Abstract
Ehrlichia canis is the most common cause of monocytotropic ehrlichiosis in dogs around the world. The purpose of the present study was to validate a new automated fluorescence system (Accuplex4™ BioCD system; Antech Diagnostics, Lake Success, New York) to detect antibodies against the E. canis immunodominant glycoprotein 36 (gp36). Sera and blood samples (ethylenediamine tetra-acetic acid) were collected from mixed sex beagles (n = 8) on days 0, 3, 7, 10, 14, 17, 21, 28, 42, 49, 56, 63, 70, 77, 84, and 98 after intravenous inoculation with culture-derived E. canis. Sera were assayed using the Accuplex4 BioCD system (Accuplex4), an E. canis indirect fluorescent antibody test (IFAT), and a commercially available kit. A complete blood cell count and a proprietary E. canis polymerase chain reaction (PCR) were performed on each blood sample. On the day thrombocytopenia was first detected for each dog, E. canis DNA was amplified from blood of all dogs. At those times, E. canis antibodies were detected in 7 of 8 dogs by the Accuplex4, 1 of 8 dogs by the commercial kit, and 4 of 8 dogs by IFAT. Ehrlichia canis DNA was amplified from blood before seroconversion in any antibody assay for 6 dogs. Antibodies against gp36 were detected by Accuplex4 within 3 days of PCR-positive test results and were detected up to 25 days sooner than the commercial kit. After starting doxycycline treatment, E. canis DNA was no longer amplified by PCR assay, but serum antibodies remained detectable by all assays.
Keywords
Ehrlichia canis is the most common cause of monocytotropic ehrlichiosis in dogs around the world and is associated with subclinical, acute, or chronic infections. 8 The organism has also been associated with clinical illness in some human beings. 14 Amplification of E. canis DNA from biological samples using polymerase chain reaction (PCR) assay is generally used to document current infection in dogs.2,8,12 Serum antibodies against E. canis can be detected by several methods, including enzyme-linked immunosorbent assay (ELISA), indirect fluorescent antibody test (IFAT), and Western blot immunoassay.1,5 Detection of serum antibodies against E. canis documents exposure but does not necessarily correlate to current infection.8,11,12 Serum antibody and PCR assays for use as aids in the diagnosis of canine ehrlichiosis are available via commercial diagnostic laboratories in many countries. In some countries, point-of-care serological assays are available for use in veterinary clinics.5,13
The immunodominant E. canis antigens from strains infecting dogs have been determined in several studies.4,6,15 Glycoprotein 36 (gp36) is one of the most conserved and immunodominant antigens. 4 New automated systems have been developed that are capable of testing for antibodies against multiple antigens using small volumes of serum. 16 Such automated systems can lessen interassay variability, and large numbers of samples can be assayed concurrently making the systems of great benefit to large commercial service laboratories. The purpose of the current study is to describe the use of an automated system to detect antibodies against E. canis gp36.a
The present study was approved by the Institutional Animal Care and Use Committee at an independent research laboratory. The mixed sex beagles (n = 8) used in the study were purchased from a laboratory animal facility and ranged in age from 12 to 13 months at the beginning of the experiment. Prior to shipment to the research facility, all dogs were shown to be negative for antibodies against Anaplasma phagocytophilum, Borrelia burgdorferi, and E. canis as well as for Dirofilaria immitis antigen by use of a commercially available kit (SNAP)b and for DNA of Ehrlichia spp. and Anaplasma spp. by PCR. 7
A field isolate of E. canis was grown on DH-82 cells and delivered to the research facility by a same-day air service while stored at ambient temperature. d The specific strain of E. canis was originally isolated from a naturally infected dog with fever in southwestern Oklahoma in 1994. 10 The dogs in the current study were inoculated with approximately 3.2 × 106 DH-82 cells that had an E. canis infection rate of approximately 80%. The dogs were premedicated with approximately 2.2 mg/kg of diphenhydramine administered subcutaneously approximately 30 min prior to inoculation with E. canis. The E. canis inoculum (1 ml) was administered by slow intravenous injection. The dogs were then observed for side effects for 1 hr after inoculation (none noted).
Blood (6 ml) was collected into a syringe by jugular venipuncture on days 0, 3, 7, 10, 14, 17, 21, 28, 42, 49, 56, 63, 70, 77, 84, and 98. After blood was collected, 1.5 ml was placed into a ethylenediamine tetra-acetic acid (EDTA) tube and then 0.25 ml was pipetted into a 1.5 Eppendorf e tube and stored at −80°C until evaluated for E. canis DNA by PCR assay. The blood remaining in the EDTA tube was maintained at 4°C until a complete blood cell count was performed. The remaining blood was placed into a tube without anticoagulant, allowed to clot, centrifuged at 1,500 × g for 10 min, and the sera stored in multiple aliquots at −80°C until assayed.
On the day of collection, the blood samples in EDTA were shipped on cold packs by overnight express to a commercial laboratory for performance of proprietary PCR assays that amplify the DNA of A. phagocytophilum, A. platys, E. canis, Ehrlichia chaffeensis, and Ehrlichia ewingii using the standard operating procedures of the laboratory. c Sera were ultimately assayed for E. canis antibodies by IFAT using slides purchased from a commercial laboratory f by SNAP following the manufacturer’s guidelines, b and by Accuplex4 using E. canis gp36 as the antigen source as described in the section that follows. a An E. canis IFAT titer of >1:80 was considered positive.
The E. canis gp36 antigen is expressed at the laboratory c using a previously described sequence. 4 The Accuplex4 is based on a silicon wafer with a thermal oxide layer. The wafer is treated with both a 3-aminopropyldimethylethoxysilane vapor deposition as well as a 1,6-diisocyanatohexane liquid deposition. A fluorescent hydrophobic mask is screen printed on the surface to create a 288-well pattern. Using a contact protein printer, 11 different markers are used to print 64 spots in a specific spot pattern in every well. Eight spots are dedicated to each peptide or protein used to capture target antibodies. The assay as currently designed detects antibodies against B. burgdorferi, E. canis, and A. phagocytophilum as well as D. immitis antigen. After printing of the peptides and antibody, the disc surface is blocked with ethanolamine vapor for 15 min at 30°C j to lessen potential for nonspecific binding and is coated in trehalose (2% by volume diluted in deionized water) g for added stability. The finished Accuplex4 disc can hold up to 274 patient samples along with 8 positive controls and 6 negative controls spread around the disc for each of the 4 antibody assays and 1 antigen assay.
Each disc is loaded onto a sample processor that is used for liquid handling and dispensing using a keyed chuck to ensure proper disc loading. h The disc is washed with a phosphate buffered saline solution (pH 7.4) with Tween-20 (PBST) for 20 sec at 9 x g and then rinsed with deionized water for 20 sec at the same speed before being centrifuged at 511 x g for 15 sec to dry. Patient serum is loaded into each reaction well (5 µl) and incubated for 30 min at 80% humidity. The disc is again washed with PBST and deionized water for 20 sec and centrifuged as described to dry. The fluorescent conjugate is dispensed into each well (6 µl) and incubated for 10 min. i The disc is then washed for the final time with PBST and deionized water for 20 sec and centrifuged as described to dry.
The dual channel reader includes both a fluorescent and interferometric detector and also contains the same keyed chuck as the sample processor to ensure proper disc orientation. j Once the disc is loaded, it is centrifuged at 1,309 x g, and a 20-mW, 532-nm laser attached to the optical stage is “stepped” across the disc in the x-orientation. As the stage sweeps across the disc, 2,351 data points are recorded through both detectors and sent to the computer workstation.
The interferometric data is used for disc image transformation and well mapping. These data points produce not only the disc image, but an individual image for each well. Using image processing, a spot pattern template is then applied to the fluorescent image where fluorescent counts are taken for each protein spot in all 288 wells. The median value of fluorescent counts is assigned to each individual immunologic reaction. The fluorescent counts for the 6 negative control wells are used to calculate the cutoffs for each assay. The median is taken from the 6 negative control wells and added to 3 standard deviations of the negative control well values (root calculation). A floor value (X) is used to prevent these numbers from falling into the noise level. The floor constant was created by evaluating known negative patients as well as increases in fluorescent counts over time postinfection in the intravenously inoculated dogs. The cutoff takes the greater value between the floor value and the root calculation. A constant, Y, is then added to this number to create the functional cutoff. The cutoff format for each immunologic reaction is MAX{(median (negative controls) + 3 STDEV (negative controls)) OR X} + Y. This allows the cutoff to adjust for minor variation in discs. This cutoff is then applied to each reaction, measured in fluorescent counts for an individual patient sample. A suspect sample is considered positive for antibodies against E. canis when the result is greater than the specified threshold.
The Accuplex4 system was optimized using positive and negative control sera used in assay titrations obtained from the dogs inoculated in the study described herein. The optimal concentration of gp36 for use in the assay was determined by assessing for the optimal signal-to-noise ratio by varying printed gp36 concentrations and buffer compositions. The cutoff point for a positive test result was determined by assay of serum from dogs with known infection status based specifically on differential responses compared with E. canis IFAT results on serum collected preinfection (negative IFAT) and postinfection (positive IFAT).
The intra-assay variation was calculated by determining the mean and standard deviation for the fluorescent counts for 20 positive control sample wells and calculating the coefficient of variation on 1 disc. This experiment was performed with the same positive control samples on separate discs on 3 different days. The interassay variation was determined by comparing the coefficient of variations among the 3 discs. Plasma, separated from the whole blood from dogs (n = 10) submitted for PCR testing and proven to be infected byE. ewingii, was collected and assayed in the Accuplex4 system to determine specificity.
Results in all 4 assays were recorded as positive or negative. The percentages of dogs that were positive in each assay on each date were analyzed using a generalized linear model with test, week, and the test by week interaction included as fixed effects in the model. Where a significant test effect was detected within a week, all pairwise comparisons were made. Group mean platelet counts for each date postinoculation with E. canis were compared with day 0 platelet counts by 2-sided Student’s t-test. The time to first positive result was compared among the SNAP, IFAT, and Accuplex4 E. canis assay using Wilcoxon rank sum test. Significance was defined as P < 0.05 in all analyses.
In the optimized Accuplex4 system, the intra-assay variation of 20 E. canis antibody–positive control wells per disc evaluated on separate discs was estimated to be 8.1%, 9.0%, and 9.3%, respectively. The interassay variation of these results among the 3 discs was estimated to be 5.8%. Plasma from the 10 dogs with PCR-documented E. ewingii infection was negative in the Accuplex4 E. canis antibody assay.
Clinical signs of fever and lethargy of more than 2 days duration were noted for 1 dog. Abnormalities were first identified on day 28 after inoculation, and doxycycline (approximately 10 mg/kg, orally, daily) was administered from day 30 through day 58. Clinical abnormalities resolved by day 32 and did not recur. None of the other 7 dogs developed obvious clinical signs of disease and were empirically administered doxycycline (100 mg/dog, orally, twice daily) from day 56 through day 84. Side effects associated with doxycycline administration were not noted.
The percentages of dogs positive in the E. canis IFAT, SNAP, Accuplex4, and the PCR assay were stratified by day after inoculation (Table 1). Six of 8 dogs were positive in the PCR assay as soon as day 14 after inoculation. All dogs were positive in the PCR assay on days 21 and 28. The dog that was administered doxycycline for suspected clinical ehrlichiosis from day 30 through day 58 was negative in the PCR assay on all samples collected after day 42. Positive PCR assay results were detected in significantly more dogs than some of the antibody tests on several days after inoculation up to day 28 (Table 1).
Distribution of positive test results in 4 Ehrlichia canis diagnostic assays performed on samples collected over time from 8 dogs experimentally inoculated with culture-grown E. canis.*
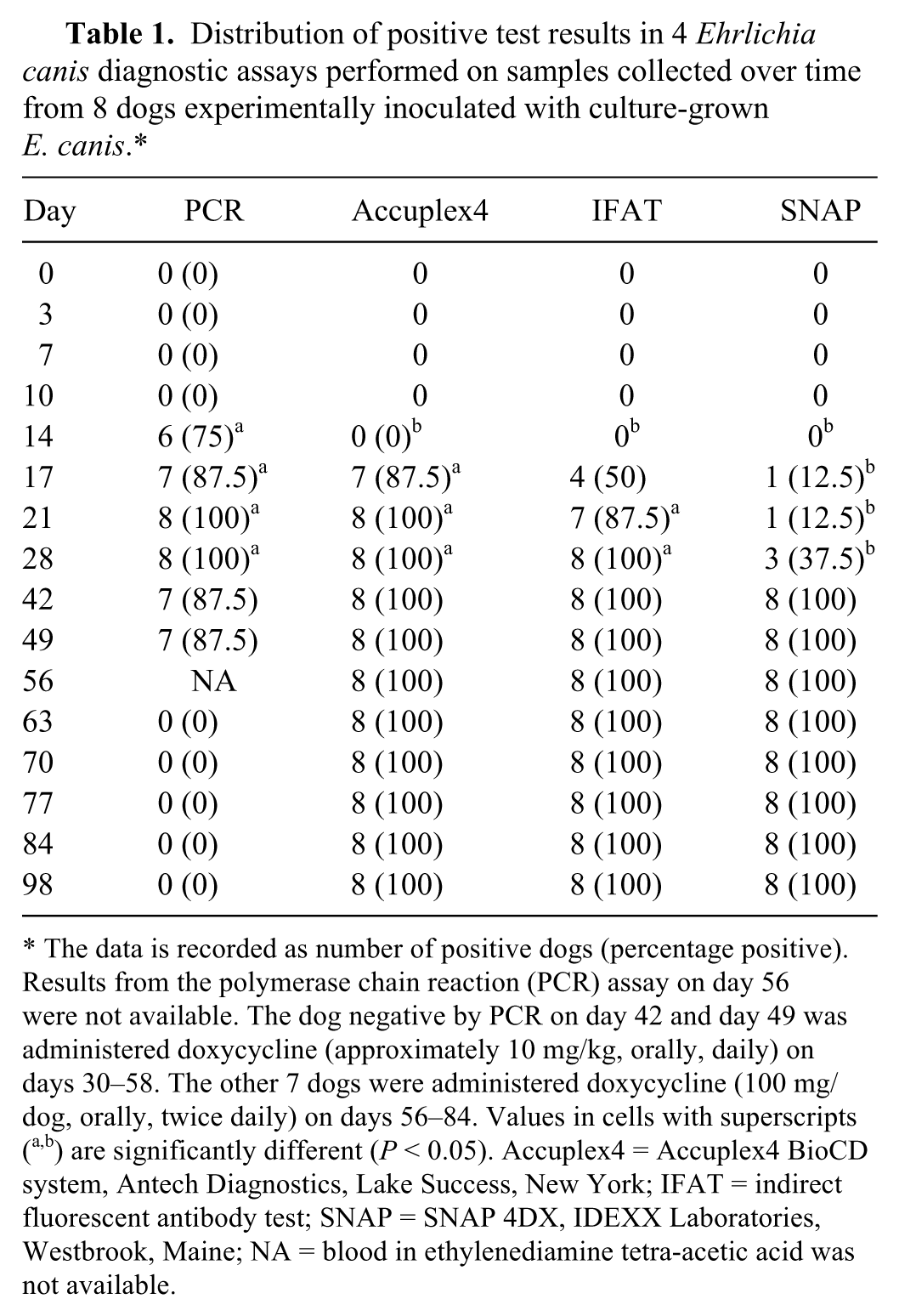
The data is recorded as number of positive dogs (percentage positive). Results from the polymerase chain reaction (PCR) assay on day 56 were not available. The dog negative by PCR on day 42 and day 49 was administered doxycycline (approximately 10 mg/kg, orally, daily) on days 30–58. The other 7 dogs were administered doxycycline (100 mg/dog, orally, twice daily) on days 56–84. Values in cells with superscripts (a,b) are significantly different (P < 0.05). Accuplex4 = Accuplex4 BioCD system, Antech Diagnostics, Lake Success, New York; IFAT = indirect fluorescent antibody test; SNAP = SNAP 4DX, IDEXX Laboratories, Westbrook, Maine; NA = blood in ethylenediamine tetra-acetic acid was not available.
Antibodies against E. canis gp36 were detected by the Accuplex4 in 7 of 8 dogs (87.5%) by day 17. When percentages of serum antibody–positive dogs in the IFAT and Accuplex4 were compared by date, there were no significant differences. However, when results from the IFAT or Accuplex4 were compared with those of SNAP, significant differences were detected on some dates (Table 1). The day to first positive test result was significantly (P < 0.05) earlier for both IFAT and Accuplex4 when compared with SNAP. After starting doxycycline administration, E. canis DNA was not amplified by PCR in any dog, but serum antibodies were detected in all dogs by all assays.
Compared with day 0 platelet counts, statistically significant reductions in mean platelet counts were detected on days 14–56 and day 70. All 8 dogs developed thrombocytopenia (platelet counts <170,000/µl) on several dates after E. canis inoculation (Fig. 1). On the day thrombocytopenia was first detected, PCR was positive for E. canis DNA in all 8 dogs and positive for antibodies to E. canis by Accuplex4 in 7 of 8 dogs, by IFAT in 4 of 8 dogs, and by SNAP in 1 of 8 dogs. The platelet counts for all dogs were >170,000/µl on day 63 and all subsequent days.
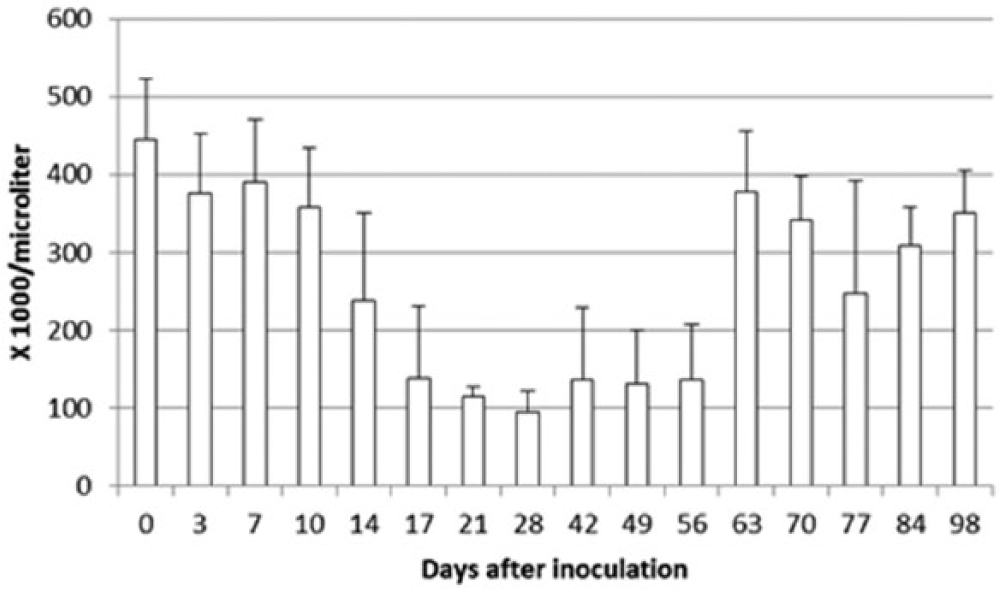
Mean platelet counts over time in 8 dogs experimentally inoculated with cell culture–grown Ehrlichia canis on day 0. One of the dogs was administered doxycycline (approximately10 mg/kg, orally, once daily) on days 30–58. The other 7 dogs were administered doxycycline (100 mg/dog, orally, twice daily) on days 56–84. Compared to day 0 platelet counts, statistically significant decreases in mean platelet counts were detected on days 14–56 and day 70.
The intra-assay and interassay variation as estimated for the Accuplex4 system for detection of E. canis antibodies show that the precision of the assay is high. The assay appears to be specific for E. canis as antibodies againstE. ewingii were not detected.3,9 However, the number of samples from dogs with E. ewingii infection were small (n = 10), and further samples should be assessed with the optimized assay. Antibodies against E. canis gp36 were reported not to cross-react with antibodies to E. chaffeensis but this was not verified in the current study. 4 In E. ewingii and E. chaffeensis endemic areas, dogs with suspected ehrlichiosis that are negative by the Accuplex4, SNAP, or IFAT should be assessed by PCR assay.
While the E. canis PCR assay results correlated best with the onset of thrombocytopenia, 3 of 8 dogs were positive for E. canis antibodies by Accuplex4 at the time of first detection by PCR, and the other 5 dogs were all positive on sampling 3 days later. Thus, dogs with suspected ehrlichiosis that are negative for E. canis antibodies should be evaluated for infection by PCR assay. Positive E. canis antibody results in the Accuplex4 are detected up to 25 days sooner than by SNAP. This earlier detection of antibodies may reflect increased sensitivity of gp36 as the test antigen. 15
The results of the IFAT, SNAP, and Accuplex4 E. canis antibody assays should be evaluated further using samples from naturally infected dogs. However, the information described in the current report has clinical benefit because in client-owned dogs, the date of infection is unknown and repeated testing prior to clinical illness or thrombocytopenia is not available, thus making it more difficult to assess differences in results among tests.
Administration of doxycycline was associated with a rapid resolution of clinical signs of ehrlichiosis in the 1 clinically affected dog. Ehrlichia canis PCR assay results were negative on samples collected after doxycycline was started on day 56 for 7 dogs and on day 30 for the 1 clinically affected dog. This finding emphasizes the recommendation to perform E. canis PCR assays on blood samples prior to the administration of drugs with anti-Ehrlichia effects 12 ; however, because the dogs were not immune suppressed, whether infection was cleared cannot be determined in the current study. Serum antibody titers persisted for the duration of the study. Because of the short-term nature of the current study, the duration of positive E. canis antibody assays cannot be determined. There was no obvious clinical disease in 7 of the 8 dogs over the course of the study. However, antibody was detected by each of the assays, and E. canis DNA was amplified from each of the 7 clinically normal dogs. The findings emphasize that because E. canis can have a variable subclinical phase, detection of DNA or antibodies does not always correlate to the presence of clinical disease. 12 Whether to treat healthy E. canis seropositive dogs with normal blood work and urinalysis is controversial; the pros and cons of treatment should be discussed with the owner. 12
Footnotes
Acknowledgements
The authors thank Jennifer Hawley, Melissa Brewer, Arianne Morris, and Amber Caress for technical assistance during completion of this study.
a.
Accuplex4 BioCD system, Antech Diagnostics, Lake Success, NY.
b.
SNAP 4DX, IDEXX Laboratories, Westbrook, ME.
c.
Antech Laboratories, Lake Success, NY.
d.
Ehrlichia canis strain OSU Eb061409; Dr. Susan Little, Oklahoma State University, Stillwater, OK.
e.
Eppendorf North America, Hauppauge, NY.
f.
Prototek Reference Laboratory, Chandler, AZ.
g.
Trehalose, Sigma-Aldrich, Analytical, St. Louis, MO.
h.
SIAS MODEL, Xantus manufactured by Sias, Hombrechtikon, Switzerland.
i.
Protein-A/Alexafluor532, Invitrogen Corp., Carlsbad, CA.
j.
BioCD reader, dual channel reader; Antech Diagnostics, West Lafayette, IN.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All the authors except Michael R. Lappin are employees of Antech Diagnostics. However, none of the authors directly benefited financially from completion of this work.
Funding
The funding for the study was provided by Antech Diagnostics.
