Abstract
An epidemiological survey for Canine parvovirus 2 (CPV-2) and Canine coronavirus (CCoV) was conducted in Albania. A total of 57 fecal samples were collected from diarrheic dogs in the District of Tirana during 2011–2013. The molecular assays detected 53 and 31 CPV- and CCoV-positive specimens, respectively, with mixed CPV–CCoV infections diagnosed in 28 dogs. The most frequently detected CPV type was 2a, whereas IIa was the predominant CCoV subtype. A better comprehension of the CPV–CCoV epidemiology in eastern European countries will help to assess the most appropriate vaccination strategies to prevent disease due to infections with these widespread agents of acute gastroenteritis in the dog.
Canine parvovirus type 2 (CPV-2) and Canine coronavirus (CCoV) are considered the main pathogens responsible for acute gastroenteritis in dogs.2–4 Canine parvovirus 2 is a small, nonenveloped, single-stranded DNA virus, which is closely related to Feline parvovirus (FPV), from which it presumably originated, and to parvoviruses of other carnivores. All carnivore parvoviruses are included in the family
Canine parvovirus can cause a severe, often fatal disease, whereas CCoV is usually associated with mild, self-limiting enteritis followed by rapid recovery. Fatal disease may occur as a consequence of mixed infections with CCoV together with CPV-2, Canine adenovirus type 1, or
Several studies have assessed the CPV and/or CCoV type distribution in different European countries,6–8,13,18,20,21 but, to date, there is no epidemiological data about the circulation of these enteric pathogens in Albania. Thus, the main purpose of the present study was to determine the frequency of CPV-2 and CCoV infections in dogs with diarrhea in Albania and to characterize the viral types that circulate in this country.
A total of 57 fecal specimens were collected from dogs presented with acute diarrhea at private veterinary clinics in the District of Tirana, Albania, during 2011–2013. The signalment for each sampled dog (i.e., age, sex, and breed) is reported in Table 1. The age of the dogs varied from 1 to 7 months; 35 animals were males, while the 22 remaining samples were from females. All sampled animals, with the exception of 2, were purebred dogs. There was no data regarding administered vaccines or immunization protocols. Analogously, there was no information concerning the severity of clinical signs and disease outcome.
Canine parvovirus and coronavirus type distribution in Albania.*
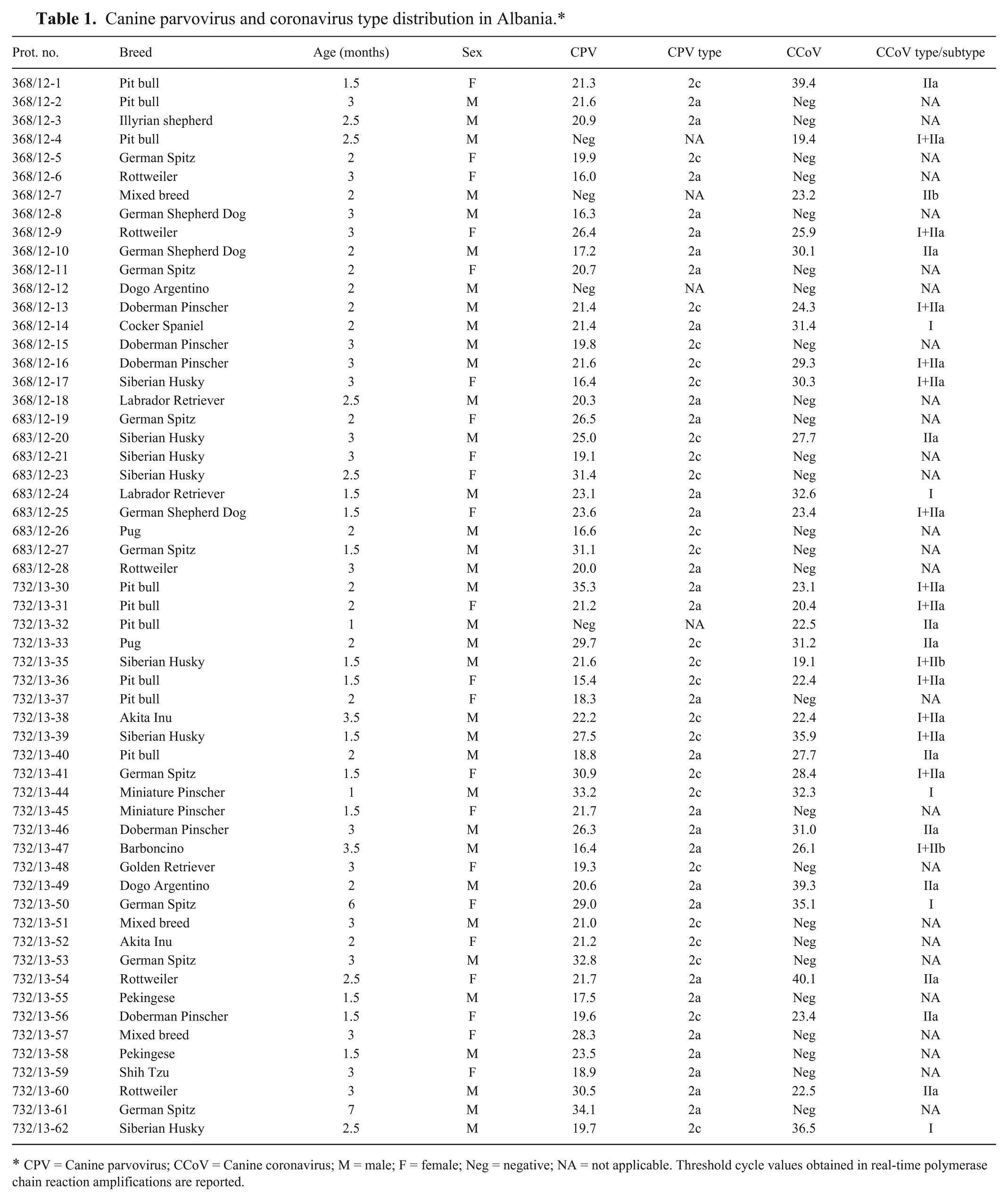
CPV = Canine parvovirus; CCoV = Canine coronavirus; M = male; F = female; Neg = negative; NA = not applicable. Threshold cycle values obtained in real-time polymerase chain reaction amplifications are reported.
Fecal samples were homogenized (10% weight/volume) in phosphate buffered saline solution (pH 7.2) and subsequently clarified by centrifugation at 8,000 ×
For CCoV detection and characterization, viral RNA was extracted from the supernatants of fecal homogenates by means of a commercial kit, b following the manufacturer’s protocol, and the RNA templates were stored at –70°C until their use. All RNA extracts were subjected to a previously established TaqMan-based real-time reverse transcription (RT)-PCR assay for rapid detection and quantification of CCoV RNA. 17 The detected CCoV strains were characterized by means of 2 distinct genotype-specific assays. 16 Samples that tested positive for CCoV-II were subjected to subtype-specific CCoV-IIa and CCoV-IIb gel-based RT-PCR assays targeting the spike-protein gene. 13 Viral RNA detection and characterization was achieved by using a commercial master mix. c
Fifty-three out of 57 samples (92.98%) tested positive for the presence of CPV by using the TaqMan assay. By means of real-time PCR with type-specific MGB probes, 29 samples were characterized as CPV-2a (54.71%) and 24 as CPV-2c (45.28%). None of the CPV strains were typed as either CPV-2b or vaccine viruses (Table 1).
By TaqMan real-time RT-PCR, CCoV was detected in 31 out of 57 tested samples (53.38%). By using genotype-specific assays, CCoV-I was detected less frequently (19 samples) than CCoV-II (26 samples). As for the subtype distribution, 23 CCoV-II strains were characterized as CCoV-IIa, whereas the remaining 3 strains were CCoV-IIb. Infections with more than 1 genotype were detected in 14 animals, but no cases of coinfections with different CCoV-II subtypes were observed. Mixed infections caused by CPV-2 and CCoV were detected in 28 out of 57 samples (49.12%), whereas a single sample tested negative for either virus (Table 1). The mean ages for CPV-2, CCoV, and dual infections were 2.46, 2.27, and 2.32 months, respectively.
The present study represents a survey for CPV-2 and CCoV infections in Albania by means of molecular techniques. Fifty-three and 31 samples tested positive for CPV and CCoV, respectively, with mixed infections detected in 28 specimens. Analogous to other geographical areas, the original type CPV-2 was shown not to circulate in dogs, whereas type 2a was the most frequently detected variant, which was in agreement with reports in other eastern European countries.7,8,18,20 However, CPV-2c was also detected in a high proportion of CPV-2 PCR-positive fecal samples not previously observed in eastern Europe. As for CCoV infection, a larger number of CCoV-IIa subtypes were detected compared with CCoV-IIb (CCoV–TGEV recombinant strains). In previous studies, a widespread circulation of CCoV-IIb was shown in Eastern Europe, mainly in Hungary, 13 but the virus was also detected in Greece albeit at a lower frequency. 22 Mixed CCoV-I and -II infections were present in several analyzed samples, which was in agreement with previous epidemiological studies.6,8,13 However, no coinfections with more than 1 CCoV-II subtype were detected in the present study.
Interestingly, a high proportion of CPV-2 and CCoV mixed infections was also observed. It is well-known that the 2 viruses act as synergistic agents in the onset and outcome of the induced clinical signs. 19 Canine parvovirus 2 infects the cells of the intestinal cryptae, whereas CCoV replicates in the enterocytes at the top of the villi. Consequently, the damaged epithelium is no longer replaced by new enterocytes developed in the cryptae, thus leading to very severe hemorrhagic enteritis. 19 Unfortunately, there was no data available about the outcome of single or double infections in Albanian dogs, which prevented any clear association of coinfections to disease exacerbation.
It is also noteworthy that almost one-half of the dog population with enteritis belonged to breeds that are regarded to have an increased risk to develop CPV infection (i.e., Rottweiler, Doberman Pinscher, German Shepherd Dog, Siberian Husky, etc.). 19 A better comprehension of the CPV–CCoV epidemiology in eastern European countries will help assess the most appropriate vaccination strategies to prevent these widespread agents of canine acute gastroenteritis.
Footnotes
a.
iTaq DNA polymerase, IQ Supermix; Bio-Rad Laboratories Srl, Milan, Italy.
b.
QIAamp viral RNA mini kit, Qiagen SpA, Milan, Italy.
c.
SuperScript III Platinum one-step qRT-PCR kit, Life Technologies Srl, Milan, Italy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
