Abstract
A 7-year-old, female Huacaya alpaca presented with a 3-week history of left-sided head tilt and mild ataxia. Despite aggressive supportive therapy with empirical antibiotic, anti-inflammatory, thiamine, and anthelminthic therapy, neurologic signs progressed to a right-sided head tilt, positional nystagmus, and episodes of severe ataxia and obtundation. On necropsy, discrete expansile to poorly delineated infiltrative masses with tan, creamy to grumous exudate were observed in the right petrous temporal bone, right middle and inner ear, right cerebellum and medulla as well as in lung, kidney, liver, spleen, lymph nodes, and the gastrointestinal tract. Impression cytology and histology revealed a myriad of intralesional yeast, 10–25 μm in diameter, with double-contoured walls, granular protoplasm, and broad-based budding. Culture characteristics, including conidial morphology and temperature-dependent yeast phase transformation, were consistent with Blastomyces sp. Positive Blastomyces antigenuria was identified on a postmortem sample of urine. Microsatellite typing and sequencing of the internal transcribed spacer region 2 identified the infecting strain as a genetic group 2 isolate of Blastomyces dermatitidis. The present report of camelid blastomycosis adds intracranial blastomycosis to the differential list for camelid neurologic disease in endemic regions.
Blastomyces dermatitidis is a fungal pathogen belonging to the taxonomic order, Onygenales. The Onygenales order is defined by the following morphologic characteristics: The sexual teleomorph stage forms rudimentary asci surrounded by a network of hyphae, and the asexual anamorph stage forms either unicellular aleuriconidia or chains of arthroconidia. 5 For B. dermatitidis, the corresponding sexual teleomorph stage is Ajellomyces dermatitidis.4,5,17 Additionally, the anamorph stage of B. dermatitidis is thermally dimorphic, existing as a mold form at cooler temperatures (25–30°C) and converting to a yeast form at warmer temperatures (approximately 37°C).4,5,17 The mold phase of B. dermatitidis is a soil saprophyte, preferentially found in warm, moist acidic soils with high organic content. 5 Endemic regions border the Mississippi, Ohio, and St. Lawrence River valleys and the Great Lakes, and also include parts of Africa and Central and South America.4,5,8 The usual route of human or veterinary infection is inhalation of conidia, although cutaneous or oral inoculation has been reported.4,5,8 In human beings, the estimated incubation period is 4–6 weeks. 5 The more frequently reported susceptible species include human beings, dogs, cats, and horses1,8; however, blastomycosis had been reported in other mammalian species, including a free-ranging black bear. 11 Clinical infection can manifest as localized pulmonary or cutaneous disease or can spread to establish disseminated infection. Lung, skin, bone, and the genitourinary tract are the most frequent sites involved. In disseminated cases, lesions can occur in any tissue including lymph node, eyes, central nervous system, liver, kidney, spleen, heart, endocrine organs, and the gastrointestinal tract.4,5,8
Previous genetic characterization of B. dermatitidis using microsatellite markers revealed that it is a genetically diverse pathogen 21 with the 2 principally divergent genetic groups being associated with different clinical phenotypes. 22 In human patients, group 1 was more frequently associated with respiratory-only infections, while group 2 was more frequently associated with disseminated infection. 22 Examination of veterinary isolates revealed that the majority of cases identified were disseminated infections caused by group 2 isolates 1 ; however, statistical analysis of associations between microsatellite group and clinical phenotype was hampered by the small number of group 1 cases. Within the past year (2013), multilocus sequence analysis has led to the proposal of 2 separate phylogenetic species. 6 The present report describes a case of disseminated B. dermatitidis, genetic group 2 infection with central nervous system involvement in a novel veterinary species, and discusses current diagnostic methods and the implications of genotyping.
A 7-year-old, female Huacaya alpaca (Vicugna pacos) presented to the University of Wisconsin, Veterinary Care (Madison, Wisconsin) with a 3-week history of left-sided head tilt and mild ataxia. On physical exam, the alpaca was dull but responsive, with intact cranial nerves and an unremarkable otoscopic exam of the external ear canals. Gastrointestinal motility was absent to minimal, and thoracic auscultation was unremarkable. Hematology and serum chemistry revealed a mild hyponatremia (144 mmol/l), hypokalemia (4.2 mmol/l), hypophosphatemia (3.4 mmol/l), and hypoalbuminemia (2.6 g/dl), moderate hyperglycemia (253 mg/dl), and elevated creatinine (2.2 mg/dl), creatinine kinase (404 U/l), and aspartate aminotransferase (356 U/l), interpreted to reflect decreased feed intake, stress, slight prerenal or renal azotemia, and recumbency, respectively. All other hematologic and biochemical parameters were within reference ranges. Cerebrospinal fluid analysis was consistent with mild nonspecific inflammation. Despite aggressive supportive therapy over 3 days with empirical antibiotic, anti-inflammatory, thiamine, and anthelminthic therapy, neurologic signs progressed. The alpaca developed a right-sided head tilt, right-sided hemiparesis, positional nystagmus, and multiple episodes of severe ataxia with a wide-based stance, obtundation, and potential seizure activity (paddling in lateral recumbency). Humane euthanasia and postmortem evaluation were elected.
At necropsy, the right petrous temporal bone, right middle ear, and tympanic bulla were infiltrated and effaced by a tan, creamy to grumous exudate. The same process extended into the cranial vault to form an expansile and infiltrative mass in the right cerebellopontine angle that displaced the cerebellum to the left, infiltrated the cerebellar peduncles, and filled the fourth ventricle (Fig. 1A). The right cerebellar peduncle was more extensively affected than the left, and the medulla was only minimally infiltrated. A similar tan, creamy to grumous exudate regionally expanded the pituitary gland. The cerebral aqueduct and the third and lateral ventricles were mildly dilated with clear fluid. The left petrous temporal bone and ear canal were unaffected. Small discrete to large coalescing, tan masses that oozed a similar creamy to grumous exudate were observed in the lung (Fig. 1B), liver, kidney (Fig. 1C), spleen, heart, retropharyngeal lymph node, and colon. Multifocal ulceration of compartment 3 was also present.
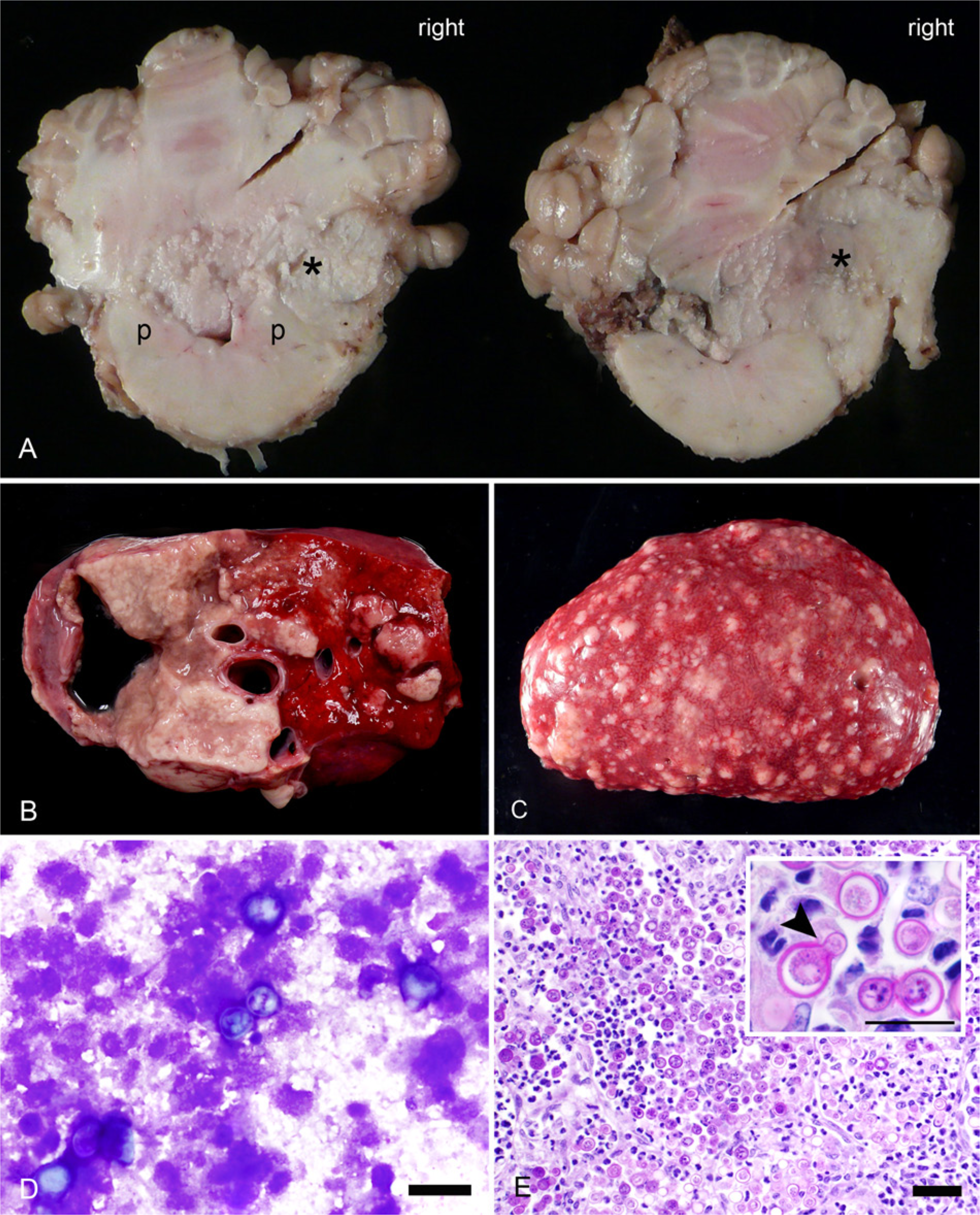
Disseminated blastomycosis in a Huacaya alpaca (Vicugna pacos).
Impression cytology from fresh lung was allowed to air dry and subsequently Wright–Giemsa stained, revealing numerous yeast intermixed within a background of degenerate cellular debris (Fig. 1D). Urine was collected to evaluate for Blastomyces antigenuria. Samples of fresh lung were frozen at −80°C for DNA extraction and fungal culture. Tissue samples were collected in 10% buffered formalin, routinely processed, and embedded in paraffin blocks. From these blocks, 5-μm-thick sections were cut and stained with hematoxylin and eosin. Additional sections were also stained with periodic acid–Schiff to aid visualization of fungal morphology.
Histology demonstrated pyogranulomatous right temporal osteomyelitis, right otitis media and interna, and right-sided rhombencephalic meningoencephalitis with parenchymal compression and reactive astrocytosis. Pyogranulomatous pituitary adenitis, pneumonia, myocarditis, cortical nephritis to pyelonephritis, hepatitis, splenitis, lymphadenitis, submucosal to mural colitis, and submucosal gastritis were also observed. Intermixed within all of the pyogranulomatous inflammation, but most numerous within the affected sections of lung, were a myriad of 10–25 μm in diameter yeast (Fig. 1E). The yeast were characterized by refractile, double-contoured walls, granular protoplasm, and broad-based budding (Fig. 1E, inset). Equal numbers were viable and nonviable (characterized by a cell wall alone, without any internal protoplasm) but only rarely were yeast phagocytosed within multinucleated giant cells.
The urine sample was Blastomyces-positive by antigen enzyme immunoassay, a reported as above the limit of quantification (>14.7 ng/ml). To confirm the identification and determine the etiologic species, DNA was extracted b from fresh-frozen lung with modifications as previously described. 21 Five microliters of nucleic acid were amplified c with 0.2 µM final concentration of the third and fourth internal transcribed spacer region (ITS3 and ITS4, respectively) primers specific to the 5.8S and 28S ribosomal DNA (rDNA) regions. 26 Amplification conditions were as follows: 15 min denaturation at 95°C; 36 cycles of 94°C for 30 sec, 55°C for 30 sec, and 72°C for 1 min; followed by a 4-min final extension at 72°C. The polymerase chain reaction (PCR) products were analyzed by gel electrophoresis on a 1.5% agarose gel with 0.5 mg/l of ethidium bromide and visualized d to confirm product size and yield. The amplified products were purified e for direct sequencing. Sequencing was carried out using dye-terminator chemistry, f electrophoresed, and detected on a capillary sequencer. g Three hundred thirty-six base pairs of bidirectional sequence, spanning a portion of the 5.8S rDNA and ITS2, was obtained and aligned h based on 4-fold coverage of the amplified region. Sequence identity was confirmed as a 100% match with Ajellomyces dermatitidis (sexual teleomorph stage of B. dermatitidis) using the Basic Local Alignment Search Tool (BLAST) program (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi). The resulting sequence was deposited in GenBank (accession no. KF543873).
Additional fresh-frozen lung tissue was minced, inoculated onto selective media, i and incubated at 28°C. Resulting colonies were subbed to potato flake agar and liquid culture medium, j and incubated at 28°C and 35°C, respectively. Typical “lollipop” conidia were visualized by tape mount with lactophenol cotton blue staining (Fig. 2A), and characteristic broad-based budding yeast (Fig. 2B) were observed using calcofluor white in 10% potassium hydroxide. DNA was extracted from the cultured Blastomyces isolate for genotyping with 27 microsatellite markers, as previously described. 21 Amplified products were sized on a capillary sequencer g by use of an internal standard. k After alleles were binned and analyzed, as previously described, 21 the isolate was confirmed as belonging to microsatellite group 2, with novel alleles identified at loci 3 and 18.
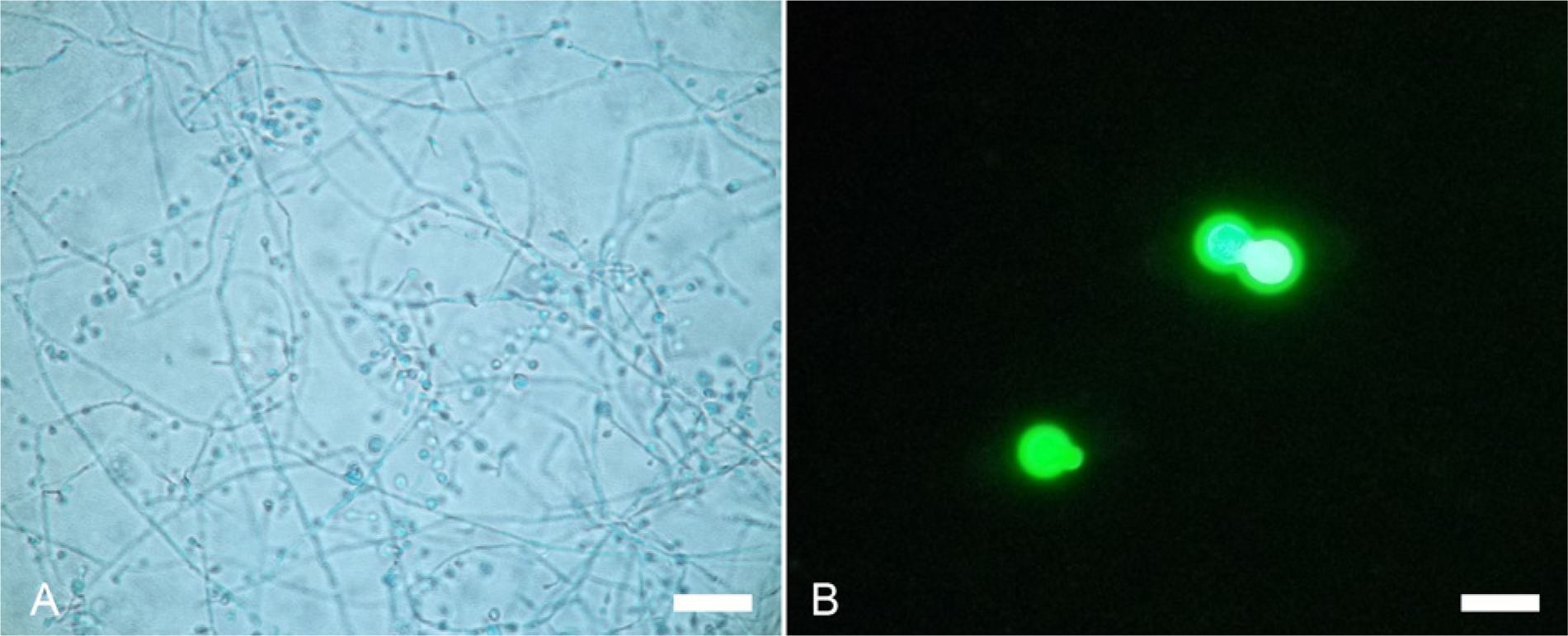
Blastomyces dermatitidis isolated from a Huacaya alpaca (Vicugna pacos).
Based on the yeast morphology, culture characteristics, positive antigenuria, and absolute ITS2 sequence identity to A. dermatitidis, the causative agent of the disseminated mycotic infection in this alpaca was confirmed to be B. dermatitidis. The infection was probably established by environmental exposure, as the alpaca was housed in an endemic region (northeast morainal region of Illinois 16 ). Fungal dissemination to the middle and inner ear, and subsequently to the cranial vault, most likely spread via the eustachian tube and ascended centripetally through foramina19,24,28 with progression to petrous temporal osteomyelitis and cerebellopontine meningoencephalitis. Direct hematogenous seeding of the petrous temporal bone or meninges could also have been possible. Based on the absence of left-sided otitis media, otitis interna and/or meningoencephalitis, the initial clinical signs of a left-sided head tilt were most likely explained by paradoxical central vestibular disease. The contribution of immunosuppression to establishment of infection, pathogen burden, and progression of disease was considered, but substantiating evidence (hematologic or globulin abnormality, cytolysis in lymphoid organs, other opportunistic infection) was not observed.
Common differential diagnoses for central vestibular disease in New World camelids, essentially alpacas and llamas (Lama glama), include listeriosis, trauma, polioencephalomalacia, verminous meningoencephalomyelitis with Parelaphostrongylus tenuis, as well as centripetal extension of otitis media and/or interna, as in the current case. 27 Meningitis and/or meningoencephalitis, as a sequel to bacterial septicemia, is also a differential; however, central nervous system involvement subsequent to disseminated fungal infection is not commonly recognized. 27 When recognized, fungal intracranial or intraspinal canal disease in New World camelids has been most frequently associated with Coccidioides immitis.12,13,23 Cryptococcus sp.,3,15 Cladophialophora bantiana, 14 Rhizopus sp., 12 and Aspergillus sp. 7 have also been reported to cause primary or secondary (compressive) cerebrospinal disease in New World camelids.
Postmortem diagnosis of blastomycosis is generally based on identification of the yeast in tissue or in cytologic preparations.4,8 Morphologic features used to differentiate Blastomyces sp. from other pathogenic fungi include size of the yeast (5–30 μm), discrete double-contoured cell walls, broad-based budding, and lack of a capsule or endosporulation.4,5,8,17 The gold standard is evaluation of culture characteristics, including mold growth at 25–28°C on selective media, growth at higher temperatures (approximately 35–37°C), and conversion to yeast phase at higher temperatures.4,5 Culture is less commonly performed because of biosafety level III requirements and the risk of laboratory personnel exposure.5,8,9,17 Alternately, a sensitive and highly specific PCR assay for the WI-1 (BAD1) virulence gene can be performed successfully on paraffin-embedded, formalin-fixed tissues. 2
For antemortem diagnosis, a commercially available titration-based enzyme immunoassay a for Blastomyces galactomannan antigen in urine or serum is available and has a demonstrated sensitivity of 93.5% by antigenuria and 87.0% by antigenemia. 25 In both human and veterinary patients, evaluation of antigenuria is a commonly used noninvasive method to establish the diagnosis and monitor response to treatment.10,25 Cross-reactivity occurs with Paracoccidioides sp., Histoplasma sp., Penicillium marneffei (anamorph of Talaromyces marneffei), and less frequently with Cryptococcus sp. and Aspergillus sp., 10 but in endemic regions and in the appropriate clinical setting, the diagnostic value of the assay compensates for the lack of specificity. 25 Diagnosis by serologic detection of antibodies is of limited utility.4,25 A qualitative agar gel immunodiffusion method for detection of immunoprecipitans to the yeast-specific A antigen is also availablel,m and can be used across species (does not require species-specific reagents) and to evaluate response to therapy.18,25
Genetic diversity within B. dermatitidis was initially established using restriction fragment length polymorphisms in mitochondrial and rDNA and random amplified polymorphic DNA array fingerprinting of the entire genome.20,29 Isolates were separated into 3 genotypes: groups A, B, and C. Later, genetic characterization using microsatellite markers revealed that B. dermatitidis is a genetically diverse pathogen with the 2 principally divergent genetic groups being associated with different clinical phenotypes in human beings.21,22 Patients infected with group 1 strains were more likely to have respiratory-only disease. Patients infected with group 2 strains were more likely to have disseminated disease. 22 Within the past year, multilocus sequence typing of Blastomyces isolates has led to the proposal of 2 separate phylogenetic species (B. dermatitidis [PS1], and B. gilchristii [PS2]). 6 Five isolates analyzed by both multilocus sequence typing and microsatellite typing suggest that PS1 (B. dermatitidis) is analogous to microsatellite group 2 and PS2 (B. gilchristii) is analogous to microsatellite group 1. 6 The previous study 6 found that B. gilchristii may be relatively geographically restricted to Ontario, Canada and the U.S. states of Wisconsin and Minnesota, and more frequently associated with outbreaks in these hyperendemic areas. This is consistent with previous findings 21 that, to date, all outbreak-associated isolates are microsatellite group 1 (B. gilchristii).
A similar epidemiologic study 1 of clinical veterinary B. dermatitidis isolates demonstrated that significantly more infections were attributable to microsatellite genetic group 2. In addition, most veterinary cases in that study 1 resulted in disseminated disease, as in the case described herein. However, associations between microsatellite group and clinical manifestation could not be statistically verified because of the small number of genetic group 1 isolates. To date, environmental exposure in endemic regions remains the single recognized risk factor for blastomycosis in veterinary species. Other disease determinants, such as genetic grouping, fungal virulence factors, or host comorbidities such as immune-suppressive conditions, remain to be fully evaluated. Expansion of the sample collection and further evaluation of the genetic diversity among clinical isolates of B. dermatitidis can only increase the current understanding of disease determinants for this potentially fatal human and veterinary pathogen. With the current case, an additional isolate from a novel host species has been added to enrich the known genetic diversity of B. dermatitidis. Additionally, disseminated blastomycosis has been added to the differential list for camelid neurologic disease.
Footnotes
Acknowledgements
The authors would like to acknowledge the excellent technical assistance provided by the staff at the Veterinary Medical Teaching Hospital, University of Wisconsin (Madison, Wisconsin) and Marshfield Labs (Marshfield, Wisconsin), with special thanks to Tammy Krier. The authors extend their gratitude to Dr. Melissa Behr for detailed review of this article and Dr. Benjamin Darien for case mentorship.
a.
316 MVista, MiraVista Diagnostics, Indianapolis, IN.
b.
QIAamp DNA mini kit, Tissue protocol; Qiagen Inc., Valencia, CA.
c.
HotStarTaq master mix kit, Qiagen Inc., Valencia, CA.
d.
Gel Doc 2000 instrument, Bio-Rad Laboratories, Hercules, CA.
e.
QIAquick PCR purification kit, Qiagen Inc., Valencia, CA.
f.
ABI BigDye v3.1, Applied Biosystems, Foster City, CA.
g.
ABI 3130xl DNA analyzer, Applied Biosystems, Foster City, CA.
h.
DNASTAR Inc., Madison, WI.
i.
Mycosel agar, BD, Franklin Lakes, NJ.
j.
7H9 broth, BD, Franklin Lakes, NJ.
k.
GeneScan-500 ROX size standard, Applied Biosystems, Foster City, CA.
l.
Gibson Laboratories, Lexington, KY.
m.
Immuno-Mycologics Inc., Norman, OK.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
