Abstract
Avian bornavirus (ABV), the cause of proventricular dilation disease in psittacine birds, has been detected in multiple tissues of infected birds using immunohistochemical staining (IHC) and reverse transcription polymerase chain reaction (RT-PCR). In the current study, real-time RT-PCR, using primers targeting the ABV matrix gene, was used to detect ABV in 146 tissues from 7 ABV-infected psittacine birds. Eighty-six percent of the samples tested positive, with crossing point values ranging from 13.82 to 37.82 and a mean of 22.3. These results were compared to the findings of a previous study using gel-based RT-PCR and IHC on the same samples. The agreement between the 2 RT-PCR techniques was 91%; when tests disagreed it was because samples were negative using gel-based RT-PCR but positive on real-time RT-PCR. Agreement with IHC was 77%; 16 out of 74 samples were negative using IHC but positive on real-time RT-PCR. The results suggest that real-time RT-PCR is a more sensitive technique than gel-based RT-PCR and IHC to detect ABV in tissues. The tissues that were ranked most frequently as having a high amount of viral RNA were proventriculus, kidney, colon, cerebrum, and cerebellum. Skeletal muscle, on the other hand, was found to have a consistently low amount of viral RNA.
Keywords
Proventricular dilation disease (PDD) is a progressive neurologic disease of birds with a high case fatality rate once clinical signs are present. 6 Although the vast majority of cases of PDD occur in psittacine birds, consistent clinical and pathological features have been described in several other avian orders.4,15 In nearly all clinical cases, birds present with neurologic or gastrointestinal signs or a combination of both.6,11 Characteristic histological lesions include lymphoplasmacytic infiltrates in the autonomic nerves, particularly of the gastrointestinal tract, in the central nervous system, and in the peripheral nerves. 16
Avian bornavirus (ABV) was proposed as the causative agent of PDD in 2008,10,12 and experimental infection trials and investigations of outbreaks provided strong supporting evidence.7,9,11 Since its initial recognition in the United States, ABV has been reported worldwide 18 and poses a significant threat to the captive breeding of endangered psittacine species (Enderlein D, Piepenbring A, Herzog S, et al.: 2011, The situation of ABV in endangered psittacines like the Spix’s macaw. In: Proceedings of the 11th European Association of Avian Veterinarians Conference, ed. Samour J, pp. 228–229).
The diagnosis of PDD has traditionally been based on the identification of histological lesions in biopsy or necropsy tissues.6,16 However, with the discovery of the association of ABV with PDD, diagnostic testing has focused on reverse transcription polymerase chain reaction (RT-PCR) detection of the virus. 6 Both gel-based RT-PCR (gbRT-PCR) and real-time RT-PCR have been successfully used to detect ABV in tissues and swabs.6,11,16 Primers have been designed to target the nucleocapsid N gene (ABVN), matrix M gene (ABVM), phosphoprotein P gene, and polymerase L gene. Assays for the M and N genes appear to have a similar high sensitivity, whereas RT-PCR for L and P genes seems generally less accurate.6,16 While the ABVN gene is the more highly expressed region of the genome, the ABVM gene is the more highly conserved region. 12
It appears that ABV is widely distributed in the body of infected birds, and the virus has been identified in many tissues using in situ hybridization, 19 immunohistochemical staining (IHC),16,18,20 gbRT-PCR,13,16,17 and real-time RT-PCR.10,14,17 The use of real-time RT-PCR has been documented in only a small number of tissues from a small number of birds, and has been used to describe viral distribution rather than viral load. 17 The objective of the current study is to describe the use of real-time RT-PCR to estimate viral load in various tissues from ABV-infected psittacine birds, and to compare these findings to the results of gbRT-PCR and IHC on the same samples. Viral load was estimated by using crossing point (Cp) as a simple measure of viral nucleic acid in the sample to facilitate comparison of viral RNA in various tissues, and not to determine an absolute number of viral particles in a sample.
The 7 parrots used in the present study were part of a research colony of 16 birds that was created to study fecal viral shedding in psittacine birds with PDD. All birds in the colony had a history of exposure to birds with confirmed PDD. The birds in the colony were euthanized and necropsied; 3 sets of tissue samples were collected, with precautions taken to reduce chances of cross-contamination among birds and tissues. The first set of samples was evaluated for the presence of ABV by gbRT-PCR detection of the ABVM and ABVN genes. The second set of samples was placed in 10% neutral buffered formalin and subjected to histologic examination and IHC using rabbit polyclonal antiserum raised against the ABVN protein. a The third set of tissues was immediately frozen in liquid nitrogen and stored at −70°C. The results of evaluation of the first and second samples from all 16 birds have been published. 16
Seven of the 16 previously evaluated birds for whom samples were available were selected on the basis of their having histologic lesions of PDD and being ABV positive. 16 Birds 1–3 and 7 had a history of neurologic signs within the year preceding euthanasia. None of the birds showed or had a history of clinical signs of gastrointestinal disease, and all had maintained good body condition. For the current study, the third set of tissues collected from the 7 birds was evaluated for the presence of ABV by real-time RT-PCR detection of the ABVM gene at the Animal Health Laboratory, University of Guelph, Guelph, Ontario, Canada. Samples were weighed, and 10% weight/volume tissue suspensions in sterile tryptose phosphate broth b were prepared and homogenized. Total nucleic acids were extracted from 50-µl aliquots of the tissue suspensions using a total RNA isolation kit c in an automated magnetic particle processor. d The real-time RT-PCR assay was a duplex test with 2 sets of primers and hydrolysis probes targeting both psittacine ABVM and geese ABVM gene sequences. The amplification was carried out in 25-µl reactions in a 96-plate–based real-time PCR platform e using a one-step RT-PCR kit. f A detailed description of the RNA extraction, real-time RT-PCR primers used, and cycling parameters has been provided previously. 4 Crossing point values less than 37.00 were reported as positive, between 37.00 and 39.99 as inconclusive, and equal to or greater than 40.00 as negative. For comparisons among different techniques (real-time RT-PCR vs. gbRT-PCR, and real-time RT-PCR vs. IHC) and for calculations of the average Cp per tissue and per bird, the inconclusive real-time RT-PCR results were considered negative.
Table 1 presents the previously published results of IHC and gbRT-PCR testing on these 7 birds, as well as the results of the current real-time RT-PCR test, with Cp values for up to 22 tissues in each bird. The identification numbers for the birds are the same as those used in the previous publication. 16 A summary of the agreements between gbRT-PCR and real-time RT-PCR and between IHC and real-time RT-PCR is presented in Table 2. The McNemar test, performed using commercial software, g indicated significant differences among the proportions of positive results (2-tailed P value = 0.0009 for real-time RT-PCR vs. gbRT-PCR, 2-tailed P value = 0.0007 for real-time RT-PCR vs. IHC). This means that the Cohen kappa test is not appropriate to measure agreement between the tests.2,5 Test agreements were thus estimated using overall percentage agreement, positive percentage agreement, and negative percentage agreement.
Results of real-time reverse transcription polymerase chain reaction (R), gel-based reverse transcription polymerase chain reaction (G), and immunohistochemical staining (I) on tissues collected from seven psittacine birds infected with Avian bornavirus.*
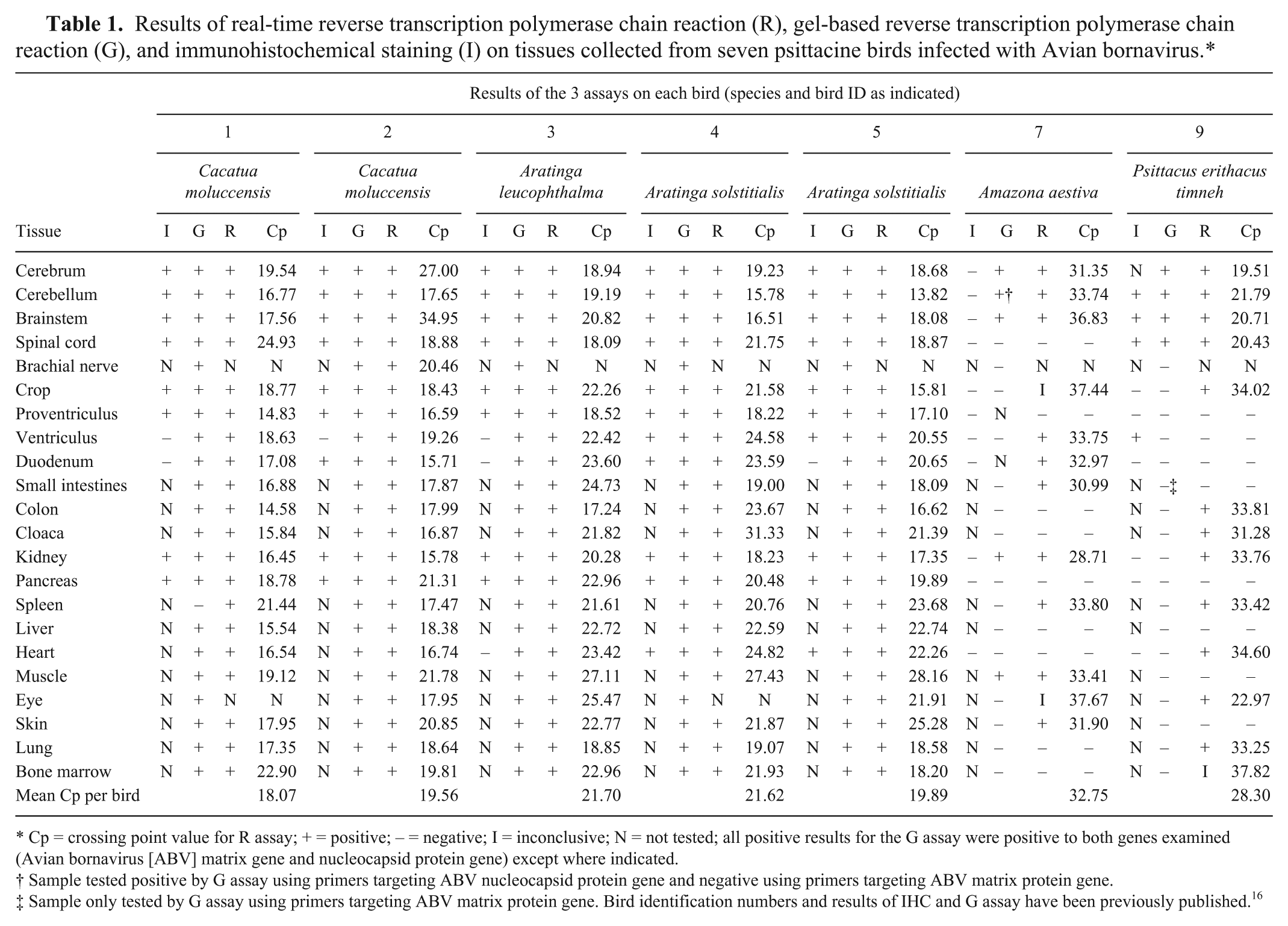
Cp = crossing point value for R assay; + = positive; – = negative; I = inconclusive; N = not tested; all positive results for the G assay were positive to both genes examined (Avian bornavirus [ABV] matrix gene and nucleocapsid protein gene) except where indicated.
Sample tested positive by G assay using primers targeting ABV nucleocapsid protein gene and negative using primers targeting ABV matrix protein gene.
Sample only tested by G assay using primers targeting ABV matrix protein gene. Bird identification numbers and results of IHC and G assay have been previously published. 16
Summary of overall, positive, and negative percentage agreements among diagnostic tests in psittacine birds infected with Avian bornavirus.*
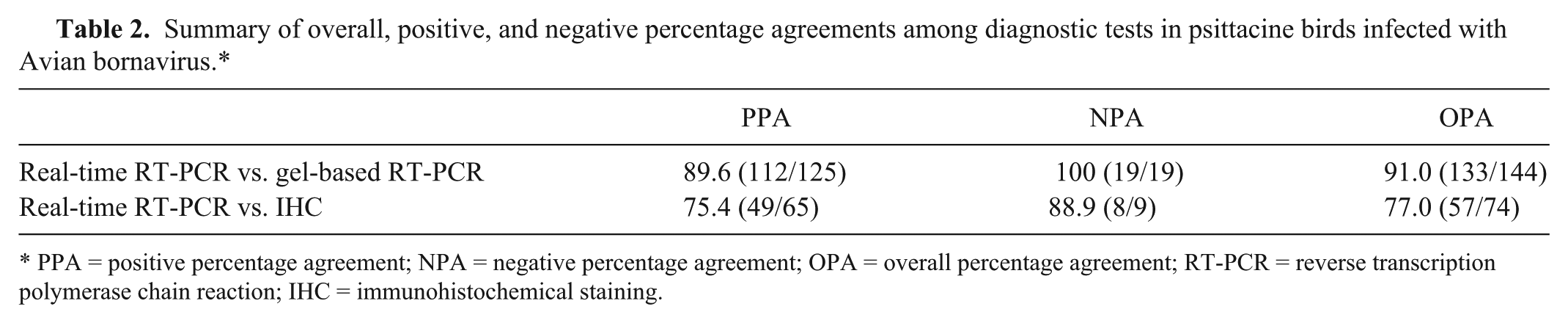
PPA = positive percentage agreement; NPA = negative percentage agreement; OPA = overall percentage agreement; RT-PCR = reverse transcription polymerase chain reaction; IHC = immunohistochemical staining.
Out of 144 samples for which both gbRT-PCR and real-time RT-PCR were performed, only 13 discrepancies were noticed, resulting in a 91.0% overall percentage agreement between the 2 tests. Where there was non-agreement, samples were negative using gbRT-PCR but positive on real-time RT-PCR. Twelve of the 13 samples where testing was not in agreement originated from the 2 birds with the lowest overall viral load (highest mean Cp for all tissues; birds 7 and 9); 11 of the 13 samples had a Cp higher than 30 (low viral load).
Comparing the results of IHC and real-time RT-PCR (Fig. 1), there was agreement in 57 out of 74 pairings (77.0% overall percentage agreement). In 16 of the 17 samples where testing was not in agreement, samples were negative using IHC but positive on real-time RT-PCR. Nine of these 16 samples again originated from birds 7 and 9, and had a Cp higher than 30. Ten of the samples with non-agreement were tissues of gastrointestinal origin, particularly ventriculus and duodenum.
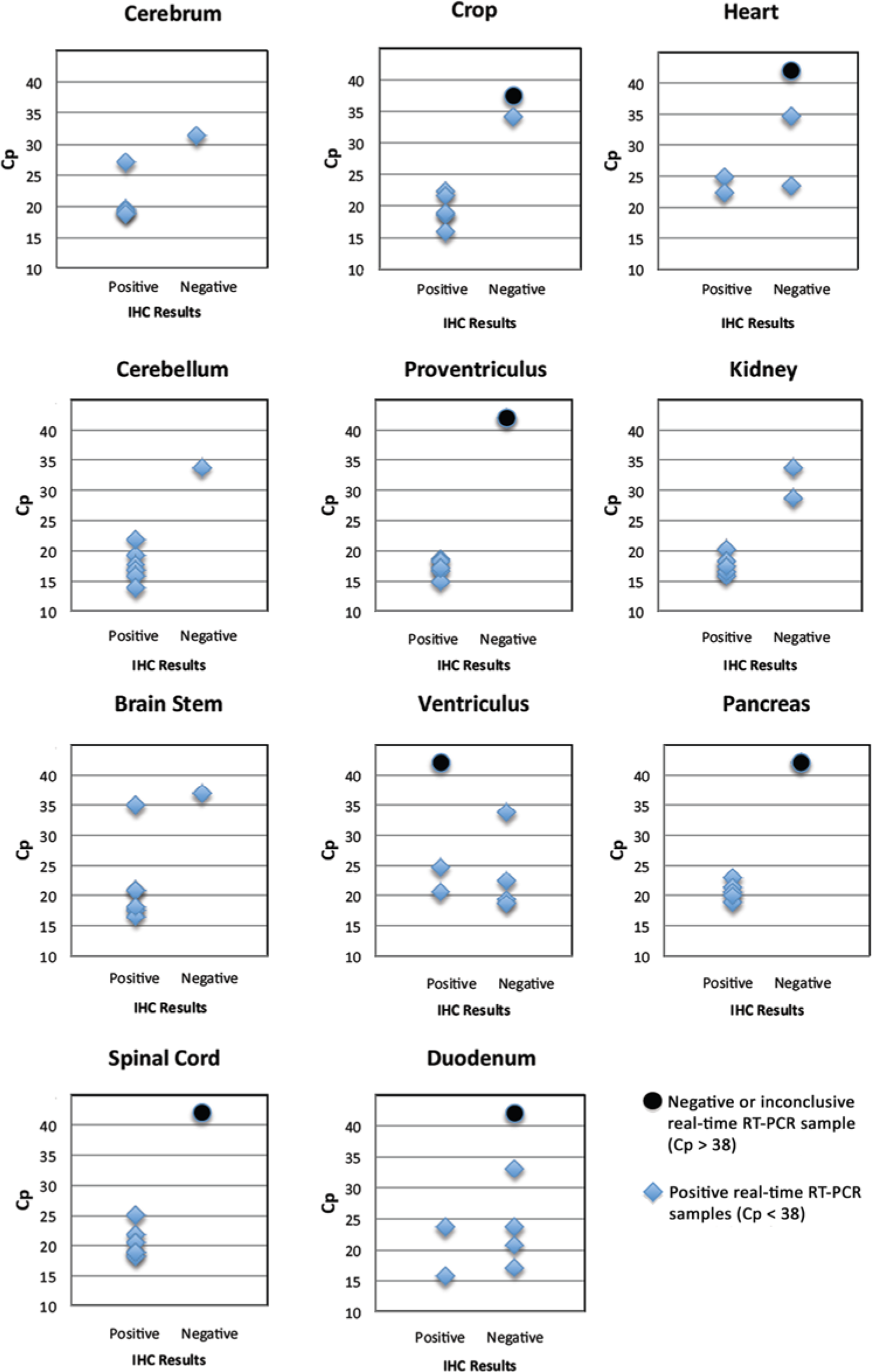
Test agreement between real-time reverse transcription polymerase chain reaction results (real-time RT-PCR), expressed as crossing point (Cp) values and immunohistochemical staining (IHC) on various tissues from parrots infected with Avian bornavirus.
The mean Cp per tissue and the probability of each tissue being among the 5 tissues with the highest and the lowest viral loads in each bird are presented in Table 3. Eighty-six percent of the samples (126/146) tested positive, with Cp values ranging from 13.82 to 36.83 and an overall mean of 21.9. The mean Cp for all tissues from a specific bird ranged from 18.07 (bird 1) to 32.75 (bird 7). The 20 negative samples originated from birds 7 and 9, which had the lowest overall viral loads (highest mean Cp for all positive tissues). The proventriculus, although positive in only 5 out of 7 birds, was the tissue with the lowest mean Cp (17.05) and was among the 5 tissues with the highest viral load (lowest Cp) in each of the 5 birds positive on this tissue. Other tissues most consistently included in the group with the highest viral load (lowest Cp) were kidney (5/7), colon (3/6), cerebrum (3/7), and cerebellum (3/7). Skeletal muscle contained the least virus (highest Cp; mean Cp = 26.17, n = 6) and tested negative or was included in the 5 tissues with the lowest viral load (highest Cp) in 6 out of 7 birds.
Mean crossing point (Cp) values of real-time reverse transcription polymerase chain reaction per tissue and probability of each tissue being included in the five tissues with the highest or lowest viral load in seven psittacine birds infected with Avian bornavirus.*
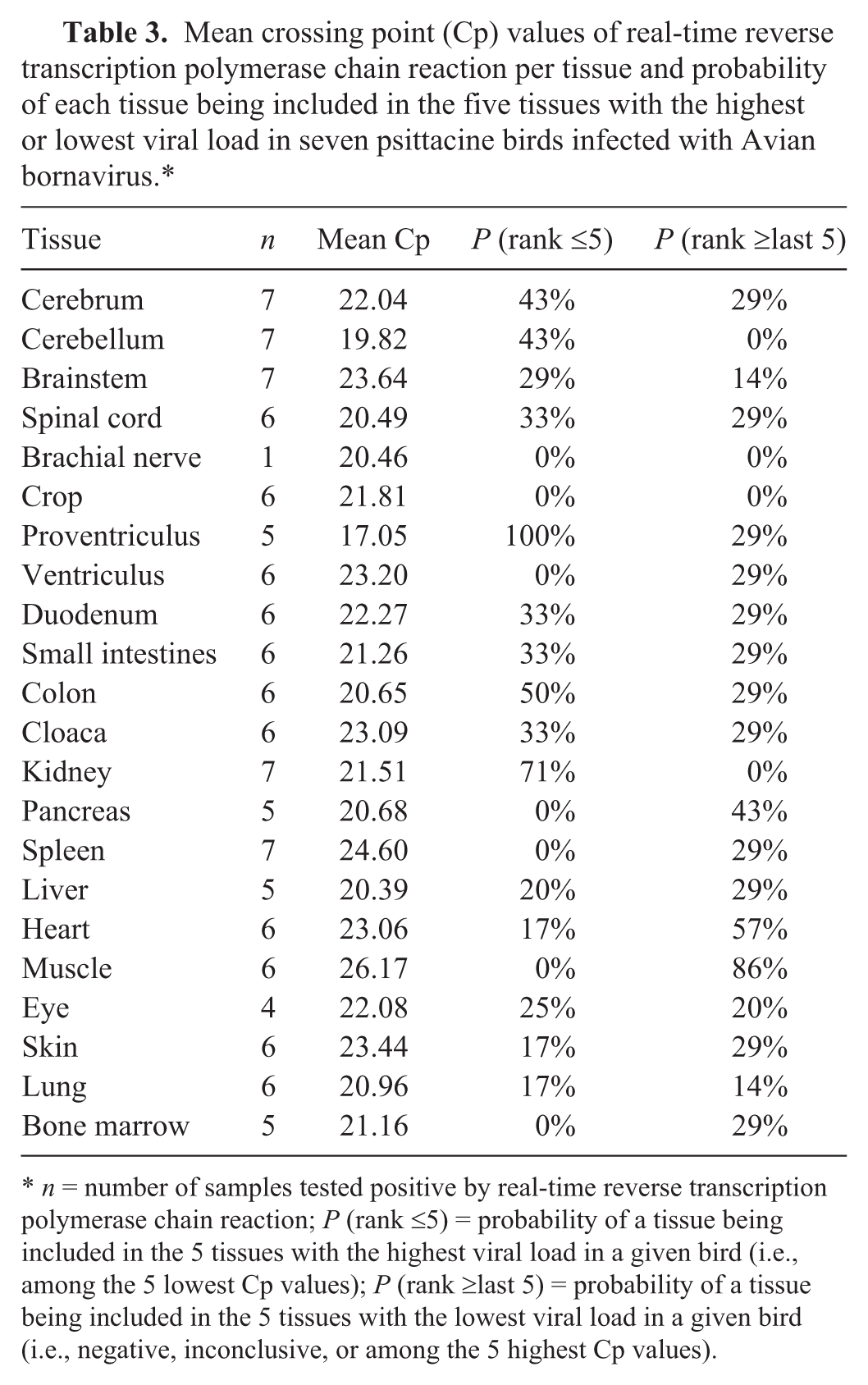
n = number of samples tested positive by real-time reverse transcription polymerase chain reaction; P (rank ≤5) = probability of a tissue being included in the 5 tissues with the highest viral load in a given bird (i.e., among the 5 lowest Cp values); P (rank ≥last 5) = probability of a tissue being included in the 5 tissues with the lowest viral load in a given bird (i.e., negative, inconclusive, or among the 5 highest Cp values).
The results of the current investigation confirm that real-time RT-PCR for the ABVM gene can be used to identify ABV-infected psittacine birds, and that widespread distribution of virus in infected birds occurs even in birds with a low viral load. As the pathogenesis of PDD as a clinical entity is still poorly understood, the relationship between the presence of ABV in tissues, especially in birds with low viral load, and clinical and pathological findings is yet to be determined.
The results presented herein suggest that real-time RT-PCR has greater sensitivity than gbRT-PCR. In 11 of the 13 samples determined to be positive by real-time RT-PCR and negative by gbRT-PCR, the Cp values were above 30, indicating low viral load. This cannot, however, be considered definitive as the extracts used for gbRT-PCR were no longer available for real-time RT-PCR assessment. While it was hypothesized that viral distribution was uniform between the samples of tissue used to prepare the 2 extracts, this cannot be proven. This hypothesis also does not likely apply to certain tissues whose composition is not uniform, and may explain the few examples of disagreement between the 2 RT-PCR techniques in birds with relatively high viral loads (low Cp values). For example, it is quite conceivable that anatomically dissimilar sections of eye were present in the 2 samples, one perhaps containing more nervous system tissue than the other. If this apparent higher sensitivity of real-time RT-PCR is borne out by future research, its use will allow for more accurate diagnosis of ABV infection and reduction of the occurrence of false-negative results.
The sensitivity of real-time RT-PCR also appeared to be greater than that of IHC. Of the 16 tissue samples that were negative on IHC but positive by real-time RT-PCR, 8 had low viral load, with Cp values greater than 30. For the 3 samples of ventriculus and 3 samples of duodenum for which IHC was negative and real-time RT-PCR had a Cp less than 30, it is possible that virus in gastric or intestinal contents was amplified by RT-PCR, but could not be identified on IHC. One sample of ventriculus was positive by IHC but negative with both RT-PCR techniques. This could have been a false positive by IHC, or perhaps the results relate to inconsistent sample division. As PCR testing was performed on frozen tissues rather than on scrolls of paraffin-embedded tissue, it is physically impossible to use the same samples for both IHC and RT-PCR.
While the tissue distribution of ABV is broad, some tissues appear to contain a greater amount of virus and thus would be better for diagnostic sampling at necropsy. A system of ranking tissues by Cp for each bird was used to determine this, and results suggested proventriculus, kidney, colon, cerebrum, and cerebellum would be the best samples for diagnosis by RT-PCR. The inclusion of proventriculus, colon, and kidney supports the prevailing thought that ABV may be shed via feces and urine.7,8,16 In the current study, Cp values were used to compare relative amounts of virus among and within birds and tissues, without providing quantitative assessments of the number of viral particles present. The determination of specific viral amounts in international units/ml, as is carried out for certain human diseases, 1 would require the use of standardized methods and laboratory controls that are not available for ABV.
The current investigation was not intended to assist with antemortem diagnosis of ABV infection, but does provide some relevant information. The identification of nonsuppurative ganglioneuritis in a crop biopsy has been the standard method to diagnose PDD antemortem, and the crop remains the most commonly sampled tissue in clinical situations. 6 However, the presence of inflammatory cells in the autonomic nervous tissue associated with the crop is inconsistent among PDD cases, and numerous technical issues can affect the quality of samples obtained. These factors explain the variable and sometimes low sensitivity reported for this diagnostic tool; ranging from 22% to 76%. 6 In the present study, the crop was positive for ABV in 5 out of 7 birds by IHC and both RT-PCR techniques, with Cp values less than 23. The crop from the remaining 2 birds was negative by IHC and gbRT-PCR, and was positive in 1 sample by real-time RT-PCR with a Cp of 34.02, and inconclusive in the second. This suggests that the use of IHC or real-time RT-PCR on crop tissue can enhance the sensitivity of crop biopsy as an antemortem diagnostic tool for ABV infection in psittacine birds.
Based on results from the current study, clinicians should consider bone marrow as an accessible antemortem sample for RT-PCR diagnosis of ABV infection. In 5 out of 7 birds evaluated, bone marrow collected at necropsy from the tibiotarsus was positive by both RT-PCR techniques, and the mean Cp for these 5 birds was 21.16. Another antemortem sample that can be easily obtained from a parrot, regardless of its size, is a skin biopsy. Skin samples were positive by real-time RT-PCR in 6 out of 7 birds, with a mean Cp of 23.44, including the bird with the lowest mean viral load (bird 7). A 2011 publication suggested the use of gbRT-PCR on feather calami to diagnose ABV infection in psittacine birds. 3 As feathers were not assessed in the current study, the authors cannot comment on viral load, but it appears reasonable that real-time RT-PCR assessment might also be a more sensitive test for this application.
In conclusion, the present study has compared the results of real-time RT-PCR, gbRT-PCR, and IHC using a large selection of tissues collected from psittacine birds infected with ABV. While overall agreement among tests was high, real-time RT-PCR appears more sensitive than the other 2 techniques in detecting virus in tissues with low viral load, and thus reducing the number of false-negative diagnostic results.
Footnotes
Acknowledgements
The authors gratefully acknowledge Nicole Zaranek, Sarah Hoyland, Jane Coventry, and Elizabeth Hillyer for their essential technical assistance in carrying out the real-time RT-PCR testing of archived samples, as well as all those involved in the initial study from which the samples originated.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Financial support was provided by the Animal Health Laboratory, University of Guelph. A graduate stipend for P. Delnatte was provided by the Toronto Zoo.
a.
ABV nucleocapsid protein provided by deRisi Laboratory, University of California, San Francisco, CA.
b.
Tryptose Phosphate Broth, Difco, Detroit, MI.
c.
MagMAXTM-96 kit, Applied Biosystems Inc., Foster City, CA.
d.
MagMAX Express-96 magnetic particle processor, Applied Biosystems Inc., Foster City, CA.
e.
LightCycler 480 real-time PCR system, Roche Diagnostics, Laval, Quebec, Canada.
f.
AgPath-ID one-step RT-PCR kit, Applied Biosystems Inc., Foster City, CA.
g.
QuickCalcs, GraphPad Software Inc., La Jolla, CA.
