Abstract
Between 2008 and 2012, commercial Swiss layer and layer breeder flocks experiencing problems in laying performance were sampled and tested for infection with Duck adenovirus A (DAdV-A; previously known as Egg drop syndrome 1976 virus). Organ samples from birds sent for necropsy as well as blood samples from living animals originating from the same flocks were analyzed. To detect virus-specific DNA, a newly developed quantitative real-time polymerase chain reaction method was applied, and the presence of antibodies against DAdV-A was tested using a commercially available enzyme-linked immunosorbent assay. In 5 out of 7 investigated flocks, viral DNA was detected in tissues. In addition, antibodies against DAdV-A were detected in all of the flocks.
Keywords
Introduction
Egg drop syndrome 1976 (EDS-76) is an economically important viral disease of chickens and quail occurring worldwide. 1 Since the first appearance of EDS-76, the disease has been described in many regions worldwide, including Europe, Asia, Africa, Latin America, and Australia (http://www.cfsph.iastate.edu/Factsheets/pdfs/egg_drop_syndrome.pdf).
Except for some vaccinated animals imported from a known EDS-76–positive country in 1999, EDS-76 has not, to date, been detected in Switzerland (RK Hoop, personal communication). The causative agent, Duck adenovirus A (DAdV-A), belongs to the genus Atadenovirus within the family Adenoviridae. 8 Infection with the virus was described for the first time in 1976 (as Egg drop syndrome 1976 virus [EDSV]) in the Netherlands in a flock of laying fowls experiencing reduced egg production. 23 Serologic examinations revealed hemagglutinating adenoviruses, 15 and subsequently the virus was identified as a duck adenovirus.3,4 Disease outbreaks were demonstrated mainly in laying hens but ducks and geese are suspected to be the natural hosts of DAdV-A. 2 Serological investigations have demonstrated the presence of antibodies against DAdV-A in various bird species such as mergansers, coots, grebes, cattle egrets, herring gulls, owls, storks, swans, and pigeons.2,10,12 The virus was successfully isolated from healthy as well as from diseased ducks3,7 and is supposed to be responsible for decreased egg production and reduced egg quality in ducks. 6 Furthermore, DAdV-A has also been isolated from young goslings suffering from acute respiratory symptoms indicating that DAdV-A is also able to induce disease in geese. 11 In addition, quail were shown to become infected with DAdV-A and to develop classical signs of EDS-76. 5
The susceptibility of chickens to infection with DAdV-A is age independent. Vertical transmission from hen to egg is the most important route of infection and is followed by horizontal spread via feces within the flock, resulting in oral uptake of the virus.1,20 After experimental oral infection of chickens, DAdV-A replicates in the nasal mucosa and then spreads through viremia into the lymphoid tissue, followed by infection of the oviduct.21,24 Viral colonization of the uterine glands leads to dysfunction of eggshell generation.22,24
Detection of DAdV-A infection by classical methods is based on electron microscopy, virus isolation, and serology.9,14,17 Molecular-based techniques such as restriction enzyme analysis 19 as well as polymerase chain reaction (PCR)13,16 have become more important for the diagnosis of EDS-76. For laboratory diagnostics, quantitative PCR (qPCR) has several advantages over conventional PCR methods including a lower risk of contamination, the possibility of easier quantification, and time savings. 18 The aim of the current study was to establish a qPCR assay for the detection of DAdV-A DNA and to screen Swiss poultry flocks with a history of lowered egg production with qPCR in combination with a commercially available enzyme-linked immunosorbent assay (ELISA).
Materials and methods
Field samples
In total, 330 organ samples, as well as 208 blood samples, originating from 7 commercial layer and layer breeder flocks (A–G) were collected. Because vaccination against DAdV-A is forbidden in Switzerland, none of the investigated animals were vaccinated. The flocks showed a decreased laying performance including reduction of egg numbers and an increase in the production of small eggs, shell-less eggs, and eggs with a thin or a rough shell.
Viral strains
In order to establish the DAdV-A qPCR assay, EDSV-127 was used as the reference strain. Furthermore, viral DNA extracted from different members of Fowl adenovirus A–E (FAdV-A through -E) as well as from strain EDSV-BC14 was used to determine the specificity of the newly developed method (Table 1).
Analytical specificity of Duck adenovirus A (DAdV-A) quantitative real-time polymerase chain reaction (qPCR).*
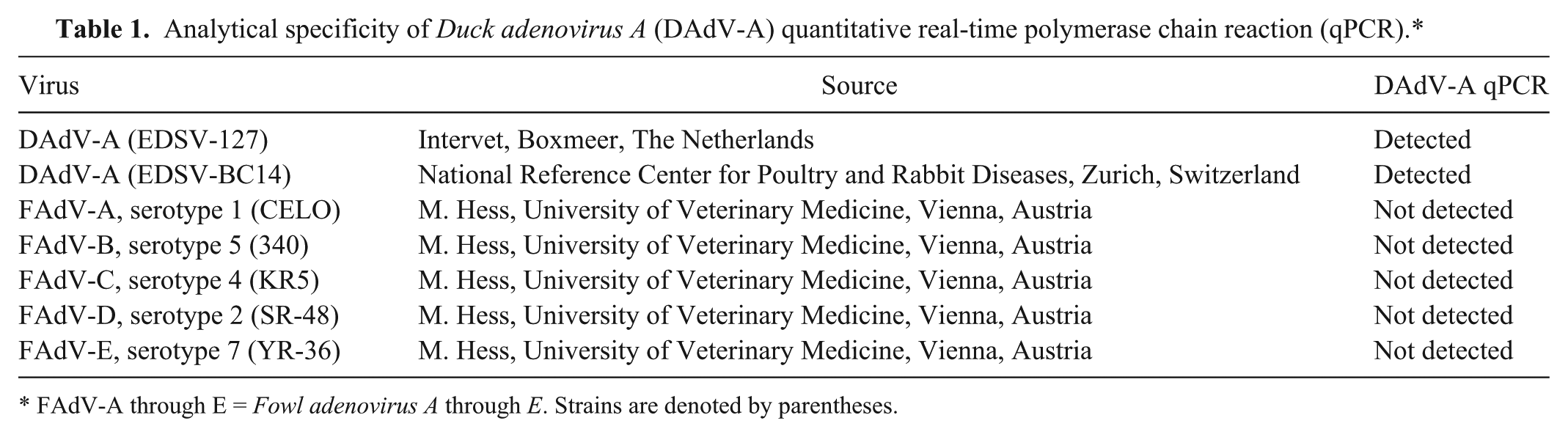
FAdV-A through E = Fowl adenovirus A through E. Strains are denoted by parentheses.
Extraction of viral DNA and quantitative real-time PCR assays
To investigate the presence of DAdV-A DNA, 330 organ samples from flocks A–G, consisting of 55 sets of organs (liver, infundibulum, magnum, trachea, kidney, and lung) were analyzed by the DAdV-A qPCR. DNA extraction from tissue samples and viruses was performed using a commercial kit a according to the manufacturer’s protocol. Primer and probe sequences for the specific detection of DAdV-A DNA were chosen within the hexon gene based on available sequence data (GenBank accession no. Y09598.1) and designed b and synthesized c (Table 2). Because the hexon gene represents one of the best characterized DAdV-A genes and most PCR assays for the detection of avian adenoviruses are designed within this region, 9 it was chosen as target sequence, resulting in an amplification fragment of 133 base pairs (bp). The DAdV-A qPCR was evaluated with strain EDSV-127.
Primer and probe sequences used in the quantitative real-time polymerase chain reaction (qPCR) assay for Duck adenovirus A (DAdV-A) and for the glyceraldehyde-3-phosphate dehydrogenase (GAPDH) gene.

For amplification of a partial copy of the glyceraldehyde-3-phosphate dehydrogenase (GAPDH) gene, used as the extraction control, primers were designed based on GenBank accession no. M11213, resulting in an amplicon with a size of 135 bp (Table 2). Diethylpyrocarbonate-treated water was used as a negative control. The reaction mixture contained 10 μl of DNA, 12.5 μl of a commercial PCR reaction mix, d 1 μl of both the forward primer and the reverse primer (final concentration 400 nM), and 0.5 μl of the probe (final concentration 200 nM). The DNA amplification used a commercial PCR system. e The thermal cycling protocol was as follows: 50°C for 2 min, 95°C for 10 min, and 40 cycles at 95°C for 15 sec and 60°C for 1 min. Results were analyzed with commercial software f using manual base line and threshold settings.
Two plasmids containing either the DAdV-A or the GAPDH amplicon were constructed using a commercial cloning kit. g The sensitivity of the DAdV-A qPCR assay was determined using ten-fold serial dilutions of the plasmid ranging from 1.35 × 1011 to 1.35 × 10−2 copies per microliter. In addition, a standard curve for the amplification of the plasmid containing the GAPDH amplicon was performed, and the GAPDH qPCR was evaluated using chicken cell culture as well as organ material.
Serology
A commercially available ELISA h for the detection of DAdV-A–specific antibodies was used according to the manufacturer’s recommendations. Briefly, sera were diluted 1:500 with sample diluent and added onto a DAdV-A antigen–coated plate. Following different incubation and washing steps, the absorbance was measured on a photometer i at 405 nm and given as optical density. Based on manufacturer’s validation results, a sample showing a sample-to-positive (S/P) ratio >0.50 was determined as positive. Samples with an S/P ratio between 0.350 and 0.499 were regarded as suspicious. A serum originating from specific pathogen–free animals and a serum containing antibodies specific to DAdV-A diluted in phosphate buffered saline containing protein stabilizers and 0.1 % sodium azide preservative were used as the negative and positive controls, respectively.
Results
Analytical specificity and sensitivity of the DAdV-A qPCR
DNA extracted from EDSV-127, EDSV-BC14, and FAdV-A through E were used to test the analytical specificity of the newly developed DAdV-A qPCR assay. DNA originating from DAdV-A strains was successfully amplified, whereas diethylpyrocarbonate-treated water as well as DNA extracted from FAdV-A through -E yielded negative results (Table 1).
Dilution experiments using plasmid copy numbers ranging from 1.35 × 105 to 1.35 × 10−1 per reaction were performed and repeated 2 times. The linear regression analysis demonstrated a linear correlation between the threshold cycle (Cq) value and the logarithm of the amount of the plasmid in the sample over a 5-log range. The coefficient of determination (R2) for the linear regression was 0.0997 (Fig. 1). In all 3 experiments using duplicate samples, the newly developed DAdV-A qPCR was able to detect 13.5 copies repeatedly. In 1 experiment, even 1.35 plasmid copies could be amplified from a duplicate whereas it was not possible to detect 0.135 copies (data not shown).
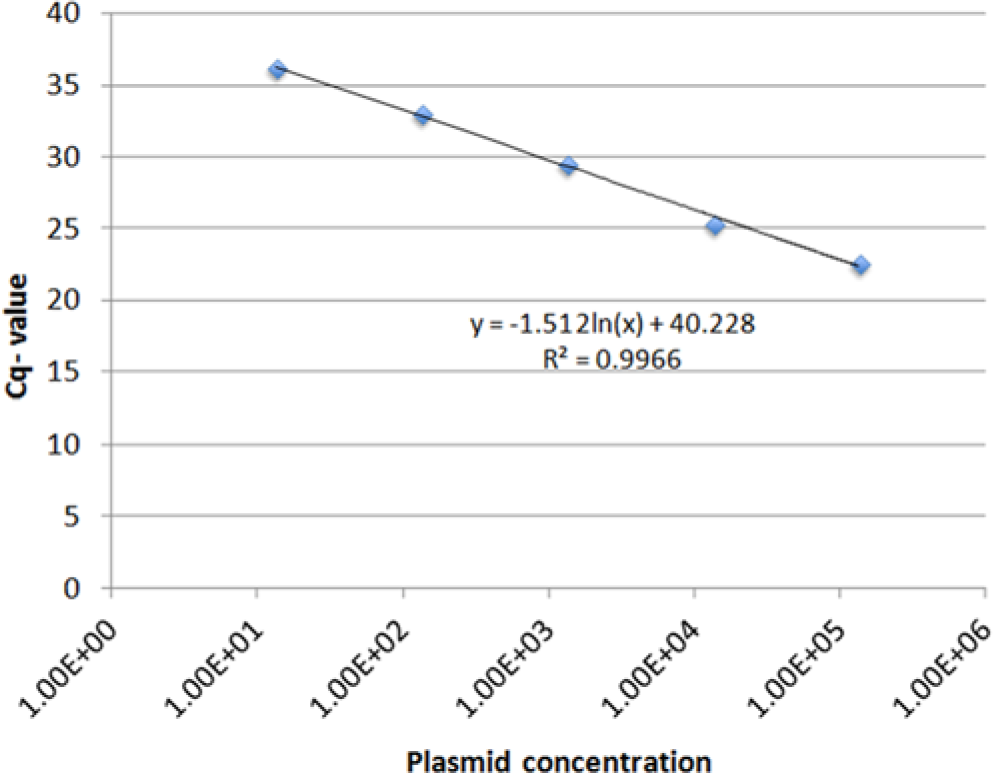
Analytical sensitivity of the Duck adenovirus A quantitative real-time polymerase chain reaction. The number of standard plasmid molecules per reaction is given on the x-axis, whereas threshold cycle (Cq) values are presented on the y-axis. The formula for the linear regression was calculated and indicated in the figure.
Detection of DAdV-A DNA in tissue samples by the DAdV-A qPCR
From all 330 organ samples, GAPDH DNA could be successfully amplified (data not shown). The DAdV-A DNA could be detected in 60 organ samples, whereas 270 samples yielded a negative result. Samples testing positive originated from 5 flocks (B, C, E, F, and G) while all samples taken from flocks A and D tested negative (Table 3). The DAdV-A qPCR successfully amplified viral DNA extracted from tissue samples originating from 3, 1, 4, 9, and 2 birds from flocks B, C, E, F, and G, respectively (Table 3). In addition, the detection of viral DNA with respect to organ localization was examined. The positive results obtained were as follows: 9 liver and trachea samples, 12 samples from each infundibulum and magnum, 14 kidney samples, and 4 lung samples (Table 3). The following Cq values were produced by the DAdV-A qPCR: 36.8–39.8 in flock B, 35 in flock C, 33.6–39.8 in flock E, 24.7–39.8 in flock F, and 38.6–39.3 in flock G. The amount of corresponding viral DNA can be estimated by comparison with Cq values obtained in the standard curve assay generated by amplification of plasmid dilutions. No standard curve was run with the diagnostic samples.
Detection of Duck adenovirus A DNA in field samples by quantitative real-time polymerase chain reaction.*
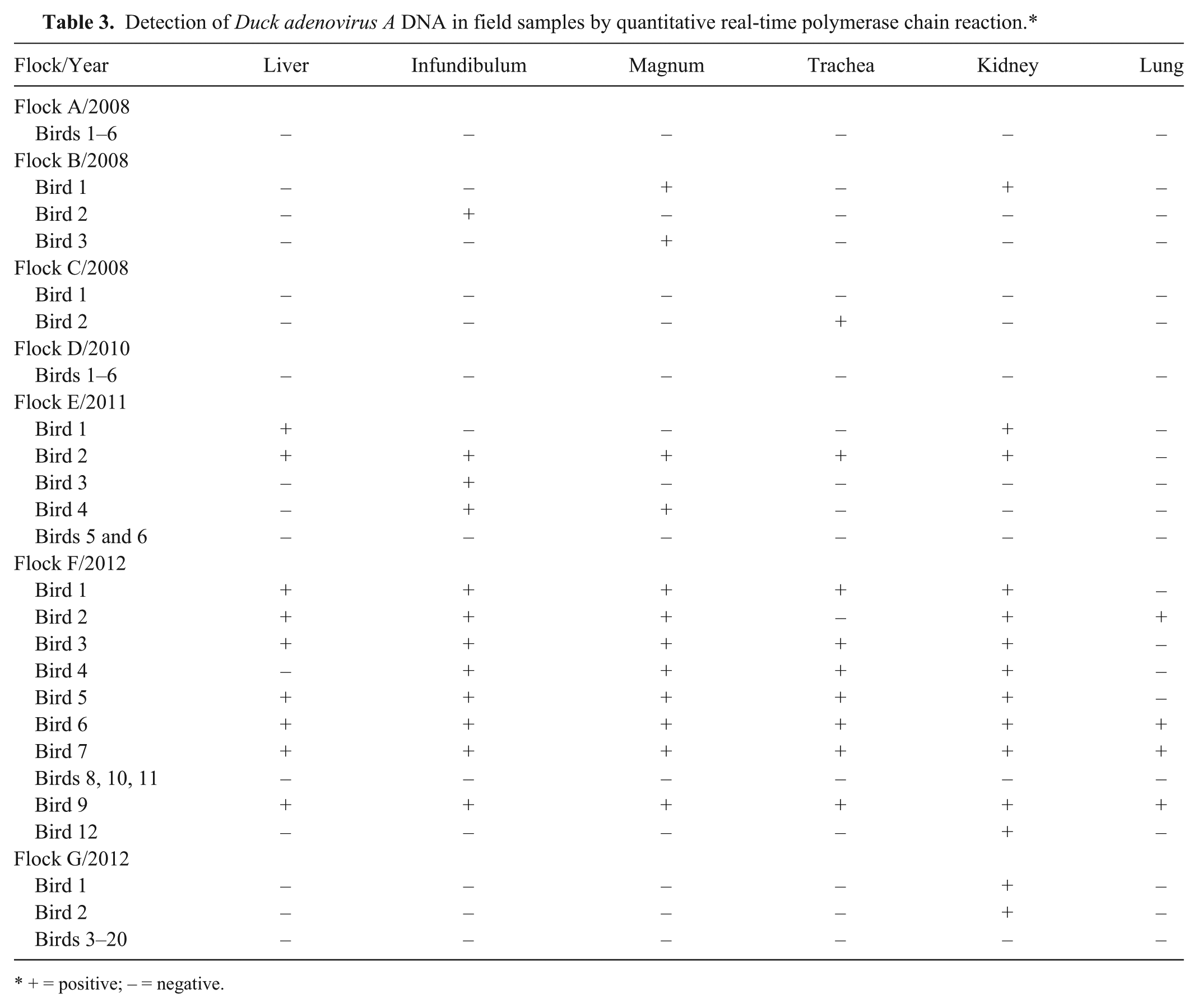
+ = positive; – = negative.
Serological analysis of blood samples
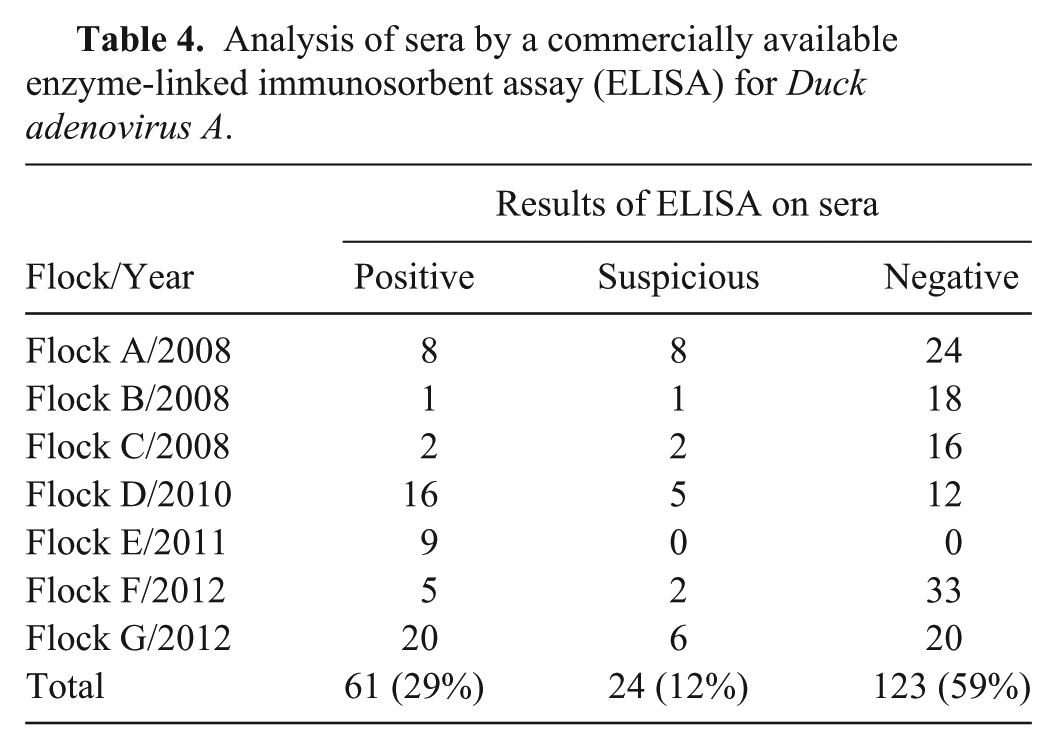
In order to determine antibody titers induced by DAdV-A, 208 sera from birds originating from flocks A–G were collected and analyzed by ELISA. A total of 61 sera were found to be positive for antibodies specific for DAdV-A, whereas 24 sera yielded suspicious and 123 yielded negative results, respectively (Table 4). The following S/P ratios were obtained by ELISA: 0.067–0.880 in flock A, 0.136–0.525 in flock B, 0.057–0.558 in flock C, 0.076–0.819 in flock D, 1.184–6.958 in flock E, 0.060–6.709 in flock F, and 0.151–3.141 in flock G.
Analysis of sera by a commercially available enzyme-linked immunosorbent assay (ELISA) for Duck adenovirus A.
Discussion
To evaluate the current situation of infections with DAdV-A in Swiss poultry flocks, blood and tissue samples were collected from 7 poultry flocks, reared in Switzerland, experiencing a drop in egg production. A newly developed DAdV-A qPCR assay and a commercially available ELISA were used to analyze the collected samples.
The successful amplification of the 2 reference strains BC14 and 127 and the fact that no signal was produced from DNA of other adenoviruses such as FAdV-A through E demonstrated the specificity of the new DAdV-A qPCR assay. A reliable sensitivity of the newly developed assay was demonstrated by a detection limit ranging between 1.35 and 13.5 copies of a plasmid containing the cloned DAdV-A fragment. Moreover, the linearity between Cq value and log of copy number persisted over a range of 1.35 × 105 to 1.35 × 101 molecules per reaction.
When samples from chickens experiencing loss in egg production were analyzed, amplification of DAdV-A DNA was achieved in samples originating from 5 out of 7 flocks. The percentage of viral detection in organ samples was as follows: kidney (25%), infundibulum and magnum (22%), liver and trachea (16%), and lung (7%). In accordance with previous findings, 1 the results indicate that DAdV-A DNA is most frequently present in kidney samples, followed by the oviduct. Because amplification of the internal control gene (GAPDH) was successful in all samples tested, negative results were assumed to not be due to an inappropriate nucleic acid extraction procedure. The Cq values obtained by the newly developed DAdV-A qPCR were >35 in most of the samples from flocks B, C, E, and G, indicating the presence of 1.35 viral copies or even less in the sample according to the standard curve. It is also worth mentioning that, in the case of such high Cq values, in most of the cases only one of the duplicates run in the assay was positive in contrast to low Cq values where both revealed a positive signal. In addition, the high Cq values obtained reflect a reaction that is no longer in the nonlinear range of the DAdV-A qPCR and should therefore be cautiously interpreted. The high Cq values could be due to the state of infection. In contrast, lower Cq values were obtained with DNA extracted from organ samples originating from flock F from which 9 out of 12 animals were tested positive by the DAdV-A qPCR. The presence of a high amount of viral DNA in combination with a majority of infected animals within a flock indicates an acute infection with DAdV-A.
In contrast to the DAdV-A qPCR–based examinations, serological analysis revealed the presence of DAdV-A–specific antibodies in all 7 flocks. In total, 29% of the sera were positive, 12% were negative, and 59% were suspicious using the commercially available ELISA. Interestingly, antibody titers found in flocks E, F, and G were higher compared with those in the other flocks. Moreover in flocks E and F, where the highest titers were found, viral DNA was detected in 4 out of 6 and 9 out of 12 animals, respectively. Thus, the simultaneous detection of viral DNA and antibodies could represent a reinfection occurring in these flocks.
In summary, the potential of a newly developed qPCR assay to detect DAdV-A DNA in clinical material was clearly demonstrated. Furthermore, the new method presented herein has a lower risk of contamination and occurrence of false-positive results as no postamplification procedures such as gel electrophoresis are needed. The presence of a DAdV-A infection in Swiss poultry flocks by the detection of antibodies against DAdV-A was shown. In addition, the newly developed DAdV-A qPCR was able to amplify viral DNA from some organs collected from animals originating from these flocks. Nevertheless, as no clearly defined positive and negative samples such as organs from experimentally infected and specific pathogen–free animals, respectively, were available, the field validation of the newly developed assay is missing. Therefore, future work is needed in order to have a completely well-established diagnostic tool at hand.
Footnotes
Acknowledgements
The authors are grateful to Corinne Nievergelt-Rutz and Regula Güttinger for collecting samples and to Kurt Tobler for critically reading the manuscript.
a.
DNAeasy blood and tissue kit, Qiagen GmbH, Hilden, Germany.
b.
wEmboss application, Swiss Institute of Bioinformatics, Lausanne, Switzerland.
c.
Microsynth AG, Balgach, Switzerland.
d.
2× TaqMan Universal PCR Master Mix, Applied Biosystems, Foster City, CA.
e.
7500 Fast real-time PCR system, Applied Biosystems, Foster City, CA.
f.
7500 Fast system SDS software version 1.4.0, Applied Biosystems, Foster City, CA.
g.
TOPO TA cloning kit, Invitrogen Corp., Carlsbad, CA.
h.
Egg drop syndrome antibody test kit, BioChek, Smart Veterinary Diagnostics, ER Reeuwjik, The Netherlands.
i.
Thermo Scientific Multiskan EX, Thermo Fisher Scientific Inc., Waltham, MA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
