Abstract
The purpose of the present study was to evaluate the use of enzyme-linked immunosorbent assay (ELISA) antigen detection in blood or fetal fluids and reverse transcription polymerase chain reaction (RT-PCR) amplification in tissues for routine laboratory diagnosis of Border disease virus (BDV) infection. Samples from 67 fetuses, 6 stillbirths, and 11 lambs from 25 commercial flocks with suspicion of BDV abortion and 3 fetuses, 7 stillbirths, and 15 lambs obtained from an experimental infection with a local isolate (BDV genotype 4) were investigated. Presence of BDV was detected by RT-PCR in 7.9% of fetuses, 50% of stillbirths, and 50% of lambs from the commercial flocks analyzed, corresponding to 8 of the 25 farms (32%). A similar percentage of the lambs and stillbirths from the experimental infection were positive by RT-PCR of tissue samples (54.5%), and the highest positivity was detected in lymph node, thyroid gland, and kidney. The current study revealed that RT-PCR analysis of stillbirths and lambs with clinical symptoms is more suitable than the analysis of fetuses to confirm the presence of BDV in a flock. Pestiviral antigen was detected by antigen ELISA in a high proportion of fetuses (24/58) and stillbirths (3/4) from commercial flocks, but in lambs, the presence of colostral antibodies masked the detection of the antigen by ELISA. Nevertheless, in lambs from the experimental infection that were not fed colostrum, antigen ELISA was less efficient than RT-PCR in detecting viral presence in stillbirths and lambs. Antigen ELISA is therefore recommended for fetuses with advanced autolysis that can adversely affect RNA integrity.
Keywords
Introduction
Pestiviruses are RNA viruses that infect a wide range of ungulate species, such as swine, cattle, sheep, and other wild ruminants. Along with Classical swine fever virus (CSFV) and Bovine viral diarrhea virus 1 and 2 (BVDV-1, BVDV-2), Border disease virus (BDV) is one of the 4 main viral species recognized within the genus Pestivirus in the family Flaviviridae. In sheep, infection of pregnant ewes with BDV during early or mid gestation results in abortion, stillbirth, or unviable lambs. Border disease (BD) is one of the main causes of abortion in Northern Spain, 4,12 where the most frequently identified genotype is BDV-4. 7,16 An experimental infection carried out in pregnant ewes with this genotype 5 led to a high number of stillbirths (32%) and a significantly reduced body-weight of lambs at lambing. Only 20% of the lambs were clinically normal, and affected lambs had tremors, gait anomalies, an inability to stand unaided, and skeletal abnormalities (e.g., brachygnathia, prognathia, arthrogryposis). 5 Nevertheless, pestiviral infections are difficult to diagnose based solely on clinical signs. Duration of outbreaks varies, depending on whether or not persistently infected (PI) animals are present, the virulence of the strain, and the management and housing conditions.
Various methods have been used to demonstrate the presence of virus in fetal tissues. For definitive diagnosis of BDV, isolation of the virus is required. Although highly specific, isolation is time consuming, expensive, and not sensitive enough when autolysis is present. Immunohistochemistry (IHC) using the monoclonal antibody C15C against the conserved envelope glycoprotein gp48 of pestiviruses has been successfully applied to bovine and ovine fetal tissues. 3,14 However, viral isolation and IHC require expertise and special laboratorial resources. A widely used technique since the 1990s is reverse transcription polymerase chain reaction (RT-PCR), which requires good-quality RNA that is not always easy to obtain from fetal tissues. 8 Presence of the viral antigen can also be investigated by antigen enzyme-linked immunosorbent assay (ELISA), which is a rapid technique mostly applied to blood samples from seronegative animals to look for PI animals, although parenchy mal tissue samples from PI animals have also tested positive by antigen ELISA. 13 However, not many reports on the use of antigen ELISA in bovine and ovine fetuses are available. Thus, the purpose of the present study was to evaluate the efficacy of antigen ELISA in identifying BDV in fetal fluids or blood from aborted fetuses, stillbirths, and lambs from commercial flocks with suspicion of BD and from an experimental infection of pregnant ewes and to compare antigen ELISA results with those obtained with RT-PCR. The current study also attempted to determine the most suitable tissues for RNA detection and the effect of autolysis as a limiting factor for virus RNA detection.
Materials and methods
Preparation of clinical samples
Natural infection. Twenty-five commercial flocks located in Northern Spain were selected based on clinical suspicion of BD and with compatible lesions (hypomyelination, hypergliosis, spongiosis, and/or vasculitis) in the brain of at least one fetus or affected lamb. A total of 67 fetuses, 6 stillbirths, and 11 lambs from these flocks were analyzed. At necropsy, several tissues (brain [cortex, mesencephalon, cerebellum] and spleen) were collected. A portion was maintained at −80°C for molecular studies. Another portion was fixed in buffered 4% formalin and embedded in paraffin wax; 4–5-μm sections were stained with hematoxylin and eosin for histopathologic examination. Microbiologic and serologic analyses performed as previously described 6 had ruled out the presence of other abortifacient agents.
Experimental infection. Three fetuses and 22 lambs were obtained in an experimental infection of 13 pregnant ewes with an ovine pestivirus (BDV-4 genotype) at different phases of the gestational period. Details of experimental infection have been described elsewhere. 5 Briefly, after an adaptation period, pregnant ewes were challenged on day 108 (5 ewes, group A), on day 76 (5 ewes, group B), and on day 55 (3 ewes, group C) of gestation by intramuscular inoculation into the hind leg muscles with 2 ml of an ovine pestivirus suspension (0502234 strain) containing 10 6 50% tissue culture infective dose (TCID50). Behavior and clinical signs of infected ewes were monitored daily before and after inoculation, and lambing was closely supervised. After clinical examination, live lambs (15) were euthanized within 24 hr of birth with sodium pentobarbital. a Examination of gross morphology was carried out using standard procedures. Tissues (brain [cortex, mesencephalon, cerebellum], spleen, kidney, thyroid, thymus, spinal cord, lymph node, and skin) from all the lambs (22) and aborted fetuses (3) were collected for viral detection by RT-PCR. To guarantee viral RNA stability, tissues were preserved either at −80°C or at −20°C submerged in a RNA stabilization reagent. b
Viral detection
Reverse transcription polymerase chain reaction. Detection of viral RNA in tissues and blood was carried out by 1-tube RT-PCR as described elsewhere 18 using primers that target the 5′-noncoding region and adding 50–100 ng of RNA template in each reaction. RNA extraction was performed with phenol-chloroform c in samples from a natural infection. For samples from the experimental infection, a commercial viral RNA kit b was used following the manufacturer's procedure but with the addition of an incubation step at 40°C for 30 min in AVL buffer containing carrier RNA (both provided in the kit) to ensure efficient viral particle lysis and RNA extraction.
Antigen enzyme-linked immunosorbent assay. Detection of pestivirus p125/p80 antigen was performed in fetal fluids or whole blood by commercial sandwich ELISA d following the manufacturer's instructions. Fetal fluids were collected from the thoracic cavity of fetuses, and whole blood was taken from the jugular vein (live lambs) or by cardiac puncture (stillbirths) in ethylenediamine tetra-acetic acid–containing tubes. Leukocytes were then extracted following a standard procedure described elsewhere. 5
Blood samples (leukocytes) showing an optical density (OD) >80%, 50–80%, or <50% of the positive control were considered positive, weakly positive, and negative for BDV antigen, respectively. Fetal fluids showing an OD >120%, 80–120%, or <80% of the positive control were considered positive, weakly positive, and negative for BDV antigen, respectively. All the samples giving positive or weakly positive results were retested, and if confirmed, both were considered positive.
Statistical analysis
Comparison of RT-PCR results between the groups of lambs of the experimental infection was performed by chisquare or Fisher exact tests of the SAS 8.0 e statistical package. A P value of <0.05 was considered significant. Complementary sensitivity of one method over the other (RT-PCR and antigen ELISA) was calculated with the following formula: 100 × (no. of samples positive by method 1 and negative with method 2/total no. of samples positive with method 2). This formula measures the additional detection efficacy of method 1 over method 2 when both have similar specificity. 9
Results
Natural infection: detection of BDV in commercial flocks
Forty-five percent of the fetuses (30/67) from the commercial flocks showed compatible lesions with BD. Lesions were restricted to the central nervous system (CNS) and included hypomyelination (43%, 13/30), gliosis (33%, 10/30), spongiosis (30%, 9/30), and vasculitis (17%, 5/30), among others. Autolysis was present in 31% (21/67) of the fetuses, and 24% (16/67) had no lesions. Thirty-eight fetuses, 4 stillbirths, and 8 lambs were analyzed by RT-PCR. Border disease virus RNA was detected in 7.9% (3/38) of the fetuses (Table 1). All positive fetuses had shown compatible lesions with BD, representing 15.8% (3/19) of the fetuses with lesions (Table 1). None of the positive animals were affected by autolysis. A reduced number of stillbirths and lambs were available for RT-PCR analysis, and 50% of the stillbirths and 50% of the lambs analyzed showed BDV RNA (Table 1). BDV RNA-positive samples were detected in 8 of the 25 farms (32%).
Results of reverse transcription polymerase chain reaction (RT-PCR) and antigen enzyme-linked immunosorbent assay (ELISA) in fetuses, stillbirths, and lambs from 25 commercial flocks.*
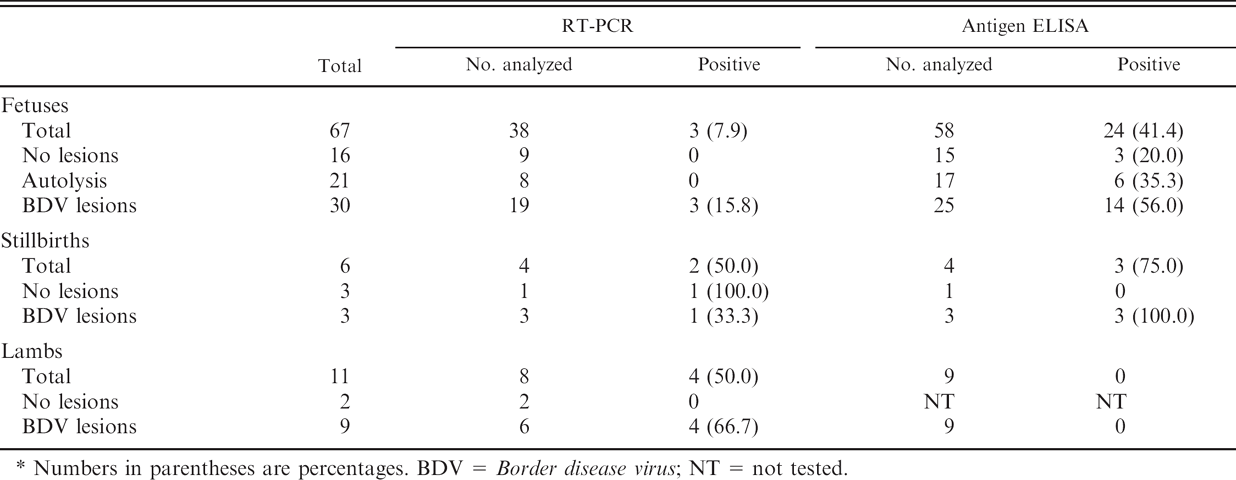
Numbers in parentheses are percentages. BDV = Border disease virus; NT = not tested.
Pestiviral antigen was detected by antigen ELISA at a higher proportion of fetuses and stillbirths than by RT-PCR (Table 1). In contrast, viral antigen was absent in lambs. In the fetuses, antigen detection was independent of the presence of autolysis or BD lesions (Table 1). Five of 24 antigen ELISA-positive fetuses showed OD values between 80% and 120%.
Experimental infection: detection of BDV in experimental animals
Fifty-four and one half percent (12/22) of the lambs and stillbirths from experimental infection were positive by RT-PCR of tissue samples, and the 3 fetuses were negative. More detailed data on each lamb are shown in Table 2. The virus was most frequently detected in lymph node (50%, 10/20), thyroid gland (50%, 11/22), and kidney (48%, 12/25). Detection of viral RNA in the different tissues varied widely among lambs; thus, whereas lamb no. 5 only had positive results in 5 of 10 tissues, others had all the tissue samples test positive (lambs no. 10 and 11). Tissues with higher lipid content, such as the CNS, thymus, and skin, were sometimes negative in animals that tested positive on other tissues. Results on the analysis of 3 different parts of the CNS (cortex, mesencephalon, and cerebellum) identified the cerebellum as the best sample for viral RNA detection.
Lambs and stillbirths from group C, infected at the beginning of the gestational period, were all positive to RT-PCR, compared with 45% and 22% of positive animals from groups B and A, respectively. The 3 fetuses and 2 of the stillbirths (animal nos. 11 and 25) presented autolysis. However, whereas the fetuses were RT-PCR negative, the 2 stillbirths were positive in all (no. 11) or most (no. 25) tested tissues (Table 2).
Regarding the different tissues analyzed, positive tissues in group A accounted for 20.9% (14/67) of the tissues analyzed belonging to 2 of the 9 animals; 37.7% (40/106) of tissues and 5 of 11 animals were positive in group B; and 79.6% (39/49) of tissues and all 5 animals in group C were RT-PCR positive. The number of positive tissues in group C was significantly higher than in groups A and B (P < 0.0001), as it was between groups B and A (P = 0.0199).
Analysis of blood (leukocytes) by RT-PCR detected viremia in a higher number of animals (68%, 15/22), because viral presence was detected in the blood of 6 animals that were RT-PCR negative in tissues. Antigen ELISA was less efficient than RT-PCR in detecting viral presence (31.8%, 7/22). The best correlation between antigen ELISA and RT-PCR results was observed in the lambs from group C.
Comparison of RT-PCR and antigen ELISA results
Considering the results of the natural and experimental infections, RT-PCR and antigen ELISA were performed in parallel on a total of 35 fetuses, 11 stillbirths, and 18 lambs. Reverse transcription polymerase chain reaction detected viral RNA in stillbirths and lambs at higher rates than antigen ELISA (54.5% vs. 45.5% for stillbirths and 55.6% vs. 27.8% for lambs) but not in fetuses (8.6% RT-PCR positive vs. 40.0% antigen ELISA positive; Table 3). However, only 2 of 3 fetuses, 3 of 6 stillbirths, and 5 of 10 lambs positive to RT-PCR were also positive to antigen ELISA. The complementary sensitivity of RT-PCR over antigen ELISA for fetuses, stillbirths, and lambs were 7.1%, 60.0%, and 100.0%, respectively. Similarly, only 2 of the 14 fetuses and 3 of the 5 stillbirths positive to antigen ELISA were positive to RT-PCR. Conversely, the 5 lambs positive to antigen ELISA were also positive by RT-PCR. The complementary sensitivity of antigen ELISA over RT-PCR for fetuses, stillbirths, and lambs was 40.0%, 33.6%, and 0%, respectively. In general, concordance of techniques was poor, and results showed a better performance of antigen ELISA in fetuses, whereas RT-PCR seemed more sensitive when processing samples from lambs and stillbirths.
Antigen enzyme-linked immunosorbent assay (ELISA) and reverse transcription polymerase chain reaction (RT-PCR) results in fetuses, stillbirths, and lambs obtained in the experimental infection study.*
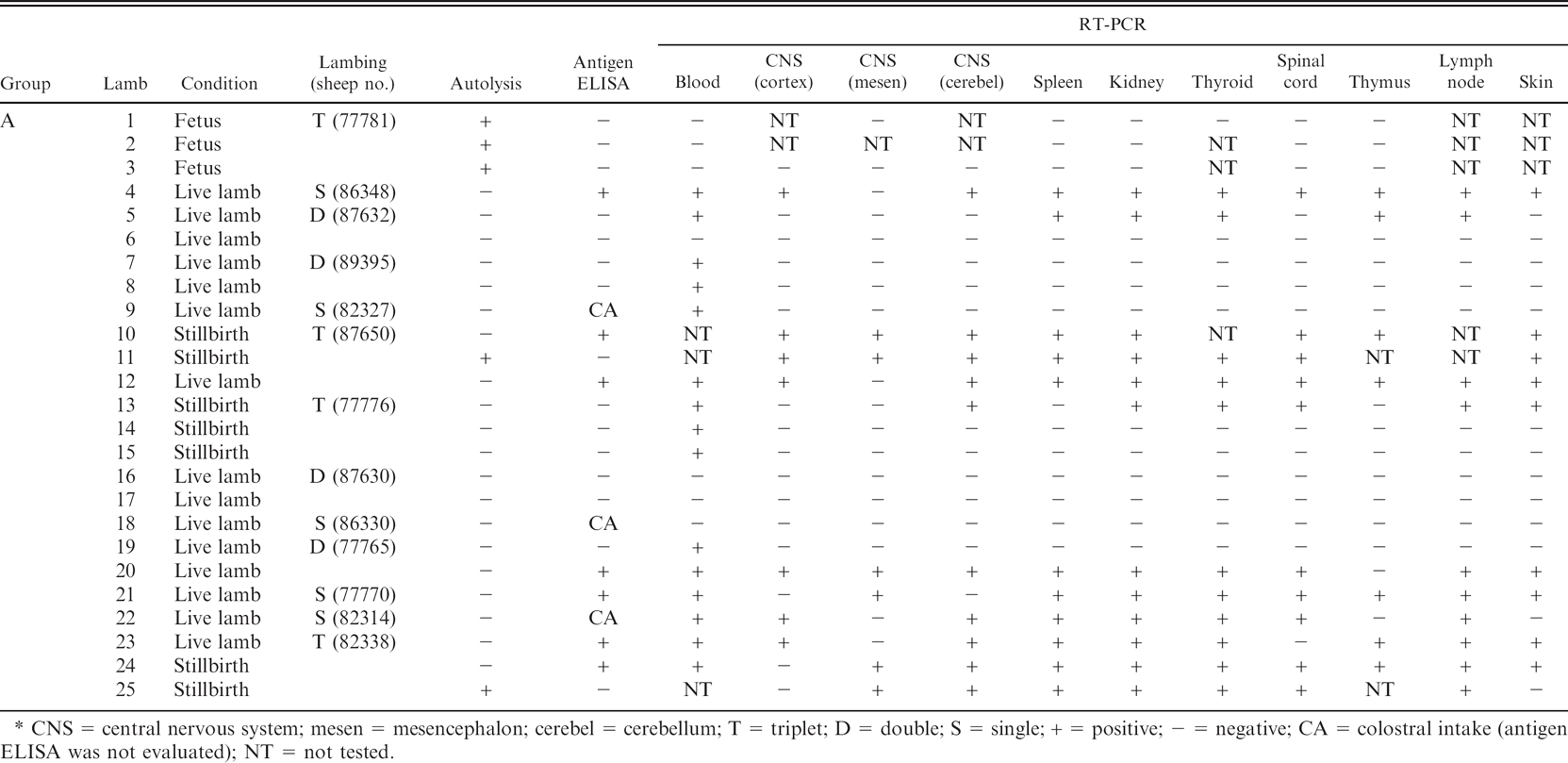
CNS = central nervous system; mesen = mesencephalon; cerebel = cerebellum; T = triplet; D = double; S = single; + = positive; - = negative; CA = colostral intake (antigen ELISA was not evaluated); NT = not tested.
Comparison of reverse transcription polymerase chain reaction (RT-PCR) and antigen enzyme-linked immunosorbent assay (ELISA) in 35 fetuses and 29 born-to-term lambs in which both techniques were performed.*
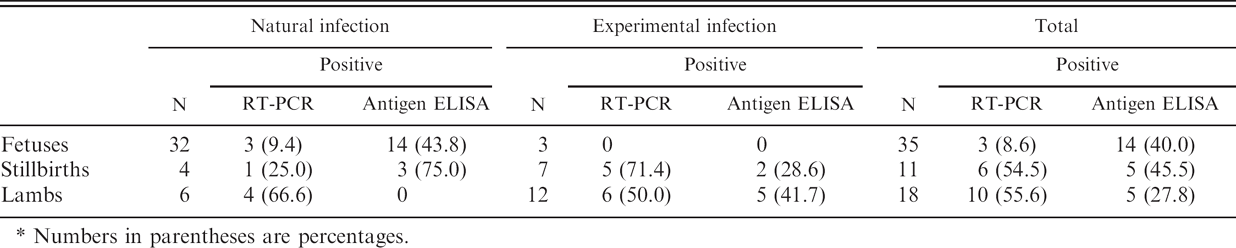
Numbers in parentheses are percentages.
Discussion
The current study provides practical data that can be directly applied to the interpretation of laboratory results for diagnosis of ovine pestivirus infection in fetuses, stillbirths, and lambs. Reverse transcription polymerase chain reaction and antigen ELISA tests were evaluated on samples from commercial flocks with suspicion of BD and from an experimental infection of pregnant ewes with a BDV-4 ovine pestivirus. Whereas the former represents the type of material usually submitted to diagnostic laboratories, including fetuses with different degrees of autolysis, the latter provides fresh material that is ideal for test performance evaluation. In addition, most of the samples from the commercial flocks consisted of fetuses (67/84, 80%), whereas in the experimental infection, the vast majority were lambs (22/25, 88%). Despite the relatively low number of animals included in the present study, this is the first report that compares both RT-PCR and antigen ELISA in tissues and fluids of fetuses and stillbirths and in tissues and blood of lambs.
Antigen ELISA is a rapid and easily automated technique that can be performed in any laboratory without the need for heavy investments. Antigen ELISA of blood or leukocytes has been widely used in cattle and provided excellent results in the detection of PI animals. 13 Its use in sheep is not so extended. However, we have reported its usefulness to detect PI animals in sheep, 1 although not to detect transitory viremia. 5 The commercial kit used in the present study d is validated for blood samples, leukocytes, and tissue extracts but not for fetal fluids. Therefore, included in the present study was an attempt to evaluate its performance on fetal fluids using a more restrictive cutoff also recommended by the manufacturer for leukocyte samples or extract tissues in order to avoid false positives. Hence, performance was good among fetuses and stillbirths from commercial flocks (natural infection), with a high percentage of positive animals (41.4% and 75.0%, respectively), but not for live lambs, which were all negative probably due to the presence of colostral antibodies masking the antigen. 19 In contrast, in lambs that had not taken colostrum (experimental infection), antigen ELISA detected BDV antigen in blood but at a lower proportion than RT-PCR. Therefore, the choice of which technique to use to confirm the presence of BDV in a flock will depend on the type of samples available; antigen ELISA can be an interesting alternative for BD diagnosis when only fetuses or/ and stillbirths with a certain degree of autolysis that could affect the stability of viral RNA are available.
Reverse transcription polymerase chain reaction has been widely applied to tissues from necropsies or to biopsies and blood samples from live animals for pestivirus detection, followed occasionally by subsequent typing of strains by sequencing analysis. 7,15,17 This procedure requires a previous extraction step to obtain pure nucleic acids free of inhibitors. Two different extraction methods were used in the present study, phenol-chloroform for total nucleic acids extraction for the naturally infected samples and a commercial kit b for viral RNA for the experimentally infected samples. Naturally infected samples originated from a previous study on the causes of ovine abortion, 12 whereas experimentally infected samples were part of a more recent study specifically designed for pestiviruses. 5 A comparative study on the performance of different extraction methods for fetal samples reported better results for RNA extraction kits than for phenol-chloroform methods. 8 The lower performance of the latter could have resulted in an underestimation of the RT-PCR results obtained for the naturally infected samples. However, this does not seem to be the case for the lambs, because the percentage of PCR-positive lambs in the natural infection (50%) was similar to the value obtained in the experimental infection (46.7%), suggesting that RNA extraction was good and that the results from both type of samples were comparable.
The study in commercial flocks (natural infection) revealed that RT-PCR analysis of stillbirths and lambs with clinical symptoms rather than fetuses was more suitable to confirm the presence of BDV in a flock. The results from the experimental infection demonstrated that RT-PCR in tissues was more accurate and effective than antigen ELISA in blood at detecting BDV in stillbirths and infected lambs. However, RT-PCR in blood was even more sensitive, detecting 6 animals that tested negative in tissues, maybe due to inhibitors present in tissues and not efficiently removed by the extraction method.
Another objective of the experimental infection study was to identify the most suitable tissues for RT-PCR detection of BDV. Best results were obtained for lymph node, thyroid gland, and kidney, tissues that, along with brain, spleen, and gut, are recommended for virus isolation. 11 In the study herein, gut was not analyzed, but spleen gave very good results (44%, 11/25), whereas results for brain varied, depending on the region analyzed (cortex, mesencephalon, or cerebellum), with best results in the cerebellum (43%, 10/23). Skin biopsies are commonly used for PI animal detection, 2,10 but in the current study, 3 animals that tested positive in other tissues were negative in skin, probably because these animals suffered transient or acute infections, difficult to detect in this type of tissue, or, alternatively, because skin would require a different extraction method to eliminate specific inhibitors.
It is noteworthy that lambs and stillbirths from group C in the experimental infection were all positive by RT-PCR in many tissues. These results, and the fact that inoculation took place 55 day after gestation, suggest that they were PI animals. 11 Other animals in groups A and B were also RT-PCR positive in many tissues and positive by antigen ELISA, indicating high viral load or that these could also be PI animals, particularly those in group B. However, the confirmation of PI status requires 2 consecutive positive analyses by RT-PCR or antigen ELISA in a 2- to 3-week interval. 13 Because the presence of viral RNA was investigated only once, PI status could not be confirmed in the experimental animals. Detection of viral RNA in only a few tissues could be the result of lower viral loads, but this would need confirmation by other techniques, such as quantitative real-time PCR.
Some animals in the experimental infection study (3 fetuses and stillbirths no. 11 and 25) showed a certain degree of autolysis that did not preclude PCR amplification in the 2 stillbirths. In the natural infection study, autolysis was only present in fetuses, and none of the 8 analyzed were positive by RT-PCR. By the time fetuses are naturally expelled a few days after death, the degree of autolysis is quite advanced in most cases. In this situation, antigen ELISA would be the technique of choice. Alternatively, IHC has also been recommended for autolytic samples, 14 but its performance compared with antigen ELISA and RT-PCR is still in progress in our laboratory.
In conclusion, the current study suggests that molecular analysis by RT-PCR of stillbirths and lambs with clinical symptoms is more suitable than the analysis of fetuses to confirm the presence of BDV in a flock. Blood is the recommended sample for RT-PCR detection of pestivirus infection, and, alternatively, lymph node, thyroid gland, and kidney are the most suitable tissues. When only fetal samples with advanced autolysis are available for diagnosis, antigen ELISA of fetal fluids constitutes a valuable alternative.
Acknowledgements
This work was supported by funding from Fondo Europeo de Desarrollo Regional (FEDER) and Instituto Nacional de Investigaión y Tecnología Agraria y Alimentaria (INIA, RTA04-057) and from the Department of Agriculture, Fisheries and Food of the Basque Government. The authors thank Mrs. L. Estévez for her help in carrying out the experimental infection procedure.
Footnotes
a.
Vetoquinol, Alcalá de Henares, Madrid, Spain.
b.
RNAlater™ RNA Stabilization Reagent, Qiagen QIAamp Viral RNA Mini Kit; Qiagen Iberia SL, Las Matas, Madrid, Spain.
c.
Sigma-Aldrich Química SA, Madrid, Spain.
d.
SERELISA BVD p80 Ag Mono-indirect, Synbiotics Europe SAS, Lyon Cedex, France.
e.
SAS Institute Inc., Cary, NC.
