Abstract
To aid in the rapid diagnosis of myxomatosis in rabbits, a real-time polymerase chain reaction (PCR) for the specific detection of Myxoma virus is described. Primers and probe were designed to amplify a 147-bp fragment within the Serp2 gene. The assay was able to detect 23 copies of a synthesized oligo indicating a reliable sensitivity. In addition, the real-time PCR did not detect the Rabbit fibroma virus used in myxomatosis vaccines. The novel PCR was shown to be able to detect Myxoma virus in fresh and paraffin-embedded rabbit tissues originating from myxomatosis cases from various regions in Switzerland.
Myxoma virus (MYXV; family Poxviridae, subfamily Chordopoxvirinae, genus Leporipoxvirus) 1 is a linear double-stranded DNA virus that specifically infects rabbits and hares. If introduced in European rabbits (Oryctolagus cuniculus), the virus causes the mostly fatal disease myxomatosis, named after the prominent mucinous skin lesions. 3 Affected rabbits display conjunctivitis, anorexia, listlessness, and fever. Animals may die within 2 days after onset of symptoms. In a more prolonged disease course, depression and myxomas in the eyelids, nose, lips, ears, vulva, or scrotum are observed. The edematous ears become heavy and droop. Animals usually die within 1–2 weeks due to emaciation and dyspnea. 4 In the course of an outbreak, the virulence is reduced, 6 and the disease is characterized by reduced mortality in young animals and seropositivity of all adult rabbits. 2 The European rabbit is highly susceptible, a fact that has been exploited in the deliberate release of MYXV strain Moses in Australia (1950) and strain Lausanne in Europe (1952) to control feral rabbits.5,10 Both strains were originally isolated in Brazil.4,10 Contrary to the fatal disease in European rabbits, MYXV only produces local fibromas in its natural host, the wild American cottontail rabbits (Sylvilagus spp.).3,10 As seen in the Australian outbreak in 1950, the virus is efficiently transmitted over large distances by blood-sucking vectors, such as mosquitoes and rabbit fleas (Spilopsyllus cuniculi). 5
In Switzerland, myxomatosis is a notifiable disease, and vaccination is strictly forbidden. 11 A total of 68 cases were reported to the Swiss Federal Veterinary Office from 1991 to 2010 (http://www.infosm.bvet.admin.ch; date of accession March 7, 2011). The diagnosis of myxomatosis is based on clinical presentation of typical symptoms and histology. In order to have a faster diagnostic tool, a quantitative real-time PCR (qPCR) was developed and validated, as described in the current study.
Primers for the MYXV qPCR were designed with the Primer3 program 9 to amplify a 147-bp fragment within the serpin (Serp2) gene (accession no. U60474). 8 Viral DNA was extracted from all viral strains, swabs, and fresh and paraffin-embedded tissues samples using a commercial kit a following the manufacturer’s protocols. In addition, after lysis of fresh and paraffin-embedded tissue, a homogenizer column b was used. Quantitative PCR assays were performed on a thermocycler c with the standard cycle protocol of 2 min at 50°C, 10 min at 95°C, followed by 40 cycles of denaturation at 95°C for 15 sec and annealing at 60°C for 1 min. The MYXV PCR was carried out in 25-µl reactions containing (final concentrations) 12.5 µl of commercial PCR reaction mixture, d 900 nM of each primer (Serp2_For: 5’-GTCGGACGTCTTCGTTTCTC-3’; Serp2_Rev: 5’-ACTCTGGATGCGACGGTTAC-3’), e 250 nM of probe Serp2_Probe (5’-FAM-CGCGCAATTACGTCTC GCCC-TAMRA-3’), e and 7 µl of extracted DNA.
Analytical sensitivity was determined using the standard curve approach. An oligo with a length of 147 bp corresponding to the amplicon size was synthesized e and designated Serp2_Oligo. For analytical specificity, the following virus strains of the genus Leporipoxvirus were used: MYXV strain Munich 1, f MYXV isolate Hasi 1998, g and Rabbit fibroma virus contained in a myxomatosis vaccine. h Furthermore, the Rabbitpox virus strain CAM.-klon.2/87 f was tested. In addition, Pasteurella multocida subsp. multocida i and Bordetella bronchiseptica i were tested.
The PCR was tested on paraffin-embedded organ samples from farm, pet, and wild rabbits as well as fresh swab and tissue samples of 4 rabbits j (Table 1). Further, nasal swabs from healthy Swiss pedigree rabbits of 3 Swiss breeds (Schweizer Fuchs, Schweizer Dreifarben-Kleinschecke, and Schweizer Feh) were taken. k The animals originated from 3 different regions in Switzerland and had not been vaccinated. A total of 12 rabbits from each region (comprised of 3 rabbits of each breed) were tested with the new myxomatosis qPCR.
Real-time Myxoma virus polymerase chain reaction (PCR) assessment of swabs and fresh and paraffin-embedded tissue samples from cases of myxomatosis in pet, pedigree, farm, and wild rabbits in Switzerland from 1999 to 2010.
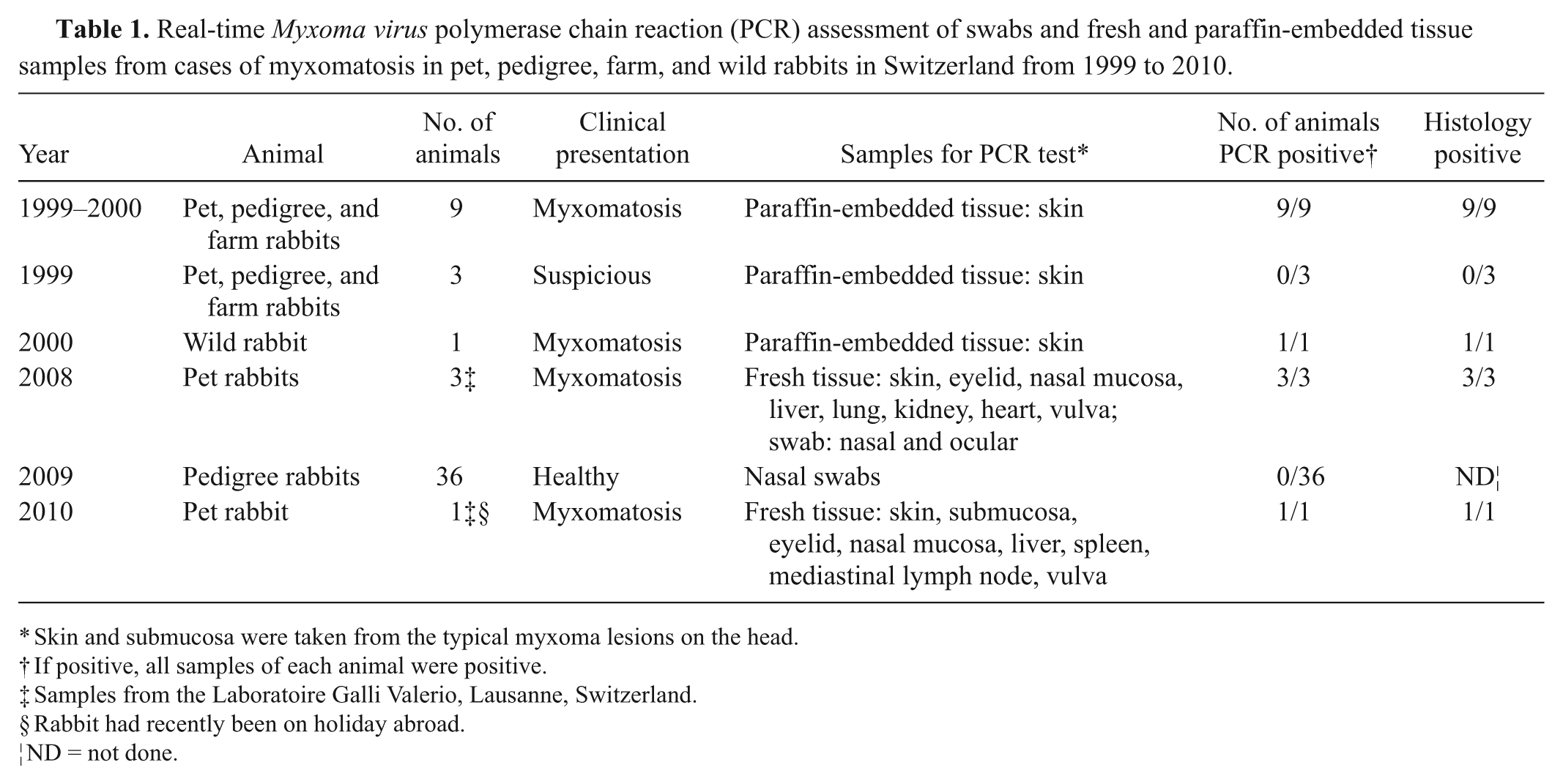
Skin and submucosa were taken from the typical myxoma lesions on the head.
If positive, all samples of each animal were positive.
Samples from the Laboratoire Galli Valerio, Lausanne, Switzerland.
Rabbit had recently been on holiday abroad.
ND = not done.
The analytical sensitivity of the MYXV qPCR was determined using 10-fold dilution series of the Serp2_Oligo. The assay was able to detect 230 copies in 100% and 23 copies in 60% of experiments. Linearity persisted over a range of 10 7 –10 3 copies. The coefficient of determination for the linear regression was 0.9977 (data not shown). The MYXV qPCR was able to detect the 2 MYXV strains tested, while no signal was produced with Rabbit fibroma virus or with Rabbitpox virus. Pasteurella multocida subsp. multocida and B. bronchiseptica were negative.
Swiss field cases of myxomatosis in farm and pet rabbits, as well as in 1 wild rabbit, occurring throughout the country within the last decade were used to test the qPCR in a diagnostic procedure. All of these cases had been confirmed by histology, and all tested positive by qPCR. Three suspicious cases, which were negative in histology, also proved negative by qPCR (Table 1). As well, 36 selected nasal swabs of pedigree rabbits were subjected to the MYXV qPCR, and all of them tested negative (Table 1).
The current study shows that the qPCR is able to detect myxomatosis cases in farm, pet, and wild rabbits using swabs and fresh or paraffin-embedded tissues from the last 10 years. Although myxomatosis is infrequently diagnosed in Switzerland (68 cases from 1991 to 2010), spillover from wild rabbit populations and importation of rabbits can provoke new outbreaks. 11 The qPCR developed in the present study simplifies the diagnosis of myxomatosis in clinical cases as well as from fresh or paraffin-embedded tissue samples. Suspicious cases can be quickly affirmed by taking a nasal, ocular, or genital swab. This is also important in the chronic form of myxomatosis, which may not present typical lesions and may be inconclusive in histology.7,11 Furthermore, protective measures for flocks in the vicinity of a disease outbreak can be implemented very quickly, such as insect control to prevent spreading by vectors and restriction of animal movements. As the test works well in paraffin-embedded tissue, myxomatosis can also be diagnosed retrospectively from archived samples. Thus, the newly developed assay will be helpful in generating a fast diagnosis for a notifiable disease.
Footnotes
Acknowledgements
The authors are grateful to the staff of the Department of Poultry Diseases and the Institute of Veterinary Pathology, especially R. Weilenmann and B. Senn for additional technical support.Dr. A. S. Waldvogel, Laboratoire Galli Valerio, Lausanne, Switzerland is thanked for providing myxomatosis cases. Furthermore, the authors acknowledge Prof. Dr. C.P. Czerny from the Bavarian Animal Health Service, Poing, Germany, and the Friedrich-Loeffler-Institut, Federal Research Institute for Animal Health, Greifswald-Insel Riems, Germany for providing virus strains. The authors are indebted to the association “Kleintiere Schweiz,” Zofingen for organizing the rabbit sampling, to the rabbit owners, and to the team of the Animal Genetics Group, Swiss Federal Institute of Technology, Zurich, the Institute of Laboratory Animal Science, University of Zurich, and to L. Konrad of the National Reference Center for Poultry and Rabbit Diseases, University of Zurich.
Notes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
