Abstract
A case of proventricular dilatation disease is described in a scarlet macaw(Ara macao) from clinical presentation to diagnosis with molecular methods. The initial clinical signs were depression progressing to head pressing over several days. A leukocytosis with toxic heterophil changes, hypoalbuminemia, and increased serum activity of aspartate aminotransferase and creatine kinase were present. Lead and zinc assays were within reference ranges, and results from Chlamydophila and polyomavirus testing were negative. Contrast-enhanced fluoroscopy revealed normal gastrointestinal transit times and motility as well as the presence of 2 small metallic foreign bodies in the ventriculus. The macaw was treated with antimicrobials, analgesics, vitamins E and B complex, force-feeding, and fluid administration with little improvement. Euthanasia was elected, and histologic examination of brain tissue revealed a perivascular lymphoplasmacytic infiltration, while the lungs had evidence of a fungal pneumonia. Tissue samples from the brain and proventriculus tested positive for the presence of Avian bornavirus genotype 2, while serology confirmed Avian bornavirus infection.
Neuropathic gastric dilation or proventricular dilatation disease (PDD) has been variably described in the veterinary literature, and was initially referred to as macaw wasting syndrome in the early 1970s. 9 The terms “neuropathic gastric dilatation” and “encephalomyelitis” are in reference to the most commonly observed clinical and pathologic features of PDD. Historically, affected birds present with varying degrees of gastrointestinal signs, including anorexia, regurgitation, or presence of undigested seeds in feces, with or without neurologic signs (e.g., ataxia or obtundation). Frequently, the clinical signs are nonspecific and reflect disturbance in a number of different organs. 17,20,22 Although PDD appears to have a predilection for psittacines, it has been presumptively identified in toucans, Canada geese, and spoonbills. 7 A viral etiology for the disease has been suspected for many years because of the epidemiologic evidence and the characteristic histopathologic changes observed in tissues of affected birds, but no specific virus was identified until the recent discovery of avian bornaviruses. 8,11,12 The present report describes a case of Avian bornavirus (ABV) in a scarlet macaw (Ara macao) exhibiting vague neurologic signs from initial presentation to identification and classification of the virus genotype.
A 1.5-year-old male scarlet macaw weighing 0.95 kg was referred to the University of Wisconsin Veterinary Medical Teaching Hospital (UW-VMTH; Madison, Wisconsin) with a 2-week history of depression and loss of appetite. The bird was housed part of the time in a pet store, which sold and boarded birds of many different species. The bird had been acquired by the pet store owner 1 year earlier from a breeder and had been apparently healthy during this period. The scarlet macaw had been examined by the referring veterinarian (rDVM) for acute onset of lethargy and anorexia per the owner, having lost nearly 8% of its body weight over 2 weeks. Over several visits to the rDVM, the bird was found to have a leukocytosis and radiographic changes, including a gas-filled proventriculus, possible mineral or metal opacities in the ventriculus, and a prominent spleen. Tests for plasma levels of lead and zinc by atomic absorption and flame atomic absorption spectrophotometry, respectively, were within normal limits. Testing for Chlamydophila psittaci by polymerase chain reaction (PCR) assay was negative. The rDVM prescribed antibiotics (enrofloxacin and doxycycline; dosages unknown) as well as initiating chelation therapy for heavy metals (ethylenediamine tetra-acetic acid [EDTA] at 40 mg/kg, single dose intramuscularly [IM] in clinic; dimercaptosuccinic acid at 35 mg/kg orally [PO] every 12 hr) while awaiting test results. By the third visit to the rDVM, the bird was increasingly lethargic despite medical therapy and was anemic as well as exhibiting toxic white cell morphology changes on blood work.
When the bird arrived at UW-VMTH, it was thin, with variable periods of alertness followed by sudden periods of recumbency and depression. No other physical abnormalities were noted. Given the history and physical findings, differential diagnoses included chemical intoxication (such as lead and zinc toxicity), PDD, Pacheco's disease (herpesvirus), polyomavirus, fungal granulomatous disease, bacterial septicemia, and hypovitaminosis E and B. The bird was placed in a warm incubator overnight for observation. The following day, symptomatic therapy was initiated: enrofloxacin (15 mg/kg, PO, every 12 hr), along with fluconazole (20 mg/kg, PO, every 48 hr), meloxicam (0.5 mg/kg, PO, every 12 hr), doxycycline (40 mg/kg, PO, every 24 hr), and electrolyte replacement a (40 ml/kg, subcutaneously, every 24 hr). Complete blood cell count (CBC) abnormalities included anemia (packed cell volume [PCV] 29%; reference [ref.] interval: 47-55%) and a pronounced leukocytosis (total white blood cell count = 37 × 103 cells/ml; ref. interval: 7 × 103-22 × 103 cells/μl) with toxic morphologic changes associated with the heterophils. Plasma biochemical abnormalities included hypoalbuminemia (<1.0 g/dl; ref. interval: 1.3-1/6 g/dl) and hyperglobulinemia (>2.6 g/dl; ref. interval: 1.3-1.9 g/dl), increased aspartate aminotransferase (1697 IU/l; ref. interval: 90-180 IU/l), and increased creatine kinase (>8050 IU/l; ref. interval: 180-500 IU/l). Fecal acid-fast staining on direct cytology revealed no acid-fast organisms and a primarily Gram-positive flora. Results of a barium contrast-enhanced gastrointestinal fluoroscopy included normal progression of contrast from the crop to the proventriculus, ventriculus, and small intestines. Normal motility of the caudal esophagus, proventriculus, and ventriculus was noted. Two small metallic opacities were observed in the ventriculus. By the afternoon of the second day at UW-VMTH, the bird began to show more pronounced neurologic signs, including periods of inability to perch, erratic head movements, and transient anisocoria. An electrocardiogram was performed to rule out cardiac rhythm abnormalities leading to syncopal episodes, but a normal sinus rhythm was measured. Assisted crop feeding b via gavage syringe and a single bolus of vitamin B complex (1 mg/kg, IM) were administered. A blood sample was submitted for polyomavirus PCR while Chlamydophila psittaci testing via direct complement fixation assay and PCR was repeated. All of these tests were negative.
A crop wash was collected for aerobic bacterial and fungal culture at the VMTH clinical pathology laboratory. Samples for aerobic culture were plated on chocolate agar, trypticase soy agar containing 5% sheep blood, eosin methylene blue agar, and colistin nalidixic agar with 5% sheep blood, and incubated at 36°C in 5% CO2. Plates were evaluated at 12, 24, and 48 hr postinoculation. Samples for fungal culture were inoculated onto blood heart infusion (BHI) agar, Sabouraud BHI agar, and mycobiotic agar, and incubated overnight at 36°C in 5% CO2. After 12 hr, fungal plates were incubated at 25°C in 5% CO2 and examined on a weekly basis for 1 month. All results were negative for abnormal flora. Supportive care was continued, including assisted feeding and subcutaneous fluid administration. By day 3 of hospitalization, the bird's mentation remained abnormal, and polyuria developed. A CBC was similar to the previous samples, although the anemia appeared to be persisting (PCV 27%). A single dose of vitamin E (0.06 mg/kg, PO) was administered, and calcium EDTA (32 mg/kg, IM, every 12 hr) therapy was started, while the plasma zinc concentration was pending (an in-house, whole blood lead test was negative c ). During days 4 and 5 of hospitalization, the bird became progressively thinner, weaker, and less responsive to handling. By this time, the bird had spent much of its time pressing its head into its food dish. Five days after presentation, the owners elected to euthanize the bird because of the poor prognosis and lack of response to therapy.
At necropsy, the bird was found to be severely emaciated with pronounced pectoral muscle atrophy. The left lung was severely and diffusely congested, and 2 discrete white nodules were noted in the parenchyma. The right lung appeared normal. The pericardium appeared thickened and opaque. The crop and proventriculus were mildly dilated. Histologic examination of the lungs revealed a severe, multifocal, necrotizing, and granulomatous fungal pneumonia in the left lung lobes. The lung lesions had myriad dimorphic fungi that were visible with both hematoxylin and eosin and Gomori methenamine silver staining (Fig. 1). The fungal hyphae had nonparallel walls with frequent branches and occasional septae suggestive of Aspergillus species. A severe tracheitis was also present. Within the brain, there were multifocal areas of moderate polioencephalitis and leukoencephalitis characterized by perivascular lymphoplasmacytic infiltration (Figs. 2, 3). The ganglia associated with the proventriculus and peripheral nerves adjacent to the proventriculus had no histopathologic lesions. However, focally, adjacent to a proventricular artery, there was mild lymphocytic infiltration between the nerve fibers suggestive of lymphocytic neuritis. Within many of the organs (esophagus, intestines, lungs, and airsacs), there were multifocal variably sized, nonrefractile, homogeneous eosinophilic foreign particles of unknown origin (Fig. 4). These histopathologic findings suggested that the macaw had developed fulminant fungal pneumonia secondary to a viral infection exemplified by the perivascular cuffing in the brain.
Samples of the brain and proventriculus were collected at necropsy and sent to Columbia University's Center for Infection and Immunity (New York, New York) along with a serum sample for molecular analyses. RNA from both tissue samples was extracted, DNase I-treated, and reverse transcribed using random hexamer primers as previously described. 11 Both the brain and proventriculus tested positive using degenerate primers developed for detection of described genotypes of ABV that target the C-terminus of the nucleoprotein and the N-terminus of the phosphoprotein. 11,12,24 The sequence of the generated 724-bp fragment was determined by automated dideoxy sequencing d and used for phylogenetic analysis (submitted to GenBank under accession no. GU249593). As shown in Figure 5, the sequence obtained from the macaw clustered with ABV genotype 2. In addition, the macaw's serum was tested for reactivity with purified recombinant ABV N and P proteins in a Western immunoblot assay. 12 Reactivity was noted for both ABV antigens at a serum dilution of 1:1,000 (Fig. 6).
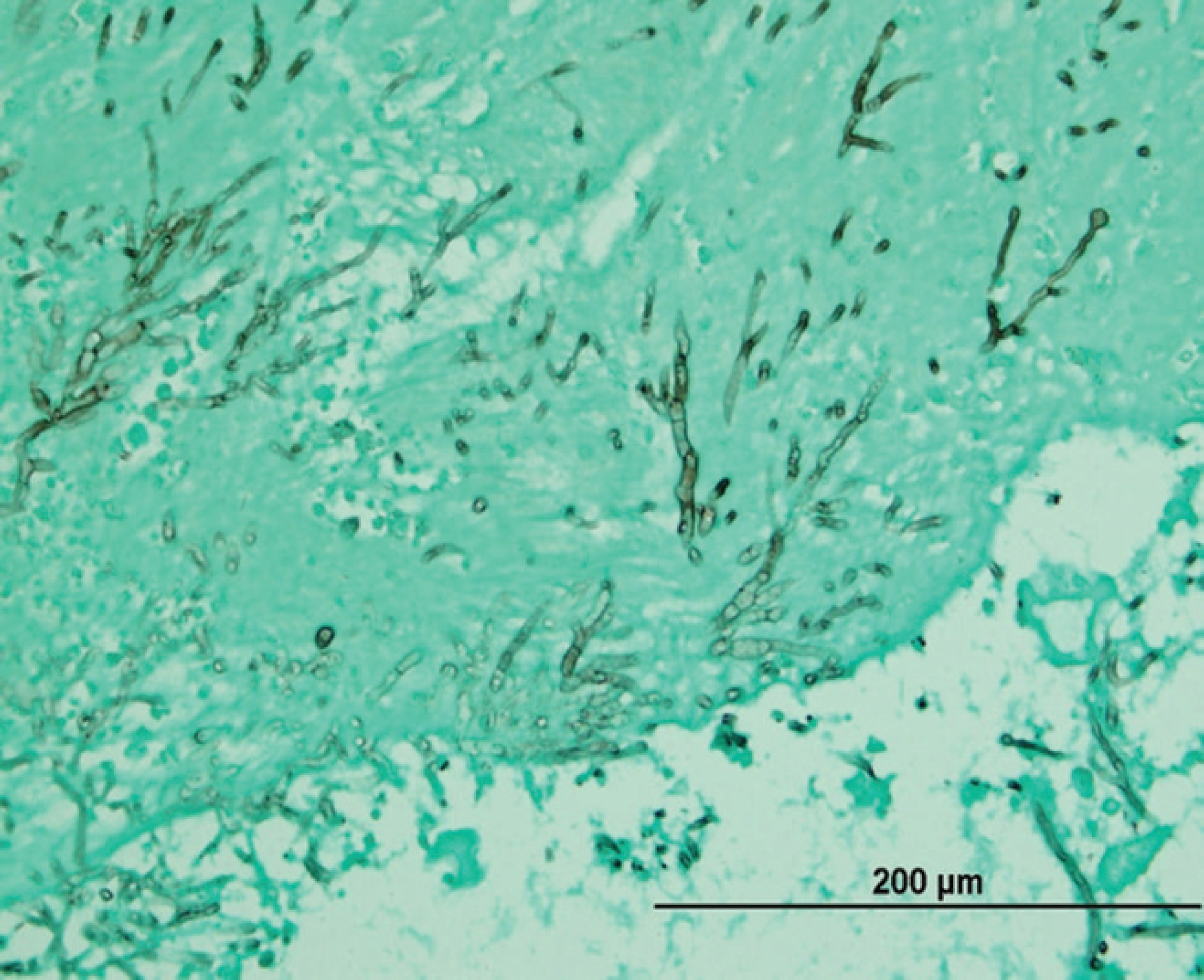
Photomicrograph of lung tissue from a scarlet macaw (Ara macao) infected with Avian bornavirus genotype 2 showing large numbers of fungal hyphae. Gomori methenamine silver. Bar = 200 μm.
Presumptive antemortem diagnosis of PDD was originally excluded as a result of ancillary testing. Typically, an enlarged proventriculus or ventriculus can be observed on plain radiographs while contrast-enhanced fluoroscopy is used to identify gastrointestinal tract dysfunction, although this is not pathognomonic for PDD. 9 More definitive diagnosis has relied on proventricular or crop biopsy of splanchnic nerves. Characteristic histopathologic changes such as lymphoplasmacytic infiltration of the gastrointestinal autonomic nerves and peripheral nerves are considered key findings in birds identified with PDD. 1,8 Unfortunately, most PDD cases are diagnosed postmortem.
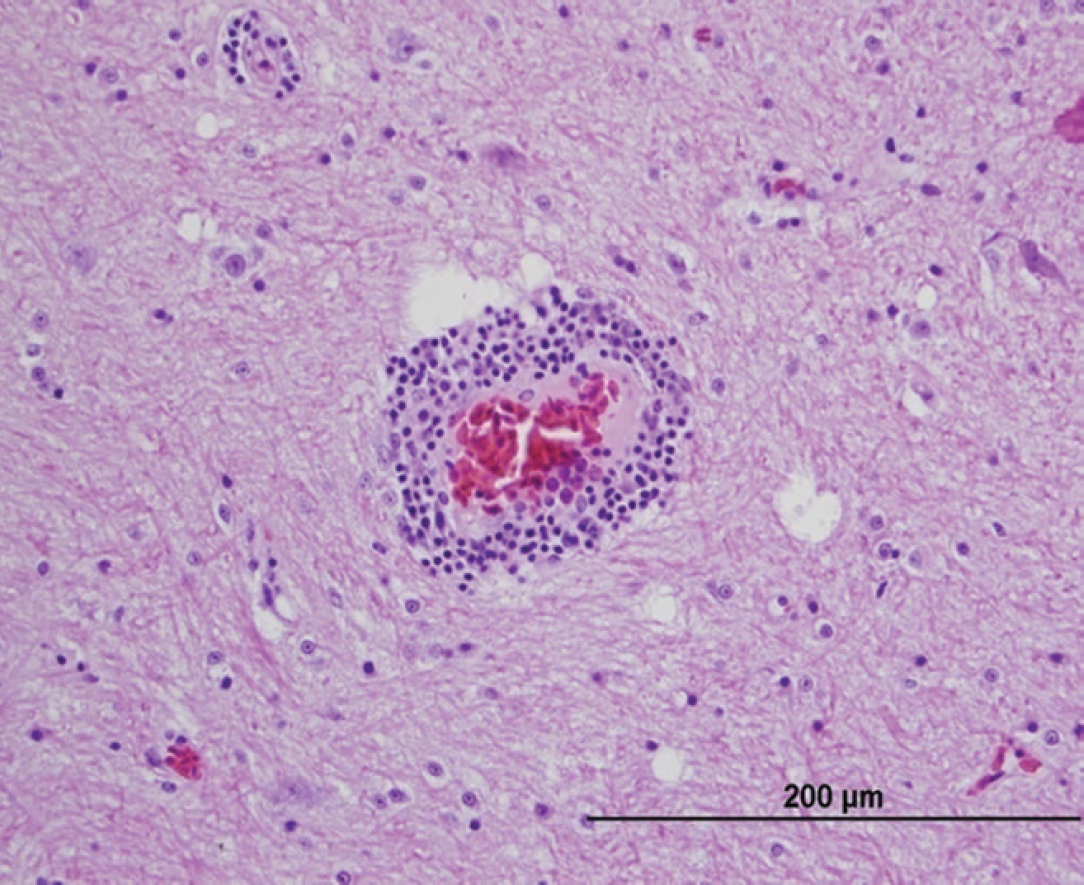
Photomicrograph of brain tissue from a scarlet macaw (Ara macao) infected with Avian bornavirus genotype 2 showing moderate lymphoplasmacytic perivascular infiltration extending into the neuropil. Hematoxylin and eosin. Bar = 200 μm.
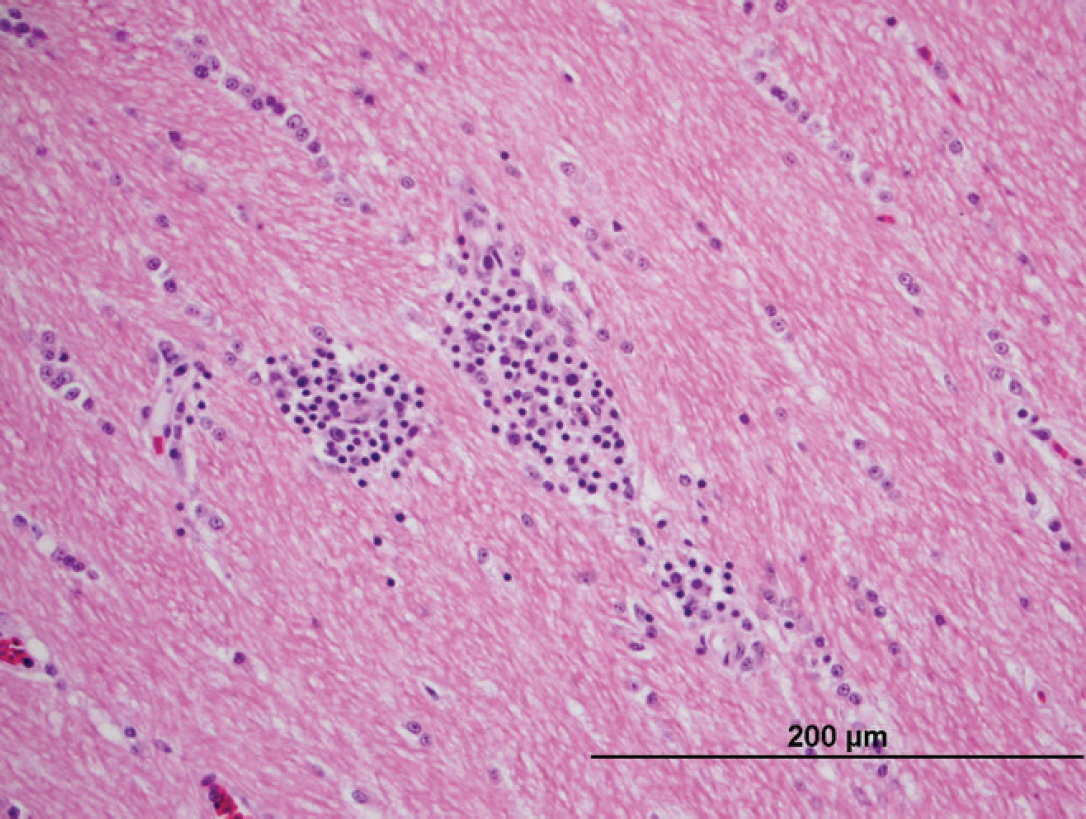
Photomicrograph of brain tissue from a scarlet macaw (Ara macao) showing leukoencephalitis with mild to moderate lymphoplasmacytic infiltration in the white matter. Hematoxylin and eosin. Bar = 200 μm.
Several etiologic agents have been proposed and rejected as the cause of PDD over the years since the disease was first described. The list includes paramyxovirus, adenovirus, and coronavirus as well as Eastern equine encephalitis virus. 5,1,10,19 In the case of the macaw, other potentially neurotropic viruses such as Avian influenza virus, Newcastle disease virus, or Western equine encephalitis virus were excluded based on patient history, gross necropsy findings, and lack of characteristic histopathologic changes associated with these agents.
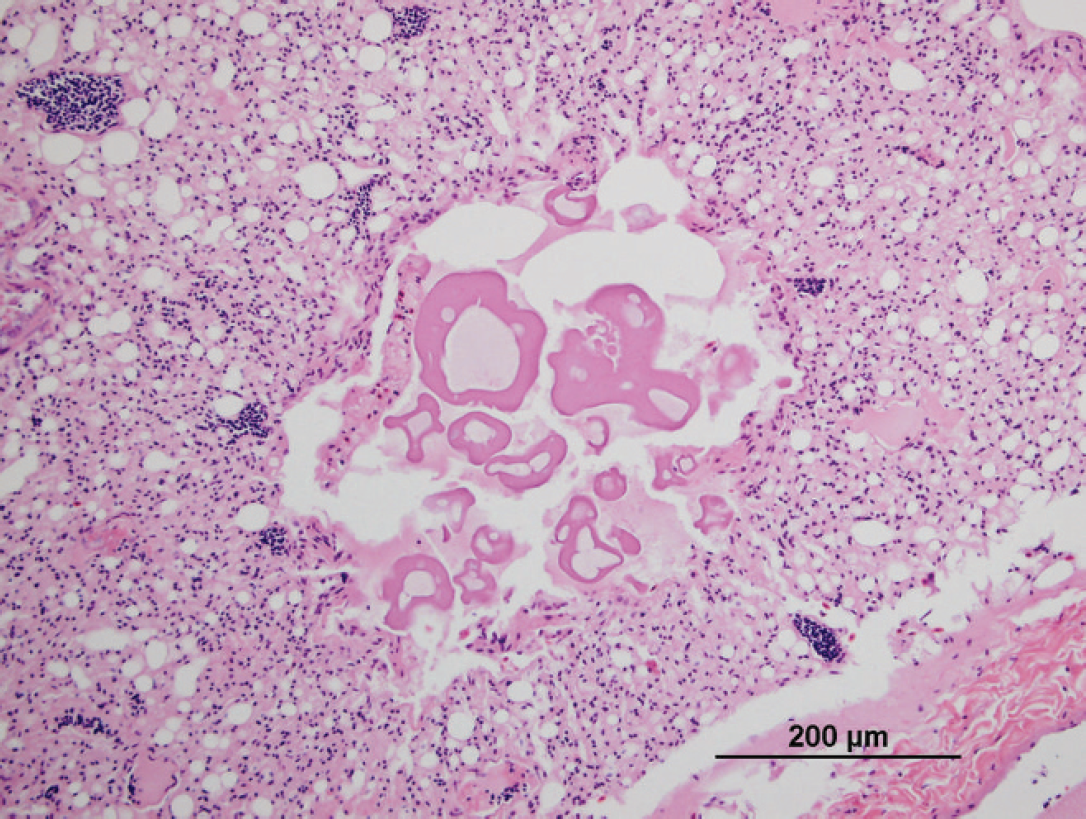
Scarlet macaw (Ara macao). Photomicrograph showing unidentified eosinophilic particles in a parabronchus. Particles were also found in the lungs, airsacs, and gastrointestinal tract. Hematoxylin and eosin. Bar = 200 μm.
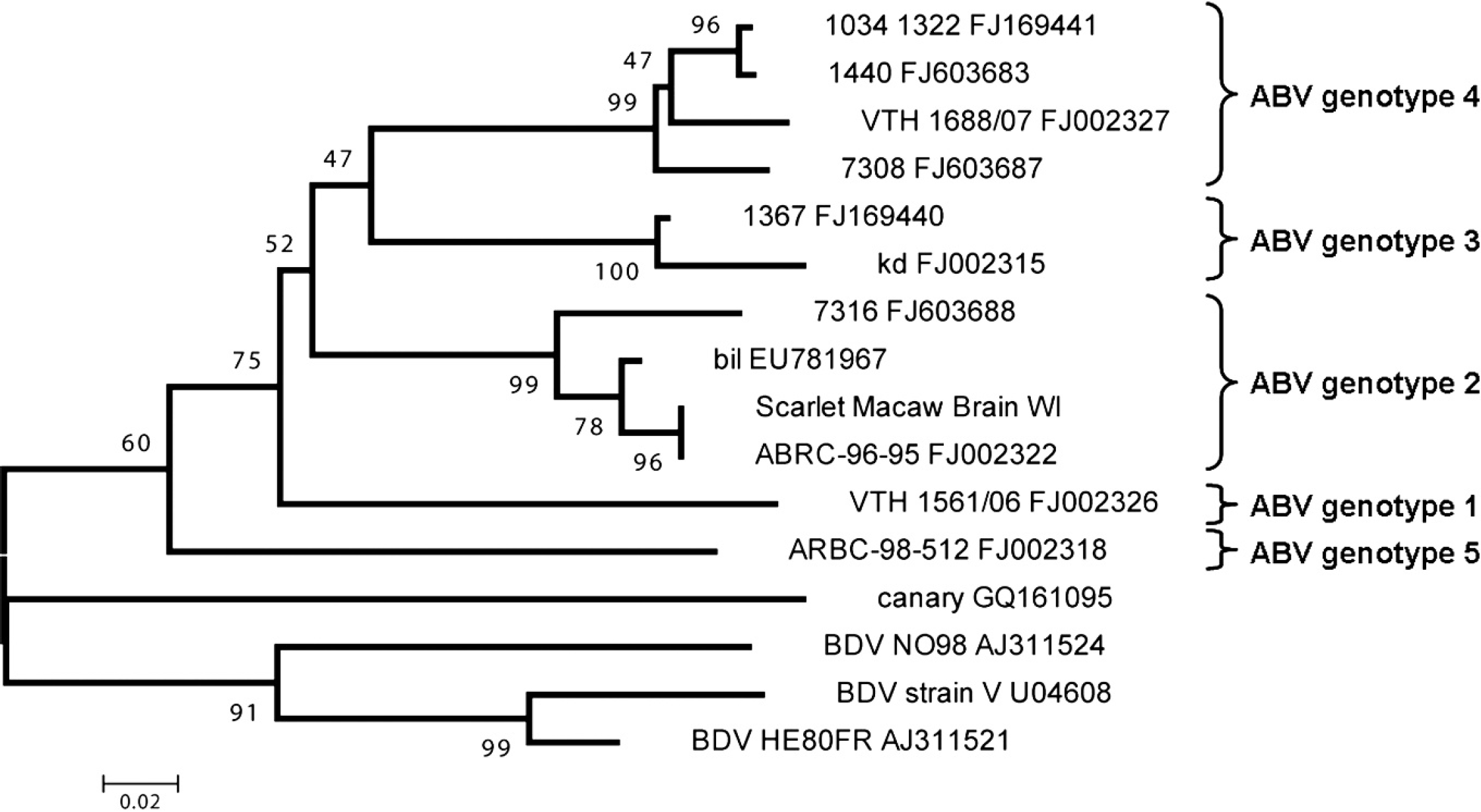
Avian bornavirus (ABV) sequence obtained from brain of a scarlet macaw (Ara macao) that clusters with ABV genotype 2. Representative ABV sequences were retrieved from GenBank (accession numbers are indicated) and aligned with that of ABV from the scarlet macaw using a multiple sequence alignment program. e Phylogenetic relationships were deduced from a 150-bp region of overlap between all sequences by neighbor-joining analysis, applying a Jukes-Cantor model, and performing 1,000 pseudoreplicate analyses. Bootstrap values are given at the respective nodes; scale bar indicates number of substitutions per site.
Recently, novel bornaviruses, belonging to a potential new genus in the family Bornaviridae, have been isolated from the tissues of birds affected with PDD. 4,11,12,21,25 These viruses have been referred to as ABV to distinguish them from Borna disease virus (BDV), which classically causes progressive neurologic disease in horses and sheep. 15 In chickens, experimental BDV infection has also been reported, and BDV has been suspected to be associated with a paresis syndrome in farmed ostriches. 16,18 Unlike BDV, which is highly neurotropic, ABV appear to have a broad tissue distribution, which may explain the variety of clinical signs observed in birds affected with PDD. 13,21,23,25 In addition, experimental transmission of ABV to naïve birds by inoculation with infected tissue homogenates from PDD-infected birds has been accomplished, with resultant PDD-like clinical signs in the inoculated animals. 4,6 Nonpsittacine birds can also be infected naturally and experimentally with ABV. In a 2009 report, ABV was demonstrated in a diseased canary. 25 To date, 6 genetic subgroups of ABV have been proposed. 24 The scarlet macaw-derived virus described herein belongs to genotype 2.
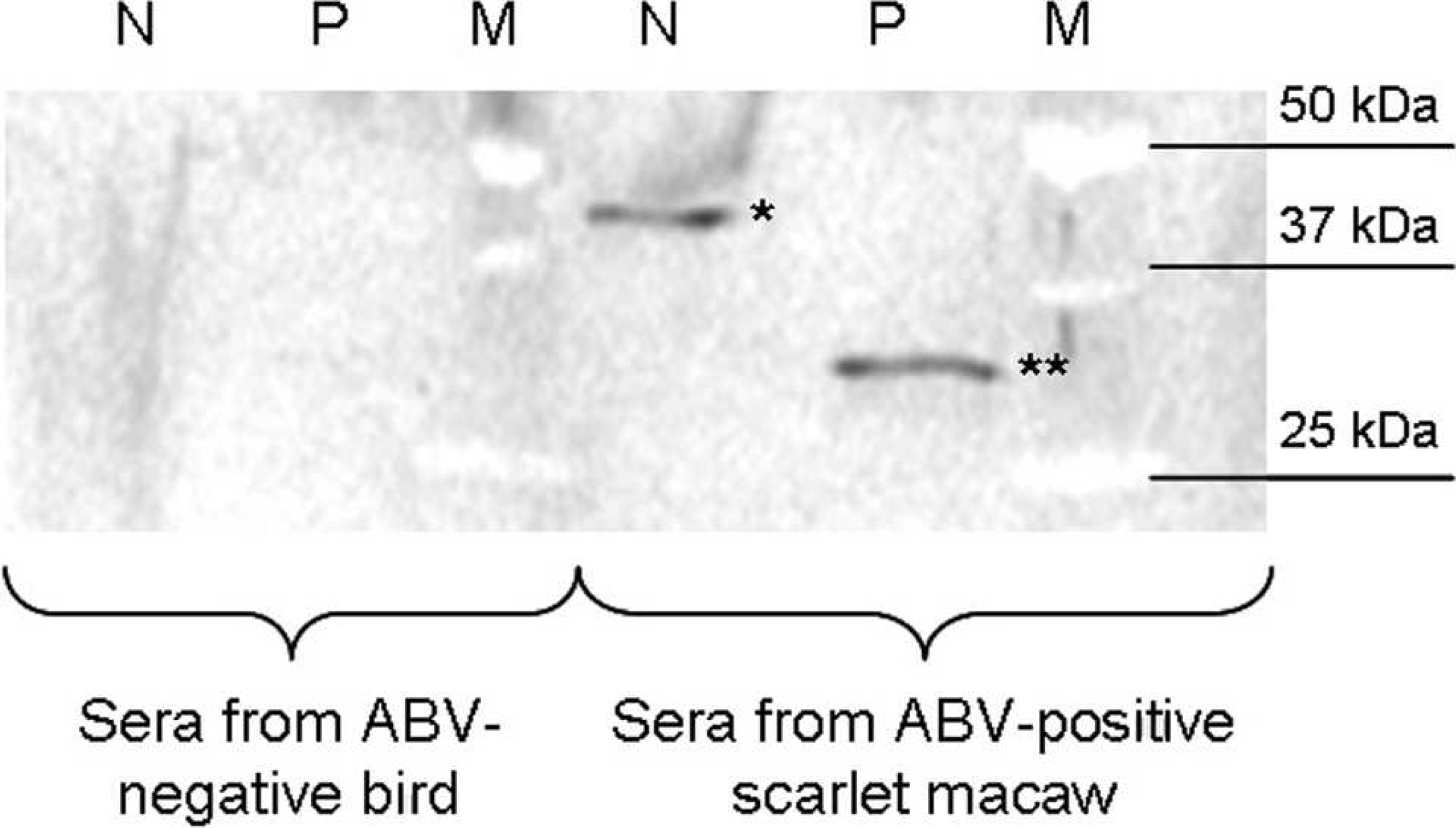
Western blot analysis of serum from the proventricular dilatation disease-affected scarlet macaw (Ara macao). Sera from the Avian bornavirus (ABV)-positive scarlet macaw and an ABV-negative control bird were analyzed at 1:1,000 dilution for reaction with recombinant HIS-tagged nucleocapsid (N, 25 ng; strain 1367, GenBank accession no. FJ169440; 49 kDa, including tag and vector sequence) and phosphoprotein (P, 25 ng; strain 1322, GenBank accession no. FJ169441; 30 kDa, including tag and vector sequence) of ABV blotted onto nitrocellulose; secondary goat anti-bird immunoglobulin G 1:2,000. Macaw antibody reaction with recombinant N (*) and P (**) proteins indicates ABV infection. M = size marker.
The lack of response to medical therapy in the current case is similar to reports in which the most severely affected PDD cases progress rapidly to death, whereas it is thought that asymptomatic cases may live for some time with the disease. 3,4,14 Palliative treatment includes antimicrobial coverage, supplemental feeding with easily digestible food, and fluid therapy. The use of anti-inflammatory medication such as celecoxib, tepoxalin, and meloxicam has become the mainstay therapy for PDD cases. Birds affected with the neurologic form of the disease have a much more guarded prognosis than those showing only gastrointestinal signs. To date, no successful treatment for the neurologic form has been developed. In the present case, it appears that the bird ultimately succumbed to a fungal pneumonia, most likely to presumptive Aspergillus sp. Birds appear to be particularly susceptible to fungal infections when immunocompromised, stressed, or on prolonged antibiotic therapy. 2 Although it is not possible to say whether the bird was infected with the fungus or ABV first, it is likely that the fungal infection was an opportunistic secondary invader. The cause of the tracheitis and the origin of the eosinophilic particles found in multiple tissues remain unclear but may have been related to regurgitation and aspiration of crop contents in this partially recumbent animal, although this was never observed. Cytologic examination of particles of the tube-feeding formula showed that these had identical staining characteristics as the eosinophilic particles found in the bird. However, an ascending pulmonary infection cannot be ruled out.
Risk factors for infection and transmission potential of ABV have yet to be elucidated, though initial studies now suggest that there is a linear progression from infection with ABV to development of PDD. 13 In addition, it is possible that the ABV genotype may affect the form of clinical disease produced. Some birds appear to be able to live with ABV-2 for more than several months without showing any clinical signs. 3,4 Age at infection may determine the rate of progression of disease in infected birds. Younger birds may have a more rapid course of disease progression than older birds. 13
Until recently, a diagnosis of PDD has been largely based on ruling out other diseases and on invasive and potentially risky biopsies of the gastrointestinal tract. With the establishment of a link between ABV and PDD, and the advent of testing for ABV in avian tissues and serum, the ability of veterinarians to diagnose birds with PDD and subsequently counsel bird owners has improved significantly.
