Abstract
Eosinophils within the central nervous system are abnormal and are usually associated with fungal or parasitic infections in horses. Causative agents include Halicephalobus gingivalis, Sarcocystis neurona, and Neospora hughesi. Confirmation of these organisms via specific testing is typically not performed, and final diagnoses are often presumptive. With molecular technology, many of these organisms can now be confirmed. This is important for emerging and zoonotic pathogens, including Angiostrongylus cantonensis, an emerging parasite of interest in the southeastern United States. We retrospectively analyzed eosinophilic encephalitides in Floridian horses for H. gingivalis, S. neurona, and A. cantonensis, applied descriptors to equine eosinophilic encephalitides, and determined if a relationship existed between these descriptions and specific etiologic agents. In a database search for horses with eosinophilic and other encephalitides submitted to the University of Florida, College of Veterinary Medicine, Anatomic Pathology Service, we identified 27 horses with encephalitis, and performed DNA isolation and rtPCR on formalin-fixed, paraffin-embedded blocks from these cases. Real-time PCR identified 6 horses positive for S. neurona and 4 horses positive for H. gingivalis; all horses were negative for A. cantonensis. All 25 control horses were negative for H. gingivalis, S. neurona, and A. cantonensis. Pattern analysis and eosinophil enumeration were not useful in differentiating among causes of eosinophilic encephalitides in horses in our study.
Keywords
Central nervous system (CNS) infections caused by viruses, bacteria, and parasites can cause devastating morbidity in horses. Except for infections with Sarcocystis neurona, little directed research has investigated CNS parasites of horses, including pathogens newly emerging in the southeastern United States, such as Angiostrongylus cantonensis, which causes eosinophilic granulomatous encephalitis in humans and animals worldwide, including in Florida.6,8 A. cantonensis has been reported in a miniature horse from Mississippi. 4 Otherwise, little work has been done to determine if this parasite is a threat to horses.4,6,8 To date, the most accurate way to diagnose parasitic encephalitis in veterinary medicine is postmortem identification of parasites within the brain. There are no antemortem tests that can be performed on horses with parasitic encephalitis, other than for S. neurona.9,13,14 With the advent of molecular technology, the identity of several neuroparasites in the horse can now be confirmed on postmortem examination.
Our goals were to: 1) apply descriptors to the inflammatory reaction in the brain, 2) determine if there was a relationship between etiology and the degree of eosinophilic inflammation, and 3) retrospectively analyze eosinophilic encephalitides in horses for the presence of the equine parasites, S. neurona, Halicephalobus gingivalis, and A. cantonensis. S. neurona and H. gingivalis were chosen because of their prevalence and commonality as determined by microscopic pathology in Floridian horses (pers. obs.). A. cantonensis, a zoonotic parasite, has been detected in non-human primates and snails in Florida; therefore, we attempted to determine if this parasite has gone undetected in verminous encephalitis cases at the University of Florida. 6
An in-house database search was performed on postmortem examination and surgical biopsy cases from 1990 to 2015 to identify eosinophilic encephalitis, eosinophilic and granulomatous encephalitis, or granulomatous encephalitis with neutrophils and eosinophils in horses. Several search strings were used, including “equine encephalitis,” “meningoencephalitis,” “granulomatous encephalitis,” “eosinophilic encephalitis,” a combination of these, as well as several specific etiologies such as “rabies virus,” “West Nile virus” (WNV), “S. neurona,” “eastern equine encephalitis virus” (EEEV), “H. gingivalis,” and other specific causes. Hematoxylin and eosin (H&E)-stained slides were obtained from the Anatomic Pathology Service at the University of Florida, College of Veterinary Medicine (Gainesville, FL) and examined by a veterinary pathology resident to verify the appropriate lesions and confirm an eosinophilic component of the inflammation. If present, the slides were retained for further evaluation. Additional cases without a significant eosinophilic component were also chosen as non-eosinophilic encephalitis controls. After confirmation of a significant inflammatory component in the affected CNS tissues, 2 formalin-fixed, paraffin-embedded (FFPE) blocks from each case with lesions were obtained from the pathology archive for molecular testing.
Fifty-two horses were divided into 3 groups: horses with eosinophilic encephalitis (n = 12), horses with non-eosinophilic encephalitis (n = 15), and neurologically normal horses without any CNS lesions (control horses; n = 25). The 12 cases in the eosinophilic encephalitis group were tested for rabies virus via direct fluorescent antibody testing performed by the Florida Department of Health. Immunohistochemistry or PCR (performed at the state diagnostic laboratory) for EEEV or WNV was performed on 8 of these 12 cases after rabies virus testing was completed. The 25 control horses that were free of clinical CNS disease (based on the provided history) lacked histologically identifiable inflammation, neoplasia, or other CNS lesions.
The examiner was blinded to the history and pathology reports on cases. The percentage of eosinophils was determined within inflammatory infiltrates. For each H&E-stained slide, 10 photomicrographs at 400× magnification were taken in areas of inflammation, including areas with eosinophils. Regions of the brain included in this process depended on the slides available and the location of the most significant inflammatory reaction (Fig. 1). A 2.8 × 2.8 cm grid was placed over areas of inflammation. Grid placement was not random but was targeted at the most inflamed areas. Within each box of the grid, the numbers of eosinophils and other inflammatory cells (lymphocytes, plasma cells, neutrophils, and macrophages) were counted, and the percentage of eosinophils within the inflammatory reaction was calculated. Characteristics of the inflammatory reaction were evaluated, including a subjective assessment of severity (mild, moderate, marked), predominant inflammatory cell population (eosinophilic, lymphoplasmacytic, granulomatous), the pattern of inflammation (multifocal, multifocal-to-coalescing, perivascular), and percentage of eosinophils (<5%, 5–20%, >20%) within the inflammatory reaction.
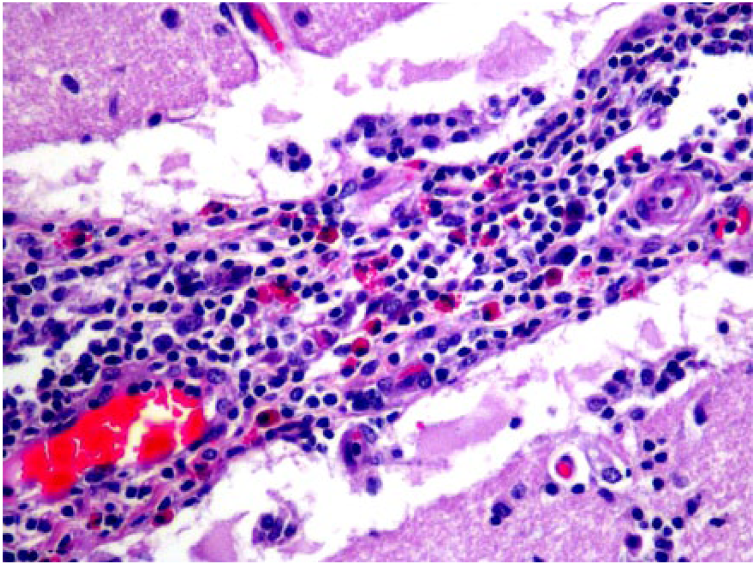
Virchow–Robins space in horse 6 expanded by a moderate-to-marked amount of a predominantly lymphoplasmacytic and eosinophilic inflammatory reaction (12.6% eosinophils). H&E. 400×.
Real-time (rt)PCR protocols were developed and validated for H. gingivalis and A. cantonensis using FFPE tissues.7,12 Real-time PCR primers for H. gingivalis and a probe were designed using Primer 3 software, with the H. gingivalis large subunit ribosomal RNA gene (LSU rRNA) as the target gene.1,10,17,18 Protocols for A. cantonensis, 12 H. gingivalis, S. neurona (Liu J. Phenotypic and mRNA profiling for investigating the pathogenesis of equine protozoal myeloencephalitis [PhD dissertation]. University of Florida, Gainesville, FL, 2015), and an equine reference gene, G3PDH, 11 were used (Table 1). The primers and probes of S. neurona were specifically designed to target the 18S rRNA gene (Liu J, 2015).
Primer and probe sets used to target Angiostrongylus cantonensis, Halicephalobus gingivalis, Sarcocystis neurona, and an equine reference gene.

The standard rtPCR reaction consisted of 10 µL of TaqMan Fast Universal master mix (Life Technologies, Carlsbad, CA) and 2 µL of the DNA sample, plus various quantities of primers and probe (Table 2). Standard cycling conditions were used for all reactions: 20 s at 95°C, and 40 repeat cycles of 95°C for 3 s, and 60°C for 30 s.
Percentages of eosinophils and overall pattern in equine encephalitis postmortem cases in which the histologic lesions exhibited an eosinophilic component (n = 12).
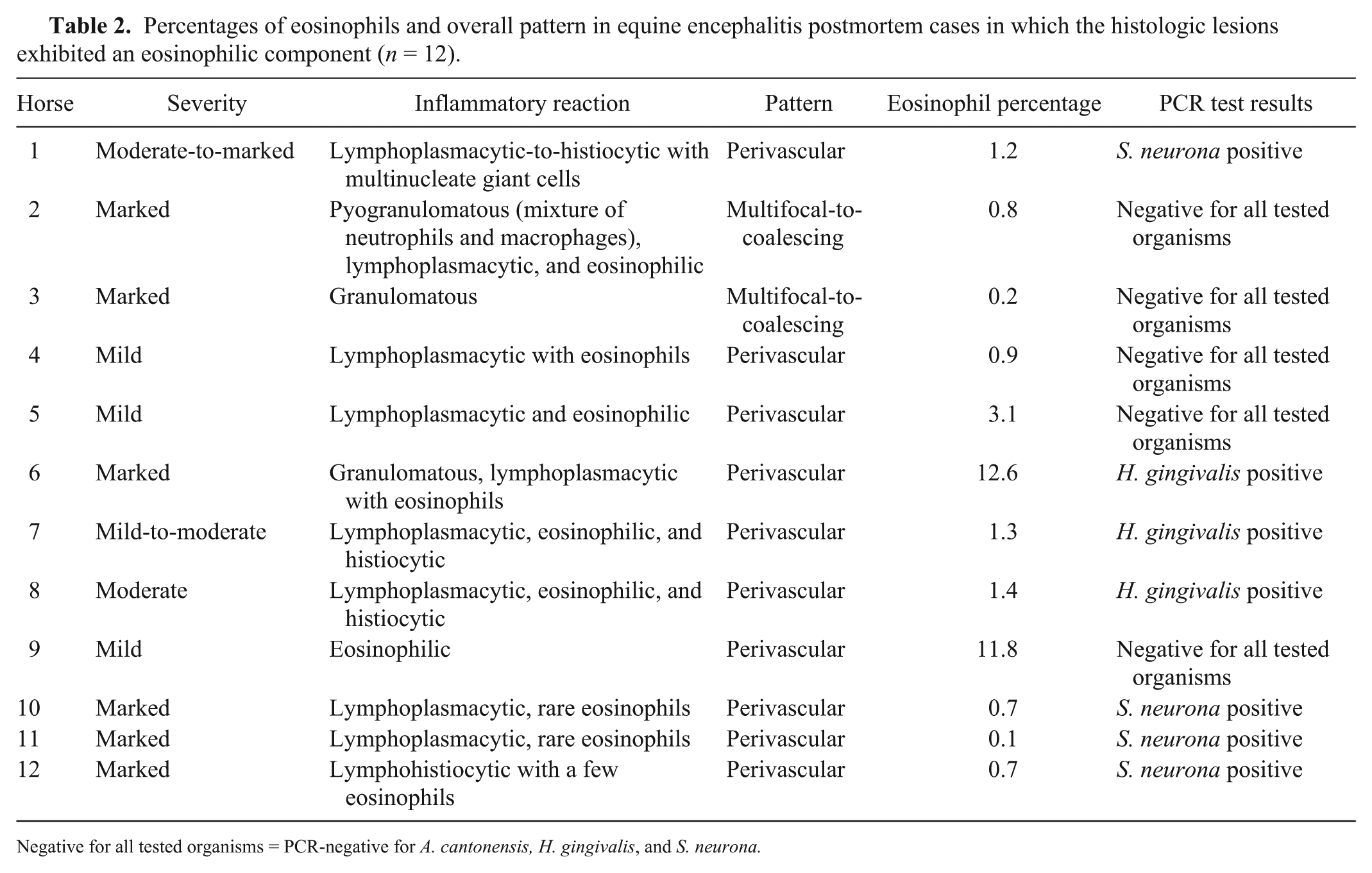
Negative for all tested organisms = PCR-negative for A. cantonensis, H. gingivalis, and S. neurona.
FFPE blocks of brain and/or spinal cord tissue were used; 2–4 ten-µm scrolls were collected from each block. Scrolls were placed into a 2-mL plastic tube, and paraffin was removed with a mild acid (CitriSolv, Thermo Fisher Scientific, Waltham, MA). 16 Samples were processed (RecoverAll total nucleic acid isolation kit, Life Technologies) according to the manufacturer’s instructions. DNA samples were placed in a –20°C freezer for storage.
All 12 eosinophilic encephalitis cases were used in the eosinophil enumeration portion of our study. H. gingivalis horses (n = 3) appeared to have the highest average percentage of eosinophils (5.1%) within the inflammatory reaction, followed by horses negative for all tested organisms (n = 5, 3.4%) and horses positive for S. neurona (n = 4, 0.7%); however, differences were not statistically significant (student t-test, p = 0.093; Table 2). There was a predominantly perivascular pattern to the inflammatory infiltrate in the 12 eosinophilic encephalitis cases.
Seven horses were suspected to be infected with H. gingivalis based on history, components of the inflammatory population (macrophages and eosinophils), and the inflammatory cell distribution. Rhabditiform nematodes were observed in 6 of the 7 suspected cases. Real-time PCR confirmed H. gingivalis in 4 of these 7 suspected cases; in 3 of the H. gingivalis suspect cases, the FFPE tissue with the primary lesions was no longer available or the tissues available were highly decomposed. The remaining available CNS tissues did not have evidence of infection. H. gingivalis cases had numerous epithelioid macrophages and multinucleate giant cells within the inflammatory reaction (Fig. 2). One H. gingivalis rtPCR–positive case (horse 1) had a very high percentage of eosinophils, whereas another high eosinophilic percentage case (horse 6) was rtPCR-negative for all 3 parasites.
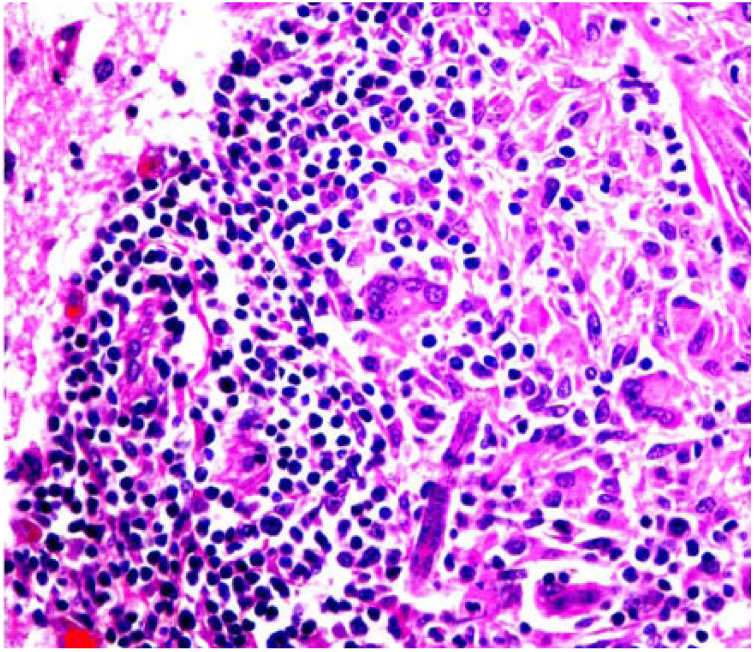
Halicephalobus gingivalis nematode in the cerebrum of horse 1, admixed with a predominantly granulomatous inflammatory component. H&E. 400×.
Seventeen horses were suspected to be infected with S. neurona based on clinical history or histologic examination (protozoal cysts within the neuropil; n = 2) and negative test results for WNV, EEEV, and rabies virus. Real-time PCR confirmed S. neurona infection in 6 of the 17 suspected cases. One case with identified organisms was negative by rtPCR for S. neurona.
A. cantonensis was not detected histologically or by rtPCR in any of the horses in our study.
Twelve horses (first group, n = 12) were suspected to be infected with rabies virus, EEEV, or WNV based on clinical signs, the provided history, and random-to-perivascular inflammatory reaction (lymphocytes, plasma cells, and neutrophils). EEEV was confirmed in 2 of the 12 horses; all 12 horses were negative for WNV and rabies virus.
The control horses, which did not have CNS lesions, were negative for viral and bacterial pathogens, S. neurona, H. gingivalis, and A. cantonensis based on the absence of inflammatory histologic lesions and negative rtPCR.
Based on histologic examination and clinical history, 7 horses were suspected to be infected with H. gingivalis. Four of these 7 horses were confirmed by rtPCR to be infected with H. gingivalis. Two horses with H. gingivalis parasites in the neuropil were not positive via rtPCR. The third rtPCR-negative case was suspected to be H. gingivalis positive based on the history and presence of macrophages, multinucleate giant cells, and eosinophils seen on histologic examination of CNS tissues. These discrepancies may have been the result of the tissue blocks selected, poor rtPCR sensitivities from FFPE tissues, severe degradation of the tissues, or that some of these parasites were actually other species.
S. neurona is one of the causative agents of equine protozoal myeloencephalitis and is a common rule-out for neurologic disease in horses, but is not commonly confirmed even on postmortem examination.3,5,13 S. neurona was confirmed by rtPCR in 6 of 17 suspected cases based on history, clinical signs, and histologic examination of CNS tissues. Two possible reasons for lack of confirmation (in addition to misdiagnosis) could have been lack of parasites within tissue blocks and prior treatment with anti-protozoal drugs limiting the presence of the organism within tissues.2,5 In our cases, of 17 suspected S. neurona cases, 6 had been treated with anti-protozoal drugs, and all 6 tested negative by quantitative PCR. In 5 of 17 suspect S. neurona rtPCR–negative cases, no treatments were reported in the clinical history. Three rtPCR-positive cases were treated with anti-inflammatories and dexamethasone, and 3 rtPCR-positive cases had been treated with anti-protozoal drugs. A second explanation for negative results by S. neurona rtPCR includes infection with a similar protozoal organism, such as Neospora hughesi or Toxoplasma gondii.5,13 These 2 organisms are considered the agents that must be differentiated from S. neurona in horses via PCR or other ancillary tests.5,13
The eosinophil enumeration used in our project was expected to help differentiate among certain etiologies of equine encephalitis, specifically S. neurona, H. gingivalis, and A. cantonensis. However, there was no statistically significant difference in the eosinophil percentage in the H. gingivalis–positive, S. neurona–positive, and negative horses. There was a predominantly perivascular pattern to the inflammatory infiltrate in the 12 eosinophilic encephalitis cases. H. gingivalis cases had numerous macrophages and multinucleate giant cells within the inflammatory reaction. There is no clear evidence that the nematode H. gingivalis produced a more robust eosinophilic inflammatory response than the protozoan S. neurona. In fact, H. gingivalis–positive cases (via histology and rtPCR) appeared to have a more histiocytic inflammatory reaction (Fig. 2), as described previously. 15 S. neurona–positive cases had a mixture of lymphoplasmacytic, histiocytic, and eosinophilic cellular components. 3 Therefore, eosinophil enumeration was not diagnostically useful in differentiating among causes of eosinophilic encephalitides in horses in our study.
Only 2 of 12 eosinophilic encephalitis cases had a high percentage of eosinophils within the inflammatory reaction. Horse 6 was positive for H. gingivalis via rtPCR, and organisms were seen on histologic examination. Horse 8 was negative for all 3 parasites; however, there was little remaining tissue in the block, and the residual tissue had minimal inflammation, limiting the case to only 4 photomicrograph sections to be evaluated.
Footnotes
Acknowledgements
The primer and probe set and positive control for S. neurona and the equine reference gene (G3PDH) were provided by J Liu. We thank Sally Beachboard for laboratory assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by a Resident Research Competitive Grant provided by the College of Veterinary Medicine Hospital at the University of Florida and by funds provided by MT Long’s laboratory.
