection was cachexia. Termination was based on a combination of body condition scores, weight loss, and overall health. The mice were housed at the University of California, Davis (UCD) and studies were conducted in compliance with the U.S. Federal Animal Welfare Act and the Health Research Extension Act and overseen by the UCD Institutional Animal Care Use Committee.
Following euthanasia, single-cell suspensions were obtained by gently pressing spleen tissue through 100-µm nylon mesh screens. b Cell preparations were washed twice in Dulbecco phosphate buffered saline (PBS) and pelleted at 5,220 × g using a microfuge. Supernatant was removed, the pellet resuspended in PBS, and cells enumerated following dilution in 4% acetic acid. The suspension was adjusted to a final concentration of 3.3 × 107 cells/ml with PBS, and 1.5 µl was evenly dispersed over each well surface of 12-well slides. c Slides were fixed in 99.6% acetone for 5 min at room temperature. Slides were air-dried and stored at −20°C.
Indirect fluorescent antibody test
Bovine fetal fluid samples were serially diluted 10-fold with PBS, beginning at 1:10 with a maximum of 4 dilutions. Fifteen-microliter aliquots of diluted fluid were added to antigen-coated wells and incubated at room temperature for 20 min. Fluids were removed by aspiration, and slides were gently washed by applying PBS in a dropwise fashion. Following a 5-min incubation in a PBS bath, goat anti-bovine IgG (heavy + light chains) fluorescent dye conjugate, d was added to each well. Slides were incubated at room temperature for 15 min, washed as before, and coverslips mounted using mounting medium. e A negative control FBS f and a positive control, consisting of a pool of fluid from 3 fetuses derived from dams inoculated with aoEBA-infected bovine tissues and diagnosed with EBA, were tested with each assay run.
Slides were viewed under both 10× and 20× objectives, representing magnifications of 100× and 200×. Fluid samples with a staining pattern consistent in size, shape, and distribution to positive control serum were deemed positive under 100× magnification and confirmed at 200×. Wells without staining of bacterial organisms under 100× magnification were considered negative for that dilution.
Determination of assay sensitivity and specificity
Specificity and sensitivity calculations were based on comparisons of serologic data from fetuses infected with aoEBA (n = 114) with those diagnosed with alternate infectious etiologies (n = 68). Samples with undetermined etiologies and negative reference sera were not included in these calculations.
The ordinal nature of the data and the minimal number of samples with titers of 10 or 100 limited the utility of a receiver operating characteristic (ROC) curve in selecting criteria for calculating sensitivity and specificity. Calculations are presented based on 1 of 3 cutoff criteria: titers ≥10, ≥100, and ≥1,000 (Table 2).
Sensitivity, specificity, and positive likelihood ratios (LR+) for epizootic bovine abortion diagnosis based on elevated serology titers and immunoglobulin (Ig)G using various criteria, alone and in combination.*
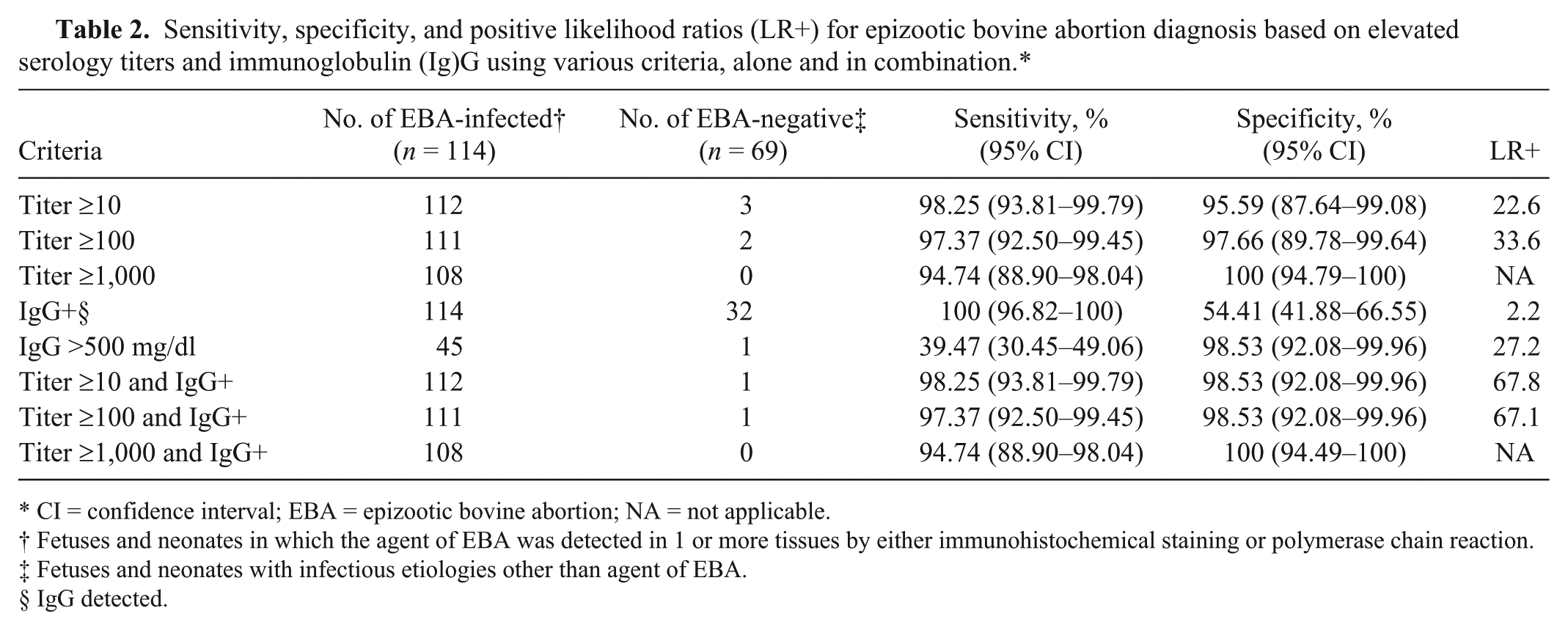
CI = confidence interval; EBA = epizootic bovine abortion; NA = not applicable.
Fetuses and neonates in which the agent of EBA was detected in 1 or more tissues by either immunohistochemical staining or polymerase chain reaction.
Fetuses and neonates with infectious etiologies other than agent of EBA.
IgG detected.
Assay repeatability
Assay repeatability was measured by conducting runs containing 6 replicates of 10 positive and 8 negative fetal fluids. Runs were performed on 7 separate occasions using slides from a single antigen preparation. Serial dilutions were prepared for positive fluids as described above while negative fluids were run at a single 1:10 dilution. Antibody titer constituted an observation. Forty-two observations per fluid sample and 756 total observations were possible.
Immunoglobulin G determination
Fluids were routinely analyzed for IgG concentration using a radial immunodiffusion assay i on all animals submitted to the CAHFS-Davis laboratory while those submitted to Tulare were selectively analyzed. Data was available for all samples with confirmed or suspected infectious etiologies and 86 of 115 fetuses with noninfectious and/or undetermined etiologies. Fetal fluids were categorized into 6 groups for comparative analysis: (1a) aoEBA-infected, transmitted by infected bovine tissue, (1b) aoEBA-infected, transmitted by infected murine tissue, (1c) aoEBA-infected, transmitted by tick bite, (2) fetuses with alternate infectious etiologies, (3a) fetuses of undetermined etiologies with aoEBA serology titers <1,000, and (3b) fetuses of undetermined etiologies with aoEBA titers ≥1,000. Immunoglobulin G concentrations were analyzed as a function of the method of transmission, clinical diagnosis, and serologic titer. Sensitivity, specificity, and likelihood of a positive diagnosis were calculated for fetal fluids with detectable IgG and for those with IgG >500 mg/dl. Data from IgG and IFAT assays were analyzed in combination to determine utility as a tool for improving diagnoses.
Statistical analysis
Sensitivity, specificity, and positive likelihood ratios
Calculations for sensitivity, specificity, and positive likelihood ratios (LR+) were conducted with commercial software k using Fisher exact test.
Assay repeatability
Variance component models for estimating reliability were applied. A cross-classified random effects model with 3 levels was applied: fetal fluid replicates (i; 1–6), fetal fluid (j; 1–18), and batch (k; 1–7), where fluid replicates were nested within fetal fluid and fetal fluid was cross-nested in batch. Variance components were estimated from a ordinal logistic multilevel model (equation [1]), with random intercepts for batch (τ k ) and fetal fluid (μ j ) to account for variability attributable to batch and cross-classification of fetal fluid, respectively (Bell AB, Owens CM, Kromrey JD, Ferron JM: 2009, Parsimony vs. complexity: a Monte Carlo investigation of hierarchical and cross-classified modeling using SAS® PROC MIXED. Paper 199-2009. SAS Global Forum 2009). 12 The outcome was ordinal (m; 1 = <10; 2 = 1,000; 3 = 10,000), and the models were fitted using PROC GLIMMIX using residual pseudo-likelihood subject specific expansion method (RSPL) with the optimization technique of Newton–Raphson with ridging. j
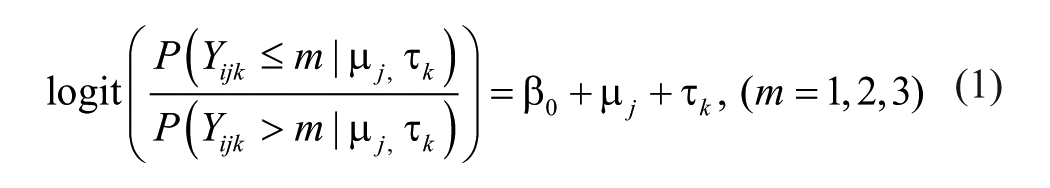
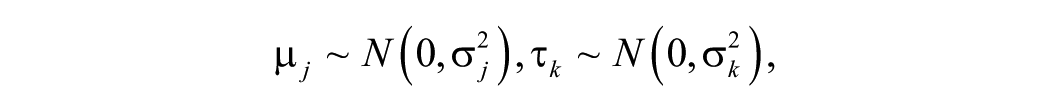
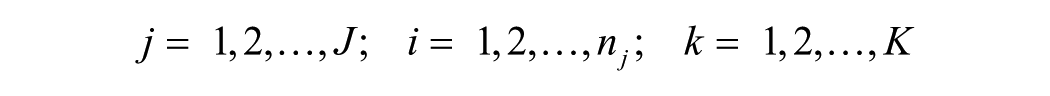
Reliability was estimated by intraclass correlation coefficient (ICC, ρ) and using a latent class approach (equation [2]).6,9,15
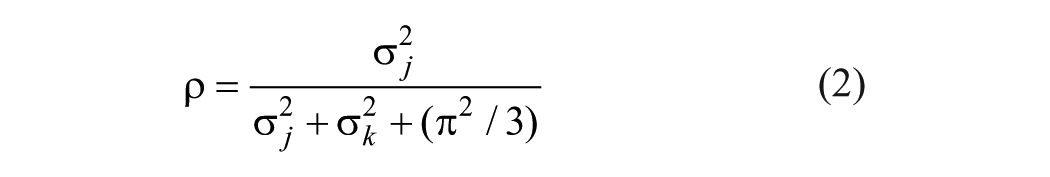
Comparison of IgG concentrations
Normal distribution was evaluated using D’Agostino–Pearson omnibus test. Statistical differences in fetal fluid IgG concentrations were determined using the Kruskal–Wallis tests, and individual groups were compared using Dunn multiple comparisons test. Samples with undetectable or trace IgG were assigned values of 0 and 150 mg/dl, respectively, while those reported as >2,748 was assigned a value of 2,800 mg/dl. Analyses were conducted using commercial software. k
Results
Clinical diagnosis
Following diagnostic evaluation, 114 of 334 fetuses submitted to CAHFS (34.1%) were determined to be infected with aoEBA. The majority (n = 102) presented with lesions characteristic of the disease, and all infections were confirmed by IHC. Bacteria were detected by IHC in 4 of 9 fetuses with lesions suggestive of, or presumptive for, EBA; the remaining 5 were confirmed by PCR. Minimal lesions were reported in 3 fetuses and all required PCR for confirmation.
Fetuses infected with a microorganism other than aoEBA constituted 20.4% of the samples (n = 68). Bacterial infections included Trueperella pyogenes (n = 7), Streptococcus spp. (n = 5), Escherichia coli (n = 4), Salmonella spp. (n = 3), Bacillus spp. (n = 2), Campylobacter spp. (n = 2), Listeria ivanovii (n = 1), Staphylococcus sp. (n = 1), Mannheimia haemolytica (n = 1), Leptospira sp. (n = 1), and 2 unidentified Gram-negative rods. Neospora caninum was responsible for all protozoal infections (n = 20), and viral etiologies were either IBR (n = 10) or BVDV (n = 3). One fetus was infected with Aspergillus fumigatus, 1 with Ureaplasma diversum, and 4 were diagnosed with mixed etiologies.
Fetuses with undetermined etiologies made up 43.7% of those studied (n = 146). Twelve fetuses presented with lesions consistent with or suggestive of EBA but without detection of the bacterium, 20 were suspected of an infectious etiology but lesions were not suggestive of EBA, and 115 lacked evidence of an infectious etiology. The 6 fetuses donating negative reference sera had no remarkable lesions. Data for all diagnostic groups are summarized in conjunction with aoEBA serology data in Table 1.
Diagnostic sensitivity and specificity of aoEBA serology
The bacterial rods identified in the current study using IFAT were of similar size and shape to those previously observed using IHC (Fig. 1). 1 Antibody titers were routinely detectable in fluids derived from aoEBA-infected fetuses but uncommon in fetal fluids with alternate infectious etiologies (Fig. 2; Table 2). All negative reference sera had titers <10. Fluid samples with titers ≥1,000 had a high degree of confidence for accurate diagnosis of fetuses infected with aoEBA (specificity 100%; 95% confidence interval [CI] = 94.79–100) while maintaining good sensitivity (94.7; 95% CI = 88.9–98.04). Setting a titer criteria of ≥100 provided the best balance in assay sensitivity and specificity; both were >97% (Table 2).
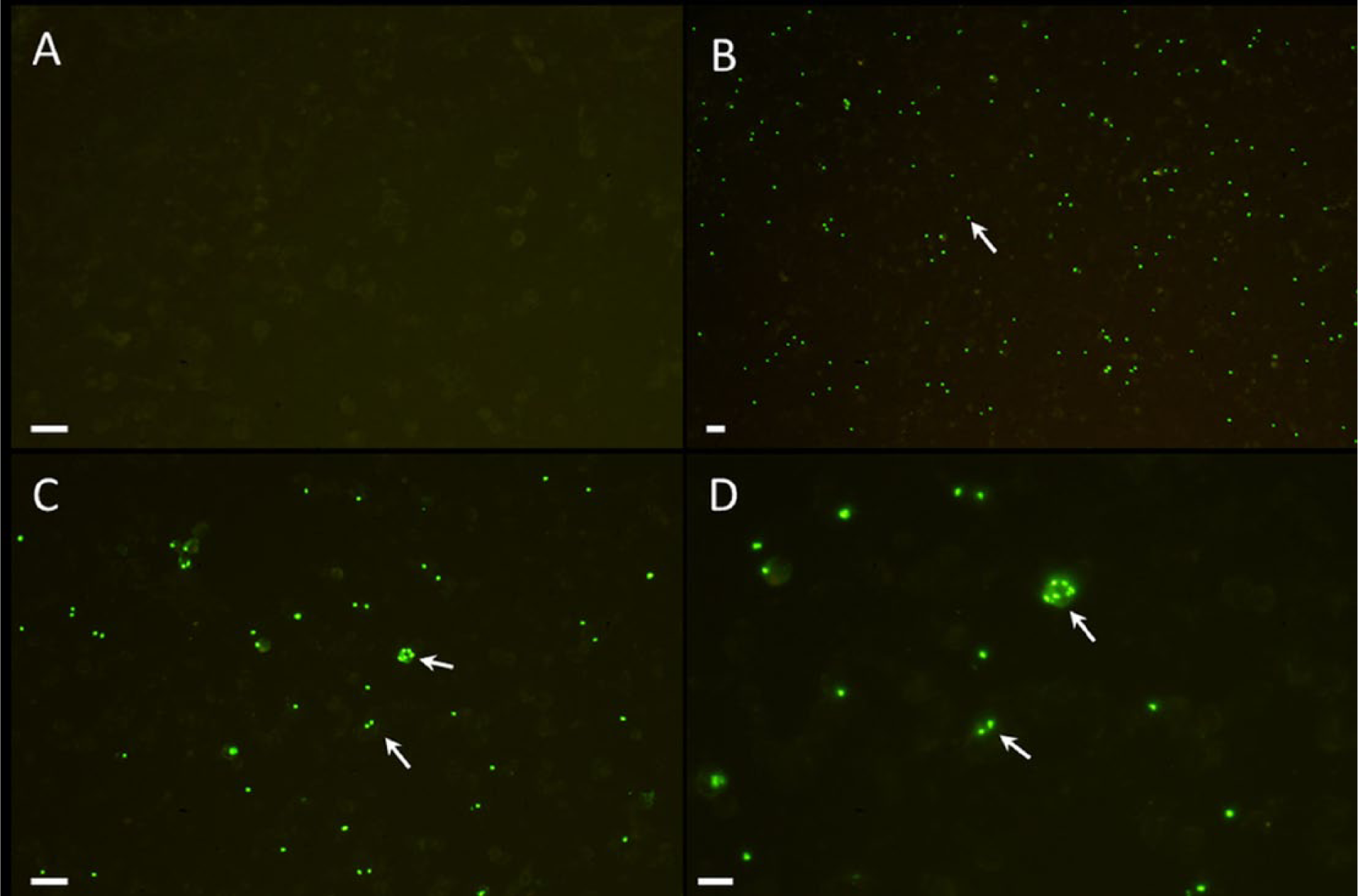
Murine spleen cell preparations infected with the agent of epizootic bovine abortion (aoEBA) were probed with either negative control fetal calf serum (
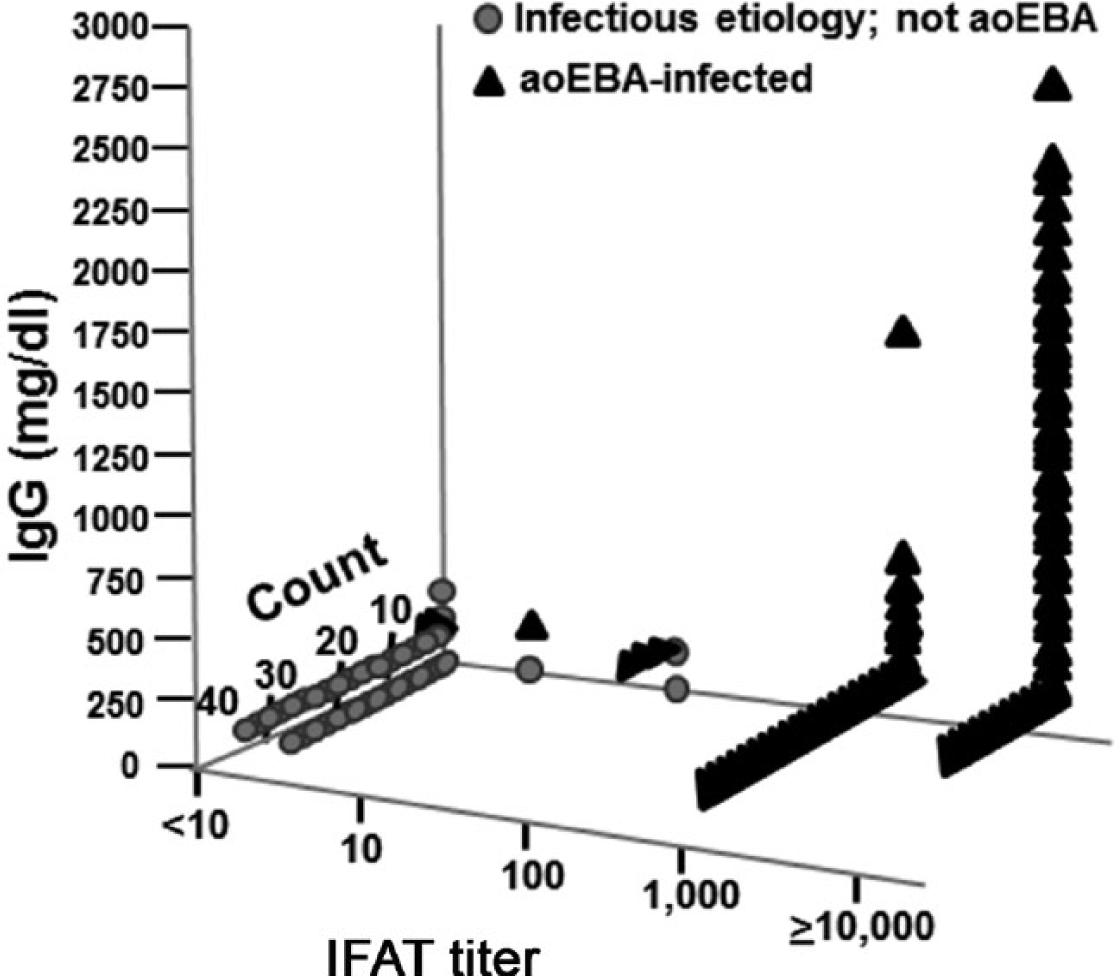
Indirect fluorescent antibody test titers and immunoglobulin (Ig)G concentration were compared between fetuses infected with the agent of epizootic bovine abortion (aoEBA) and those diagnosed with an alternate infectious etiology.
Antibody cross-reactivity
Fluids from 3 fetuses with Gram-positive bacterial etiologies were positive by aoEBA IFAT. Titers of 100 were detected in 1 fetus infected with L. ivanovii and 1 fetus infected with Staphylococcus sp. A titer of 10 was detected in 1 fetus infected with an unidentified Streptococcus sp.
Antibody titers in fetuses with undetermined etiologies
Antibody titers ≥1,000 were detected in 16 of 146 fetuses classified with undetermined etiologies (Table 1). The majority were from those with lesions suggestive of EBA (n = 11). Tissues were available for PCR analysis from 7 fetuses in the group suggestive of EBA and 2 in the group not suggestive of EBA.
Assay repeatability
Seven hundred fifty observations were available to evaluate assay repeatability; data from 6 samples were discarded due to lack of antigen in the wells. Three hundred thirty-six observations were made from 8 negative sera, and all had titers <10. Five positive sera had titers of ≥10,000 in 206 of 208 observations. The 2 discrepant results were titers of 1,000 and both were from the same fetal fluid sample. The remaining 5 positive fluids had titers of 1,000 in 204 of 206 observations. Two discrepant observations of ≥10,000 were from the same fetal fluid sample. The variability in EBA titers attributable to fetal fluid and batch was 98.83% (95% CI = 95.98–101.68) and 0.065% (95% CI = 0–0.53), respectively. The ICC (ρ) within laboratory was 0.97 indicating that the similarity of results in replicates of fetal fluid samples of different batches was high.
Immunoglobulin G quantitation
Immunoglobulin G was detected in all fluids from aoEBA-infected fetuses and those suspected of infection based on antibody titers ≥1,000 (groups 1a–c and 3b; Fig. 3). The majority of fetuses with alternate infectious etiologies, undetermined etiologies, or those not suspected of aoEBA infection, had either no detectable IgG or IgG was detected in trace amounts but not quantifiable (groups 2 and 3a; Fig. 3). Quantifiable levels of IgG were detected in 4 fetuses in these later 2 groups; 1 each infected with Streptococcus sp., BVDV, U. diversum, and an unknown agent (254, 362, 601, and 382 mg/dl, respectively). Immunoglobulin G was not detected in the 6 negative reference sera collected experimentally. Purchased FBS all contained <50 mg/dl of IgG, based on vendor-provided documentation.
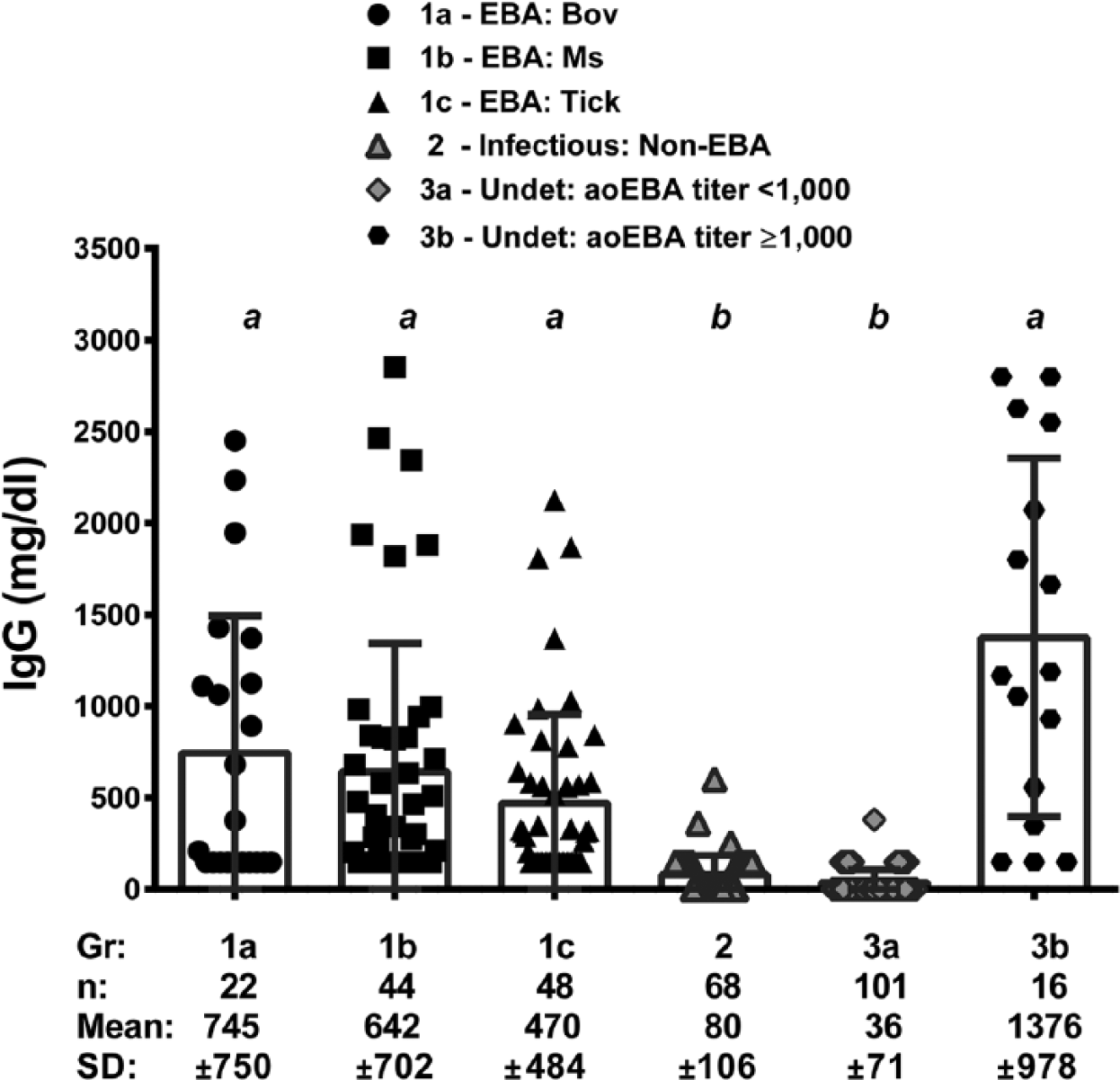
Comparison of immunoglobulin (Ig)G in fluids from fetuses and neonates as a function of the mode of the agent of epizootic bovine abortion (aoEBA) transmission and diagnosis. Fetuses in which aoEBA was detected in 1 or more tissues (EBA) were compared to those with other infectious (infectious: non-EBA) and undetermined (undet) etiologies. Fetuses infected with aoEBA were subdivided by experimental inoculation of aoEBA-infected bovine tissue preparations (EBA: bov), aoEBA-infected murine tissue preparations (EBA: ms), and natural tick bite (EBA: tick). Submissions with undetermined etiologies were divided based on aoEBA antibody titers (<1,000 vs. ≥1,000). Samples with undetectable or trace IgG were assigned values of 0 and 150 mg/dl, respectively, while those reported as >2,748 was assigned a value of 2,800 mg/dl. Groups with significant differences (P < 0.001) are labeled with different lowercase letters (a vs. b).
Immunoglobulin G data was not normally distributed, and comparisons were made using nonparametric methods. No significant differences were observed in IgG between the 4 groups with confirmed or suspected aoEBA infections or between the 2 groups lacking evidence of infection with aoEBA (P > 0.05; Fig. 3). Groups confirmed with or suspected of an EBA diagnosis were significantly different from those lacking the diagnosis (P < 0.001; Fig. 3).
Immunoglobulin G sensitivity and specificity
Detection of IgG was a sensitive indicator of EBA, but lacked specificity. Conversely, IgG levels >500 mg/dl correlates to good specificity but poor sensitivity (Table 2). Diagnostic sensitivity was not altered, regardless of titer criteria, when IgG was examined in conjunction with antibody titer (Fig. 2, Table 2). The addition of IgG data does not change diagnostic specificity when applied to fetuses with titers ≥1,000, already at 100%. However, when taken together, diagnostic specificity was increased for fetuses with antibody titers of 10 or 100. Fetuses with elevated titers and detectable IgG are 3 times more likely to have been infected with aoEBA as compared with those evaluated by titer alone (LR+ of 67.8 and 22.6, respectively; Table 2).
Discussion
The IFAT-based serology assay discussed in the current study is 100% specific (95% CI = 94.79–100) and 94.74% sensitive (95% CI = 88.9–98.04) when a titer criteria of ≥1,000 is applied. Titers of ≥10,000 are common in aoEBA-infected fetuses (Fig. 2) and indicate that this assay is capable of detecting low concentrations of antibody specific for the aoEBA. Antibody titers of ≥1,000 confirmed aoEBA infections in 11 of 12 fetuses that had been classified as presumptive for EBA based on histology and elevated Ig in the absence of identifiable bacteria. This is consistent with previous demonstrations that an infected fetus can mount a robust immune response that is likely responsible for bacterial clearance prior to abortion. 4
Data from the current study suggests EBA is underdiagnosed. Three aoEBA-infected fetuses with minimal lesions and 5 fetuses diagnosed with undetermined etiologies not suggestive of EBA were determined to have antibody titers ≥1,000. All 8 of these submissions were beef cattle; 4 were known to have been pastured in tick habitat early in gestation and 3 were recovered from dams experimentally inoculated with aoEBA. Immunoglobulin G was elevated in all, and concentrations of >500 mg/dl were detected in 2 aoEBA-infected fetuses and 3 with undetermined etiologies. Taken together, data suggests these 5 undetermined fetuses represent true EBA positives.
Fluids from 4 fetuses with undetermined etiologies and without evidence of infection had antibody titers of 10. All were dairy cattle, and vector exposure was unlikely. Two of these fetuses had no detectable IgG, and 2 had trace levels of IgG. Taken together, it is unlikely these fetuses were infected with aoEBA.
Fluid from fetuses with alternate infectious etiologies, representing at least 15 different genera of microorganisms, demonstrated limited antibody binding to aoEBA. Fluids derived from fetuses infected with 1 of 3 Gram-positive organisms (L. ivanovii, an unidentified Staphylococcus, or an unidentified Streptococcus) had aoEBA titers of either 10 or 100 (Table 1). The fetus infected with L. ivanovii was from a dam known to have been pastured in O. coriaceus habitat, and therefore an underlying infection with aoEBA cannot be ruled out. The other 2 dams were dairy cattle and, while vector exposure is unknown, contact between the dams and ticks was unlikely. A previous study demonstrated that antibody derived from aoEBA-infected fetuses was weakly cross-reactive with S. aureus but not cross-reactive with Streptococcus agalactiae or Listeria monocytogenes. 2 The cross-reaction with S. aureus was hypothesized to be the result of nonspecific binding of Ig to protein A on the bacterial surface, but data presented herein suggests that it may have been due to specific binding. In summary, IFAT titers ≥1,000 would appear diagnostic for EBA.
Increased IgG in fluids from EBA fetuses is common knowledge among diagnosticians familiar with the disease. However, the range in concentrations of IgG in fluids from EBA as compared to non-EBA abortions is lacking. Elevated levels of IgG in EBA fetal fluids were first noted in 1973 13 and were reported as mean IgG concentrations of approximately 350 mg/dl. This data was compared to IgG in fetuses experimentally infected with chlamydia, anaplasmosis, Q fever (Coxiella burnetii), Campylobacter fetus, BVDV, and Bluetongue virus infections. Fetuses diagnosed with EBA presented with higher IgG on average, but neither the range nor standard deviations were provided. The data presented herein supports a high degree of confidence that fetuses infected with aoEBA will have elevated IgG and that IgG >500 mg/dl correlates well with EBA diagnosis. Combining the presence of elevated levels of total IgG with aoEBA antibody titer enhanced the diagnostic specificity of the latter (Table 2).
To summarize, the IFAT-based serologic assay described herein is specific and sensitive. Turnaround time is rapid, and fluid quality does not appear to affect the results. The high reliability coefficient indicates the test is repeatable. While the assay does not easily lend itself to automation, several dozen samples can be assayed within a few hours by experienced personnel and thus can easily keep pace with demand. Diagnosticians in EBA-endemic regions should consider routine submission of fetal and precolostral neonatal fluid for determination of total IgG in fetuses with an undetermined etiology of abortion. Follow-up with aoEBA serology in fetuses with elevated IgG will improve diagnosis, especially in fetuses where pathologic lesions are not characteristic of the disease.
Footnotes
Acknowledgements
The authors would like to thank the staff of the Davis and Tulare branches of the California Animal Health and Food Safety Laboratory System, University of California for assistance in providing samples and diagnostic data for this study. The authors would also like to thank the staff at the University of Nevada Main Station Field Laboratory in Reno, Nevada and the Sierra Foothill Research and Experimental Station at the University of California for animal care as well as assistance in animal handling and sample collection.
a.
The Jackson Laboratory, Bar Harbor, ME.
b.
BD Biosciences, San Jose, CA.
c.
Teflon-coated slides, Cel-Line, Thermo Fisher Scientific Inc., Waltham, MA.
d.
DyLight 488, Jackson ImmunoResearch Laboratories, West Grove, PA.
e.
Shandon Immu-mount, Thermo Fisher Scientific Inc., Waltham, MA.
f.
Atlanta Biologics, Lawrenceville, GA.
g.
Gemini Bio-products, West Sacramento, CA.
h.
Gibco, Invitrogen Corp., Carlsbad, CA.
i.
Triple J Farms, Bellingham, WA.
j.
Version 9.3, SAS Institute, Cary, NC.
k.
Prism version 6.02, GraphPad Software Inc., San Diego, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported in part by funds provided by the Center for Food Animal Health, School of Veterinary Medicine, University of California, Davis; USDA Formula Funds, USDA Western Regional Research Project W-112 and the California Cattlemen’s Association.
