Abstract
Epizootic bovine abortion (EBA), also commonly known as “foothill abortion,” is a late-term abortion primarily in beef cattle with significant economic impacts in California, Nevada, and Oregon. The causative agent is a novel deltaproteobacterium (aoEBA) closely related to the order Myxococcales and vectored by the soft-shelled tick Ornithodoros coriaceus. Historically, diagnosis has relied upon the pathologic examination of the fetus and the presence of elevated fetal serum immunoglobulins. Identification of the etiologic agent, a unique deltaproteobacterium, permitted the development of a quantitative duplex real-time polymerase chain reaction (qPCR) using a unique 90-bp sequence of aoEBA 16S ribosomal RNA gene in conjunction with an 88-bp sequence of the bovine β-actin gene. Reaction efficiencies were 100.9% for the 16S aoEBA gene and 93.1% for the bovine β-actin gene. Application of the duplex TaqMan to a set of aoEBA-infected fetal bovine necropsy tissues demonstrated the assay to be robust in quantitatively identifying the aoEBA bacteria and establishing host-tissue pathogen load. Consistent with previously reported immunohistochemical data, organized lymphoid tissue generally carried the heaviest bacterial load as compared to non-lymphoid tissue. The newly developed duplex TaqMan assay will facilitate diagnosis in difficult cases and provide an invaluable tool for delineating the pathogenesis of EBA.
Introduction
Epizootic bovine abortion (EBA), also commonly known as “foothill abortion,” has been a major contributor of economic loss to the beef cattle industry for many years in California, Nevada, and Oregon.3,5,6 The etiologic agent of the disease has recently been identified as a deltaproteobacterium, most closely related to the order Myxococcales. 10 The bacterial agent is transmitted to naïve heifers by the bite of the soft-shelled tick, Ornithodoros coriaceus (Loomis EC, Schmidtmann ET, Oliver MN: 1974, A summary review on the distribution of Ornithodoros coriaceus Koch in California (Acarina: Argasidae). Calif Vector Views 21:57–62), 13 and preferentially resides in the tick’s salivary gland. 4
Epizootic bovine abortion is characterized by a late-term abortion or birth of a weak calf; fetal pathology is unique to the disease and serves as the primary diagnostic. The characteristic lesions, which develop in the third trimester, include mucosal and thymic hemorrhages, lymphadenopathy, splenomegaly, hepatomegaly, and ascites.6-9 A loss of cortical thymocytes and extensive infiltration of macrophages into the medulla of the fetal thymus are pathognomonic for EBA.1,7,9 Fetuses also have elevated levels of immunoglobulins (Ig) that are specific for the bacterium.2,12 The development of a reliable bovine challenge model in which lesions indistinguishable from those observed in naturally infected animals provided a much needed experimental system with which to identify the etiologic agent and advance EBA pathogenesis studies.1,9,16
Upon identification of a novel deltaproteobacterium as the causative agent of EBA (aoEBA) in 2005, new methodologies were developed to identify the bacterium in fetal and tick tissue1,4,10,17; sensitivity and specificity of a polymerase chain reaction (PCR)-based diagnosis approached 88% and 100%, respectively. 10 While diagnostic capabilities have improved, techniques for quantitative assessment of bacterial load in fetal tissues are needed as this bacterium has yet to be cultivated in vitro. 16
The present study was designed to develop a quantitative real-time PCR (qPCR) for the purposes of facilitating pathogenesis studies of aoEBA and disease diagnosis. To this end, a maximally unique 16S ribosomal RNA (rRNA) sequence was duplexed to the bovine β-actin (BACTB) gene to allow for absolute quantification of gene copy number based upon a plasmid standard curve. The assay was then used to assess pathogen load in a set of tissues harvested from experimentally infected EBA fetuses.
Materials and methods
Duplex TaqMan and standard curve design
A BLASTn search (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi) on the nucleotide collection (nr/nt) database was performed using the 16S rRNA gene sequence derived from aoEBA (AF503916) to identify the most closely related 16S gene sequences. Three uncultured deltaproteobacteria (DQ676313, AY395395, and AY922068) and 9 uncultured environmental bacteria (EU589312, DQ906906, EF516043, EF516279, EU669621, DQ451480, EF516312, EF516611, and AY662017) were selected for the primer-design alignment based on their highest homology with the aoEBA 16S rRNA. Sorangium cellulosum (previously Polyangium cellulosum, AY039304) and Chondromyces pediculatus (AJ233940), the closest identified relatives at the time of aoEBA identification, 10 were also included in the assembly and alignment. a Primers and probes were designed to amplify a maximally unique sequence of aoEBA, as compared to these closely related bacteria (Table 1).
Primer and probe sequences used in the duplex TaqMan in the current study.*
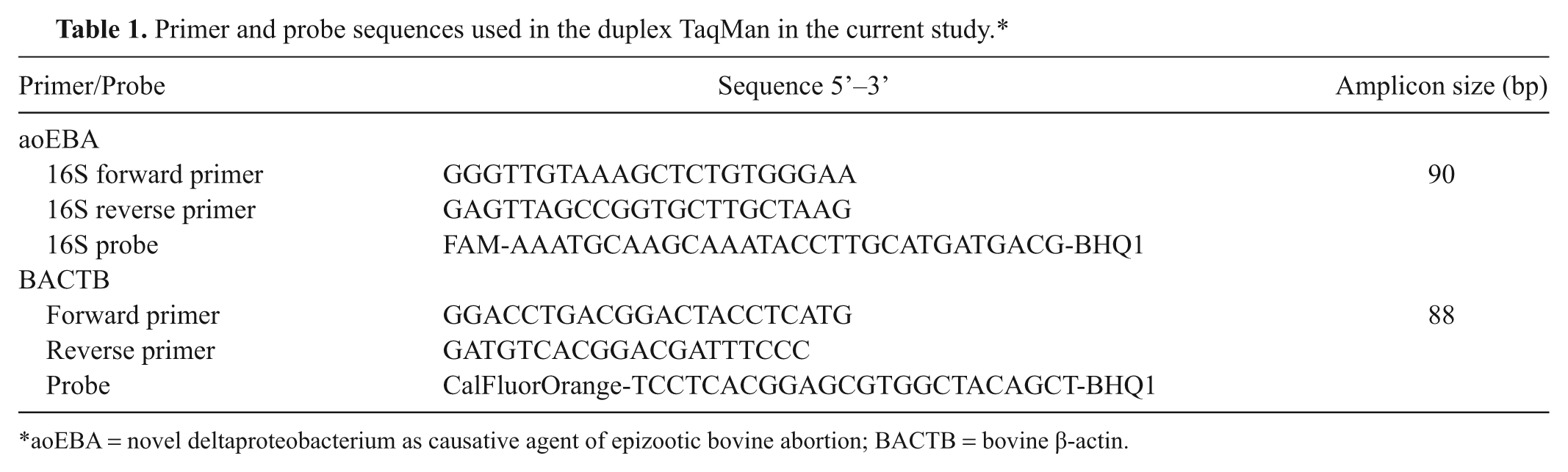
aoEBA = novel deltaproteobacterium as causative agent of epizootic bovine abortion; BACTB = bovine β-actin.
The complete BACTB gene sequence was obtained from the University of California, Santa Cruz Genome Bio-informatics Site (http://genome.ucsc.edu/). Software b was used to generate primers and probe (Table 1). All primers c and probes d were purchased from commercial vendors.
Twenty-five–microliter PCR reactions were performed using 12.5 µl of master mix containing ROX as a passive reference, e 3 µl of DNA, 100 nM of BACTB forward and reverse primers, 900 nM of aoEBA primers, and 250 nM of each probe. Amplification and detection were performed using a real-time thermocycler. f Data were analyzed using the corresponding software. The following conditions were used: 2 min at 50°C, 10 min at 95°C, and 40 cycles of 15 sec at 95°C and 1 min at 60°C. Efficiencies of both the single-plex reaction and duplex reaction were determined using serial dilutions of genomic DNA extracted from a heavily infected fetus.
Additional primers were designed to generate the plasmid for the standard curves by amplifying 442 bp of the aoEBA 16S gene and 387 bp of the BACTB gene, each containing their respective TaqMan amplification site. The forward primer sequence for aoEBA was 5’-GGATCCCGGCCCATCA-3’ and the reverse was 5’-GAATTCCCTCTCCGATACTCT AGC-3’. The forward primer sequence for the BACTB gene was 5’-GAATTCAGACCTTCAACACCCC-3’ and the reverse primer sequence was 5’-TCTAGATGACCTGCCC GTCAG-3’. Amplicons were separated on 1.5% agarose gel and purified, g then cloned individually. h Single strands of both plasmids were sequenced to ensure amplicon integrity.
The aoEBA and BACTB plasmids were combined and diluted (nuclease-free water) according to the Applied Biosystems plasmid standard curve protocol (www6.appliedbiosystems .com/support/tutorials/pdf/quant_pcr.pdf) resulting in 7 diluted standards ranging from 300,000 to 0.3 copy number. Standards were aliquoted and stored at −80°C. Aliquots were thawed once and immediately used. A standard curve was run on each plate for quantification purposes. Each standard was run in triplicate; the 3 threshold cycles (Ct) were averaged to generate the standard curve graph. Gene copy number was determined using the equation 10[(gene Ct-Y intercept)/slope]. Results are reported as the ratio of the aoEBA 16S gene copy number to the BACTB copy number. If a tissue aoEBA Ct did not fall upon the standard curve, it was assigned a pathogen-to-host ratio of 5.0e-5.
Animals and sample collection
Eight naïve pregnant heifers were experimentally inoculated with aoEBA between 102 and 111 days gestation using aliquots of an infected pooled bovine fetal spleen and thymus homogenate as previously described. 16 Two pregnant negative control heifers were inoculated at 102 and 117 days gestation with noninfected bovine fetal thymus homogenate. Cattle were maintained on irrigated pasture at the Main Station Field Laboratory at the University of Nevada (Reno, Nevada). All animal research was conducted in compliance with the United States Federal Animal Welfare Act and the Health Research Extension Act and under approval of the University of Nevada Institutional Animal Care Use Committee. Four principal fetuses were recovered following abortion at 114, 120, 121, and 125 days post-challenge (PC), 4 principal fetuses were collected following dam euthanasia at 119, 119, 126, and 126 days PC, and the 2 control fetuses were collected at 122 and 136 days PC. Complete necropsies, abortion panels to rule out other abortifactive agents, serum Ig levels, and aoEBA-specific immunohistochemistry (IHC) were performed on each fetus to confirm EBA diagnosis. Tissues from 6 infected and 2 control fetuses were processed for duplex TaqMan analysis; 2 principal fetuses (114 and 120 days PC) were not included due to extensive postmortem tissue degradation. The following tissues were aseptically removed from each fetus for DNA analysis: blood, mesenteric lymph node, prescapular lymph node, Peyer patch, skin, spleen, tongue, and thymus. Tissues were stored at −80°C prior to DNA extraction.
Immunohistochemistry, DNA extraction, assay specificity, and pathogen load in tissues
Immunohistochemistry was performed as previously described on all collected tissues. 1 Genomic DNA was extracted from cryopreserved samples according to manufacturer’s instructions i with the following modifications: 40 μl of proteinase K was used during the overnight digestion of tissues. Purified DNA was eluted from spin columns using 100 μl of nuclease-free water. Extraction controls were run with each DNA extraction to ensure that buffers and samples were not contaminated with bovine or bacterial DNA during the extraction process. Each tissue sample was run in triplicate, and the average Ct was calculated.
Specificity for the aoEBA duplex assay was assessed using DNA extracted from a combination of necropsy tissues derived from non-EBA fetal bovine abortions and pure bacterial cultures of established bovine pathogens. Tissue samples were obtained from the California Animal Health and Food Safety Diagnostic laboratory from fetal bovine abortion cases with Campylobacter fetus subsp. venerealis (lung), Staphylococcus sp. (lung)., Pasteurella multocida (spleen), Neospora sp. (spleen), and Bovine herpesvirus 1 (BHV-1; spleen) as causative agents. Additionally, DNA was extracted from 4 pure cultures of bacteria and included the smooth variant of Brucella abortus strain 19 (alphaproteobacteria), Listeria monocytogenes (class Bacilli), Streptococcus agalactiae (class Bacilli), and Mannheimia sp. (gammaproteobacteria).
Statistical analysis
Pathogen-to-host gene-copy ratios were transformed by the natural log for data analysis. A linear mixed effects model was used to test the differences in pathogen load based on tissue type. Tissue type was included as a fixed effect, and a compound symmetry structure was used to model dependence in the measurements taken from a single animal. Model fit was assessed using graphical analysis of the residuals and a Shapiro–Wilk test for normality. Variances were tested using Levene test and found to be equal. The Sidak method was used for post-hoc comparisons at the 0.05 level. j
Results
Duplex TaqMan performance
Reaction efficiencies were calculated using extracted genomic DNA to ensure that tissue-specific PCR inhibitors were not affecting the reactions. Prior to duplexing, the reaction efficiencies were calculated for each of the single-plex reactions. The single-plex aoEBA efficiency was determined to be 99.7% using 10-fold serial dilutions over a 4-log range. The single-plex BACTB efficiency was determined to be 99.3% using 10-fold serial dilutions over a 5-log range. Duplex reaction efficiencies, using 5-fold serial dilutions over a 4-log range of genomic DNA, were determined to be 100.9% for the 16S gene and 93.1% for the bovine β-actin gene (R2 > 0.99).
The slopes of the standard curves were used to monitor the integrity of each reaction plate (Fig. 1). An acceptable slope for either gene was between −3.1 and −3.6, otherwise the run was repeated. The reaction routinely detected as little as 3 copy numbers for both genes. The 0.3 copy number dilution was sporadically detected as expected, indicating that the standard curve dilutions had been performed correctly. Additionally, mixing high copy numbers (3e5) of 1 plasmid with low copy numbers (3) of the other plasmid showed that higher concentrations of an individual gene did not inhibit the amplification of the other (data not shown).
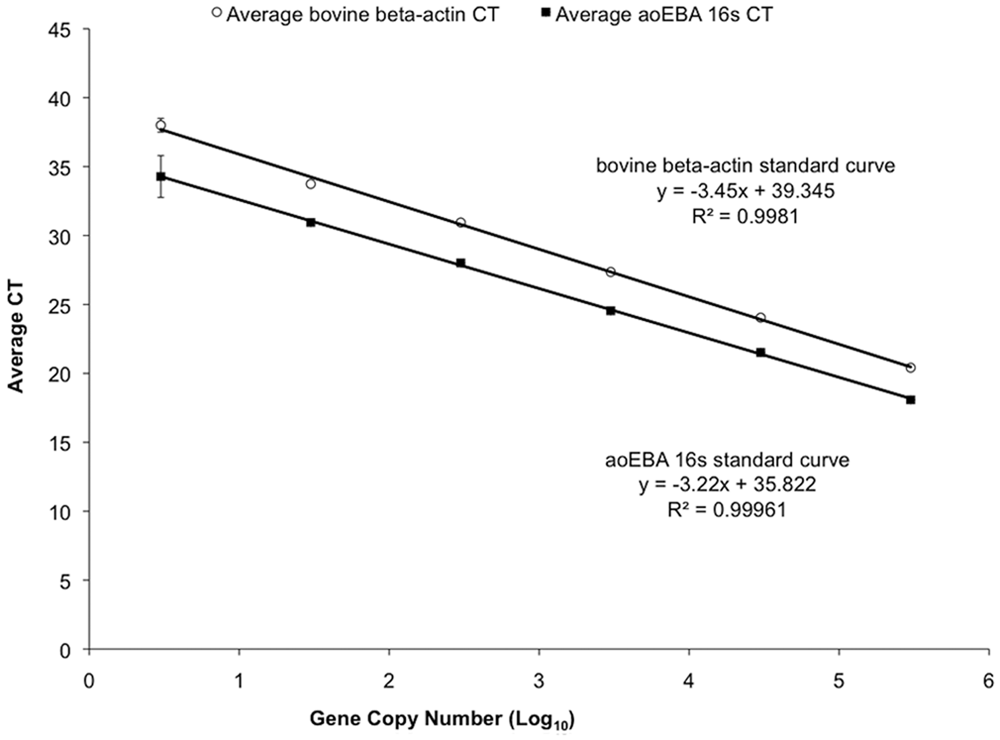
Example of plasmid standard curves used for absolute quantification of aoEBA (novel deltaproteobacterium as causative agent of epizootic bovine abortion) 16S and bovine β-actin genes. The arithmetic mean of triplicate reactions is shown in addition to the equations (y) used for calculations and correlation coefficients (R2). Error bars represent standard deviation.
The duplex was tested for cross reactivity with a few select bovine pathogens and was determined to be non–cross-reactive to C. fetus subsp. venerealis (epsilonproteobacteria), Staphylococcus sp. (class Bacilli), P. multocida (gammaproteobacteria), Neospora sp., BHV-1, the smooth variant of B. abortus strain 19 (alphaproteobacteria), L. monocytogenes (class Bacilli), S. agalactiae (class Bacilli), and Mannheimia sp. (gammaproteobacteria). The TaqMan assay amplified the bovine β-actin gene in DNA extracted from tissues, thus confirming DNA integrity and lack of PCR inhibitors. Universal 16S bacterial primers successfully amplified DNA extracted from the pure bacteria cultures to ensure DNA integrity.
Epizootic bovine abortion fetus TaqMan results
Ten fetuses were included in the present study; 8 were experimentally infected with aoEBA (animals 1–5, 7–9) and 2 served as negative controls (animals 6, 10). Animals are numbered in the order in which they were collected. The 2 negative controls were aoEBA negative by qPCR in all 8 tissues examined, had no detectable serum Ig, and had no lesions suggestive of EBA.
Seven (animals 1, 2, 4–9) of the 8 experimentally infected fetuses were confirmed to be EBA-positive by pathologic examination, fetal serum Ig quantitation, and IHC. Animals 1 and 4 were not subjected to TaqMan analysis due to severe autolysis. The remaining 5 fetuses were positive by qPCR in all 8 tissues taken for analysis. The eighth experimental fetus, animal 3, did not have any conclusive EBA lesions, was negative by IHC, and had no serum Ig. However, this animal was qPCR positive in all tissues except for the skin and the tongue.
Organized lymphoid tissue carried higher burdens than non-lymphoid tissues. Both the prescapular lymph node and mesenteric lymph node had significantly higher pathogen loads as compared to blood, Peyer patch, and tongue (P < 0.05). Spleen and thymus carried significantly higher loads as compared to tongue (P < 0.05; Fig. 2). The greatest variability in pathogen load was identified in thymus, skin, and Peyer patch. Distinct skin lesions were grossly evident on 2 fetuses (animals 7, 8). All tissues that were positive by IHC were also positive by TaqMan, while 14 out of 16 negative IHC tissues had detectable bacterial DNA (data not shown).
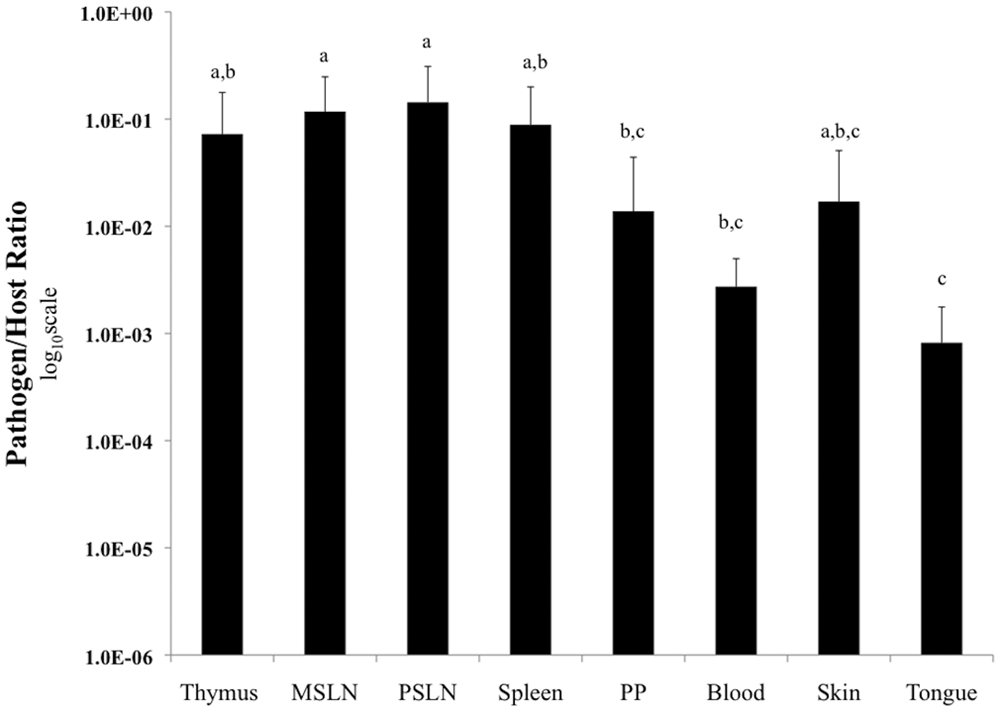
Linear mixed effects model using tissue as a fixed effect and a compound symmetry structure to compare average pathogen load in each tissue type (n = 6 infected fetuses). Tissues without a letter in common are significantly different (P < 0.05). Bars represent standard deviation. MSLN = mesenteric lymph node; PSLN = prescapular lymph node; PP = Peyer patch.
While the group of fetuses in the current study was small, the authors used the opportunity to preliminarily identify potential relationships between pathogen load and fetal lesion severity; fetuses were graded based upon histologic severity of lesions in 10 different tissues as previously specified. 9 Three distinct groups were created as follows: 1) inconclusive or non-diagnostic lesions, 2) mild lesions, and 3) moderate to severe lesions. All animals in groups 2 and 3 had histologic lesions consistent with EBA (Fig. 3).
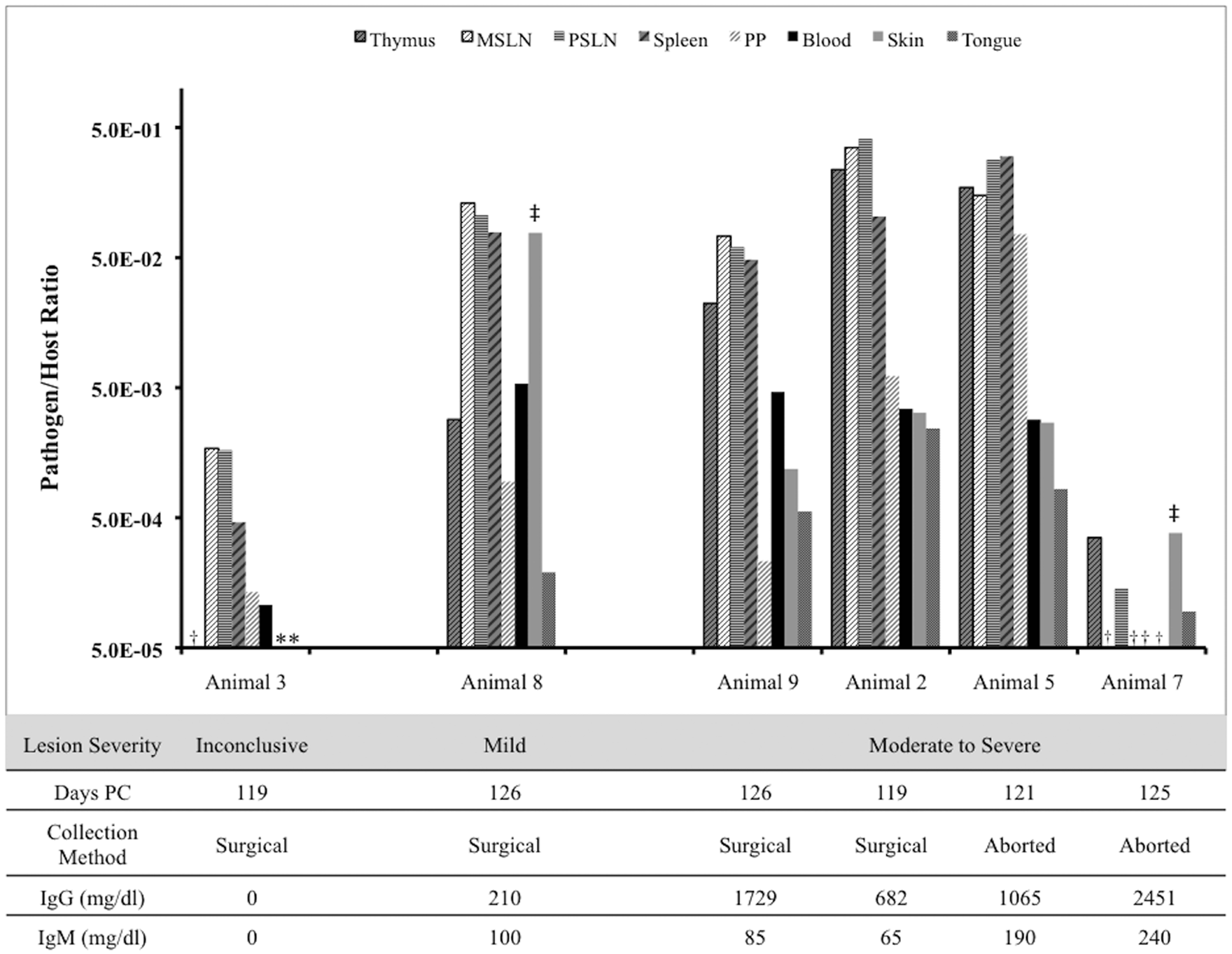
Pathogen load (log10 scale) in non-autolyzed aoEBA (novel deltaproteobacterium as causative agent of epizootic bovine abortion)-infected fetuses as a function of tissue type, lesion severity, days post-challenge (PC), fetal collection method, and immunoglobulin levels. Asterisk (*) indicates aoEBA not detected; dagger (†) indicates trace levels of aoEBA detected (assigned pathogen-to-host ratio of 5e-05); double dagger (‡) indicates visible skin lesion sampled. MSLN = mesenteric lymph node; PSLN = prescapular lymph node; PP = Peyer patch; IgG, IgM = immunoglobulin G and M, respectively.
Though statistical analysis was not applied to these data to differentiate fetal groups, minimal differences in pathogen load profiles between animals 2, 5, 8, and 9 were evident. Two animals (animals 3, 7) with the lowest pathogen loads were in distinct lesion severity groups and had the lowest and highest immunoglobulin concentrations. Diagnostic lesions were only identified in animals with detectable Ig, while bacterial DNA was identified in all infected fetuses.
Discussion
Historically, diagnosis of EBA has relied upon dam history (exposure in the first and second trimesters to habitat containing the tick vector, O. coriaceus) and compatible pathology in fetuses. Recent development of an immunohistochemical assay to visualize the bacterium in tissue has increased the specificity of diagnosis, 1 and conventional PCR has increased both sensitivity and specificity of diagnosis. 10 From a diagnostic perspective, IHC is laborious and has limited sensitivity while conventional PCR requires inclusion of a second PCR reaction to confirm DNA integrity (bovine reference gene), post-amplification processing, and sequencing for specificity confirmation. An aoEBA TaqMan assay has been previously described and applied to the identification of the bacterium in the tick vector. 17 The TaqMan primer–probe pair described in the aforementioned study 17 was not employed in the current study. Recently available deltaproteobacterial sequences were utilized in the current study to generate a maximally unique primer–probe pair for TaqMan analysis. This DNA-based assay provides quantification of total pathogen load. While a quantitative RNA-based assay would better address the live bacterial load, the varied quality of diagnostic samples and extremely short half-life of bacterial RNA11,15 considerably neutralize this advantage. Thus, the aoEBA DNA-based assay described herein represents a robust diagnostic assay with the additional benefit of bacterial quantification.
From a diagnostic perspective, the superior sensitivity of the TaqMan assay was evident in the aoEBA-infected fetus (animal 3) in which serum Ig, IHC, and histologic lesions indicative of EBA were all absent. The analysis of pathogen:host load included a variety of tissues typically harboring lesions in EBA-positive fetuses. The data provided insight for establishing a hierarchy of samples best suited for detection of aoEBA, and therefore for diagnostic submission. Organized lymphoid tissue (thymus, lymph nodes, and spleen) generally carried higher bacterial burdens as compared to Peyer patches, blood, and tongue (Fig. 2). The aoEBA load in skin and Peyer patch were highly variable. The Peyer patch results may have been influenced by sampling error whereby some samples of the small intestine were unknowingly collected from areas without well-developed gut-associated lymphoid tissue. Variability in skin pathogen load was more easily explained as only 2 fetuses (animals 7, 8) had visible skin lesions that were directly sampled. To summarize, the current tissue pathogen load data supports previous findings of organized lymphoid organs being the tissue of choice for identifying the presence of aoEBA1,16 and further establishes the predilection of aoEBA for organized lymphoid tissue and the widespread nature of the infection.
Based upon the varied method of fetal collection (abortion vs. surgical removal) and the small number of animals, the current study was obviously not designed to fully address the pathogenesis of EBA. However, the authors took the opportunity to use these fetuses to gain insight into the relationship between pathogen load and fetal pathology. Animals 2, 5, 8, and 9 all had similar pathogen load profiles and were definitively diagnosed using standard methods. Pathogen loads in animals 3 and 7 were unique as compared to the rest of the infected animals. Animal 3 had minimal and non-diagnostic lesions, no detectable Ig, and low levels of bacteria. Animal 7 had moderate to severe lesion development, extremely high levels of Ig, and low levels of bacteria. The authors hypothesize that these 2 animals have different etiologies for their low pathogen loads and represent the early and late stages of infection. Thus, the low pathogen load of animal 3 is due to initial bacterial growth within the fetus while the low pathogen load in animal 7 is due to fetal immune-mediated clearance of the bacteria.
The underlying reason(s) for the varying degree of disease progression in this group of pregnant heifers, all of which were challenged with an identical dose of aoEBA at essentially the same time in gestation, is open to speculation. Innate susceptibility of the individual heifers could influence both the timing and the bacterial load to which the developing fetuses were exposed. An alternate, or additional hypothesis, focuses upon variation in acquisition of immunologic competence in the infected fetus with lesion development being driven by the generation of an aoEBA-specific antibody response and associated bacterial opsonization. 2
In summary, the pathogenesis of EBA is highly complex and undoubtedly influenced by multiple factors including dam susceptibility, a unique and apparently intracellular bacterial pathogen, and an active fetal immune response in a continual state of maturation. 14 Development of this quantitative duplex TaqMan PCR represents an improved and simplified diagnostic for EBA while also allowing for establishment of pathogen load in necropsy tissues. The attributes of this duplex TaqMan will provide a valuable tool for future epidemiological and pathogenesis studies directed at this unique bacterium.
Footnotes
Acknowledgements
The authors would like to thank Dr. Emir Hodzic for his guidance and support during the development of the TaqMan, and Drs. Barbara Byrne, Guillermo Rinaldi, John Adaska, and Pat Blanchard for providing the specificity-control bacteria and tissue samples.
Notes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This study was supported in part by funds provided by the Center for Food Animal Health, School of Veterinary Medicine, University of California, Davis; USDA Western Regional Research Project W-112; and the generous support of the California Cattlemen’s Association. Graduate student support was provided by the UC Davis School of Veterinary Medicine and Pfizer Animal Health.
