Abstract
An unusual case of osteomyelitis caused by Nocardia cyriacigeorgica infection and resulting in mandibular osteomyelitis and cellulitis (lumpy jaw) is described in a young cat. A 1-cm hard nodular mass was an incidental finding in the right mandible of a 14-month-old cat during routine physical examination. The lesion was fast growing, reaching up to 6 cm in its largest dimension over a 5-week period. A core biopsy of the affected mandible revealed foci of osteolysis, woven bone formation, and a few large clusters of filamentous bacteria surrounded by fine eosinophilic amorphous material bordered by neutrophils, plasma cells, macrophages, and occasional multinucleated giant cells. Pure cultures of acid-fast variable, Gram-positive filamentous bacteria were recovered on blood and chocolate agar plates at 48-hr postinoculation. On amplification and sequencing of the 16S ribosomal RNA and 65-kDa heat shock protein genes, the microorganisms were identified as N. cyriacigeorgica, within the actinomycetes.
Members of the genus Nocardia are Gram-positive, acid-fast variable, aerobic, saprophytic bacteria frequently associated with pyogranulomatous inflammation in a wide range of terrestrial and aquatic animals.9,11 The species of the genus Nocardia in the family Nocardiaceae form a homogenous cluster among the Corynebacterineae, a suborder of the order Actinomycetales. 2 Usually referred to as filamentous bacteria, Nocardia are capable of producing branching filaments and clinically are indistinguishable from other actinomycetes, including members of the genus Actinomyces. There are more than 50 different bacterial species reported as members of the genus Nocardia; however,N. asteroides is the species frequently associated with diseases in animals.3,9,11
Lumpy jaw is a common term used to describe progressive pyogranulomatous osteomyelitis involving the mandible or the maxilla of human beings and animals. In cattle and human beings, the agents commonly associated with this condition are members of the genus Actinomyces. 8 In cats, lumpy jaw or lumpy jaw–like conditions have rarely been reported, and actinomycetomas are usually associated with subcutaneous lesions due to trauma or with pneumonias in animals undergoing immunosuppression.6,9,10
In the current report, a case of mandibular osteomyelitis (lumpy jaw) caused by N. cyriacigeorgica, diagnosed in a rescued 14-month-old stray cat presented to the Ross University School of Veterinary Medicine (RUSVM) Teaching Hospital (Basseterre, St. Kitts, West Indies) for a routine wellness exam and vaccination is described. The cat weighed 3.2 kg and had no history of trauma. Physical examination was normal except for a raised, firm, 1 cm nodular mass with smooth surface palpable on the ventral aspect of the right mid-mandible. The mass was nonpainful to direct firm digital pressure. The following month, the cat returned to the hospital for what the owner perceived to be a mandibular abscess. Clinical examination revealed progressive enlargement of the previously described mass, which was approximately 6 cm in its largest dimension and involved most of the body of the right mandible. The mass remained nonpainful to direct palpation, but was characterized by multifocal areas of ulceration on the buccal surface of the gingiva along the molar portion of the mandible. According to the owner, despite the increased size of the mass and the ulceration, the cat was still eating and drinking normally. The results of blood chemistry analysis, a complete blood cell count, b and urinalysis c were within normal ranges.
The cat was sedated for dental and skull radiographic imaging (Fig. 1), and a bone biopsy was obtained from the core of the lesion and submitted to the RUSVM Diagnostic Laboratory for histological and microbiological analysis. Recovery from anesthesia was unremarkable, and the cat was discharged late that afternoon. The cat was prescribed buprenorphine d (0.02 mg/kg, transmucosally under the tongue, twice a day) for 7 days, and amoxicillin trihydrate–clavulanate potassium e (15 mg/kg, orally, twice a day) for 10 days.
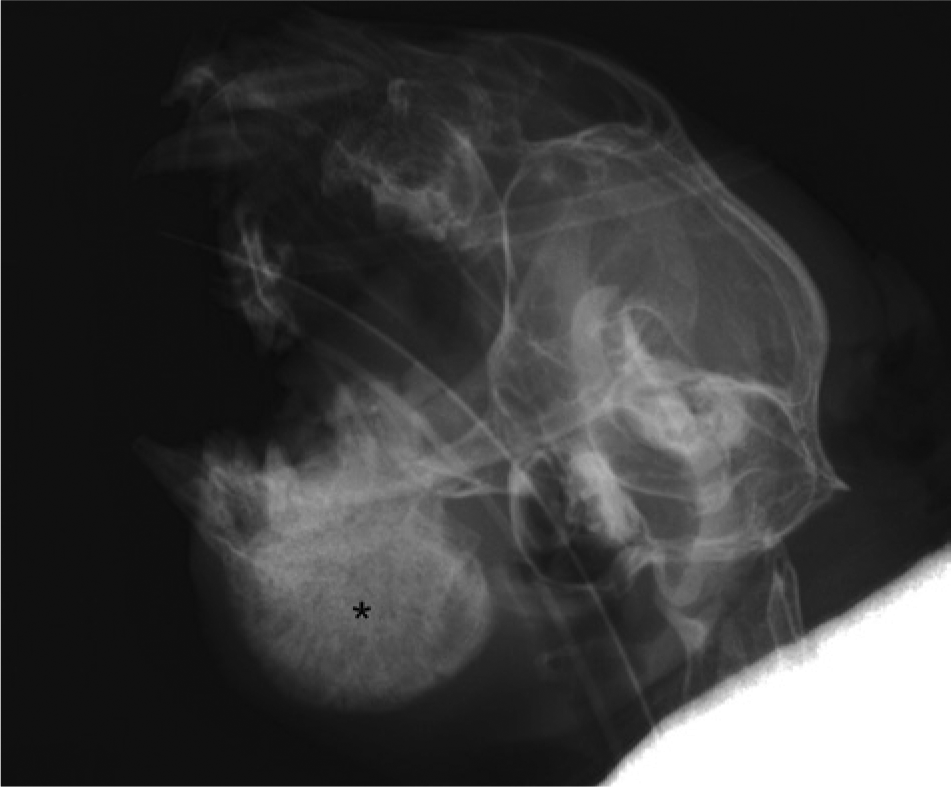
Mandible; young cat with mandibular osteomyelitis. The mass was radiodense (*). Lateral oblique view, radiograph.
Microscopic examination of routine hematoxylin f and eosin g –stained sections from the paraffin-embedded core biopsy revealed woven bone formation around areas of osteolysis and inflammation containing a few large, irregularly shaped, faintly basophilic clusters of filamentous bacteria surrounded by fine eosinophilic amorphous material. Bacterial colonies were surrounded by neutrophils, fewer plasma cells, macrophages, and occasional multinucleated giant cells embedded in variable amounts of fibrous connective tissue. Periosteal proliferation and bone remodeling, manifested by increased osteoclastic activity within surviving bone and the presence of anastomosing trabeculae of newly deposited woven bone, were present around inflammatory foci.
Biopsy samples were used to inoculate tryptic soy agar supplemented with 5% sheep blood (blood agar), h modified Thayer–Martin, i chocolate, j and MacConkey k agar plates. The inoculated plates were incubated for 5 days at 37°C under aerobic and anaerobic conditions. Only 1 colony morphotype was found in the chocolate and blood agar plates incubated in aerobic conditions. The isolate recovered from the chocolate agar plate was found to be undistinguishable from the one recovered in blood agar. Single colonies of the isolate were frozen at −80°C in 1× phosphate buffered saline (PBS) l containing 20% glycerol m for later use.
A loopful of bacteria was suspended in 500 µl of sterile 1× PBS to a McFarland standard of 0.5, and subjected to DNA extraction n following the manufacturer’s suggested protocol for Gram-positive bacteria. The extracted DNA was stored at −20°C until further use. Molecular identification of the isolates was determined by amplification and sequencing of the 16S ribosomal RNA (rRNA) and 65-kDa heat shock protein (hsp65) genes following published protocols and primers.7,13 Amplicons for sequencing were purified o according to the manufacturer’s suggested protocol, and were submitted for sequencing to Davis Sequencing (Davis, California). The sequences were compared with those stored in GenBank using a BLASTn search of the nonredundant nucleotide (nr/nt) database of the National Center for Biotechnology Information.
The colonies were white and dry and were observed at 48-hr postinoculation. The isolates were Gram-positive, acid-fast–negative filamentous bacteria that, on further molecular diagnosis provided by amplification and sequencing the 16S rRNA and hsp65 genes, were confirmed as N. cyriacigeorgica. The isolate showed 98% and 97% identity to N. cyriacigeorgica strain American Type Culture Collection (ATCC) 14759 16S rRNA and hsp65 genes (GenBank accession nos. EF127493.1 and EF127503.1, respectively).
The minimal inhibitory concentrations (MICs) of 15 antimicrobial agents to N. cyriacigeorgica and a quality control strain (Escherichia coli ATTC 25922) were tested using a commercial system, p following the manufacturer’s suggested protocol and the Clinical and Laboratory Standards Institute published protocols.4,5 The MIC value was defined as the lowest concentration exhibiting no visible growth. Each antibiotic was run in triplicate. The isolate was susceptible to imipenem, gentamicin, trimethoprim–sulfamethoxazole, cefepime, and amikacin; intermediate to levofloxacin; and resistant to erythromycin, ceftazidime, tetracycline, ampicillin, amoxicillin, vancomycin, oxacillin, clindamycin, and chloramphenicol. The quality control E. coli produced results within acceptable published ranges for the antimicrobial agents. 4
Because of the extensive bone involvement, surgical removal of the affected portion of the mandible and long-term antibiotic therapy was recommended to the client. Based on the in-vitro susceptibility of N. cyriacigeorgica to trimethoprim–sulfamethoxazole (MIC < 0.02 µg/ml) and the previously published antimicrobial treatment success in the treatment of nocardiosis infections,15,17 a treatment protocol of 60 mg/kg orally twice a day was initiated. q At the same time, the cat was started on human folic acid r supplement to prevent anemia associated with trimethoprim–sulfamethoxazole treatment in cats. The client declined the surgical option and discontinued the medication approximately 2 weeks after starting therapy. The cat’s overall physical condition worsened, and 2 months later it was brought back to the RUSVM Teaching Hospital for evaluation. Because of the poor body condition and prognosis as a result of the unsuccessful antibiotic therapy, humane euthanasia was recommended.
The main postmortem findings were confined to the right mandible and regional lymph nodes. A few small areas of alopecia and ulceration, covered by variable amounts of dry crusty exudate, were present on the overlying skin (exudative dermatitis, draining fistulous tracts; Fig. 2). The ramus and molar portion of the right mandible were markedly and diffusely enlarged, up to 5 cm in width, firm but slightly brittle and could be sectioned with the necropsy knife. Sterile swabs from the mandibular lesion were sent to bacteriology for testing. The right lower gingiva was diffusely red, firm, and markedly swollen. This swelling extended to the right sublingual intermandibular space. Sections of the enlarged mandible revealed numerous multifocal brown-tan nodules, interspersed with small cavities less than 2 mm in diameter that oozed small amounts of brownish granular material and gave the bone a “honeycomb” appearance. The right mandibular and parotid lymph nodes were markedly enlarged (up to 2 cm in the largest dimension), firm, and wet. Microscopically, the enlarged mandible was characterized by prominent periosteal bone proliferation and had similar findings to those described in the core biopsy (Fig. 3). Bacterial colonies were numerous and stained positive with Coates modified Fite Nocardia stain. 1 Microscopically, the regional lymph nodes revealed moderate follicular and parafollicular hyperplasia with medullary plasmacytosis and erythrophagocytosis. No bacteria or areas of necrosis or inflammation were detected in the lymph node sections examined. The final morphologic diagnosis for the lesions in the cat was severe pyogranulomatous osteomyelitis of the right mandible, with intralesional colonies of filamentous bacteria, and chronic lymphoid hyperplasia of the regional (mandibular and parotid) lymph nodes. Molecular testing of the mandibular swabs taken at necropsy confirmed the presence of N. cyriacigeorgica. Maceration of the mandibles further demonstrated the severity and chronicity of the inflammatory lesions within the right mandible (Fig. 4).
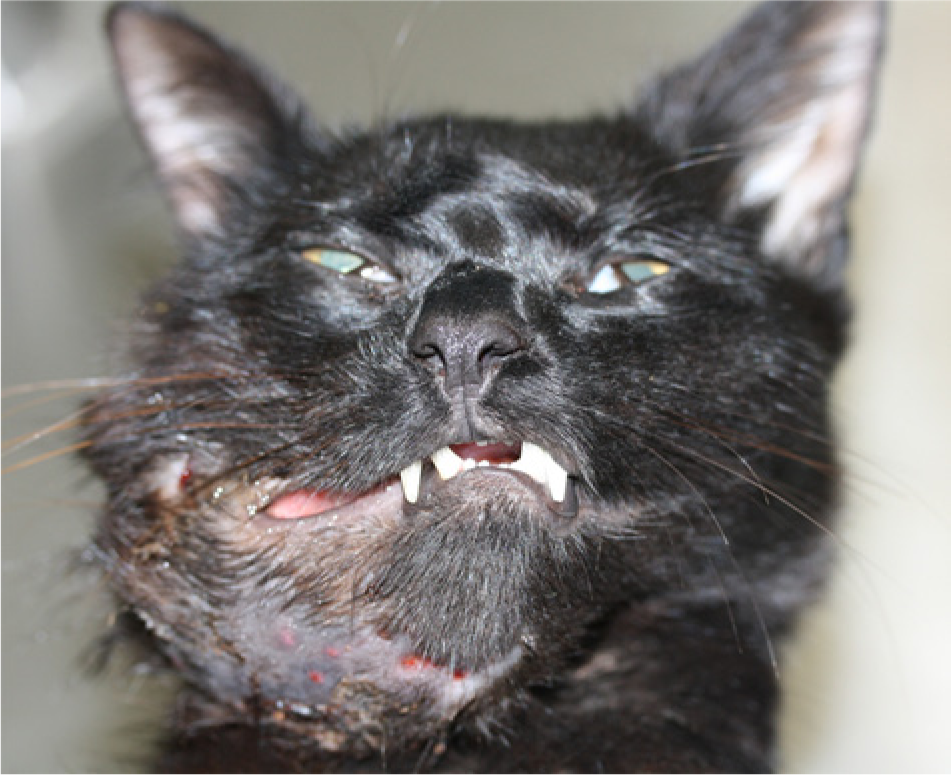
Areas of alopecia and ulceration covered by variable amounts of dry crusty exudate were present on the skin overlying the markedly enlarged right mandible.
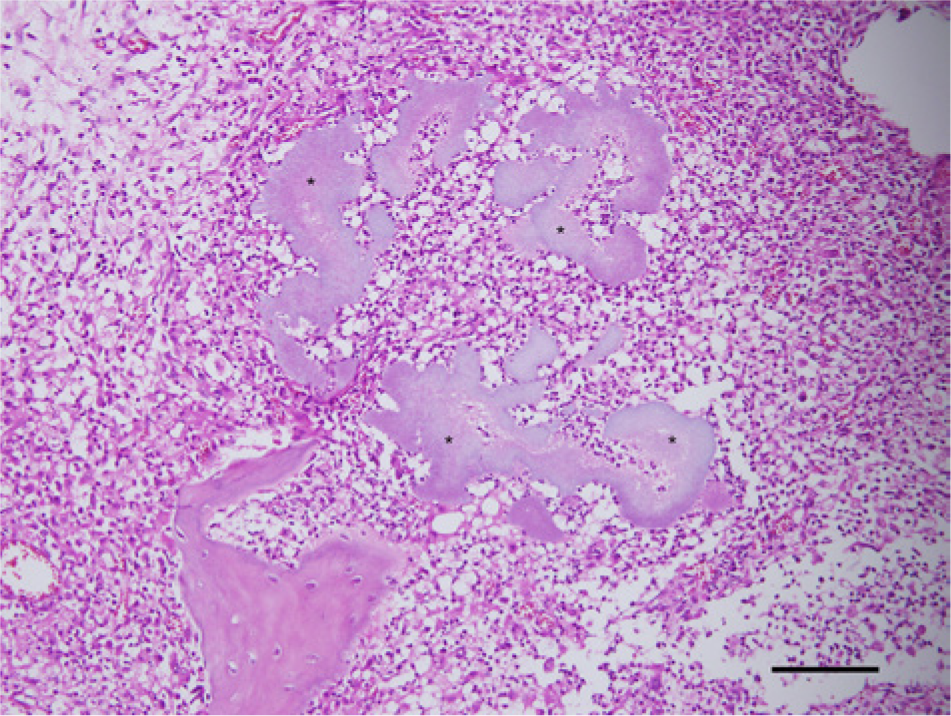
Right mandible; young cat with mandibular osteomyelitis. Pyogranulomatous osteomyelitis with large, irregularly shaped, faintly basophilic clusters of filamentous bacteria (*). Hematoxylin and eosin. Bar = 50 µm.
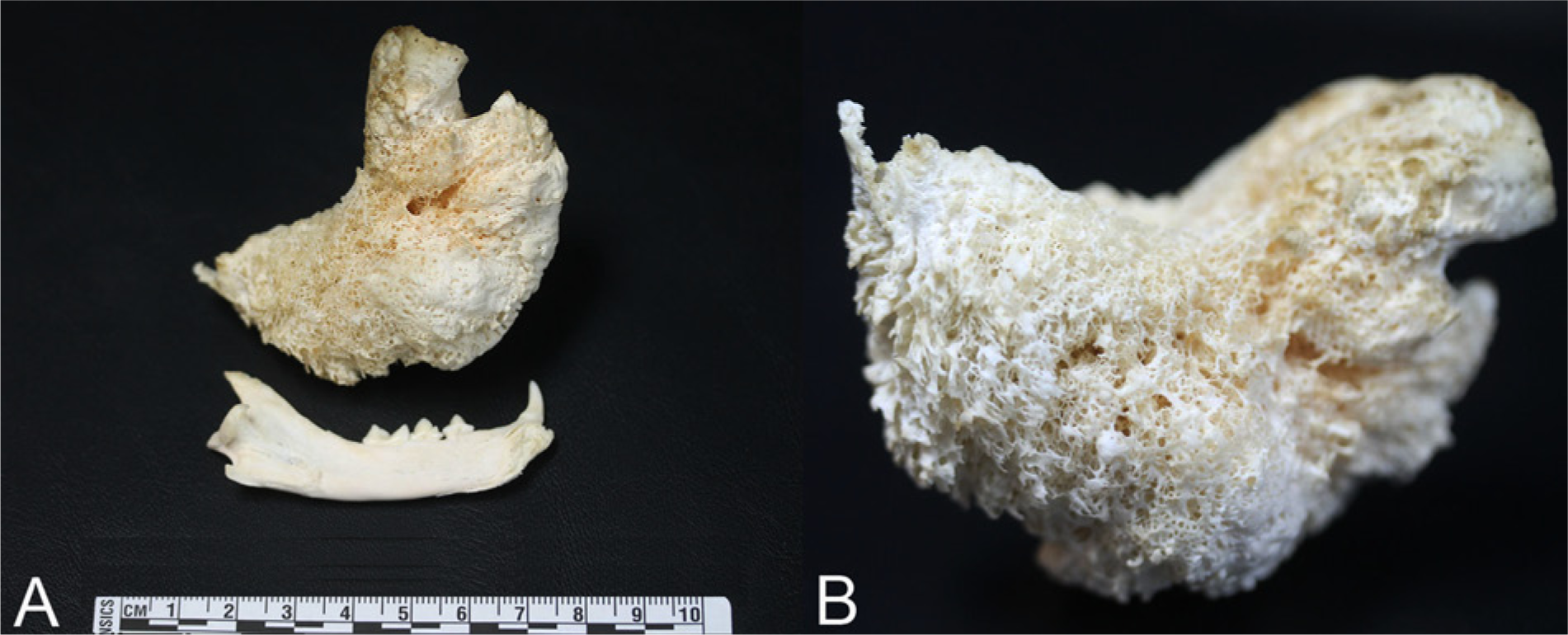
Young cat with mandibular osteomyelitis.
Initially, in addition to bacterial or fungal osteomyelitis, the differential diagnoses for this case included callus formation as a sequel to mandibular trauma and/or fracture. After radiographic assessment of the mandible, in spite of the young age of the patient, a primary bone neoplasm was also considered. Mandibular osteomyelitis (lumpy jaw) due to N. cyriacigeorgica was diagnosed by microscopic assessment and bacteriology molecular testing of a core bone biopsy of the affected mandible. A similar case of mandibular osteomyelitis in a cat has been reported from Brazil. 6 In that case, N. africana was identified as the etiologic agent, and the authors provided no description of the pathologic findings within the affected mandible. Nocardia cyriacigeorgica was described as a new species within the actinomycetes in 2001. 3 Since this first description, N. cyriacigeorgica has been recovered from human patients worldwide3,14 and appear to account for up to 35% of Nocardia strains recovered from patients in the southern United States. 3 Because of the increased diagnosis of N. cyriacigeorgica as the causative agent of diseases in human beings, as well as the diagnosis of this bacterium in new geographical areas, it is now regarded as an emergent pathogen.12,14 By contrast, only a few reports mention this bacterium as the causative agent of disease in veterinary medicine.2,8,16,18
Genotypic and phenotypic comparison between members of the genus Nocardia revealed that human isolates of N. cyriacigeorgica and N. asteroides type VI strains share identical partial 16S rRNA gene sequences and similar antimicrobial susceptibility.3,14 It seems possible that bacteriologic misdiagnosis due to the genetic homology between these 2 Nocardia species may partially account for the scarcity of reported cases of N. cyriacigeorgica as the causative agent of pyogranulomatous disease in veterinary medicine. Human isolates have been found to be susceptible to broad-spectrum cephalosporins, amikacin, imipenem, linezolid, and few to ciprofloxacin, but resistant to penicillins, clarithromycin, and ciprofloxacin. The isolate recovered from the cat in the current report had similar antibiotic susceptibility to that reported in human beings.
Infections caused by members of the genus Nocardia are usually associated with immunosuppression 9 ; interestingly, no evidence of immunosuppression was detected in the young cat in the current report. It is likely that the organism was traumatically introduced into the gingiva and led to the severe osteomyelitis that grossly and microscopically closely resembled mandibular actinomycosis (lumpy jaw) in cattle.
Footnotes
Acknowledgements
The authors acknowledge Mr. David Hilchie and Ms. Candita Chapman at the Ross University, School of Veterinary Medicine, Department of Biomedical Sciences, for their technical support.
a.
VetScan VS2, Abaxis North America, Union City, CA.
b.
VetScan HM5, Abaxis North America, Union City, CA.
c.
VetLab UA analyzer, IDEXX Laboratories Inc., Westbrook, ME.
d.
Buprenex, Reckitt Benckiser, Slough, Berkshire, United Kingdom.
e.
Clavamox, Pfizer, New York, NY.
f.
Hematoxilin Platinum Line hematoxylin stain solution (Harris’ modified), Mercedes Medical Inc., Sarasota, FL.
g.
Eosin E511-100, Fisher Scientific, Fair Lawn, NJ.
h.
Tryptic soy agar supplemented with 5% sheep blood, Remel Inc., Lenexa, KS.
i.
BBL modified Thayer-Martin (MTM II) agar, BD Diagnostic Systems, Sparks, MD.
j.
Chocolate agar, Remel Inc., Lenexa, KS.
k.
MacConkey agar, Remel Inc., Lenexa, KS.
l.
Phosphate buffer saline, Fisher Scientific, Fair Lawn, NJ.
m.
Glycerol, Thermo Fisher Scientific Inc., Waltham, MA.
n.
DNeasy kit, Qiagen Inc., Valencia, CA.
o.
QIAquick PCR cleanup kit, Qiagen Inc., Valencia, CA.
p.
E-test, bioMérieux Inc., Durham, NC.
q.
Sulfatrim suspension, Alpharma, Baltimore, MD.
r.
Folic acid, Nature Made, San Fernando, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was supported by Ross University, School of Veterinary Medicine (RUSVM) Research Program.
